Abstract
Mast cells accumulate in the perivascular tissue during atherosclerotic plaque progression and contribute to plaque destabilization. However, the specific triggers for mast cell activation in atherosclerosis remain unresolved. We hypothesized that psychological stress-induced activation of mast cells may contribute to plaque destabilization. To investigate this, apoE−/− mice on Western-type diet were exposed to 120′ restraint stress. A single episode of restraint caused a significant increase in mast cell activation in the heart. In addition to a rise in serum corticosterone and changes in circulating leukocyte populations, we observed an increase in the circulating pro-inflammatory cytokine interleukin (IL)-6 in the stressed mice. Subsequent characterization of the atherosclerotic plaques revealed a high incidence and larger size of intraplaque hemorrhages in stressed mice. In mast cell-deficient apoE−/− mice, restraint stress affected circulating leukocyte levels, but did not increase plasma IL-6 levels. Furthermore, we did not observe any intraplaque hemorrhages in these mice upon stress, strongly indicating the involvement of a mast cell-dependent response to stress in atherosclerotic plaque destabilization. In conclusion, we demonstrate that acute stress activates mast cells, which induces the incidence of intraplaque hemorrhage in vivo, identifying acute stress as a risk factor for atherosclerotic plaque destabilization.
Similar content being viewed by others
Introduction
Acute cardiovascular syndromes (ACS), such as acute myocardial infarction and stroke remain principle causes of death worldwide1,2. In addition to traditional risk factors for ACS such as dyslipidemia, diabetes and obesity, psychological stress is receiving increased attention as both a contributing factor to various disorders, including cardiovascular disease3. For instance, the INTERHEART study indicated that the presence of chronic psychosocial stressors, such as work stress, increases the risk of myocardial infarction4, and also acute stress has been associated with an increase incidence of ACS5. Very recently, activity of the amygdala, an area of the brain involved in stress, was seen to associate with the incidence of ACS via bone marrow activation and arterial inflammation6. Although these data remain associative, the immune system seems to actively participate in stress-induced ACS. Previously, we and others have established a key role for the inflammatory mast cell in atherosclerosis and especially in the destabilization of advanced atherosclerotic plaques7,8. Furthermore, these experimental animal data are in line with human data, correlating perivascular mast cell numbers and activation status with disease progression and the incidence of ACS9. As mast cells are shown to accumulate in and near atherosclerotic lesions, these cells are uniquely located to respond to acute triggers and subsequently release their pro-atherogenic content. Being in close proximity to perivascular neurons10 and expressing different types of neuropeptide and hormone receptors, a neuron-mast cell connection is likely to play an exacerbating role in cardiovascular diseases. Combined, the epidemiological and experimental data suggest a pro-atherogenic role for stress-induced mast cell activation. However, the direct effects on advanced atherosclerosis, plaque stability and its implications for CVD have not yet been established. In this study we therefore evaluated mast cell-dependent effects of acute stress on atherosclerotic lesion stability in mice.
Results
Restraint stress activates perivascular mast cells
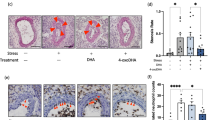
To evaluate the effect of acute physical and psychological stress on perivascular mast cell activation we subjected male apoE−/− mice to a restraint stress protocol. A time course experiment was performed to determine the optimal stress time resulting in mast cell activation. Restraint stress induced a quick and strong increase in circulation glucocorticoid levels (P < 0.05, Fig. 1A), indicating HPA-axis activation. Furthermore, as shown before11, we observed a significant decrease in total white blood cell counts (Fig. 1B), which was caused by a reduction in blood monocyte and lymphocyte numbers, while the amount of circulating neutrophils was not affected by stress (Fig. 1C). The relative leukocyte composition in blood shifted to an increased percentage of neutrophils and a reduction in lymphocytes and monocytes (Fig. 1D). Serum IL-6 levels were significantly higher in the stressed mice compared to unstressed mice (Fig. 2A). Next, we assessed perivascular mast cell numbers and activation status by immunohistochemical staining of heart cross-sections at the level of the aortic root. While absolute mast cell numbers in the perivascular area of the aortoc root were similar in all groups, the percentage of activated mast cells, scored by the presence of granules deposited outside the mast cell (Fig. 2B), was significantly increased upon stress exposure (37.3 ± 1.8% unstressed vs 50.7 ± 5.0% in 120′ stressed mice, Fig. 2C, P < 0.05). Serum levels of the mast cell-granule derived mediator β-hexosaminidase significantly correlated with the percentage of activated mast cells (Fig. 2D). As apoE−/− mice subjected to 2 hours of restraint stress showed a prominent increase in perivascular mast cell activation we used this experimental setup for subsequent experiments.
(A) Restraint stress significantly increased plasma corticosterone levels (n = 6 per group, one-way ANOVA followed by Dunnett’s multiple comparisons test). (B) Automated differential cell counting analysis of blood from non-stressed and 120′ stressed apoE−/− mice showed a significant decrease in circulating WBC levels, which was mainly caused by a reduction in (C) lymphocytes and monocytes directly after stress exposure, both in numbers and as percentage of the total WBC population. Circulating neutrophil numbers were increased. n = 6 per group, *P < 0.05, **P < 0.01, ***P < 0.001. All data in B and C were analyzed using a two-sided unpaired student T-test.
(A) Plasma IL-6 levels were significantly increased in stressed versus non-stressed mice (n = 6 per group, Mann-Whitney test). (B) Representative images of resting (non-activated, arrows) and degranulating (activated, arrow heads) perivascular mast cells in the aortic root. (C) Restraint stress increased mast cell activation in the aortoc root (n = 6 per group, one-way ANOVA followed by Sidak’s multiple comparisons test). (D) The percentage of activated aortic root mast cells significantly correlated with serum β-hexosaminidase activity levels. (Spearman’s correlation, all mice pooled, n = 30). *P < 0.05.
Stress-induced mast cell activation in atherosclerosis
Next, we investigated the effects of acute stress-induced mast cell activation in an atherosclerotic setting. Western-type diet fed apoE−/− mice were sacrificed three days after exposure to the stressor, a time point after which mast cell activation was been shown to have the most prominent effects on the composition of the atherosclerotic lesion7 (Fig. 3A). Again, 120′ restraint produced a strong stress response, indicated by a rise in glucocorticoid levels (Fig. 3B). As expected at this time-point, lesion size in the aortic root was similar between the groups (Fig. 3C). Also, at this timepoint after the stress protocol, we did not observe a significant increase in the percentage of activated perivascular mast cells in the stressed mice anymore (Fig. 3D). Apoptotic cell numbers or relative necrotic core area did not differ between stressed and unstressed mice (data not shown). Relative collagen content did not differ between the groups (Fig. 3E). Interestingly, we observed intraplaque hemorrhages (IPH), a hallmark of unstable plaques characterized by the presence of intimal erythrocytes in 5 out of 13 mice in the stress group (Fig. 3F). In contrast, only one IPH was detected in the unstressed mice. In addition, when we quantified the relative area of the intimal erythrocytes, the IPH in the stressed mice were significant larger compared to control mice (Fig. 3F).
(A) Schematic overview of the experimental setup to evaluate the mast cell contribution to acute stress-induced cardiovascular complications. (B) Stress-induced increase in circulating corticosterone level in apoE−/− mice (n = 13 per group, one-way ANOVA followed by Tukey’s multiple comparisons test). Atherosclerotic lesion size (C), mast cell activation status (D) or collagen content (E) did not differ between the groups at 3 days after the stress protocol (n = 13, two-sided unpaired student T-test). (F) Representative pictures of an intraplaque hemorrhage (IPH), displaying erythrocytes inside the atherosclerotic plaque. Amount and percentage of intraplaque hemorrhages in each group (Fisher’s exact test) and quantification of the IPH area demonstrates significantly larger bleedings on the stressed mice compared to the controls (Mann-Whitney test).
To firmly establish the contribution of mast cells to the stress-dependent effects on plaque stability, we next evaluated the effects of acute stress on plaque stability in the mast cell deficient apoE−/−KitW-sh/W-sh mice. In this experiment, Western-type diet fed apoE−/−KitW-sh/W-sh mice were exposured to the stressor. A subset of mice was bled immediately after the stress protocol and as expected, stress exposure induced similar effects in the WBC populations as compared to the apoE−/− mice (Fig. 4A). Strikingly however, IL-6 levels did not rise upon acute stress in the apoE−/−KitW-sh/W-sh mice (Fig. 4B), and also did not differ between the groups at the time of sacrifice (i.e. 3 days after the stress protocol, Fig. 4C). No significant differences in plaque size were observed between the stressed and non-stressed apoE−/−KitW-sh/W-sh mice (Fig. 4D), while we also did not observe a significant difference in relative collagen content of the plaques (34 ± 2% in the non-stressed group versus 31 ± 3% in the stressed group). Strikingly, no intraplaque hemorrhages could be detected in any of the aortic root plaques of either the non-stressed or stressed mice, suggesting that stress-induced plaque destabilization is, at least partly, mediated by a mast cell-dependent response (Fig. 4E).
(A) Automated differential cell counting analysis of bloodsamples from non-stressed and 120′ stressed apoE−/−KitW-sh/W-sh mice (n = 4/5 per group, unpaired two-sided student T-test). (B) In the apoE−/−KitW-sh/W-sh mice, circulating IL-6 levels were not induced immediately upon stress exposure (n = 5/6 per group, unpaired two-sided student T-test), and (C) also after 3 days, IL-6 levels did not differ between the groups (control: n = 7, stress: n = 13 unpaired two-sided student T-test). (D) Plaque size in apoE−/−KitW-sh/W-sh was not affected at 3 days after the stress protocol (control n = 7, stress n = 11, two-tailed unpaired student T-test). (E) Amount and percentages of IPH in apoE−/−KitW-sh/W-sh mice (Fishers exact test).
Discussion
Plaque inflammation and degradation of the fibrous cap are prerequisites for plaque rupture and identifying risk factors fueling the inflammatory response is key in preventing ACS12. The stress response and its main mediators (glucocorticoids, adrenalin and noradrenalin) are among the most potent immune modulatory agents known to men. Considerable evidence exist for a detrimental and bi-directional role for psychological stress in allergies, asthma and skin diseases13,14. However, such implications for ACS are less well understood. In the current study, we show that restraint stress, a mouse model for acute physical and psychological stress, activates aortic root mast cells resulting in a more unstable atherosclerotic plaque phenotype.
In the human arterial wall, mast cells have been shown to reside in the perivascular tissue10, but also in the intima of the human atherosclerotic plaque mast cells has been detected15. More specifically, mast cells were shown to accumulate in the shoulder region of coronary atheroma. There, these mast cells may degranulate and release both chymase and tryptase, as intraplaque mast cells were seen to contain both proteases16. These proteases, and other mast cells mediators such as chemokines and cytokines, have been shown to enhance smooth muscle cell apoptosis17, collagen degradation18, vascular leakage and overall inflammatory status of the plaque7, thereby potentially contributing to plaque instability. In addition, the mast cell proteases can activate matrix metalloproteinases19, which in turn can degrade extracellular matrix molecules, thus providing an additional mechanism via which mast cells contribute to plaque destabilization. Previous work from our group and others has demonstrated there are several neuropeptides that can induce mast cell activation in atherosclerosis, such as Substance P20 and Neuropeptide Y21. Furthermore, a recent literature review indeed showed that stress can precipitate acute coronary syndromes and demonstrated an important role for coronary mast cell activation through the stress hormone corticotropin-releasing hormone as well as other neuropeptides22.
Our data are in line with previous results showing cardiac mast cell degranulation and histamine secretion upon acute stress exposure23. We also show that restraint stress increases plasma IL-6 levels, which corresponds to observations in human studies, in which circulating IL-6 levels are associated with stress-related disorders3. Circulating IL-6 levels have also been shown to associate with cardiovascular disease24,25, and plasma IL-6 levels were actually reduced in the CANTOS trials upon treatment with canakinumab26. In our experimental study, IL-6 levels failed to rise upon stress exposure in the mast cell deficient mice, suggesting that the mast cell is an important source of IL-6 in episodes of stress. These data comply with a previous study, in which serum IL-6 levels were increased upon ischemia-reperfusion in wild-type mice, which did not occur in mast cells deficient W/Wv mice27. In our study, we measured increased mast cell activation by analyzing the amount of degranulating mast cells, however cytokines, including IL-6, can be released from mast cells via non-degranulation pathways as well. For example, IL-1 can stimulate mast cells to release IL-6 without granule release and thus active degranulation28.
In our study, restraint stress resulted in an increase in the amount of intraplaque hemorrhages, indicative of a more unstable plaque phenotype. Importantly, these stress-induced effects were abolished in the mast cell deficient apoE−/−KitW-sh/W-sh, suggesting that mast cells actively participate in stress-induced intraplaque hemorrhage. In human atherosclerosis, mast cells have been shown to colocalize with intraplaque microvessels29 and by release of basic fibroblast growth factor, mast cells may induce the growth of these immature microvessels in the plaque30. The number of mast cells in carotid artery plaques also associated with the number of microvessels and interestingly, with the incidence of intraplaque hemorrhage9. We here speculate that focal mast cell activation may, by the release of histamine and its proteases, result in leakiness of these small and immature vessels, subsequently leading to intraplaque hemorrhage. By the deposition of erythrocytes and cholesterol these hemorrhages in turn contribute to plaque expansion and destabilization31.
Together, the release of mast cell mediators is likely to contribute to the induction of mast cell dependent plaque destabilization in this stress model, however this remains to be further established, also at later time points after stress exposure. The exact mechanisms via which mast cells are activated during episodes of stress remain to be elucidated as well.
In conclusion, we here demonstrate that a single episode of restraint stress, is sufficient to significantly activate perivascular mast cells, resulting in an increase in serum IL-6 and β-hexosaminidase levels. Such acute stress-induced mast cell activation in mice with established atherosclerotic lesions negatively affected plaque stability by inducing the intraplaque hemorrhage incidence. Combined these results provide further evidence for the important role of mast cells in modulating atherosclerotic plaque stability and highlight the acute stress response as a ACS risk factor.
Methods
Animals
All animal work was approved by the Leiden University Animal Ethics committee and performed in compliance with the Dutch government guidelines and the Directive 2010/63/EU of the European Parliament. 10–12 weeks old male apoE−/− mice and mast cell deficient apoE−/−KitW-sh/W-sh were obtained from the local animal breeding facility and all animals had access to food and water ad libitum.
Restraint-stress model
All stress experiments were performed between 9:00 AM and noon, a period in which individual differences in corticosterone levels in mice are relatively small11. 24 male apoE−/− mice were subjected to restrained stress by immobilization in a well-ventilated 50 mL Corning tube for 30′, 60′, 90′ or 120′ (n = 6 per time point) while control mice were left undisturbed in their home cage. Directly after the indicated stress-time the mice were terminally anaesthetized by subcutaneous injections with ketamine (100 mg/mL), sedazine (25 mg/mL) and atropine (0.5 mg/mL). Deep anesthesia was established by toe pinch reflex, after which the mice were sacrificed. Total cell count, neutrophils, monocyte and lymphocyte counts in blood were analyzed using an automated XT-2000iV veterinary hematology analyzer (Sysmex Europe GMBH, Norderstedt, Germany). Serum IL-6 levels were determined by ELISA according to the manufacturer’s protocol (BD Biosciences). Hearts were excised and briefly fixed in 3.7% neutral-buffered formalin (Formal-Fixx; Shandon Scientific Ltd, UK) before further processing.
Serum analysis
Blood samples for basal corticosterone analysis were drawn by tail cut between 9:00 AM and noon. Levels of corticosterone were determined using a 125-I radioimmunoassay (RIA) with a lower detection limit of 5 ng/ml, according to the manufacturer’s specifications (MP Biomedicals, Illkirch-Graffenstaden, France). To measure β-hexosaminidase levels 50 μL of serum was added to 50 μL 2 mM 4-nitrophenyl N-acetyl-b-D-glucosaminide (Sigma, Germany) in 0.2 M citrate (pH 4.5) and incubated at 37 °C for 2 hours. After addition of 150 μL 1 M Tris (pH 9.0), absorbance was measured at 405 nm.
Atherosclerosis
Atherosclerotic lesion formation was induced by feeding male apoE−/− mice (n = 39) a Western-type diet (0.25% cholesterol and 15% cocoa butter; Special Diet Services, Witham, Essex, UK) for 6 weeks. Blood samples were taken at the start of the experiment, at 2 and 5 weeks of WTD feeding by tail cut and plasma was obtained by centrifugation at 8,000 rpm for 10 min. Levels of total plasma cholesterol were measured spectrophotometrically using enzymatic procedures (Roche Diagnostics, Almere, the Netherlands). At 6 weeks mice were randomly assigned to either the undisturbed control or the 120′ restraint stress (n = 13 per group) and subjected to the stress protocol. Three days after stress all mice were terminally anaesthetized by subcutaneous injections with ketamine (100 mg/mL), sedazine (25 mg/mL) and atropine (0.5 mg/mL). Deep anesthesia was established by toe pinch reflex, after which their vascular system was perfused with PBS at a continuous low flow via heart puncture in the left ventricle. Blood was processed for Sysmex or ELISA/RIA as described above. Heart, skin and lung tissue were fixated in 3.7% neutral-buffered formalin (Formal-fixx; Shandon Scientific Ltd, UK) before further processing. A similar experimental setting was applied to evaluate the effects of stress in mast cell deficient male apoE−/−KitW-sh/W-sh mice (no stress: n = 7, 120′ stress: n = 13).
Plaque morphometry and immunohistochemistry
Harvested hearts were fixated in 3.7% neutral-buffered formalin solution (Formal-fixx; Shandon Scientific Ltd, UK) and embedded in O.C.T compound (Tissue-Tek, Sakura Finetek, CA, USA). Once the aortic root was identified by the appearance of aortic valve leaflets, transverse 10-µm sections were prepared on a Leica CM 3050 S Cryostat (Leica Instruments, Nassloch, Germany) and mounted on gelatin-coated slides. Mean lesion area (in µm2) was calculated from six Oil-Red-O stained sections in distal direction starting at the point where all three aortic valve leaflets first appeared. Leica Qwin image analysis software was used for morphometric analysis of the atherosclerotic burden. Mast cells were visualized by chloroacetate esterase staining according to manufacturer’s protocol (CAE, Sigma, Germany) and degranulation status was assessed manually by bright-field microscopy as described previously7. Mast cells were identified and counted in the perivascular tissue of the aortic root at the site of atherosclerosis. A mast cell was considered resting when all granula were maintained inside the cell, while mast cells were assessed as activated when granula were deposited in the tissue surrounding the mast cell.
In addition, immunohistochemical stainings were performed to assess plaque composition and stability. Sections were stained for collagen (picrosirius red) and macrophages (MOMA-2) as described previously32. Necrotic core area was defined as the a-cellular, debris-rich plaque area and represented as percentage of total plaque area. Apoptosis was visualized using a terminal dUTP nick-end labelling (TUNEL) kit (Roche Diagnostics). As another measure of lesion destabilization the amount and surface area of intraplaque hemorrhages (IPH) were determined as described before7. Presence of masses of intimal erythrocytes, free in the plaque matrix or filling the necrotic core, was classified as intraplaque hemorrhage33. For detection of intraplaque hemorrhage, we analyzed all hematoxylin & eosin-stained sections (at 50 µM intervals) for each mouse having atherosclerotic plaques. Furthermore, erythrocytes were visualized by fluorescence microscopy (erythrocyte autofluorescence at 560 nm emission wavelength). All morphometric analyses were performed by blinded independent operators (I.B. and H.M.L.).
Statistical analysis
Data are expressed as mean ± SEM. A two-tailed student T-test was used to compare normally distributed data between individual groups, while non-parametric data were analyzed using a Mann-Whitney test. For comparison of three or more different groups, data were analyzed with a one-way ANOVA followed by a Tukey’s, Dunnett’s or Sidak’s multiple comparisons test. Correlations were analyzed using a Spearman’s correlation test. Frequency data analysis was performed by means of the Fisher’s exact test. Probability values of P < 0.05 were considered significant.
Data Availability
The data generated during and/or analysed in this study are available from the corresponding author on reasonable request.
References
WHO. Global atlas on cardiovascular disease prevention and control. WHO Available at, http://www.who.int/cardiovascular_diseases/publications/atlas_cvd/en/.
Libby, P. Inflammation in Atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 32, 2045–2051 (2012).
Lagraauw, H. M., Kuiper, J. & Bot, I. Acute and chronic psychological stress as risk factors for cardiovascular disease: Insights gained from epidemiological, clinical and experimental studies. Brain. Behav. Immun. 50, 18–30 (2015).
Rosengren, A. et al. INTERHEART investigators. Association of psychosocial risk factors with risk of acute myocardial infarction in 11119 cases and 13648 controls from 52 countries (the INTERHEART study): case-control study. Lancet. 364, 953–692 (2004).
Steptoe, A. & Brydon, L. Emotional triggering of cardiac events. Neurosci. Biobehav. Rev. 33, 63–70 (2009).
Tawakol, A. et al. Relation between resting amygdalar activity and cardiovascular events: a longitudinal and cohort study. Lancet. 389, 834–845 (2017).
Bot, I. et al. Perivascular mast cells promote atherogenesis and induce plaque destabilization in apolipoprotein E-deficient mice. Circulation. 115, 2516–2525 (2007).
Sun, J. et al. Mast cells promote atherosclerosis by releasing proinflammatory cytokines. Nat. Med. 13, 719–724 (2007).
Willems, S. et al. Mast cells in human carotid atherosclerotic plaques are associated with intraplaque microvessel density and the occurrence of future cardiovascular events. Eur. Heart J. 34, 3699–3706 (2013).
Laine, P., Naukkarinen, A., Heikkilä, L., Penttilä, A. & Kovanen, P. T. Adventitial mast cells connect with sensory nerve fibers in atherosclerotic coronary arteries. Circulation. 101, 1665–1669 (2000).
Dalm, S. et al. Age-related changes in hypothalamic-pituitary-adrenal axis activity of male C57BL/6J mice. Neuroendocrinology. 81, 372–380 (2005).
Otsuka, F., Joner, M., Prati, F., Virmani, R. & Narula, J. Clinical classification of plaque morphology in coronary disease. Nat. Rev. Cardiol. 11, 379–389 (2014).
Dhabhar, F. S. & McEwen, B. S. Acute stress enhances while chronic stress suppresses cell-mediated immunity in vivo: a potential role for leukocyte trafficking. Brain. Behav. Immun. 11, 286–306 (1997).
Kitagaki, H., Hiyama, H., Kitazawa, T. & Shiohara, T. Psychological stress with long-standing allergic dermatitis causes psychodermatological conditions in mice. J. Invest. Dermatol. 134, 1561–1569 (2014).
Kaartinen, M., Penttilä, A. & Kovanen, P. T. Accumulation of activated mast cells in the shoulder region of human coronary atheroma, the predilection site of atheromatous rupture. Circulation. 90, 1669–1678 (1994).
Kaartinen, M., Penttilä, A. & Kovanen, P. T. Mast cells of two types differing in neutral protease composition in the human aortic intima. Demonstration of tryptase- and tryptase/chymase-containing mast cells in normal intimas, fatty streaks, and the shoulder region of atheromas. Arterioscler. Thromb. 14, 966–972 (1994).
den Dekker, W. K. et al. Mast cells induce vascular smooth muscle cell apoptosis via a toll-like receptor 4 activation pathway. Arterioscler. Thromb. Vasc. Biol. 32, 1960–1969 (2012).
Bot, I. et al. Mast cell chymase inhibition reduces atherosclerotic plaque progression and improves plaque stability in ApoE−/− mice. Cardiovasc. Res. 89, 244–252 (2011).
Johnson, J. L., Jackson, C. L., Angelini, G. D. & George, S. J. Activation of matrix-degrading metalloproteinases by mast cell proteases in atherosclerotic plaques. Arterioscler. Thromb. Vasc. Biol. 18, 1707–1715 (1998).
Bot, I. et al. The neuropeptide substance P mediates adventitial mast cell activation and induces intraplaque hemorrhage in advanced atherosclerosis. Circ. Res. 106, 89–92 (2010).
Lagraauw, H. M. et al. Vascular neuropeptide Y contributes to atherosclerotic plaque progression and perivascular mast cell activation. Atherosclerosis. 235, 196–203 (2014).
Alevizos, M., Karagkouni, A., Panagiotidou, S., Vasiadi, M. & Theoharides, T. C. Stress triggers coronary mast cells leading to cardiac events. Ann. Allergy Asthma Immunol. 112, 309–316 (2014).
Huang, M., Pang, X., Letourneau, R., Boucher, W. & Theoharides, T. C. Acute stress induces cardiac mast cell activation and histamine release, effects that are increased in Apolipoprotein E knockout mice. Cardiovasc. Res. 55, 150–160 (2002).
Deliargyris, E. N. et al. Sites of interleukin-6 release in patients with acute coronary syndromes and in patients with congestive heart failure. Am. J. Cardiol. 86, 913–918 (2000).
Raymond, R. J., Dehmer, G. J., Theoharides, T. C. & Deliargyris, E. N. Elevated interleukin-6 levels in patients with asymptomatic left ventricular systolic dysfunction. Am. Heart J. 141, 435–438 (2001).
Ridker, P. M. et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N. Engl. J. Med. 377, 1119–1131 (2017).
Bhattacharya, K. et al. Mast cell deficient W/Wv mice have lower serum IL-6 and less cardiac tissue necrosis than their normal littermates following myocardial ischemia-reperfusion. Int. J. Immunopathol. Pharmacol. 20, 69–74 (2007).
Kandere-Grzybowska, K. et al. IL-1 induces vesicular secretion of IL-6 without degranulation from human mast cells. J. Immunol. 171, 4830–4836 (2003).
Kaartinen, M., Penttilä, A. & Kovanen, P. T. Mast cells accompany microvessels in human coronary atheromas: implications for intimal neovascularization and hemorrhage. Atherosclerosis. 123, 123–131 (1996).
Lappalainen, H., Laine, P., Pentikäinen, M. O., Sajantila, A. & Kovanen, P. T. Mast cells in neovascularized human coronary plaques store and secrete basic fibroblast growth factor, a potent angiogenic mediator. Arterioscler. Thromb. Vasc. Biol. 24, 1880–1885 (2004).
Kolodgie, F. D. et al. Intraplaque hemorrhage and progression of coronary atheroma. N. Engl. J. Med. 349, 2316–2325 (2003).
Bot, I. et al. Serine protease inhibitor Serp-1 strongly impairs atherosclerotic lesion formation and induces a stable plaque phenotype in ApoE−/−mice. Circ. Res. 93, 464–471 (2003).
Rosenfeld, M. E. et al. Advanced atherosclerotic lesions in the innominate artery of the ApoE knockout mouse. Arterioscler. Thromb. Vasc. Biol. 20, 2587–2592 (2000).
Acknowledgements
This work was supported by grants 2010B244 (H.M.L.), 2010B029 (A.W.) and 2012T083 (I.B.) from the Dutch Heart foundation. We acknowledge the support from the Netherlands CardioVascular Research Initiative”: the Dutch Heart Foundation, Dutch Federation of University Medical Centres, the Netherlands Organisation for Health Research and Development and the Royal Netherlands Academy of Sciences” for the GENIUS project “Generating the best evidence-based pharmaceutical targets for atherosclerosis” (CVON2011-19).
Author information
Authors and Affiliations
Contributions
H.M.L., A.W., D.v.d.V. and I.B. performed the experiments. H.M.L., J.K. and I.B. wrote the main manuscript text and H.M.L. and I.B. prepared the figures. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lagraauw, H.M., Wezel, A., van der Velden, D. et al. Stress-induced mast cell activation contributes to atherosclerotic plaque destabilization. Sci Rep 9, 2134 (2019). https://doi.org/10.1038/s41598-019-38679-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-38679-4
This article is cited by
-
Effects of lifestyle factors on leukocytes in cardiovascular health and disease
Nature Reviews Cardiology (2024)
-
Cellular and Molecular Mechanisms of Mast Cells in Atherosclerotic Plaque Progression and Destabilization
Clinical Reviews in Allergy & Immunology (2024)
-
Effects of Psychological Stress on Vascular Physiology: Beyond the Current Imaging Signal
Current Cardiology Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.