Abstract
Evidence on behavioural abnormalities in children exposed to secondhand smoke is limited. This study examined the relation between infant/ toddler cotinine concentration, a biomarker of secondhand smoke exposure, and behavioural problems in preschoolers who were unexposed to maternal smoking during gestation. A prospective cohort of 301 non-smoking mothers with their young children aged ≤18 months visiting postnatal primary care clinics in Hong Kong was enrolled in 2012 and followed by telephone survey 3 years afterwards. Saliva was collected at baseline for cotinine assay. Child behavioural health at 3-year follow-up was assessed by the parent-reported Strengths and Difficulties Questionnaire (SDQ). We conducted multivariable linear regressions to compute regression coefficients (b) of SDQ scores in relation to salivary cotinine level. Mean ± SD age of children at follow-up was 3.7 ± 0.5 years and 50.8% were boys. After adjusting for age, sex, birthweight, household income, housing type, maternal education and depressive symptoms, greater cotinine concentrations during early childhood were associated with greater conduct problems (b = 0.90, 95% CI 0.03–1.76) and hyperactivity/ inattention (b = 1.12, 95% CI 0.07–2.17) at preschool age. This study corroborates previous findings on the potential role of secondhand smoke in development of child behavioural problems.
Similar content being viewed by others
Introduction
Despite growing literature on the associations of secondhand smoke (SHS) exposure with behavioural problems in children, evidence is inadequate to infer causality1,2. Recent reviews suggested SHS exposure may contribute to development of hyperactivity/ inattention and conduct problems in children but did not identify any prospective study that used an objective measure of SHS (e.g. cotinine)3,4. Many studies were also limited by suboptimal control for maternal smoking during pregnancy (MSP)5, which may have independent effect on child behavioural problems6. Given up to 40% of the world’s children are exposed to SHS7, more robust evidence on its harm is imperative to advocate for stronger tobacco control measures.
It is challenging to separate the effect of postnatal SHS exposure from MSP on child health outcomes since mothers who smoked during pregnancy are likely to continue smoking postnatally2. Statistical control for MSP (e.g. by multivariable regression) may yield misleading estimates due to high collinearity between prenatal and postnatal maternal smoking5. A better approach is to restrict the analysis to offspring of non-smoking mothers5. However, among the few studies that followed the recommended approach, one was cross-sectional8 and the others relied on subjective maternal report of child SHS exposure9,10, which is prone to underestimation and potentially biased11,12.
Hong Kong is a densely-populated city where MSP is rare (3.4%)13, but SHS is pervasive due to the crowded living environment and multiunit housing14. An increased exposure to SHS in children at home was observed after smoking was outlawed in most public areas in 200715. We were not aware of any study on SHS exposure in children aged ≤18 months, who typically reside at home most of the time, and subsequent risk of behavioural problems. Therefore, using a prospective cohort of young children whose mothers were non-smokers in Hong Kong, we examined the association of SHS exposure during early childhood with behavioural health at preschool age.
Methods
Study design
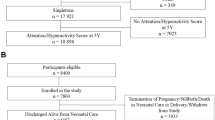
A cross-sectional study was conducted from April to August 2012 in 4 of the 33 Maternal and Child Health Centres (MCHC) in Hong Kong, which provide child health assessments and immunisation services to the general public. Details of the baseline study have been reported elsewhere16. Nurses on duty in the MCHC screened the eligibility of mothers with their children based on the following criteria: (1) the mother is a non-smoker; (2) the child is under 18 months; (3) the parents and the child live together in the same household; (4) the mother is able to communicate in Cantonese/Putonghua; and (5) both the mother and the child are Hong Kong residents. A total of 692 non-smoking mothers with an infant/toddler (mean ± SD age 6.2 ± 5.7 months) completed a self-administered questionnaire on their child’s health and family socio-demographics and provided contact information. In 2015, an a posteriori telephone follow-up survey was conceived 3 years after the baseline study, which included measures of child behavioural problems using the Strengths and Difficulties Questionnaire (SDQ).
The study protocol was approved by the Institutional Review Board of the University of Hong Kong/Hospital Authority West Cluster. The conduct of the study follows the Declaration of Helsinki. Informed consent was obtained from all mothers prior to participation.
Measures
Cotinine, a major metabolite of nicotine, is the biomarker of choice to quantify SHS exposure17. We collected saliva as it can be sampled easily and non-invasively relative to urine and blood sampling from young children. To collect saliva, trained research assistants placed a sorbette (a sponge-tip shaft) under the child’s tongue for 15 to 30 seconds. The sorbette soaked with saliva was immediately stored in a 2 mL microcentrifuge tube and then frozen at −20 °C using ice pads in a cooler box for transportation. Salivary cotinine concentrations (ng/mL) were analysed at the National University of Singapore using an enzyme-linked immunosorbent assay kit (Salimetrics), which has a minimum detectable limit of 0.05 ng/mL. Salivary samples were also obtained from the mothers to verify their non-smoking status.
The child’s behavioral health at the 3-year follow-up was assessed using 25 items on psychological attributes of the parental-report version of the SDQ18, which has been validated locally19. The 25 items are divided into 5 subscales: emotional symptoms, conduct problems, hyperactivity/inattention, peer relationship problems and prosocial behaviour. Each item has a 3-point rating scale of 0 to 2, which are summed to give an overall score ranging from 0 to 10 for each subscale. The total difficulties score is the sum of all but the prosocial behaviour subscales. The parental-report version of the SDQ has also been validated as a screening tool for attention deficit/hyperactivity disorder (ADHD) and conduct disorder in preschoolers at ages 3 to 4 years20.
Potential confounders that may influence both SHS exposure and child behavioural problems were also assessed. At baseline, we recorded the date of birth, sex and birth weight (kg) of the children, maternal education level, monthly household income and housing type. At follow-up, we assessed maternal depressive symptoms using the Patient Health Questionnaire-9 (PHQ-9), which has been validated in the local Chinese general population21. Higher composite scores in the PHQ-9 (range 0–27) indicate greater severity of depressive symptoms.
Statistical analyses
Cotinine concentration was analysed as a continuous variable with logarithmic-transformation (log10) because of its skewed distribution, which is also a standard method used in previous studies8,22. Baseline characteristics of subjects with complete data on cotinine and SDQ were compared with those without complete data on cotinine and SDQ using t-test, Mann-Whitney U test and chi-square test as appropriate. Since SDQ scores were normally distributed, linear regressions were used to examine the associations of sample characteristics with SDQ subscale scores and total difficulties scores.
The associations between SDQ scores and log-transformed salivary cotinine concentration were also examined using linear regressions. A regression coefficient (b) of 1.00 means the SDQ scores increase by 1 unit with 10 times increase in salivary cotinine concentration. In addition to child’s age and sex, potential confounders including birthweight, maternal education, household income and housing type were adjusted in model 123,24. Few previous studies on SHS and child behavioural health controlled for maternal mental health25,26. In model 2, we further adjusted for maternal depressive symptoms (PHQ-9)23,24. As a sensitivity test, we repeated the multivariable regression analyses with exclusion of infants whose mothers had salivary cotinine concentration of ≥12 ml ng/mL, which suggests active smoking27.
Incomplete data on log-transformed infant salivary cotinine (n = 112) were imputed using multivariate normal imputation on a missing at random assumption28. The imputation model included all potential confounders and outcome variables29. Fifty datasets were created. The distributions of the imputed data compared well to the observed data as assessed by diagnostic plots30, which were corroborated by the results of Kolmogorov-Smirnov tests (P < 0.05 only in 2 out of the 50 imputed data sets). The mean ± SD log-transformed cotinine level of the imputed data was also very similar to that of the observed data (0.0489 ± 0.45 vs 0.0485 ± 0.38). Complete case analysis was also performed. All analyses were conducted using Stata/IC version 13.1 with 2-sided P < 0.05 denoting statistical significance.
Results
Of the 692 mother-child pairs recruited at baseline, 445 (64.3%) mothers were successfully re-contacted to participate in the telephone follow-up survey. Loss to follow-up were mostly due to failure to contact (n = 177) and refusal (n = 66). The present study analysed data collected from 301 mother-infant dyads who completed the SDQ, which was included in the follow-up questionnaire as an optional component. Of these 301 dyads, 189 provided valid infant saliva sample for cotinine assay at baseline. The unavailability of cotinine data was mostly due to insufficient saliva sample collected by the sorbette for cotinine assay, especially in younger children (5.7 vs 6.2 months old).
Table 1 shows that subjects with (n = 189) or without (n = 503) complete data on cotinine and SDQ were similar with regard to all baseline characteristics including salivary cotinine concentration, age, sex, birthweight, household monthly income, housing type and maternal education (all P ≥ 0.11; effect sizes ≤0.094). The analytic sample (subjects with data in SDQ; n = 301) were also similar to the excluded sample (n = 391) in terms of all baseline characteristics (P = 0.11 to 0.68; effect sizes 0.016 to 0.163).
The children stayed at home most of time at baseline (mean ± SD hours per day in the past week = 21.5 ± 3.8). At the 3-year follow-up, the mean ± SD age of children was 3.7 ± 0.5 years and 50.8% were boys. Most mothers (n = 292/301) reported none to minimal depressive symptoms (PHQ-9 score ≤4) with mean ± SD score of 0.8 ± 1.6. Table 2 shows that maternal depressive symptoms were strongly associated with hyperactivity/ inattention, peer relationship problems and total difficulties of the children.
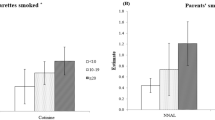
Table 3 shows that after adjusting for age, sex, birthweight, monthly household income and maternal education (model 1), higher cotinine concentration were associated with higher scores in conduct problems (b = 0.93, 95% CI 0.07 to 1.78) and hyperactivity/ inattention (b = 1.27, 95% CI 0.22 to 2.32) subscales. The associations were attenuated but remained statistically significant after further adjusting for maternal depressive symptoms (model 2). No significant association was observed between salivary cotinine level and other SDQ domains in the adjusted models.
Results from the complete case analyses were very similar to those of imputed data analyses (Table 3), although the associations for conduct problem were not statistically significant (P = 0.057 in model 3) probably due to insufficient statistical power.
For the sensitivity test, only 3.1% of mothers (8/ 253) were found to have salivary cotinine concentrations of ≥12 ng/ml. Exclusion of their children from the regression analyses (Model 3) did not change the observed associations of cotinine concentration with conduct problems (b = 1.05, 95% CI 0.09 to 2.02) and hyperactivity/ inattention (b = 1.17, 95% CI 0.12 to 2.22).
Discussion
To our knowledge, this is the first study prospectively examining the association of SHS exposure with child behavioural problems using an objective biomarker of SHS exposure. We found that exposure to SHS during early childhood was associated with conduct problems and hyperactivity/ inattention after about a 3-year follow-up. The associations remained significant after controlling for several key confounders including birthweight, maternal education and depression23,24.
Comparison with earlier studies is hampered by their vastly diverse research methodologies including different tools used to assess child behavioural health. The well-established SDQ was among the most commonly used instruments in recent years, although results were inconsistent. A survey found parent-reported child SHS exposure at home was associated with SDQ-measured hyperactivity/ inattention in children26. Another study further showed an association with conduct problems using salivary cotinine31. However, neither of these studies accounted for MSP. Two cross-sectional studies found parental report of household smoking associated with child hyperactivity/ inattention, conduct and emotional problems after statistical control for MSP32,33, but the results were not replicated in a prospective cohort using similar measures34. Other prospective studies using maternal-reported measures of postnatal SHS exposure did not find significant associations between exposure and behavioural problems in children of non-smoking mothers9,10. Our study addressed these limitations and found cotinine-measured SHS exposure in children ≤18 months was associated with conduct problems and hyperactivity/inattention. The results were consistent with other cross-sectional studies which, after adjusting for MSP, found cotinine-measured SHS to be linked with child behavioural problems8,22,25,35.
The validity of our findings was further strengthened by excluding mothers who smoked to preclude the potential effect of MSP2,5. Self-reported smoking status by Chinese mothers with young children in Hong Kong has been validated previously36. Our findings did not change after restricting the analyses to children whose mothers were non-smoking with biochemically validation. Although we were unable to separate mothers who were former smokers from never-smoking mothers in our data, MSP is rare in Hong Kong (3.4%)13. It is also very unlikely that mothers who formerly smoked during pregnancy subsequently participated in our study, since few mothers who continued smoking during pregnancy quit after delivery of the newborn2.
Our study accounted for maternal depressive symptoms, which contribute to both child SHS exposure and behavioural outcomes. We observed a strong bivariable associations between maternal depressive symptoms and child behavioural problems. However, it is also possible that child behavioural problems may contribute to or aggravate maternal mental health problems. Further research using a longitudinal design and repeated measurements of child and maternal mental health is warranted.
The biological mechanisms by which postnatal SHS may contribute to child behavioural problems has remained an area of active research. Gospe and colleagues first showed the independent biological effect of postnatal SHS on neurodevelopment in rodent models: postnatal SHS exposure reduced cellular density and increased cell size in the brains of rats unexposed to tobacco smoke prenatally37. More recent studies have suggested exposure to nicotine in SHS leads to disruption of the cholinergic system, which may underlie the development of child behavioural problems38. Epigenetic processes have also been suggested to mediate the relationship between SHS and hyperactivity/inattention, although further research is needed to test the hypothesis39.
This study has some limitations. First, due to incomplete data and loss to follow-up, numerous cases were excluded from the study. However, our attrition analyses using both significance tests and effect size calculations showed that all measured baseline characteristics were comparable between subjects with and without complete data on salivary cotinine and SDQ, suggesting that the risk of selection bias was likely minimal. Missing data on exposure were also imputed with multiple imputation diagnostics to increase the precision of the estimates. Complete case analyses also produced similar results. Notably, the analytic sample tended to have better, though insignificant, socioeconomic profile than the excluded sample. This might skew our observed associations toward the null since children with lower socioeconomic positions are more vulnerable to SHS exposure. Second, data on child behavioural health was maternal-reported. However, the parental version of SDQ has been validated to screen for conduct disorder and ADHD in preschoolers and predicts clinical endpoints 2 years later20. The parental-report version of SDQ in Chinese has also been validated locally19. Third, although we adjusted for several key confounders, unmeasured confounding by inherited factors and residual confounding by parental and family environmental factors remain a possibility. Nevertheless, known genetic variants that predict child behavioural problems are relatively rare with weak effect sizes40. Finally, while our study accounted for MSP, we were not able to rule out the potential effect of maternal exposure to SHS during pregnancy on child behavioural problems41.
In conclusion, SHS exposure during early childhood was associated with conduct problems and hyperactivity/inattention at preschool age after precluding MSP. Young children typically reside at home most of the time throughout early childhood and are unable to protect themselves from passive smoking. Our findings, if replicated by further studies, can be a new warning for parents to quit smoking or to establish smoke-free home rules, and back implementation of smoke-free policy to safeguard the health of children.
Data Availability
The dataset generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Samet, J. M. Could secondhand smoke exposure harm the mental health of children? Arch. Pediatr. Adolesc. Med. 165, 370–372 (2011).
US Department of Health and Human Services. The health consequences of involuntary exposure to tobacco smoke: a report of the Surgeon General. Atlanta, GA: US Department of Health and Human Services, Centers for Disease Control and Prevention, Coordinating Center for Health Promotion, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health (2006).
Zhou, S. et al. Physical, behavioral, and cognitive effects of prenatal tobacco and postnatal secondhand smoke exposure. Curr. Probl. Pediatr. Adolesc. Health Care 44, 219–241 (2014).
Pagani, L. S. Environmental tobacco smoke exposure and brain development: The case of attention deficit/hyperactivity disorder. Neurosci. Biobehavl. Rev. 44, 195–205 (2014).
Eskenazi, B. & Castorina, R. Association of prenatal maternal or postnatal child environmental tobacco smoke exposure and neurodevelopmental and behavioral problems in children. Environ. Health. Perspect. 107, 991–1000 (1999).
Tiesler, C. M. & Heinrich, J. Prenatal nicotine exposure and child behavioural problems. Eur. Child Adolesc. Psychiatry 23, 913–929 (2014).
Öberg, M., Jaakkola, M. S., Woodward, A., Peruga, A. & Prüss-Ustün, A. Worldwide burden of disease from exposure to second-hand smoke: a retrospective analysis of data from 192 countries. The Lancet 377, 139–146 (2011).
Cho, S.-C. et al. Association between urine cotinine levels, continuous performance test variables, and attention deficit hyperactivity disorder and learning disability symptoms in school-aged children. Psychol. Med. 43, 209–219 (2013).
Tiesler, C. M. et al. Passive smoking and behavioural problems in children: results from the LISAplus prospective birth cohort study. Environ. Res. 111, 1173–1179 (2011).
Yang, S., Decker, A. & Kramer, M. S. Exposure to parental smoking and child growth and development: a cohort study. BMC Pediatr. 13, 104, https://doi.org/10.1186/1471-2431-13-104 (2013).
Boyaci, H., Etiler, N., Duman, C., Basyigit, I. & Pala, A. Environmental tobacco smoke exposure in school children: parent report and urine cotinine measures. Pediatr. Int. 48, 382–389 (2006).
Wilkinson, J. D., Arheart, K. L. & Lee, D. J. Accuracy of parental reporting of secondhand smoke exposure: The National Health and Nutrition Examination Survey III. Nicotine Tob. Res. 8, 591–597 (2006).
Lok, K. Y. W., Bai, D. L. & Tarrant, M. Predictors of breastfeeding initiation in Hong Kong and Mainland China born mothers. BMC Pregnancy Childbirth 15, 286, https://doi.org/10.1186/s12884-015-0719-5 (2015).
Luk, T. T. et al. Exposure to secondhand smoke infiltration at home amongst Hong Kong residents: a population-based study. Tob. Induc. Dis. 16, A143, https://doi.org/10.18332/tid/84086 (2018).
Ho, S. Y. et al. Comprehensive smoke-free legislation and displacement of smoking into the homes of young children in Hong Kong. Tob. Control 19, 129–133 (2010).
Wang, M. P. et al. Paternal smoking and maternal protective behaviors at home on infant’s saliva cotinine levels. Pediatr. Res. 83, 936–942 (2018).
Avila-Tang, E. et al. Assessing secondhand smoke using biological markers. Tob. Control 22, 164–171 (2013).
Goodman, R. The Strengths and Difficulties Questionnaire: a research note. J. Child Psychol. Psychiatry 38, 581–586 (1997).
Lai, K. Y. et al. Validation of the Chinese version of the strengths and difficulties questionnaire in Hong Kong. Soc. Psychiatry Psychiatr. Epidemiol. 45, 1179–1186 (2010).
Croft, S., Stride, C., Maughan, B. & Rowe, R. Validity of the strengths and difficulties questionnaire in preschool-aged children. Pediatrics 135, e1210–e1219 (2015).
Yu, X., Tam, W. W., Wong, P. T., Lam, T. H. & Stewart, S. M. The Patient Health Questionnaire-9 for measuring depressive symptoms among the general population in Hong Kong. Compr. Psychiatry 53, 95–102 (2012).
Bandiera, F. C., Richardson, A. K., Lee, D. J., He, J.-P. & Merikangas, K. R. Secondhand smoke exposure and mental health among children and adolescents. Arch. Pediatr. Adolesc. Med. 165, 332–338 (2011).
Galéra, C. et al. Early risk factors for hyperactivity-impulsivity and inattention trajectories from age 17 months to 8 years. Arch. Gen. Psychiatry 68, 1267–1275 (2011).
Russell, A. E., Ford, T., Williams, R. & Russell, G. The association between socioeconomic disadvantage and attention deficit/hyperactivity disorder (ADHD): a systematic review. Child Psychiatry Hum. Dev. 47, 440–458 (2016).
Yolton, K. et al. Environmental tobacco smoke exposure and child behaviors. J. Dev. Behav. Pediatr. 29, 450–457 (2008).
Padrón, A. et al. Exposure to secondhand smoke in the home and mental health in children: a population-based study. Tob. Control 25, 307–312 (2016).
Jarvis, M. J., Fidler, J., Mindell, J., Feyerabend, C. & West, R. Assessing smoking status in children, adolescents and adults: cotinine cut‐points revisited. Addiction 103, 1553–1561 (2008).
Lee, K. J. & Carlin, J. B. Multiple imputation for missing data: fully conditional specification versus multivariate normal imputation. Am. J. Epidemiol. 171, 624–632 (2010).
Moons, K. G., Donders, R. A., Stijnen, T. & Harrell, F. E. Using the outcome for imputation of missing predictor values was preferred. J. Clin. Epidemiol. 59, 1092–1101 (2006).
Eddings, W. & Marchenko, Y. Diagnostics for multiple imputation in Stata. Stata J. 12, 353–367 (2012).
Hamer, M., Ford, T., Stamatakis, E., Dockray, S. & Batty, G. D. Objectively measured secondhand smoke exposure and mental health in children: evidence from the Scottish Health Survey. Arch. Pediatr. Adolesc. Med. 165, 326–331 (2011).
Chastang, J. et al. Postnatal environmental tobacco smoke exposure related to behavioral problems in children. PloS One 10, e0133604, https://doi.org/10.1371/journal.pone.0133604 (2015).
Twardella, D., Bolte, G., Fromme, H., Wildner, M. & Von Kries, R. Exposure to secondhand tobacco smoke and child behaviour–results from a cross‐sectional study among preschool children in Bavaria. Acta Paediatrica 99, 106–111 (2010).
Rückinger, S. et al. Prenatal and postnatal tobacco exposure and behavioral problems in 10-year-old children: results from the GINI-plus prospective birth cohort study. Environ. Health Perspect. 150–154 (2010).
Joo, H. et al. Secondhand smoke exposure and low blood lead levels in association with attention-deficit hyperactivity disorder and its symptom domain in children: a community-based case–control study. Nicotine Tob. Res. 19, 94–101 (2016).
Mak, Y. W., Loke, A. Y., Lam, T. H. & Abdullah, A. S. M. Validity of self-reports and reliability of spousal proxy reports on the smoking behavior of Chinese parents with young children. Addict. Behav. 30, 841–845 (2005).
Gospe, S. M., Zhou, S. S. & Pinkerton, K. E. Effects of environmental tobacco smoke exposure in utero and/or postnatally on brain development. Pediatr. Res. 39, 494–498 (1996).
Slotkin, T. A. Cholinergic systems in brain development and disruption by neurotoxicants: nicotine, environmental tobacco smoke, organophosphates. Toxicol Appl. Pharmacol. 198, 132–151 (2004).
Schuch, V., Utsumi, D. A., Costa, T. V. M. M., Kulikowski, L. D. & Muszkat, M. Attention deficit hyperactivity disorder in the light of the epigenetic paradigm. Front. Psychiatry 6, 126 (2015).
Thapar, A., Cooper, M., Eyre, O. & Langley, K. Practitioner review: what have we learnt about the causes of ADHD? J. Child. Psychol. Psychiatry 54, 3–16 (2013).
Leung, C. Y., Leung, G. M. & Schooling, C. M. Early second‐hand smoke exposure and child and adolescent mental health: evidence from Hong Kong’s ‘Children of 1997’birth cohort. Addiction 110, 1811–24 (2015).
Acknowledgements
We thank the mothers and children for their participations in this study.
Author information
Authors and Affiliations
Contributions
M.P.W., S.Y.N., D.S.K., T.H.L. and S.S.C. conceived and designed the study. M.P.W., S.Y.N. and D.S.K. collected the data. T.T.L. and M.P.W. analysed the data. T.T.L. wrote the first draft of the manuscript. All authors critically revised and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Luk, T.T., Wang, M.P., Suen, Y.N. et al. Early childhood exposure to secondhand smoke and behavioural problems in preschoolers. Sci Rep 8, 15434 (2018). https://doi.org/10.1038/s41598-018-33829-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-33829-6
Keywords
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.