Abstract
There has been a recent debate that boron nanotubes can outperform carbon nanotubes on many grounds. The most stable boron nanotubes are made of a hexagonal lattice with an extra atom added to some of the hexagons called ∝-boron nanotubes. Closed forms of M-polynomial of nanotubes produce closed forms of many degree-based topological indices which are numerical parameters of the structure and determine physico-chemical properties of the concerned nanotubes. In this article, we compute and analyze many topological indices of ∝-boron nanotubes correlating with the size of structure of these tubes through the use of M-polynomial. More importantly we make a graph-theoretic comparison of indices of two types of boron nanotubes namely triangular boron and ∝-boron nanotubes.
Similar content being viewed by others
Introduction
Mathematical chemistry provides tools such as polynomials and functions to capture information hidden in the symmetry of molecular graphs and thus predict properties of compounds without using quantum mechanics. A topological index isa numerical parameter of a graph and depicts its topology. It describes the structure of molecules numerically and are used in the development of qualitative structure activity relationships (QSARs). Most commonly known invariants of such kinds are degree-based topological indices. These are actually the numerical values that correlate the structure with various physical properties, chemical reactivity and biological activities1,2,3,4,5. It is an established fact that many properties such as heat of formation, boiling point, strain energy, rigidity and fracture toughness of a molecule are strongly connected to its graphical structure.
Hosoya polynomial, (Wiener polynomial)6, plays a pivotal role in distance-based topological indices. A long list of distance-based indices can be easily evaluated from Hosoya polynomial. A similar breakthrough was obtained recently by Klavzar et. al.7, in the context of degree-based indices. Authors in7 introduced M-polynomial in, 2015, to play a role, parallel to Hosoya polynomial to determine closed form of many degree-based topological indices8,9,10,11,12. The real power of M-polynomial is its comprehensive nature containing healthy information about degree-based graph invariants. These invariants are calculated on the basis of symmetries present in the 2d-molecular lattices and collectively determine some properties of the material under observation.
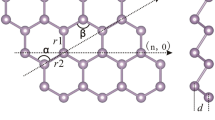
Because of increasing interests and developments of new nanomaterials, computations have minimized the burden of experimental labor to some extent. Amongst the nanomaterials, nanocrystals, nanowires and nanotubes, constitute three major categories, the last two being one-dimensional. Boron nanotubes are becoming increasingly interesting because of their remarkable properties like structural stability, work function, transport properties, and electronic structure13. Triangular Boron is derived from a triangular sheet as shown in Fig. 1. The first boron nanotubes were created, in 2004, from a buckled triangular latticework13,14,15.
These tubes are discussed recently in15,16. Other well-known type, α-boron, is derived from α-sheet. Irrespective of their structures and chiralities, both types are more conductive than carbon nanotubes14,17,18,19. Figure 2 describes basic structure of ∝- boron nanotube. Following Fig. 3 also presents different views of ∝- Boron nanotube, (a) is the planar view whereas (b) is the tabular view.
As for as structure of both tubes are concerned, ∝- Boron nanotube is more complicated than Triangular boron nanotubes with addition of an extra atom to the center of some of the hexagons15. In15, authors proved that this is the most stable known theoretical structure for a boron nanotube. They also showed that, with this pattern, boron nanotubes should have variable electrical properties: wider ones would be metallic conductors, but narrower ones should be semiconductors. So, these tubes boron tubes will be used in Nano devices similar to the diodes and transistors that have already been made from carbon nanotube. In16 authors computed some computational facts which are similar in both types of boron nanotubes and carbon nanotubes. Munir et al. computed M-polynomial and related indices of triangular boron nanotubes in11, polyhex nanotubes in12, nanostar dendrimers in8, titania nanotubes in9 and M- Polynomials and topological indices of V-Phenylenic Nanotubes and Nanotori in10. In all above mentioned articles we presented an analysis of these indices against the parameters of structure involved. In this article, we compute general form of M-polynomial for ∝- boron nanotube. Then we derive closed forms of many degree-based topological indices for these tubes. We also draw some conclusions about both types of boron tubes.
Basic Definitions and Literature Review
Throughout this article, we assume G to be a connected graph, V (G) and E (G) are the vertex set and the edge set respectively and dv denotes the degree of a vertex v.
Definition 1
. The M-polynomial of G is defined as: \(M(G,x,y)=\sum _{\delta \le i\le j\le \Delta }{m}_{ij}(G){x}^{i}{y}^{j}\) where δ = Min{dv|v ∈ V (G)}, Δ = Max{dv|v ∈ V (G)}, and mij(G) is the edge vu ∈ E(G) such that where ≤j7.
Wiener index and its various applications are discussed in20,21,22. Randić index, R−1/2(G), is introduced by Milan Randić in 1975 defined as: \({R}_{-1/2}(G)=\sum _{uv\in E(G)}\frac{1}{\sqrt{{d}_{u}{d}_{v}}}.\) For general detains about R−1/2(G) and its generalized Randić index, \({R}_{\alpha }(G)=\sum _{uv\in E(G)}\frac{1}{{({d}_{u}{d}_{v})}^{\alpha }},\) please see23,24,25,26,27,28 and the inverse Randić index is defined as \(R{R}_{\alpha }(G)=\sum _{uv\in E(G)}{({d}_{u}{d}_{v})}^{\alpha }.\) Clearly R−1/2(G) is a special case of Rα(G) when \(\alpha =-\,\frac{1}{2}\). This index has many applications in diverse areas. Many papers and books such as29,30,31 are written on this topological index as well. Gutman and Trinajstić introduced two indices defined as: \({M}_{1}(G)=\sum _{uv\in E(G)}({d}_{u}+{d}_{v})\) and \({M}_{2}(G)=\sum _{uv\in E(G)}({d}_{u}\times {d}_{v})\). Thesecond modified Zagreb index is defined as: \({}^{m}M_{2}(G)=\sum _{uv\in E(G)}\frac{1}{d(u)d(v)}.\) We refer32,33,34,35,36 to the readers for comprehensive details of these indices. Other famous indices are Symmetric division index: \({\rm{SDD}}({\rm{G}})=\sum _{uv\in E(G)}\{\frac{{\rm{\min }}({d}_{u},{d}_{v})}{{\rm{\max }}({d}_{u},{d}_{v})}+\frac{{\rm{\max }}({d}_{u},{d}_{v})}{{\rm{\min }}({d}_{u},{d}_{v})}\}\) harmonic index defined \(H(G)=\sum _{vu\in E(G)}\frac{2}{{d}_{u}+{d}_{v}}.\) Inverse sum index i: \(I(G)=\sum _{vu\in E(G)}\frac{{d}_{u}{d}_{v}}{{d}_{u}+{d}_{v}}\) and augmented Zagreb index: \(A(G)=\sum _{vu\in E(G)}{\{\frac{{d}_{u}{d}_{v}}{{d}_{u}+{d}_{v}-2}\}}^{3},\)37,38.
Tables presented in7,8,9,10,11 relates some of these well-known degree-based topological indices with M-polynomial with following reserved notations
Computational Results
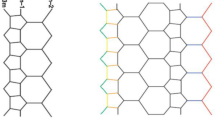
In this section, we give our computational results. In terms of chemical graph theory and mathematical chemistry, we associate a graph with the molecular structure where vertices correspond to atoms and edges to bonds. Following the same lines, we represent a ∝- boron nanotube, by a planar graph, BNTt[m, n] of order n × m, as the in Fig. 3 demonstrates. Clearly, from Fig. 4, there are \(\frac{3nm}{2}\) vertices and \(\frac{3}{2}mn+3{m}^{2}+\frac{9}{2}m+n-3\) edges in 2D graph model of ∝-boron nanotubes.
Theorem 1.
Let BNTα[m, n] is ∝-boron Nanotubes. Then
Proof.
Let BNTα[m, n] be ∝-boron nanotubes, where m is the number of columns and n is the number of rows. From Fig. 4 it is easy to observe that the graph of BNTα[m, n] has \(\frac{4nm}{3}\) number of vertices and \(\frac{3}{2}mn+3{m}^{2}+\frac{9}{2}m+n-3\) edges where n is multiple of 3.
The edge set of BNTα[m, n] has following three partitions,
And
and the vertex set V(BNTα[m, n]) of BNTα[m, n] has six partitions:
Now
and
Thus the M-polynomial of BNTα[m, n] is:
Above Fig. 5 presents the Maple 13 plot of the M-polynomial of ∝- Boron Nanotubes. Clearly, values drastically increases as Χ → ±1, Y → ±2 For the most part of [−1, 1] × [−2, 2], values remain stable whereas triangular boron nanotubes show opposite trends11, shown in Fig. 6 below
Now we derive formulas for many degree-based topological indices using M-polynomial.
Proposition 1
Let BNTα[m, n] is the boron ∝- nanotube, then
-
1.
\({M}_{1}(G)=94m+5m(3n-11)+33{m}^{2}+12n-36.\)
-
2.
\({M}_{2}(G)=220m+\frac{25}{2}m(3n-11)+90{m}^{2}+36n-108.\)
-
3.
\({}^{m}M_{2}(G)=\frac{1}{10}{m}^{2}+(\frac{943}{3600}+\frac{3}{50}n)m+\frac{1}{36}n-\frac{1}{12}.\)
-
4.
\({R}_{\alpha }(G)=3\times {16}^{\alpha }m+2m{20}^{\alpha }+4m{24}^{\alpha }+\frac{1}{2}{25}^{\alpha }m(3n-11)+3{m}^{2}{30}^{\alpha }+{36}^{\alpha }(m+n-3).\)
-
5.
\({R}_{\alpha }(G)=\frac{3}{{16}^{\alpha }}m+\frac{2}{{20}^{\alpha }}m+\frac{4}{{24}^{\alpha }}m+\frac{1}{2\times {25}^{\alpha }}m(3n-11)+\frac{3}{{30}^{\alpha }}{m}^{2}+\frac{1}{{36}^{\alpha }}(m+n-3).\)
-
6.
\(SSD(G)=\frac{623}{30}m+m(3n-11)+\frac{61}{10}{m}^{2}+2n-6.\)
-
7.
\(H(G)=\frac{6}{11}{m}^{2}+(\frac{191}{180}+\frac{3}{10}n)m+\frac{1}{6}n-\frac{1}{2}.\)
-
8.
\(I(G)=\frac{90}{11}{m}^{2}+(\frac{1673}{180}+\frac{15}{4}n)m+3n-9.\)
-
9.
\(A(G)=\frac{1000}{9}{m}^{2}+(\frac{35698735591}{395136000}+\frac{46875}{1024}n)m+\frac{5832}{125}n-\frac{17496}{125}.\)
Proof.
Let
Then
-
1.
First Zagreb Index
$${M}_{1}(G)={({D}_{x}+{D}_{y})f(x,y)|}_{x=y=1}=94m+5m(3n-11)+33{m}^{2}+12n-36.$$(31) -
2.
Second Zagreb Index
$${M}_{2}(G)={{D}_{y}{D}_{x}(f(x,y))|}_{x=y=1}=220m+\frac{25}{2}m(3n-11)+90{m}^{2}+36n-108.$$(32) -
3.
Modified Second Zagreb Index
$${}^{m}M_{2}(G)={{S}_{x}{S}_{y}(f(x,y))|}_{x=y=1}=\frac{1}{10}{m}^{2}+(\frac{943}{3600}+\frac{3}{50}n)m+\frac{1}{36}n-\frac{1}{12}.$$(33) -
4.
Generalized Randic’ Index
$$\begin{array}{rcl}{R}_{\alpha }(G) & = & {{D}_{x}^{\alpha }{D}_{y}^{\alpha }(f(x,y))|}_{x=y=1}=3\times {16}^{\alpha }m+2m{20}^{\alpha }+4m{24}^{\alpha }\\ & & +\frac{1}{2}{25}^{\alpha }m(3n-11)+3{m}^{2}{30}^{\alpha }+{36}^{\alpha }(m+n-3).\end{array}$$(34) -
5.
Inverse Randic’ Index
$$\begin{array}{rcl}R{R}_{\alpha }(G) & = & {{S}_{x}^{\alpha }{S}_{y}^{\alpha }(f(x,y))|}_{x=y=1}=\frac{3}{{16}^{\alpha }}m+\frac{2}{{20}^{\alpha }}m\\ & & +\frac{4}{{24}^{\alpha }}m+\frac{1}{2\times {25}^{\alpha }}m(3n-11)+\frac{3}{{30}^{\alpha }}{m}^{2}+\frac{1}{{36}^{\alpha }}(m+n-3).\end{array}$$(35) -
6.
Symmetric Division Index
$$SSD(G)={({S}_{y}{D}_{x}+{S}_{x}{D}_{y})(f(x,y))|}_{x=y=1}=\frac{623}{30}m+m(3n-11)+\frac{61}{10}{m}^{2}+2n-6.$$(36) -
7.
Harmonic Index
$$H(G)=2{S}_{x}J(f(x,y)){|}_{x=1}=\frac{6}{11}{m}^{2}+(\frac{191}{180}+\frac{3}{10}n)m+\frac{1}{6}n-\frac{1}{2}.$$(37) -
8.
Inverse Sum Index
$$I(G)={S}_{x}J{D}_{x}{D}_{y}{(f(x,y))}_{x=1}=\frac{90}{11}{m}^{2}+(\frac{1673}{180}+\frac{15}{4}n)m+3n-9.$$(38) -
9.
Augmented Zagreb Index
Conclusions and Discussion
In the present article, we computed closed form of M-polynomial for α-boron nanotubes and then we derived many degree-based topological indices as well. Some other degree based topological indices of boron nanotubes are given in39. Topological indices thus calculated for these nanotubes can help us to understand the physical features, chemical reactivity, and biological activities. In this point of view, a topological index can be regarded as a score function which maps each molecular structure to a real number and is used as descriptors of the molecule under testing. These results can also play a vital part in the determination of the significance of ∝- boron nanotubes in electronics and industry. We also want to remark that a thorough comparison of α-boron nanotubes can be made with triangular boron Nanotubes11. For the rest of this article we reserve symbol T for Triangular boron tube and P for α-boron nanotube.
We give a detailed comparative analysis of degree-based topological indices of both boron tubes. It has been experimentally verified that the first Zagreb index is directly related with total π -electron energy of the structure33,40 and references therein. So structure having high values of First Zagreb Index have higher total π -electron energy. From the following Fig. 6 it is evident that total π -electron energy of alpha-Boron nanotube is less than triangular Boron tubes for m ≤ 9 and for m ≥ 10, total π -electron energy of alpha-Boron nanotube rises sharply as compared to triangular Boron tubes with increase in m.
Similarly the given Figs 7 and 8 elaborates that total π -electron energy of alpha-Boron nanotube is larger than triangular Boron tubes for n ≤ 11 and for n ≥ 11, total π -electron energy of triangular Boron tubes rises sharply as compared to alpha Boron tubes with increase in n.
Similarly Randic index is useful for determining physio-chemical properties of alkanes as noticed by chemist Melan Randic in 1975. He noticed the correlation between the Randic index R and several physico–chemical properties of alkanes like, enthalpies of formation, boiling points, chromatographic retention times, vapor pressure and surface areas. Following Fig. 9 is adapted from21 relating to boiling point of some Alkanes and its correlation with Randic index.
Now we give some comparative remarks showing some correlations. The next Fig. 10 clearly depicts that green color rises sharply as compared with red indicating that alpha tube have significant correlation coefficients of above said properties over the triangular boron tubes with the rise in n and m.
It is noticeable from above Fig. 11 that boiling point and other above properties are correlated with Randic index. Subsequent years of research showed that Randic index has a variety of applications specially in medicinal and pharmacological issues. For the results about Triangular boron, we refer to11. We give comparative analysis of both tubes for Augmented Zagreb index, Inverse sum index and Harmonic index denoted by A, I and H respectively. Remaining can be traced out in similarly. We start with A(G), the Augmented Zagreb index. Recently it has been proved that this index has relatively high correlation coefficient so it can be used for designing quantitative structure-property relations. Figures 12 and 13 shows the graphs of A(G) for both tubes. We use red color for graphs of α-boron nanotubes whereas blue is used for triangular boron tubes. Figure 12 shows that A(G) decrease linearly with the rise in m for Triangular boron whereas it rises sharply for α-boron nanotubes. One who wishes to select a boron tube with high A(G) should naturally choose α-boron nanotubes. Figure 13 shows that if we fix m instead of n, A(G) rises linearly with both types of tube although for n < 4, A(T) < A(P) and A(T) > A(P) for n > 4. Critical fact is the value n = 4 where A(T) = A(P). These figures also suggest the length and width of tubes for the desired values of A(G).
Now we discuss the Inverse sum index.
As the Fig. 14 suggests that I(P) rises sharply with the rise in m but I(T) slopes downward very slowly with the same rise if we fix n = 2. Whereas both I(P) and I(T) rise linearly with rise in n although rise in I(T) seems to be negligible see Fig. 15.
For H(G), we can see that again H(P) ≥ H(T) for all positive values of m with n = 2, see Fig. 16. Fixing m = 2, we can see that for n > 3, H(P) ≥ H(T) and H(P) < H(T) for n > 3 but for n = 3, we get H(P) = H(T), see Fig. 17. We believe that all above computed indices show more or less similar results, so in the end we conclude that α-boron nanotubes are far better in obtaining higher values of degree-based topological indices than triangular boron nanotubes.
Above graphs of topological indices show the correlation of M1(G), Rα(G), A(G), I(G) and H(G) with m and n. It is clear that all topological indices have linear relation with n whereas graphs are parabolas in relation to m. These facts give an insight idea to control topological indices with length and width of these tubes. Moreover we can find extreme values of topological indices for some definite values of m and n. We give structural analysis for only three indices as all other indices discussed above show similar trends. We also conclude that α-Boron nanotubes have high correlation coefficient regarding Randic index41,42,43,44.
References
Rücker, G. & Rücker, C. On topological indices, boiling points, and cycloalkanes. Journal of chemical information and computer sciences 39(5), 788–802 (1999).
Klavžar, S. & Gutman, I. A comparison of the Schultz molecular topological index with the Wiener index. Journal of chemical information and computer sciences 36(5), 1001–1003 (1996).
Brückler, F. M., Došlić, T., Graovac, A. & Gutman, I. On a class of distance-based molecular structure descriptors. Chemical physics letters 503(4–6), 336–338 (2011).
Deng, H., Yang, J. & Xia, F. A general modeling of some vertex-degree based topological indices in benzenoid systems and phenylenes. Computers & Mathematics with Applications 61(10), 3017–3023 (2011).
Zhang, H. & Zhang, F. The Clar covering polynomial of hexagonal systems I. Discrete applied mathematics 69(1–2), 147–167 (1996).
Gutman, I. Some properties of the Wiener polynomials. Graph Theory Notes New York 125, 13–18 (1993).
Deutsch, E. & Klavzar, S. M-Polynomial and degree-based topological indices. Iran. J. Math. Chem. 6, 93–102 (2015).
Munir, M., Nazeer, W., Rafique, S. & Kang, S. M. M-polynomial and related topological indices of nanostar dendrimers, Symmetry, 8, 1–12, Article ID 97 (2016).
Munir, M., Nazeer, W., Nizami, A. R., Rafique, S. & Kang, S. M. M-polynomials and topological indices of titania nanotubes. Symmetry 8(11), 117 (2016).
Kwun, Y., Munir, M., Nazeer, W., Rafique, S. & Kang, S. M. M-polynomial and degree-based topological indices of V-phenalinic nanotubes and nanotori. Scientific Reports 7, 8756 (2017).
Munir, M., Nazeer, W., Rafique, S., Nizami, A. & Kang, S. Some computational aspects of boron triangular nanotubes. Symmetry 9(1), 6 (2017).
Munir, M., Nazeer, W., Rafique, S. & Kang, S. M. M-polynomial and degree-based topological indices of polyhex nanotubes. Symmetry 8(12), 149 (2016).
Munir, M., Nazeer, W., Shahzadi, Z. & Kang, S. Some invariants of circulant graphs. Symmetry 8(11), 134 (2016).
Manuel, P. Computational aspects of carbon and boron nanotubes. molecules 15(12), 8709–8722 (2010).
Bezugly, V. et al. Highly conductive boron nanotubes: transport properties, work functions, and structural stabilities. ACS nano 5(6), 4997–5005 (2011).
Singh, A. K., Sadrzadeh, A. & Yakobson, B. A. Ab initio prediction of stable boron sheets and boron nanotubes: Structure, stability, and electronic properties.
Sun, M. L., Slanina, Z. & Lee, S. L. Square/hexagon route towards the boron-nitrogen clusters. Chemical physics letters 233(3), 279–283 (1995).
Slanina, Z., Sun, M. L. & Lee, S. L. AM1 stability prediction: B36N24 > B36P24 > Al36N24 > Al36P24. Journal of Molecular Structure: THEOCHEM 334(2–3), 229–233 (1995).
Slanina, Z., Sun, M. L. & Lee, S. L. Computations of boron and boron-nitrogen cages. Nanostructured materials 8(5), 623–635 (1997).
Yang, X., Ding, Y. & Ni, J. Ab initio prediction of stable boron sheets and boron nanotubes: structure, stability, and electronic properties. Physical Review B 77(4), 041402 (2008).
Wiener, H. Structural determination of paraffin boiling points. Journal of the American Chemical Society 69(1), 17–20 (1947).
Dobrynin, A. A., Entringer, R. & Gutman, I. Wiener index of trees: theory and applications. Acta Applicandae Mathematica 66(3), 211–249 (2001).
Gutman, I. & Polansky, O. E. Mathematical concepts in organic chemistry. (Springer Science & Business Media, 2012).
Randic, M. Characterization of molecular branching. Journal of the American Chemical Society 97(23), 6609–6615 (1975).
Bollobás, B. & Erdös, P. Graphs of extremal weights. Ars Combinatoria 50, 225–233 (1998).
Amić, D., Bešlo, D., Lučić, B., Nikolić, S. & Trinajstić, N. The vertex-connectivity index revisited. Journal of chemical information and computer sciences 38(5), 819–822 (1998).
Hu, Y., Li, X., Shi, Y., Xu, T. & Gutman, I. On molecular graphs with smallest and greatest zeroth-order general Randic index. MATCH Commun. Math. Comput. Chem 54(2), 425–434 (2005).
Caporossi, G., Gutman, I., Hansen, P. & Pavlović, L. Graphs with maximum connectivity index. Computational Biology and Chemistry 27(1), 85–90 (2003).
Li, X. & Gutman, I. Mathematical Chemistry Monographs No. 1. Kragujevac. (2006).
Kier, L. Molecular connectivity in chemistry and drug research (Vol. 14) (Elsevier, 2012).
Kier, L. B. & Hall, L. H. Molecular connectivity in structure-activity analysis. Research Studies (1986).
Gutman, I. & Furtula, B. (Eds). Recent results in the theory of Randić index. University, Faculty of Science. Univ. Kragujevac, Kragujevac, pp. 9–47 (2008).
Nikolić, S., Kovačević, G., Miličević, A. & Trinajstić, N. The Zagreb indices 30 years after. Croatica chemica acta 76(2), 113–124 (2003).
Gutman, I. & Das, K. C. The first Zagreb index 30 years after. MATCH Commun. Math. Comput. Chem 50(1), 83–92 (2004).
Das, K. C. & Gutman, I. Some properties of the second Zagreb index. MATCH Commun. Math. Comput. Chem 52(1), 3–1 (2004).
Trinajstić, N., Nikolić, S., Miličević, A. & Gutman, I. O. Zagrebačkim indeksima. Kemija u industriji: Časopis kemičara i kemijskih inženjera Hrvatske 59(12), 577–589 (2010).
Huang, Y., Liu, B. & Gan, L. Augmented Zagreb index of connected graphs. Match-Communications in Mathematical and Computer Chemistry 67(2), 483 (2012).
Furtula, B., Graovac, A. & Vukiˇcevi´c, D. Augmented Zagreb index. J. Math. Chem. 48, 370–380 (2010).
Liu, J. B., Shaker, H., Nadeem, I. & Hussain, M. Topological Aspects of Boron Nanotubes. Advances in Materials Science and Engineering, 2018 (2018).
Li, X. & Shi, Y. A survey on the Randic index. MATCH Commun. Math. Comput. Chem 59(1), 127–156 (2008).
Li, X., Gutman, I. & Randić, M. Mathematical aspects of Randić-type molecular structure descriptors. University, Faculty of Science. Univ. Kragujevac, Kragujevac (2006).
Gutman, I. & Furtula, B. (Eds). Recent results in the theory of Randić index. University, Faculty of Science. Univ. Kragujevac, Kragujevac, (2008).
Randić, M. On history of the Randić index and emerging hostility toward chemical graph theory. MATCH Commun. Math. Comput. Chem 59, 5–124 (2008).
Randić, M. The connectivity index 25 years after. Journal of Molecular Graphics and Modelling 20(1), 19–35 (2001).
Author information
Authors and Affiliations
Contributions
Y.C. Kwun designed the problem, M. Munir, and W. Nazeer proved the results and S. Rafique and S.M. Kang verified the results and wrote his paper.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kwun, Y.C., Munir, M., Nazeer, W. et al. Computational Analysis of topological indices of two Boron Nanotubes. Sci Rep 8, 14843 (2018). https://doi.org/10.1038/s41598-018-33081-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-33081-y
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.