Abstract
Zic3 encodes a zinc finger protein essential for the development of meso-ectodermal tissues. In mammals, Zic3 has important roles in the development of neural tube, axial skeletons, left-right body axis, and in maintaining pluripotency of ES cells. Here we characterized cis-regulatory elements required for Zic3 expression. Enhancer activities of human-chicken-conserved noncoding sequences around Zic1 and Zic3 were screened using chick whole-embryo electroporation. We identified enhancers for meso-ectodermal tissues. Among them, a mesodermal enhancer (Zic3-ME) in distant 3′ flanking showed robust enhancement of reporter gene expression in the mesodermal tissue of chicken and mouse embryos, and was required for mesodermal Zic3 expression in mice. Zic3-ME minimal core region is included in the DNase hypersensitive region of ES cells, mesoderm, and neural progenitors, and was bound by T (Brachyury), Eomes, Lef1, Nanog, Oct4, and Zic2. Zic3-ME is derived from an ancestral sequence shared with a sequence encoding a mitochondrial enzyme. These results indicate that Zic3-ME is an integrated cis-regulatory element essential for the proper expression of Zic3 in vertebrates, serving as a hub for a gene regulatory network including Zic3.
Similar content being viewed by others
Introduction
Zic family of zinc finger proteins is a versatile toolkit for metazoan development1. They are involved in the regulation of gene expression during cell-fate decision, regulation of cell proliferation and physiology2,3,4. Vertebrates possess five Zic-related genes (Zic1-5) except teleost fishes, which possess six Zic genes1, among which Zic1-3 are proposed to be generated in a vertebrate ancestor5, and play essential roles in the development of the nervous system and mesodermal derivatives, including axial skeleton and somites.
Zic3 is required for the maintenance of pluripotency in embryonic stem (ES) cells6, neuroectoderm and mesoderm differentiation, and determination of left-right axis of the internal organs7. In humans, Zic3 is a causal gene for heterotaxy and VACTERL association8,9,10,11,12,13,14. The other vertebrate-specific Zic subtypes, Zic1 and Zic2, contribute differentially to vertebrate development. Zic1 plays essential roles in neural development2. It enhances neural differentiation and its deficiency causes the cerebellar dysgenesis in humans and mice15; whereas, Zic2 is required for medial forebrain development in humans and mice16.
The apparently different roles of the Zic genes in vertebrate development may be partly explained by their differential expression profiles17,18,19 because similar protein functions are revealed in some comparative studies20,21. As a basis for the spatiotemporally defined Zic expression profiles, several upstream signaling and trans-acting factors have been described for Zic1 and Zic3. For instance, suppression of the BMP signal is a common upstream factor for the upregulation of Xenopus Zic1 and Zic3 during neuroectodermal differentiation22,23. In addition, Sonic Hedgehog acts as an inhibitory factor for mouse Zic1 expression in the ventral side24. At later stages, the involvement of Meis1/Pbx induces Zic3 expression in the generation of neural crest cells25. Moreover, Brachyury upregulates the mesodermal expression of Xenopus Zic326.
Several studies have addressed the role of cis-regulatory elements, along with the associated signaling and transcription factors (TFs), in the regulation of Zic gene expression. Examples of these cis-regulatory elements include- (1) The 5′ flanking region of mouse Zic1 that controls the dorsal spinal cord expression27; (2) The BMP inhibitor-responsive promoter region in Xenopus Zic128. (3) The midbrain-hindbrain enhancer for zebrafish (Danio rerio) Zic2 and Zic529; (4) The neural plate border enhancer for zebrafish Zic3 and Zic630, and (5) The somite-mesodermal enhancer for Zic1 and Zic431. However, these regulatory elements still do not fully explain the Zic expression profiles, and none of these elements has been shown to be necessary for Zic expression during development.
To understand the mechanism underlying the regulation of Zic gene expression, we analyzed the cis-regulatory elements required for the expression of vertebrate Zic1 and Zic3, and characterized a developmentally critical Zic3 mesodermal enhancer in terms of its activity in chicken and mouse embryos, potential trans-acting factors, and its evolutionary history.
Results
Zic1 and Zic3 expression in early chick development
To identify enhancers critical for embryonic Zic expression, we used chicken embryos as an experimental system. In chicken embryos, expression of Zic1–Zic4 has been described in neuroectoderm, paraxial mesenchyme, brain, spinal cord, neural crest, inner ear, and limb buds24,32,33,34. We chose Zic1 and Zic3, whose regulatory signaling cascades and cis-elements are partly known, as targets of analysis.
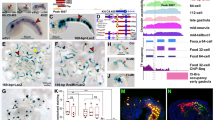
We examined the expression profiles of Zic1 and Zic3 during early chicken development by whole-mount in situ hybridization (Fig. 1). At the Hamburger and Hamilton (HH)35 stage 4 (definitive streak stage, Fig. 1A,G), Zic3 expression can be seen in the epiblast of the prospective anterior neuroectodermal region and along the primitive streak, but Zic1 expression was faint at an area near the anterior primitive streak. At the head fold stage (HH6, Fig. 1B,H), both Zic1 and Zic3 were broadly expressed in the neural plate. Zic1 expression was enhanced in the anterior region, whereas Zic3 expression increased posteriorly. A dense Zic3 signal could be seen at the node and the moderate expression continued at the primitive groove. At HH7 (Fig. 1C,I), Zic1 and Zic3 expression in the neural folds showed anterior-posterior profiles similar to those in the HH6 stage, but the signal was enhanced in the lateral region of the neural plate as seen in the cross sections (Fig. 1a–c,g–i). Moreover, Zic3 expression was strong at the prechordal plate and paraxial mesoderm. At the following stage (HH8, Fig. 1D,J), Zic3 expression was clearly observed at the somites. At later stages (HH9, Fig. 1E,K; HH11, Fig. 1F,L) of nervous systems, Zic1 expression was accentuated at the anterior end of the telencephalic vesicle, diencephalon and hindbrain region and Zic3 expression was enhanced in the broad region of the telencephalic vesicle, mesencephalic region, and some segments in hindbrain region. Dorsal restriction of the expression at the hindbrain and spinal cord was commonly observed for both Zic1 and Zic3 (Fig. 1d–f,j–m). In the mesoderm derivatives, Zic3 expression was observed strongly at both somites and unsegmented mesoderm, and modest Zic3 expression was observed at the notochord progenitors; whereas, Zic1 expression was observed at dorsomedial somites. These expression profiles were consistent with previous studies which described Zic1 and Zic3 mRNA distribution at later stages [e.g. at HH10–2433, at HH7–1334] and distribution of the Zic proteins at HH8–2324.
Expression of chicken Zic1 and Zic3 during development (A–F, a–f) Zic1, (G–L, g–m) Zic3. (A,G) HH4, (B,H) HH6, (C,I) HH7, (D,J) HH8, (E,K) HH9, (F,L) HH11. (a–m) Cross section images at the positions indicated in (C,F,I and L). Arrowheads, prechordal plates. Arrows, notochords. Open arrowhead in L, notochord progenitors. Scale bars, (A–L) 1 mm; (a–m) 100 μm.
In mesodermal development, the Zic3 expression is predominant at these stages. The expression in unsegmented mesoderm was shown to be enhanced at presomitic mesoderm at HH934. During somite development, Zic3 is expressed in two or three of the most recently formed somites34. The expression initially decreased in somites followed by detectable expression in the most anterior mature somites during HH12/13 stages, whereas detectable expression was observed in the dorsomedial dermomyotome and sclerotome34.
Enhancer screening in chicken embryos
We carried out embryo electroporation36,37 to identify enhancers of chicken Zic1 and Zic3. The analysis focused on evolutionary conserved noncoding element (CNE) between human and chicken genome. Twenty-five and twenty-eight CNEs within 150 kb flanking regions were selected for Zic1 and Zic3, respectively (Fig. 2A). The selection of distal CNEs (more than 50 kb apart) was based on the conservation between human and teleost fish sequences. The CNEs were placed upstream of the herpes simplex virus thymidine kinase (TK) promoter-driven EGFP reporter vector. TK promoter was shown to be neutral in terms of the expression in chick embryos in previous studies36. The CNE-TK-EGFP reporters were co-electroporated with a control vector (EF-LacZ) that directs ubiquitous expression. Electroporation was carried out at HH4, and the reporter gene expression profiles were monitored until HH11.
Enhancer screening in Zic1 and Zic3 CNEs. (A) Structure of human Zic4/Zic1 and Zic3. Top indicates the scales in kilobase, centered at the transcription initiation of Zic1 or Zic3 the + (plus) direction is that of transcription. Blue thin vertical lines below gene names indicate the protein coding exons. Clustered green vertical lines indicate the presence of sequence similarity with the human sequence at each position of the sequences of animals indicated at the left side (mouse, chicken, Xenopus, and zebrafish). Extent of conservation is indicated as the height of bars. The extent of conservation among the 100 vertebrate species selected at UCSC genome browser is indicated by 100 vert. Black vertical lines in the CNE line indicate the position of tested CNEs in this study. Ol-a-d and Dr-e1, known regulatory elements identified in teleost fishes30,31. (B) Structure of the reporter vector. TK, herpes simplex virus thymidine kinase promoter (minimal activity by itself); CMV, human cytomegalovirus immediate early enhancer and promoter (strong ubiquitous activity). Pictures, representative results of the reporter expression in chicken embryos at HH11 for CMV, TK, Zic1-NE, Zic1-ME, Zic3-NE1, Zic3-NE2, and Zic3-ME. (Top) Bright field views. (Middle) Reporter GFP signal at HH11 (24 h after electroporation). (Bottom) β-galactosidase activity staining for co-electroporated EF-LacZ to indicate the transfected cells. Scale bar, 1 mm. Frequencies of the reporter GFP expression/Zic1 or Zic3 expressing region are indicated above the pictures. Numbers indicate those of tested embryos. (C) Reporter GFP signals at HH9 (12 h after electroporation). Expression patterns at additional time points are shown in Supplementary Fig. S2. Scale bar, 1 mm. (D) Spatial distribution of enhancer activity in a summary.
As a result, we observed clear enhancer activities for two CNEs for Zic1 and three CNEs for Zic3 (Fig. 2). These CNEs showed spatially restricted enhancer activities. In the neural tissue, Zic1-NE (neural enhancer), Zic3-NE1 (neural enhancer 1), Zic3-NE2 (neural enhancer 2) directed the reporter gene expression (Fig. 2B–D). As to the anterior-posterior (A-P) extent of expression in the HH11 neural tube (Fig. 2B,D), Zic1-NE enhancer activity was observed all along the A-P axis with enhancement in the hindbrain and the spinal cord region. Zic3-NE1 and Zic3-NE2 activities were observed in the hindbrain and the anterior spinal cord region. Along the dorsal-ventral (D-V) axis, Zic1-NE directed wide expression at the HH7 neural plate, but the expression was dorsally restricted at HH16 neural tube as determined by electroporation of the reporter constructs into neural tube at HH11 (Supplementary Fig. S1). The change in expression mimicked that of Zic proteins in the chicken neural tube development24. Zic3-NE1 and Zic3-NE2 directed wide expression along D-V axis in the neural tube at all the stages (Supplementary Fig. S1). In the mesodermal derivatives, Zic1-ME (mesodermal enhancer) reporter expression was observed weakly at somites, whereas Zic3-ME reporter expression was strong at both unsegmented paraxial mesoderm and somites (Fig. 2B–D, Supplementary Fig. S1), mimicking distribution of Zic1 and Zic3 transcripts (Fig. 1). The spatially restricted activities of all five enhancers can be traced back to 7 hours after transfection (HH6) when the enhancer activity became evident, and it continued until 24 hours post-transfection (HH11) (Supplementary Fig. S2, Fig. 2B).
Among the five CNEs, Zic1-NE homologous region in mouse genome was included in the cochlear nucleus and dorsal spinal cord enhancer region within the 2.9 kb region of Zic1 transcription start site (Fig. 2A) that was characterized in a previous study27. The other CNEs were thought to be novel. Zic1-ME was located in an intron of Zic4, which is placed 13 kb upstream of Zic1 in a head-to-head manner (Fig. 2A). Zic3-NE1, Zic3-NE2, and Zic3-ME were located in the 3′ flanking region of Zic3 (Fig. 2A). In chicken, they are 45, 50, and 65 kb apart from Zic3, respectively. The Zic1-NE (0.9 kb), Zic1-ME (1.3 kb), Zic3-NE1 (0.4 kb), and Zic3-NE2 (0.6 kb) sequences are conserved between human and chicken but not between human and zebrafish, and Zic3-ME (1.9 kb) sequence is conserved among human, chicken, and zebrafish (Fig. 2A).
Enhancer activities of the five CNEs in mouse embryos
To characterize the five CNEs, we tested if the homologous region in the mouse genome shows enhancer activities in mouse embryos by a transgenic reporter assay. We prepared transgenes in which the mouse CNE homologues were placed upstream of the Heat shock protein 68 (Hsp68) promoter driven-LacZ. The transgene expression profiles were examined by β-galactosidase activities in the primary transgene recipient mice at embryonic day (E) 10.5 or 11.5.
In the mouse neural tissue, Zic1-NE gave the most robust expression in spinal cord and frequently at dorsal forebrain and midbrain (Fig. 3A,B,L). Zic3-NE2 also showed frequent expression in hindbrain and spinal cord, and Zic3-NE1 transgenes showed frequent expression in the spinal cord (Fig. 3C,D,L). All three CNEs directed the reporter gene expression in spinal cord. Although Zic1-NE transgene showed consistent activity in the dorsal spinal cord where the Zic1 was expressed, Zic3-NE1 and Zic3-NE2 were not consistent with the endogenous Zic3 expression17,38. In the mouse mesoderm derivatives, Zic3-ME frequently showed expression in the axial mesoderm including the unsegmented mesoderm, newly generated somites, and the dermomyotome of the anteriorly located mature somites (Fig. 3I,J,L), and less frequently at external layer of brain (Fig. 3I,K,L). Zic1-ME directed expression in the somite less frequently and weakly (Fig. 3G,H,L).
Expression of reporter gene driven by Zic1 and Zic3 enhancers in transgenic mice. Hsp68 promoter-LacZ was placed downstream of mouse Zic1-NE (A,B), Zic3-NE1 (C,D), Zic3-NE2 (E,F), Zic1-ME (G,H), and Zic3-ME (I–K). (A,C,E,G,I) Lateral views of E10.5 transgenic mouse embryos after X-Gal staining to detect reporter β-galactosidase expression. Scale bar, 1 mm. (B,D,F,H,J,K) Transverse sections along the thin lines indicated in (A,C,E,G and I), respectively. Scale bars, 100 μm. (L) Frequencies of the reporter expression in brain, spinal cord, and somites. Denominators indicate the total number of the tested embryos. (M,N) Whole mount in situ hybridization for E10.5 mice to show Zic1 (M) or Zic3 (N) mRNA distribution as references for the reporter gene assay. Scale bar, 1 mm.
Thus, the expression patterns of the mouse Zic1 and Zic3 CNE-driven reporters were similar to those in chicken embryos, indicating that the enhancer functions are evolutionary conserved between mouse and chicken. However, the expression may not always be limited to the region of mouse Zic1 and Zic3 expression at the corresponding stage (Fig. 3M,N). In this regard, Zic3-ME-driven reporter appears to reliably mimic the Zic3 expression in chicken as well as mouse17,39. We therefore focused on the analysis of Zic3-ME in the following experiments.
Zic3-ME was required for the Zic3 expression in the mesodermal tissue
We next addressed the significance of Zic3-ME by generating Zic3-ME knockout (KO) mice. The mice were generated by replacing entire Zic3-ME by a loxP sequence using homologous recombination in ES cells (Supplementary Fig. S3). We examined Zic3 expression in the E6.5 and E8.5 in Zic3-ME-deficient (Zic3-ME−/Y) and wild type (Zic3-ME+/Y) mice. By in situ hybridization, we found loss of Zic3 transcript in the mesoderm of E6.5 Zic3-ME-deficient embryos (5/5), and an intact expression in the epiblast (Fig. 4B,D,F,J, Supplementary Fig. S4). At E8.5, the Zic3 expression at somites was lost (4/4) (Fig. 4N,P, Supplementary Fig. S4). These results indicate that the Zic3-ME contains cis-regulatory elements required for the proper Zic3 expression at mesoderm and the somites in mouse embryos at the corresponding stages38,40.
Analysis of Zic3-ME-deficient mice. Wild type (Zic3-ME+/Y, A,C,E,G,I,K,M,O,Q and S) and Zic3-ME-deficient mice (Zic3-ME−/Y, B,D,F,H,J,L,N,P,R and T) were collected at E6.5 (A–L, mid- to late-streak stage) and E8.5 (M–T, 9 somite-stage). In situ hybridization was carried out for whole-mount (A–D and M–T), and section (E–L) specimens using Zic3 (A–H,I,J,M–P), Zic2 (G,H), and Brachyury (K,L, Q–T) probes. an, anterior side; po, posterior side; rt, right side; lt, left side. (E,F) are the sections along red lines in (C,D), respectively. (G–I–K and H–J–L) are adjacent sections. Asterisk indicates the mesoderm where Zic3 expression was lost. Curved lines in (O,P) indicate the location of somites (continuous lines) and unsegmented paraxial mesoderm (broken lines) at left side. Scale bars, 200 μm. Additional results are shown in Supplementary Fig. S4.
In the adult Zic3-ME-deficient mice, we did not observe the kinky-tail-like abnormality that is observed in Zic3-deficient mice41,42. Zic2 expression in the mesoderm and the epiblast (Fig. 4H) and Brachyury expression in the primitive streak at E6.5 (Fig. 4L, Supplementary Fig. S4), the notochord, and the caudal mesoderm at E8.5 (Fig. 4R,T) remained intact in the Zic3-ME-deficient embryos. These results indicate that the altered Zic3 expression is not due to the changes in embryonic architecture, but reflects the direct regulation Zic3 gene expression by Zic3-ME.
Delineation and characterization of the core region in Zic3-ME
Since our findings revealed the biological significance of Zic3-ME, we defined the core region of Zic3-ME. For the deletion mutants of chicken Zic3-ME (Fig. 5), the enhancer activity was assayed using whole-embryo electroporation. Sequential deletions from both ends revealed two critical segments within a 633 bp region, which is highly conserved among vertebrates (Figs 5 and 6). We named the 633 bp region as Zic3-ME core. Zic3-ME core sequence was then queried against public DNase I hypersensitive site (DNase-HS) and chromatin-immunoprecipitation sequencing (ChIP-seq) databases.
Deletion analysis of Zic3-ME in chicken electroporation assay. (A) Mapped Zic3-ME core region and the deletion constructs used for the assay. Right columns indicate the frequencies of the reporter expression in somite or neural tube. Segment 1 and segment 2 indicate the regions, of which deletion abolished the somite expression. (B) Representative results of the deletion assay. Top, bright field view; middle, reporter GFP signals; bottom, β-galactosidase activity staining to indicate the transfected cells. Scale bar, 1 mm.
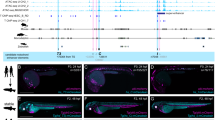
DNase HS, conservation and TF binding sites of the Zic3-ME. (A) Top block, location of DNase I HS signals. DNase I HS peaks in each cell line [top to bottom, K562 (erythroleukemia), NHLF (lung fibroblast), HSMM (myoblast), HUVEC (vein endothelial cell), NHEK (epidermal keratinocyte), HepG2 (liver cancer), GM12878 (lymphoblastoid cell), H7-ES (ES cell), and HeLa-S3 cells (cervix cancer)]. Bottom block, conservation in 100 vertebrate species (summary) and conservation in rhesus monkey, mouse, dog, elephant, chicken, X. tropicalis, and zebrafish. Red open boxes indicate the Zic3 enhancers identified in this study (Zic3-NE1/2 and Zic3-ME) and in zebrafish Dr-E1 and Dr-E230. (B) Higher magnification of Zic3-ME. Red thick bar indicates Zic3 ME core, From top to bottom, histone modification (H3K4Me1), DNase HS signal, and each animal species (mouse, zebra finch, chicken, X. tropicalis, medaka, and zebrafish). At the bottom, location of the indicated TF binding sites defined by the prediction algorithm and ChIP-seq peaks. ChIP-seq peaks of P300, Rad21, Sin3a, Hdac2, and Chd7 were identified at Zic3-ME in ES cells without predicted binding sites. Asterisks at TF indicate the ChIP-seq peaks identified in both human and mouse whereas no asterisks indicate the peaks identified in either human or mouse ChIP-seq studies. Double asterisks indicate the presence of the binding motifs only in the chicken sequence. Bold letters indicate the verification by both ChIP-seq peaks and TF binding site prediction, and non-bold letters indicate verification by either of the methods. (C) Representative DNase-seq and ChIP-seq results of human and mouse ZIC3/Zic3-ME. Box with dotted lines indicate the entire Zic3-ME regions and boxes with solid lines indicate Zic3-ME core regions. Tcf12 (ES-mesendoderm) and Tcf12 (ES-m.e.(mesendoderm) + Nodal block) indicate the embryoid body treated for 2 days with 100 ng/ml Activin and 100 ng/ml Activin plus 10 μM SB431542 (inhibitor of Smad2/3 phosphorylation) respectively65. The overall profiles including other known Zic3 enhancers and their derivations are indicated in Supplementary Fig. S6 and Table S1 respectively.
We found that, Zic3-ME core was included in the DNase I-HS in ES cells, mesodermal cells, and developing neural cells of both human and mouse (Fig. 6A–C). In the mature tissues, brain, retina, and spinal cord cells showed Zic3-ME core in DNase-HS, but not in the heart, skeletal muscle, fat, blood, vascular cells, gut, lung, or liver (ENCODE database)42.
In terms of transcription factor (TF) binding, as evidenced by ChIP-seq experiments, Zic3-ME core was bound by pluripotency-associated TFs (Nanog, Oct4 [Pou5f1]), meso-endodermal differentiation associated TFs (Brachyury [T], Eomes [Tbr2], LEF1), neural differentiation-associated TFs (Zic2, Eomes), and silencer/chromatin-looping/chromatin-remodeling associated factors (Rad21, Sin3a, Hdac2, Chd7), and transcriptional cofactor (Ep300) (Fig. 6C, Supplementary Fig. S5). Accordingly, Zic3-ME contains the predicted TF binding sites for Nanog, Oct4, T, Eomes, LEF1, and Zic proteins at the sites of their ChIP-seq peaks (Figs 6 and 7, Supplementary Figs S5 and S6). In addition, we found putative TF binding sites for Twist cotrolling mesodermal differentiation43, Snai2 also known as slug, controlling mesoderm patterning44, Nr5a2 essential for primitive streak morphogenesis45, and Zfx controlling self-renewal of ES cells46; although, their ChIP-seq results are not yet available (Fig. 7).
Orthologues and paralogues of Zic3-ME core sequence. (A) Multiple sequence alignment of most conserved region in Zic3-ME-core. From top, human, mouse, chicken, Xenopus, medaka, and zebrafish Zic3-ME nucleotide sequences are aligned. At the bottom two lines, the sequences similar to Zic3-ME in the mouse and human 3′ flanking regions of Zic2 (Zic3-ME-like) are indicated. Bold letters indicate the conserved elements between Zic3-ME and its paralogue in the Zic2 flanking region. Underline indicates the exon of PCCA (propionyl-CoA carboxylase alpha subunit), a heterodimeric mitochondrial enzyme. (B) MCCC1 (methylcrotonoyl-CoA carboxylase 1) is a paralogue of PCCA. Translated amino acid sequences of PCCA and MCCC1 are alighned. (C) The positions of Zic3-ME and its paralogues (open boxes) in the human genome. Arrows indicate the transcription start site and direction in each gene.
The binding sites for Nanog, TCF-LEF, Zic2, Twist, and Myc were clustered at the most strongly conserved region where the highest DNase-HS peak lies (Figs 6 and 7). By contrast, TF binding sites for two T-box factors (T and Eomes) were located outside of the peak and included in the segment 2 (Fig. 6B,C), the deletion of which abolishes the somite expression directing activity. The segment 2 sequence is conserved in vertebrates, but not in the teleost fishes (Medaka and Zebrafish, Fig. 6). Although another region, which is necessary for the somite expression in the chicken embryos (segment 1), contained the predicted binding sites for Oct4 and Twist, the sites are not clearly conserved in mammalian species.
Positions of ChIP-seq peaks matched to those of the binding motifs (Fig. 6B,C). However, Ep300, Chd7, Hdac2, Rad21, and Usf1 showed their ChIP-seq peaks irrespective of the binding sequences in agreement with their known roles as cofactor or in chromatin remodeling. Tcf12 (a basic helix-loop-helix protein, also known as Heb) showed the presence of additional peaks besides the peak on the predicted binding sites. Presence of the binding-site-independent-peak depended on the Nodal signal, raising a potential controlling mechanism of Zic3-ME (see Discussion, Figs 6C and 8D, and Supplementary Figs S5 and S7). Interestingly, positions of the DNase-HS peaks with Zic3-ME are different between ES cells and fetal brain tissue both in human and mouse (Fig. 6C). Brain DNA-HS peaks are placed at the distal end of the Zic3-ME core region, overlapping the Eomes and T ChIP-seq peaks. ES DNase-HS peaks coincide with the peaks of Nanog/Oct4/Zic2/Tcf3. The difference of DNase-HS suggest that the Zic3-ME higher order chromatin structure dynamically changes during the differentiation from ES cells into immature neural cells.
Hypotheses concerning the regulatory mechanism and evolution of Zic3-ME. (A) Zic3-ME and Zic3-E1 may associate with the transcriptional regulatory complex on the Zic3 coding region, based on their openness in chromatin-associating proteins, and functionalities as enhancers. This status was highly predictable in ES cells where many ChIP-seq results available, and may be involved in the cell fate specification of ES cells. (B) Zic3-ME was targeted by pluripotency-associated factors, mesodermal differentiation-associated factors, and neural differentiation-associated factors. They are likely to control Zic3 expression in ES, mesodermal, and neural cells in embryonic development. (C) Roles of Zic3-ME, based on the results of this study and the known regulatory relationships between Zic3 and T (top) and between Zic3 and Nanog (bottom). Top, Zic3-T feedback loop may control the expansion of immature mesodermal cells. Bottom, Zic3-Nanog feedback loop may be required for the maintenance of pluripotency by inhibiting endodermal inhibition. In these regulatory loops, Nanog and T associate with Zic3-ME, which in turn activates Zic3 expression. (D) Zic3-ME acts as a switch between mesodermal and endodermal cell fates. Activation of Nodal signaling induces mesendoderm formation, and subsequent high levels of Nodal signaling induces differentiation into definitive endoderm. Binding of Tcf12 to the T-binding site in Zic3-ME is dependent on Nodal signal and suppresses Zic3 expression. (E) It has been proposed that Zic paralogues appeared because of a tandem gene duplication and a subsequent quadruplication in the early vertebrate. This study predicts the presence of a common ancestral gene for PCCA, MCCC1, and Zic3-ME next to the two Zic genes as indicated in the second line. After quadruplication, PCCA, MCCC1, and Zic-ME may have diverged respectively. Ur indicates the common ancestor of the following genes. Red indicates the enhancer property, and black indicates the protein-coding property.
Sequence similar to Zic3-ME core exists in the 3′ flanking region of Zic2
In bioinformatics analysis of Zic3-ME, we noticed the presence of Zic3-ME-related sequences in vertebrate genomes. A BLAST homology search against human genome identified a sequence similar to Zic3-ME, which resides 270 kb downstream of Zic2 (named as Zic3-ME-like, Fig. 7A,C). The sequence alignment showed that the Zic3-ME-like is similar to the most strongly conserved region of Zic3-ME core where the predicted binding sites for Myc and LEF1 are present (Fig. 7A). Surprisingly, a 59 bp region downstream of the Zic3-ME-like included the protein coding exon of propionyl-CoA carboxylase alpha subunit (PCCA) that is transcribed from the transcription start site located 100 kb downstream of Zic2 (Fig. 7C). The human genome also contained a paralogue of PCCA, known as methylcrotonoyl-CoA carboxylase 1 (MCCC1). MCCC1 was found to be located 38 Mb downstream of Zic1 (Fig. 7C). As the PCCA amino acid sequence encoded in Zic3-ME-like is highly conserved in MCCC1 protein (Fig. 7B), a traceable homology existed between Zic3-ME and Zic3-ME-like in the MCCC1 region. The juxtaposition of Zic2-PCCA is conserved widely in the vertebrate species including teleost fish (zebrafish) and cartilaginous fish (elephant shark), and the tandem array of Zic1-MCCC1 was partially conserved in amniotes, including at least cow and chicken (UCSC genome browser, https://genome.ucsc.edu/). However, we did not find any traits of the enhancers (DNase-HS and histone modification) in the ENCODE database. Based on these results, we inferred the evolutionary history of Zic3-ME (See Discussion and Fig. 8E).
Discussion
This study revealed enhancers critical for Zic1 and Zic3 expression in chicken and mouse embryos. The five enhancers, Zic1-NE, Zic1-ME, Zic3-NE1, Zic3-NE2, and Zic3-ME directed the spatially restricted expression in neural or mesodermal tissue. We hypothesize that they are coordinately involved in the regulation of Zic1 and Zic3 expression during embryonic development. However, they may be only a few pieces of enhancers necessary for the proper expression of Zic1 and Zic3. This is because we targeted only the evolutionary conserved region between human and chicken genomes. In this regard, studies on Sox2 enhancer are supportive. Full scanning of upstream and downstream 100 kb of chicken Sox2 regions identified 26 enhancers, of which most (25 out of 26) corresponded to a fraction of conserved sequence blocks between chicken and mammalian genomes36,47. The exceptional one enhancer was proposed to be unique to the chicken47. Therefore, the conserved sequence-targeted enhancer screening as in this study would be effective to identify developmentally critical enhancers if the comprehensiveness is not absolutely needed.
Cis-regulatory elements for Zic1 have been investigated in regions 2.9 kb upstream of mouse Zic1 transcription start site27, 5.0 kb upstream of Xenopus Zic1, 73 kb upstream and 45 kb downstream of zebrafish Zic148, and 106 kb upstream and 36 kb downstream of medaka fish (Oryzias latipes) Zic1 gene31. These studies identified dorsal spinal cord and cochlear nucleus enhancer (Zic1-NE) and core promoter in mouse27, BMP inhibitor-responsive element in Xenopus28, and neural tube and mesodermal enhancer31. We showed that Zic1-NE enhancer activities are conserved in chicken and mouse. Whether or not the enhancer activities are conserved in vertebrates awaits further investigation. However, it is noteworthy that that major mesodermal enhancers in teleost fish reside distantly (11–60 kb) in the 5′ flanking region of Zic1 and they contain the four blocks of sequences conserved among teleost fishes and mammals31 (Ol-a-d in Fig. 2A). Insertion of a large transposon between the teleost mesodermal enhancers and Zic1 results in mesoderm-specific loss of Zic1 expression31. If the Ol-a-d enhancer activities are conserved in chicken or in mouse, Zic1-ME may control the mesodermal expression cooperatively with them.
Two cis-regulatory elements for have been reported in the distant region upstream of zebrafish Zic330. These enhancers are distinct from Zic3-NE1, Zic3-NE2, and Zic3-ME analyzed in this study. Zic3-E1 (16 kb upstream of zebrafish Zic3, Dr-e1 in Fig. 2A) directs reporter gene expression at the neural plate border, and E2 (53 kb upstream) enhancer activity is present in the neural plate border, anterior neural plate, and posterior paraxial mesoderm at an early stage (12 hours post fertilization), anterior dorsal neural tube, and posterior paraxial mesoderm at a later stage (24 hours post fertilization). E1, but not E2, shows clear enhancer signatures in terms of an epigenetic modification peak, summed TF-binding peak, and DNase-HS peaks in human or mouse genomes (Fig. 6 and Supplementary Fig. S5). Zic3-ME and Zic3-E1 may associate with the transcriptional regulatory complex on the Zic3 coding region in mammalian embryos (Fig. 8A), because humans and/or mouse Zic3-E1, Zic3 coding region, and Zic3-ME are all in the open chromatin structure as evidenced by DNase-HS peaks (Supplementary Fig. S5) and are all associated with the active and primed enhancer signature (H3K4Me1)49 (ENCODE database). Furthermore, all three regions are bound by proteins associated with chromatin remodeling factors (Chd7 and Hdac2) or the factor defining higher order chromatin architecture (Rad21)50,51 in ES cells (Fig. 6 and Supplementary Fig. S5).
In the differentiation processes of mesoderm and its derivatives, Zic3-ME binding of TFs related to the mesodermal differentiation may play important roles (Fig. 8B). These include the LEF-TCF TFs that mediate Wnt-β-catenin signaling, which have well known roles in mesodermal differentiation52,53,54. Brachyury (T), a direct target of Wnt-β-catenin signaling55, is a T-box type mesodermal TF that directly activates the mesodermal Zic3 expression in Xenopus embryos26,56. Another T-box factor, Eomes, is essential for mesoderm formation and the recruitment of prospective mesodermal cells to the primitive streak57. Zic2, another member of the Zic family, has been shown to increase the paraxial mesoderm progenitors at the primitive streak cooperatively with Zic338. In addition, Zic2 and Zic3 are also required for precise somitogenesis38. These results indicate that Zic3-ME acts as a highly integrated hub to control mesodermal development.
In previous studies, Zic3 overexpression suppresses Xenopus Brachyury expression and Wnt-β-catenin signaling in mesoderm, resulting in an impaired notochord development58, whereas Brachyury upregulates Zic3 mesodermal expression26. On the other hand, both mouse Zic2 and Zic3 are required to increase the paraxial mesodermal cells in mice38. The results are consistent if we assume that the role of Zic3 is temporally limited to enhancing mesoderm generation, and not in the generation of the notochord at a later stage. This idea is also supported by the expression profile of the chicken Zic3 that is expressed in the notochord progenitor but not in the notochord itself. Collectively, the Zic3-Brachyury (T) regulatory loop (Fig. 8C) may be critically involved in controlling mesodermal development.
Zic3-ME also contains the brain enhancer activity (Fig. 3I,K). The expression of Zic3 in the brain was detected at E10.5 (Fig. 3N, and ref.17), and Eomes (Tbr2) is expressed in the brain beginning around E10.059. Together with the shift of a ChIP-seq peak to the Eomes binding site during neural differentiation (Fig. 6C), we speculate that Eomes could be involved in the brain enhancer activity of Zic3-ME (Fig. 8B).
Although this was not directly assayed in this study, Zic3-ME may be involved in controlling pluripotent stem cells. Because many DNase-HS studies indicate that Zic3-ME is in the open chromatin structure in human and mouse ES/iPS cells (Fig. 6, Supplementary Fig. S5, ENCODE database), and Zic3-ME is bound by pluripotency-associated TFs (Nanog and Oct4) (Fig. 6, Supplementary Fig. S5). In ES cells, knockdown of Oct4, but not of Nanog, suppresses Zic3 expression60,61. Oct4 interacts with Nanog and associates with multiple transcriptional repression complexes including Sin3A complex62. Zic3-ME is likely to be targeted by the Sin3A/deacetylase complex, which has been shown to cooperate functionally with Nanog to promote pluripotency63. Therefore, we speculated that Nanog was involved in fine-tuning Zic3-ME function.
On the other hand, Zic3 protein binds to a promoter region of Nanog and directly upregulate Nanog expression in ES cells64 (Fig. 8C). Moreover, Zic3 and Nanog prevent endodermal lineage specification, and Zic3 expression was required for ES cell pluripotency6. Thus, the Zic3 and Nanog feedback loop as a whole would be associated with the establishment of pluripotency (Fig. 8C). Further investigation on the regulation of Zic3-ME in ES cells would be beneficial for better understanding of the mechanism underlying pluripotency.
Additionally, the ChIP-seq results clarified the role of Zic3-ME regulation in the mesoderm-definitive endoderm bifurcation (Fig. 8D). Tcf12 (also called Heb, a basic helix-loop-helix [bHLH] E protein that forms a heterodimer with another bHLH TF) binds Zic3-ME in a partially Nodal signaling-dependent manner (Fig. 6C, Supplementary Fig. S5)65. Depletion of Tcf12 in mesendodermal cell results in mesodermal differentiation65. Tcf12 has been proposed to be a Smad2/3 cofactor66 that link Nodal signaling65. T also interacts and collaborates with Smad2/3, but mediates mesoderm formation67. Tcf12 and T could alternatively associate with Zic3-ME, since we found ChIP-seq peaks for Smad2/3, Tcf12, and T at the same site in Zic3-ME (Fig. 6C, Supplementary Fig. S5). Furthermore, Tcf12 knockdown increases Zic3 expression in mesendodermal cells (Supplementary Fig. S7). Combining the fact that Zic3 knockdown in ES cells results in the endodermal differentiation6, Zic3-ME may be involved in the switching between mesodermal and endodermal differentiation of ES cells (Fig. 8D).
Finally, based on the presence of additional Zic3-ME-related sequences, we hypothesize the origin of Zic3-ME as follows. Ancestral Zic3-ME may have existed in the vertebrate ancestor after the tandem head-to-head duplication5 (Fig. 8E). At this point, prototypal ME may have coexisted with a protein coding exon of common ancestor gene for PCCA/MCCC1 in the 3′ flanking region of Zic1/2/3 common ancestor. The whole genome quadruplication and subsequent loss of one copy5 may have generated two additional sets of the “head-to-head tandem Zic genes”, PCCA/MCCC1/ME. Thereafter, diversification of PCCA/MCCC1/ME may have occurred where Zic3-ME kept and presumably acquired additional TF binding sites sequences, PCCA exon retained the protein coding sequence with remnant TF binding sequences, and MCCC1 retained the protein coding information, but lost all of the TF binding sequences. We think Zic3-ME may provide us an intriguing model of enhancer evolution, awaiting further validation by experimental and computational molecular phylogenetic analyses.
Methods
Animals
All animal experiments were approved by Animal Experiment Committees at the RIKEN Brain Science Institute and Animal Care and Use Committee of Nagasaki University, and carried out in accordance with the guidelines for animal experimentation in RIKEN and Nagasaki University.
Bacterial Artificial Chromosome (BAC) clones and sequencing
Original chicken Zic cDNA fragments were obtained by low stringency screening of chicken embryo cDNA library24. Zic BAC clones- CH261-95N3 (Zic1), CH261-11A12 (Zic2), CH-98E11 (Zic3) were isolated by high-stringency hybridization using high-density colony hybridization filters; the BAC filter and clones were purchased from BACPAC Resources (Children’s Hospital Oakland Research Institute, Oakland, CA, USA). The hybridization, sequencing, and sequence analysis were performed as described previously5.
Plasmid construction
We used pd2EGFP-1 in which a herpes simplex virus thymidine kinase promoter from pRL-TK (Promega) was inserted at its 5′ multiple cloning site. CNEs were PCR cloned from chicken BAC clones and inserted upstream of the TK-d2EGFP unit. Control EF-LacZ vector was generated by inserting LacZ cassette from pMC1871 (Amersham) and oligonucleotide containing an initiation methionine into pEF-BOS vector68. For the transgenic assay in mouse, CNEs were inserted into an Hsp promoter-driven β-galactosidase reporter cassette69.
In situ hybridization
In situ hybridization (ISH) was performed essentially as described38,58.
Chicken embryo electroporation and enhancer assay
For the description of the chicken developmental stages, Hamburger and Hamilton stages were used35. Chicken embryo electroporation and whole embryo culture was carried out essentially as described36,37. Fertilized chicken eggs were purchased from Inoue Egg Farm (Kanagawa, Japan). Briefly, fertilized eggs were incubated at 38 °C for 26 hours. The chicken embryos at HH4 were excised, and attached to a sterilized paper filter with hole. The yolks attached on embryos were rinsed with Hank’s balanced salt. The embryos were placed upside down on a 2 × 2 mm platinum plate electrode (cathode) on a dish (CUY700P, Nepagene). After one microliter of transfection cocktail (2 μg/μl CNE-TK-d2EGFP, 1 μg/μl of pEF-LacZ, 0.5 μg/μl fast green was injected into the space between the blastoderm and vitelline membrane using a glass pipette, electric pulses (10 V, 50 ms, 100 ms intervals, five times) were delivered using the electroporator (CUY21, Nepagene). The transfected embryos were placed on the agarose-albumin plate36, covered with yolk supernatant diluted in Hank’s solution and incubated at 38 °C.
Generation and analysis of transgenic and Zic3-ME KO mice
Transgenic mice were generated at Nihon SLC (Shizuoka, Japan). A BAC clone containing Zic3 was purchased from the BACPAC Resources of the Children’s Hospital, Oakland Research Institute. Zic3-ME targeting vector was constructed to replace Zic3-ME with a neomycin resistance gene cassette flanked by a loxP sequence (Neo). Homologous genomic DNA with the Neo cassette was joined with a diphtheria toxin A cassette for negative selection. Linearized targeting vectors were electroporated into C57BL/6J ES cells (EmbryoMax, Millipore) and homologous recombinants were isolated by G418 selection. The ES clones were screened by Southern blot analysis (Supplementary Fig. S3). Correctly targeted ES clones were injected into blastocysts of C57BL/6J mice, which were then used to produce chimeric mice. After confirmation of germ line transmission, the Neo cassette was removed by crossing mice that had germ line transmission with transgenic mice expressing Cre recombinase in germ cells70. The Cre recombination was confirmed by PCR and Southern blot analyses. Mutant animals were genotyped by PCR using DNA and the following primers: Forward primer F1 (5′-CTATGCTCATCGCTTTCGCCATCTAA-3′) and Reverse primer R1 (5′-ATTTTCACGGCCAGCAGTGTTGATAG-3′) for the knockout (KO) allele; and Forward primer F2 (5′-TCTGTGAGGGGATGTTGGAT-3′), and Reverse primer R2 (5′-CCCTGCAGCATGGAGATAAG-3′) for the wild type (WT) allele.
Bioinformatics analysis
ChIP-seq peaks were displayed using the Cistrome database (http://cistrome.org/db/#/)71, ENCODE database (https://www.encodeproject.org/)72, and GTRD (http://gtrd.biouml.org)73. A computer-assisted Zic family binding sites search was carried out at JASPAR database (http://jaspar.genereg.net/) and using the MatInspector program (Genomatix, Munich, Germany). A homology search was carried out with the NCBI BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi). General analysis was done using UCSC genome browser (https://genome.ucsc.edu/) and Ensembl genome browser (https://www.ensembl.org/).
Data Availability
The sequences newly defined in this study were deposited at the DDBJ/GenBank/EMBL database under the following accession numbers: LC377844 (CH261-98E11), LC377845 (CH261-95N3), LC377846 (CH261-11A12), LC377847 (Zic3-ME), LC377848 (Zic3-NE1), LC377849 (Zic3-NE2), LC377850 (Zic1-NE), and LC377851 (Zic1-ME).
References
Aruga, J. & Hatayama, M. Comparative Genomics of the Zic Family Genes. Adv Exp Med Biol 1046, 3–26, https://doi.org/10.1007/978-981-10-7311-3_1 (2018).
Aruga, J. The role of Zic genes in neural development. Mol Cell Neurosci 26, 205–221, https://doi.org/10.1016/j.mcn.2004.01.004 (2004).
Houtmeyers, R., Souopgui, J., Tejpar, S. & Arkell, R. The ZIC gene family encodes multi-functional proteins essential for patterning and morphogenesis. Cell Mol Life Sci 70, 3791–3811, https://doi.org/10.1007/s00018-013-1285-5 (2013).
Hatayama, M. & Aruga, J. Role of Zic Family Proteins in Transcriptional Regulation and Chromatin Remodeling. Adv Exp Med Biol 1046, 353–380, https://doi.org/10.1007/978-981-10-7311-3_18 (2018).
Aruga, J. et al. A wide-range phylogenetic analysis of Zic proteins: implications for correlations between protein structure conservation and body plan complexity. Genomics 87, 783–792, https://doi.org/10.1016/j.ygeno.2006.02.011 (2006).
Lim, L. S. et al. Zic3 is required for maintenance of pluripotency in embryonic stem cells. Molecular biology of the cell 18, 1348–1358, https://doi.org/10.1091/mbc.E06-07-0624 (2007).
Bellchambers, H. M. & Ware, S. M. ZIC3 in Heterotaxy. Adv Exp Med Biol 1046, 301–327, https://doi.org/10.1007/978-981-10-7311-3_15 (2018).
Ahmed, J. N. et al. A murine Zic3 transcript with a premature termination codon evades nonsense-mediated decay during axis formation. Dis Mod Mech 6, 755–767, https://doi.org/10.1242/dmm.011668 (2013).
Chhin, B. et al. Elucidation of penetrance variability of a ZIC3 mutation in a family with complex heart defects and functional analysis of ZIC3 mutations in the first zinc finger domain. Hum Mutat 28, 563–570, https://doi.org/10.1002/humu.20480 (2007).
Chung, B. et al. From VACTERL-H to heterotaxy: variable expressivity of ZIC3-related disorders. Am J Med Genet A 155A, 1123–1128, https://doi.org/10.1002/ajmg.a.33859 (2011).
Ma, L., Selamet Tierney, E. S., Lee, T., Lanzano, P. & Chung, W. K. Mutations in ZIC3 and ACVR2B are a common cause of heterotaxy and associated cardiovascular anomalies. Cardiol Young, 1–8, https://doi.org/10.1017/S1047951111001181 (2011).
Tzschach, A. et al. Heterotaxy and cardiac defect in a girl with chromosome translocation t(X;1)(q26; p13.1) and involvement of ZIC3. Eur J Hum Genet 14, 1317–1320, https://doi.org/10.1038/sj.ejhg.5201707 (2006).
Ware, S. M. et al. Identification and functional analysis of ZIC3 mutations in heterotaxy and related congenital heart defects. Am J Hum Genet 74, 93–105, https://doi.org/10.1086/380998 (2004).
Wessels, M. W. et al. Polyalanine expansion in the ZIC3 gene leading to X-linked heterotaxy with VACTERL association: a new polyalanine disorder? J Med Genet 47, 351–355, https://doi.org/10.1136/jmg.2008.060913 (2010).
Aruga, J. & Millen, K. J. ZIC1 Function in Normal Cerebellar Development and Human Developmental Pathology. Adv Exp Med Biol 1046, 249–268, https://doi.org/10.1007/978-981-10-7311-3_13 (2018).
Barratt, K. S. & Arkell, R. M. ZIC2 in Holoprosencephaly. Adv Exp Med Biol 1046, 269–299, https://doi.org/10.1007/978-981-10-7311-3_14 (2018).
Nagai, T. et al. The expression of the mouse Zic1, Zic2, and Zic3 gene suggests an essential role for Zic genes in body pattern formation. Dev Biol 182, 299–313, https://doi.org/10.1006/dbio.1996.8449 (1997).
Fujimi, T. J., Mikoshiba, K. & Aruga, J. Xenopus Zic4: conservation and diversification of expression profiles and protein function among the Xenopus Zic family. Dev Dyn 235, 3379–3386, https://doi.org/10.1002/dvdy.20906 (2006).
Diamand, K. E. M., Barratt, K. S. & Arkell, R. M. Overview of Rodent Zic Genes. Adv Exp Med Biol 1046, 179–207, https://doi.org/10.1007/978-981-10-7311-3_10 (2018).
Nakata, K., Nagai, T., Aruga, J. & Mikoshiba, K. Xenopus Zic family and its role in neural and neural crest development. Mech Dev 75, 43–51 (1998).
Mizugishi, K., Aruga, J., Nakata, K. & Mikoshiba, K. Molecular properties of Zic proteins as transcriptional regulators and their relationship to GLI proteins. J Biol Chem 276, 2180–2188, https://doi.org/10.1074/jbc.M004430200 (2001).
Nakata, K., Nagai, T., Aruga, J. & Mikoshiba, K. Xenopus Zic3, a primary regulator both in neural and neural crest development. Proc Natl Acad Sci USA 94, 11980–11985 (1997).
Mizuseki, K., Kishi, M., Matsui, M., Nakanishi, S. & Sasai, Y. Xenopus Zic-related-1 and Sox-2, two factors induced by chordin, have distinct activities in the initiation of neural induction. Development 125, 579–587 (1998).
Aruga, J., Tohmonda, T., Homma, S. & Mikoshiba, K. Zic1 promotes the expansion of dorsal neural progenitors in spinal cord by inhibiting neuronal differentiation. Dev Biol 244, 329–341, https://doi.org/10.1006/dbio.2002.0598 (2002).
Maeda, R. et al. Xmeis1, a protooncogene involved in specifying neural crest cell fate in Xenopus embryos. Oncogene 20, 1329–1342, https://doi.org/10.1038/sj.onc.1204250 (2001).
Kitaguchi, T., Mizugishi, K., Hatayama, M., Aruga, J. & Mikoshiba, K. Xenopus Brachyury regulates mesodermal expression of Zic3, a gene controlling left-right asymmetry. Dev Growth Diff 44, 55–61 (2002).
Aruga, J., Shimoda, K. & Mikoshiba, K. A 5′ segment of the mouse Zic1 gene contains a region specific enhancer for dorsal hindbrain and spinal cord. Mol Brain Res 78, 15–25 (2000).
Tropepe, V., Li, S., Dickinson, A., Gamse, J. T. & Sive, H. L. Identification of a BMP inhibitor-responsive promoter module required for expression of the early neural gene zic1. Dev Biol 289, 517–529, https://doi.org/10.1016/j.ydbio.2005.10.004 (2006).
Nyholm, M. K., Wu, S. F., Dorsky, R. I. & Grinblat, Y. The zebrafish zic2a-zic5 gene pair acts downstream of canonical Wnt signaling to control cell proliferation in the developing tectum. Development 134, 735–746, https://doi.org/10.1242/dev.02756 (2007).
Garnett, A. T., Square, T. A. & Medeiros, D. M. BMP, Wnt and FGF signals are integrated through evolutionarily conserved enhancers to achieve robust expression of Pax3 and Zic genes at the zebrafish neural plate border. Development 139, 4220–4231, https://doi.org/10.1242/dev.081497 (2012).
Moriyama, Y. et al. The medaka zic1/zic4 mutant provides molecular insights into teleost caudal fin evolution. Curr Biol 22, 601–607, https://doi.org/10.1016/j.cub.2012.01.063 (2012).
Lin, J. C. & Cepko, C. L. Granule cell raphes and parasagittal domains of Purkinje cells: complementary patterns in the developing chick cerebellum. J Neurosci 18, 9342–9353 (1998).
Warner, S. J. et al. Expression of ZIC genes in the development of the chick inner ear and nervous system. Dev Dyn 226, 702–712, https://doi.org/10.1002/dvdy.10262 (2003).
McMahon, A. R. & Merzdorf, C. S. Expression of thezic1, zic2, zic3, and zic4 genes in early chick embryos. BMC Res Notes 3, 167, https://doi.org/10.1186/1756-0500-3-167 (2010).
Hamburger, V. & Hamilton, H. L. A series of normal stages in the development of the chick embryo. J Morphol 88, 49–92 (1951).
Uchikawa, M., Ishida, Y., Takemoto, T., Kamachi, Y. & Kondoh, H. Functional analysis of chicken Sox2 enhancers highlights an array of diverse regulatory elements that are conserved in mammals. Dev Cell 4, 509–519 (2003).
Uchikawa, M., Nishimura, N., Iwafuchi-Doi, M. & Kondoh, H. Enhancer Analyses Using Chicken Embryo Electroporation. Methods Mol Biol 1650, 191–202, https://doi.org/10.1007/978-1-4939-7216-6_12 (2017).
Inoue, T., Ota, M., Mikoshiba, K. & Aruga, J. Zic2 and Zic3 synergistically control neurulation and segmentation of paraxial mesoderm in mouse embryo. Dev Biol 306, 669–684, https://doi.org/10.1016/j.ydbio.2007.04.003 (2007).
Inoue, T., Ogawa, M., Mikoshiba, K. & Aruga, J. Zic deficiency in the cortical marginal zone and meninges results in cortical lamination defects resembling those in type II lissencephaly. J Neurosci 28, 4712–4725, https://doi.org/10.1523/JNEUROSCI.5735-07.2008 (2008).
Elms, P. et al. Overlapping and distinct expression domains of Zic2 and Zic3 during mouse gastrulation. Gene Expr Patterns 4, 505–511, https://doi.org/10.1016/j.modgep.2004.03.003 (2004).
Purandare, S. M. et al. A complex syndrome of left-right axis, central nervous system and axial skeleton defects in Zic3 mutant mice. Development 129, 2293–2302 (2002).
Klootwijk, R. et al. A deletion encompassing Zic3 in bent tail, a mouse model for X-linked neural tube defects. Hum Mol Genet 9, 1615–1622 (2000).
O’Rourke, M. P. & Tam, P. P. Twist functions in mouse development. Int J Dev Biol 46, 401–413 (2002).
Mayor, R., Guerrero, N., Young, R. M., Gomez-Skarmeta, J. L. & Cuellar, C. A novel function for the Xslug gene: control of dorsal mesendoderm development by repressing BMP-4. Mech Dev 97, 47–56 (2000).
Labelle-Dumais, C., Jacob-Wagner, M., Pare, J. F., Belanger, L. & Dufort, D. Nuclear receptor NR5A2 is required for proper primitive streak morphogenesis. Dev Dyn 235, 3359–3369, https://doi.org/10.1002/dvdy.20996 (2006).
Galan-Caridad, J. M. et al. Zfx controls the self-renewal of embryonic and hematopoietic stem cells. Cell 129, 345–357, https://doi.org/10.1016/j.cell.2007.03.014 (2007).
Okamoto, R., Uchikawa, M. & Kondoh, H. Sixteen additional enhancers associated with the chicken Sox2 locus outside the central 50-kb region. Dev Growth Diff 57, 24–39, https://doi.org/10.1111/dgd.12185 (2015).
Sassa, T., Aizawa, H. & Okamoto, H. Visualization of two distinct classes of neurons by gad2 and zic1 promoter/enhancer elements in the dorsal hindbrain of developing zebrafish reveals neuronal connectivity related to the auditory and lateral line systems. Dev Dyn 236, 706–718, https://doi.org/10.1002/dvdy.21084 (2007).
Rada-Iglesias, A. Is H3K4me1 at enhancers correlative or causative? Nat Genet 50, 4–5, https://doi.org/10.1038/s41588-017-0018-3 (2018).
Nitzsche, A. et al. RAD21 cooperates with pluripotency transcription factors in the maintenance of embryonic stem cell identity. PLoS One 6, e19470, https://doi.org/10.1371/journal.pone.0019470 (2011).
Zuin, J. et al. Cohesin and CTCF differentially affect chromatin architecture and gene expression in human cells. Proc Natl Acad Sci USA 111, 996–1001, https://doi.org/10.1073/pnas.1317788111 (2014).
Lindsley, R. C., Gill, J. G., Kyba, M., Murphy, T. L. & Murphy, K. M. Canonical Wnt signaling is required for development of embryonic stem cell-derived mesoderm. Development 133, 3787–3796, https://doi.org/10.1242/dev.02551 (2006).
Nusse, R. Wnt signaling and stem cell control. Cell Res 18, 523–527, https://doi.org/10.1038/cr.2008.47 (2008).
Hrckulak, D., Kolar, M., Strnad, H. & Korinek, V. TCF/LEF Transcription Factors: An Update from the Internet Resources. Cancers (Basel) 8, https://doi.org/10.3390/cancers8070070 (2016).
Yamaguchi, T. P., Takada, S., Yoshikawa, Y., Wu, N. & McMahon, A. P. T (Brachyury) is a direct target of Wnt3a during paraxial mesoderm specification. Genes Dev 13, 3185–3190 (1999).
Gentsch, G. E. et al. In vivo T-box transcription factor profiling reveals joint regulation of embryonic neuromesodermal bipotency. Cell Rep 4, 1185–1196, https://doi.org/10.1016/j.celrep.2013.08.012 (2013).
Russ, A. P. et al. Eomesodermin is required for mouse trophoblast development and mesoderm formation. Nature 404, 95–99, https://doi.org/10.1038/35003601 (2000).
Fujimi, T. J., Hatayama, M. & Aruga, J. Xenopus Zic3 controls notochord and organizer development through suppression of the Wnt/beta-catenin signaling pathway. Dev Biol 361, 220–231, https://doi.org/10.1016/j.ydbio.2011.10.026 (2012).
Bulfone, A. et al. Expression pattern of the Tbr2 (Eomesodermin) gene during mouse and chick brain development. Mech Dev 84, 133–138 (1999).
Loh, Y. H. et al. The Oct4 and Nanog transcription network regulates pluripotency in mouse embryonic stem cells. Nat Genet 38, 431–440, https://doi.org/10.1038/ng1760 (2006).
Sharov, A. A. et al. Identification of Pou5f1, Sox2, and Nanog downstream target genes with statistical confidence by applying a novel algorithm to time course microarray and genome-wide chromatin immunoprecipitation data. BMC Genomics 9, 269, https://doi.org/10.1186/1471-2164-9-269 (2008).
Liang, J. et al. Nanog and Oct4 associate with unique transcriptional repression complexes in embryonic stem cells. Nat Cell Biol 10, 731–739, https://doi.org/10.1038/ncb1736 (2008).
Saunders, A. et al. The SIN3A/HDAC Corepressor Complex Functionally Cooperates with NANOG to Promote Pluripotency. Cell Rep 18, 1713–1726, https://doi.org/10.1016/j.celrep.2017.01.055 (2017).
Lim, L. S., Hong, F. H., Kunarso, G. & Stanton, L. W. The pluripotency regulator Zic3 is a direct activator of the Nanog promoter in ESCs. Stem cells 28, 1961–1969, https://doi.org/10.1002/stem.527 (2010).
Yoon, S. J., Foley, J. W. & Baker, J. C. HEB associates with PRC2 and SMAD2/3 to regulate developmental fates. Nat Commun 6, 6546, https://doi.org/10.1038/ncomms7546 (2015).
Yoon, S. J., Wills, A. E., Chuong, E., Gupta, R. & Baker, J. C. HEB and E2A function as SMAD/FOXH1 cofactors. Genes Dev 25, 1654–1661, https://doi.org/10.1101/gad.16800511 (2011).
Faial, T. et al. Brachyury and SMAD signalling collaboratively orchestrate distinct mesoderm and endoderm gene regulatory networks in differentiating human embryonic stem cells. Development 142, 2121–2135, https://doi.org/10.1242/dev.117838 (2015).
Mizushima, S. & Nagata, S. pEF-BOS, a powerful mammalian expression vector. Nucleic Acids Res 18, 5322 (1990).
Sasaki, H. & Hogan, B. L. Enhancer analysis of the mouse HNF-3 beta gene: regulatory elements for node/notochord and floor plate are independent and consist of multiple sub-elements. Genes Cells 1, 59–72 (1996).
Sakai, K. & Miyazaki, J. A transgenic mouse line that retains Cre recombinase activity in mature oocytes irrespective of the cre transgene transmission. Biochem Biophys Res Commun 237, 318–324 (1997).
Mei, S. et al. Cistrome Data Browser: a data portal for ChIP-Seq and chromatin accessibility data in human and mouse. Nucleic Acids Res 45, D658–D662, https://doi.org/10.1093/nar/gkw983 (2017).
Consortium, E. P. An integrated encyclopedia of DNA elements in the human genome. Nature 489, 57–74, https://doi.org/10.1038/nature11247 (2012).
Yevshin, I., Sharipov, R., Valeev, T., Kel, A. & Kolpakov, F. GTRD: a database of transcription factor binding sites identified by ChIP-seq experiments. Nucleic Acids Res 45, D61–D67, https://doi.org/10.1093/nar/gkw951 (2017).
Acknowledgements
We thank Hisato Kondoh, Hiroyuki Mishima, and Takashi Inoue for helpful advice on experiments and Hiroshi Sasaki for providing plasmids; Yayoi Nozaki, Chieko Nishioka, and technical staff of the Sequencing Technology Team RIKEN GSC, and the Research Resource Center RIKEN BSI for their technical assistance. The study was supported by RIKEN BSI funds, MEXT grants (grant numbers 16027261, 16390086, 21657034, 25640026, 15K15019), and Smoking Research Foundation.
Author information
Authors and Affiliations
Contributions
J.A. conceived and designed the study; Y.S.O., T.T., A.T. and J.A. performed the experiments; J.A. performed bioinformatics analysis; and J.A. wrote the paper.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Odaka, Y.S., Tohmonda, T., Toyoda, A. et al. An Evolutionarily Conserved Mesodermal Enhancer in Vertebrate Zic3. Sci Rep 8, 14954 (2018). https://doi.org/10.1038/s41598-018-33235-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-33235-y
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.