Abstract
Postoperative complication III-V is closely related with hepatectomy-related mortality for hepatocellular carcinoma (HCC) patients. The aim of the study was to investigate the relationship between CONUTS and postoperative complication III-V. 1334 HCC patients who underwent hepatectomy were divided into two groups: high CONUTS group (early postoperative CONUTS ≥ 8, n = 659) and low CONUTS group (early postoperative CONUTS < 8, n = 675). The characteristics and clinical outcomes were compared and analyzed. Risk factors for postoperative complication III-V were evaluated by univariate and multivariate analysis. early postoperative CONUTS showed a good prediction ability for postoperative complication III-V (AUROC = 0.653, P < 0.001), with the cut-off value of 8. The high CONUTS group had higher incidence of postoperative pulmonary complications (12.0% vs 7.9%, P = 0.011), bile leakage (2.6% vs 0.9%, P = 0.018), intra-abdominal hemorrhage (4.9% vs 1.6%, P = 0.001), postoperative liver failure Grade C (3.6% vs 1.0%, P = 0.002), complication III-V (15.6% vs 6.2%, P < 0.001), length of ICU stay > 48 hours (9.4% vs 4.1%, P < 0.001) and mortality in 90 days (2.6% vs 0.4%, P = 0.001), longer period of postoperative hospitalization (10 (8–13) vs 9 (7–11) days, P < 0.001). Multivariable analysis revealed that early postoperative CONUTS ≥ 8 (OR = 2.054, 95%CI = 1.371–3.078, P < 0.001) was independently associated with postoperative complication III-V. Early postoperative CONUTS ≥ 8 was identified as a novel risk factor for postoperative complication III-V, and should be further evaluated as a predictive marker for who are to undergo liver resection.
Similar content being viewed by others
Introduction
Liver cancer was the fourth leading cause of cancer death according to the Global Burden of Disease Study 20151. The most common type of primary liver cancer is hepatocellular carcinoma (HCC), followed by cholangiocarcinoma2. Surgical partial hepatectomy is widely regarded as the preferred curative treatment for patients with HCC. Postoperative mortality and morbidity for HCC patients have been reduced significantly, however the recurrence rate in 5 years remains close to 70%3. Previous studies4,5 suggested that perioperative nutritional supplementation could reduce the postoperative complications and shorten the duration of hospitalization of patients who undergo liver resection for cancer. Hsieh CE et al.6 reported that postoperative nutritional support could reduce pulmonary complications, promote the recovery of liver function and shorten length of stay in adult liver donors. As we know, malnutrition is considered to be associated with worse outcome of critical illness and appropriate nutritional intervention can improve outcomes for inpatients. There has been no uniform definition of malnutrition, different nutritional evaluation methods were reported and widely used. However, few studies investigated the relationship between nutritional assessment scores and postoperative outcomes.
Several immune-nutritional factors were reported as potential predictor for the outcomes of HCC patients after hepatectomy, such as aspartate aminotransferase to platelet ratio index (APRI)7, fibrosis index based on the four factors index (FIB-4)8, albumin-bilirubin score (ALBI)9, prognostic nutritional index (PNI)10. The Controlling Nutritional Status score (CONUTS), first validated and reported by de Ulíbarri J, I. et al. in 2005, as a screening tool for early detection and continuous control of hospital undernutrition11. The formula, consists of serum albumin, cholesterol levels, and total lymphocyte count, is shown in Fig. 1A. Previous studies have been reported that preoperative CONUTS could predict the poor prognosis in patients who with colorectal cancer12, hepatocellular carcinoma13, or esophageal squamous cell carcinoma14. However, hemodynamics and blood constituents would be affected, due to intraoperative blood loss and fluid infusion. Hence, we hypothesized that early postoperative CONTS would be superior to preoperative CONUTS in predictive ability. A retrospective cohort study was carried out to investigate the relationship between early postoperative CONUTS and complication III-V after hepatectomy in HCC patients.
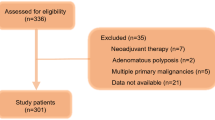
(A) Assessment of undernutrition status by the CONUT score. Ignacio de Ullibarri J. et al. Nutr Hosp11. (B) Flow diagram for 1334 consecutive patients who meet the inclusion criteria.
Materials and Methods
Study population
The study included 1334 consecutive HCC patients who underwent treatment at West China hospital of Sichuan university between January 2011 and December 2013. Hepatocellular carcinoma diagnosis was confirmed based on the current EASL15 or AASLD16 HCC management guidelines. The inclusion criteria followed: (1) Pathological diagnosis confirmed hepatocellular carcinoma, (2) received partial hepatectomy by open or laparoscopic hepatectomy, (3) patients >18 years. Exclusion criteria included the following: (1) patients with obstructive jaundice, (2) combined with portal vein tumor thrombus, (3) combined with extrahepatic metastasis, (4) liver function of Child-Pugh grade C, (5) loss to postoperative follow-up within 90 days, (6) poor data integrity. The flowchart was revealed in Fig. 1B. We collected the medical records containing the demographics, preoperative laboratory values, imaging examination data and postoperative clinical outcomes from the clinical liver cancer database of the department of Liver Surgery & Liver Transplantation Center, West China Hospital of Sichuan university.
Perioperative management
Careful history analysis, physical examination and routine preoperative laboratory measurements were performed for all patients. Routine imaging examination to evaluate the tumor and cardiopulmonary function evaluation was carried out before surgery as we previously described8. Antiviral drugs were administered to patients with positive HBV-DNA before the operation, and postoperative continuous antiviral therapy follow the current guidelines17. Patients were operated under general anesthesia and intraoperative ultrasonography was used routinely. Hepatic vascular inflow occlusion (hemihepatic or total hepatic blocking) or the Pringle maneuver was utilized according to the surgeon’s preference in most patients as those previously described18,19. Hepatectomy was performed using the clamp crushing or hooking with ligation method, ultrasonic dissector with coagulator. Based on preoperative and intraoperative condition, patients were transferred to the intensive care unit for treatment when necessary.
Parameter definition
Early postoperative CONUTS was calculated from the first postoperative serum albumin level, lymphocytes count and total cholesterol level, the blood samples were obtained within 12 hours after operation. Postoperative CONUTS change was calculated by subtracting the preoperative CONUTS from the early postoperative CONUTS. Clinically relevant portal hypertension (PHT) is defined as the presence of esophageal varices and/or a platelet count of less than 100 × 109/L in association with splenomegaly20. The Clavien–Dindo complication classification system21 was used for postoperative complication grading and grade III-V complications were defined as severe complications. Postoperative liver failure22 (PLF) and biliary leakage23 were defined in accordance with the criterion of International Study Group of Liver Surgery. PLF was classified into three categories (grade A, B, and C)22. Liver resection with more than three segments was defined as major resection, or as minor resection. Mortality was defined as any death occurring from the time of surgery up to 90 days after hepatectomy.
Statistical Analysis
Scientific research secretaries were trained to take the collection and analysis responsibilities. Continuous variables were reported as mean (standard deviation [SD]) or median (interquartile range [IQR]). Student t test for continuous variables with parametric distribution. Mann–Whitney U test or Kruskal–Wallis H test for those with nonparametric distribution. Categorical variables were reported as numbers and percentages, and compared using Pearson χ2 analyses or Fisher exact test. The predictive ability of potential factors for postoperative complication III-V was assessed by the corresponding area under the receiver operating characteristic (AUROC) curve. Youden index was utilized to choose the optimal cut-off value, which set as the value maximizing the sum of sensitivity and specificity. To identify risk factors for postoperative severe complications, all significant factors in the univariate analysis were used for multivariate analysis by the forward stepwise logistic regression. All statistical analyses were performed using IBM SPSS Statistics software 21.0, and statistical significance was set at P < 0.05, with two-tailed.
Result
1334 consecutive patients were included in this study, including 1136 (85.2%) males and 198 (14.8%) females. 1208 (90.6%) patients with chronic HBV infection. Positive HBV-DNA load was detected in 436 (32.7%) patients. The median age of patients was 50 years old. Total tumor diameter was 7.3 ± 3.2 cm. The preoperative liver function of 1295 (97.1%) patients was classified as Child–Pugh A. 354 (26.5%) patients developed clinically relevant PHT. The other clinical parameters were shown in the Table 1. Several prognostic factors, including APRI, FIB-4, ALBI, PNI, Child score and CONUTS, were compared with ROC curve (Fig. 2A). The area under curves (AUCs), standard error, P-value and 95% Confidence Interval of factors in predicting complication III-V are shown in Fig. 2B. Early postoperative CONUTS (AUC = 0.654, P < 0.001) was selected as an optimal index and 8 was identified as the cut-off value. Then, patients were divided into the two groups: high CONUTS group (early postoperative CONUTS ≥ 8) with 659 patients and low CONUTS group (early postoperative CONUTS < 8) with 675 patients. There is no significant differences in HBV infection, HBV-DNA load, Child-Pugh grade, incidence of CSPH, platelet, tumor number, total tumor diameter, ASA grade, microvascular invasion, tumor differentiation and R-0 resection were observed between the two groups. Patients with early postoperative high CONUTS had more intraoperative blood loss (P < 0.001), transfusion rate (P < 0.001), occupancy rate of ICU (P < 0.001), longer hospitalization time (P < 0.001) and mortality in 90 days (P = 0.001). The details were shown in the Table 1.
Postoperative complications
A total of 444 patients suffered the postoperative complications, grade III-V complications appeared in 145 patients. 13 patients died of the severe complications, died causes were cardiovascular accident (1 case), secondary abdominal hemorrhage (2 cases), abdominal infection (3 case), liver failure grade C (3 cases), multiple organ dysfunction syndrome (4 cases). Patients with early postoperative high CONUTS had higher incidence of complication III-V (P < 0.001), pulmonary complications (P = 0.011), bile leakage (P = 0.018), intra-abdominal hemorrhage (P = 0.001), and liver failure Grade C (P = 0.002). The details were shown in the Table 2.
Risk factors for postoperative complication III-V
In order to identify the risk factors for postoperative complication III-V, a univariate analysis was carried out. Preoperative cholesterol (P = 0.002), preoperative albumin (P < 0.001), Child-Pugh score (P < 0.001), early postoperative CONUTS ≥ 8 (P < 0.001), early postoperative albumin (P < 0.001), early postoperative cholesterol (P < 0.001), intraoperative blood loss (P < 0.001), transfusion (P < 0.001), major liver resection (P = 0.001), pathological indexes included tumor differentiation (P = 0.006) and cirrhosis (P = 0.039) were identified as the significant factors for postoperative complication III-V. The details were shown in the Table 3.
In order to control the potential confounding factors, multivariate logistic regression analysis was performed. Model 1, which included Child score, preoperative albumin, preoperative cholesterol, early postoperative CONUTS ≥ 8, blood loss, transfusion and major liver resection as independent variables, showed that early postoperative CONUTS ≥ 8 (OR = 2.054, 95% CI = 1.371–3.078, P < 0.001) and transfusion (OR = 3.235, 95% CI = 2.159–4.847, P < 0.001) were identified as the independent risk factors of postoperative complication III-V. Model 2, in which early postoperative CONUTS ≥ 8 was replaced with early postoperative albumin and cholesterol, demonstrated that transfusion (OR = 3.159, 95% CI = 2.054–4.859, P < 0.001), early postoperative albumin (OR = 1.054, 95% CI = 1.013–1.097, P = 0.009) and cholesterol (OR = 1.693, 95% CI = 1.265–2.264, P < 0.001) as the significant and independent factors associated with postoperative complication III-V. The details were shown in the Table 4.
Discussion
Severe postoperative complications would prolong the length of hospital stay, increase the morbidity and worsen the prognosis. The main complications after hepatectomy include pulmonary complication, neurological compilation, intractable ascites, bile leakage, intra-abdominal hemorrhage and liver failure. Nutritional intervention could improve the tolerance of patients for chemotherapy and surgery, decrease postoperative complications, and improve the prognosis4,24,25. Recently there’s increasing study8,9,10,26,27 focus on the nutritional status of patients and found that complication III-V after hepatectomy was not only associated with liver function reserve, but also with nutritional status.
The CONUT score is composed of serum albumin, cholesterol level and the lymphocyte count, which were associated with immune response, infection, inflammation, tissue repair and regeneration26. Hypoalbuminemia and hypocholesterolemia could predict postoperative complications and poor prognosis9,28. Lymphocyte is associated with immune response and tumor progression29,30,31. Previous studies13,32 indicated that HCC patients with preoperative high CONUTS had significantly lower recurrence-free survival (P = 0.011) and overall survival (P = 0.006) rates, however further analysis of severe postoperative complications were not carried out. Surgical trauma, blood loss and organic consumption may be closely correlated with postoperative hemodynamics and abnormal blood constituents, which may affect the predictive ability of preoperative CONUTS for postoperative outcomes. Complex liver structure combined with cirrhosis usually lead to more blood loss, then more fluid infusion or blood components transfusion were carried out. A series of prospective observational trials about evaluating the prognostic value of different nutritional scores in major abdominal surgery included hepatic surgery are ongoing. One study33, from the University of Heidelberg, has reported that none of the nutritional assessment scores before operation defined malnutrition relevant to complications after pancreatic surgery. Therefore, we hypothesized that early postoperative CONUTS may become a more meaningful risk factor for postoperative complication III-V than preoperative index.
In order to investigate the relationship between the early postoperative CONUTS and complications, we conducted a comparison of multiple ROC curves included Child score, APRI, FIB-4, ALBI and PNI. The results confirmed what we supposed, early postoperative CONUTS, not preoperative or postoperative change CONUTS, was chosen as an optimal index. The postoperative high CONUTS group had more intraoperative blood loss, transfusion, postoperative morbidity, mortality and longer hospital stay after operation. Univariate and multivariate analysis suggested that early postoperative CONUTS ≥ 8 (OR = 2.054, 95%CI = 1.371–3.078, P < 0.001) was identified as an independent risk factor for complication III-V. Once early postoperative high CONUTS appears, attention should be paid and took required measure.
Transfusion was identified as another independently risk factor for complication III-V in the study. Operative trauma, blood composition consumption and coagulation derangements are important factors, which were associated with transfusion. HCC patients with cirrhosis who usually had lower tolerance to general anesthesia and surgical trauma. Several studies reported that transfusion was an independent risk factor for postoperative complications after hepatectomy, which was consistent with our results. Intraoperative excessive bleeding would cause liver ischemia and worsen intestinal ischemia-reperfusion injury. The impairment of intestinal barrier function would lead to the bacterial translocation, increase the incidence of postoperative infections, prolong the time of hospitalization, and influence patients’prognosis34,35. The liver parenchyma bleeding, blood vessel injury and tumor hemorrhage are important factors leads to transfusion. Therefore, accurate operation, meticulous hemostasis, and shorten operation time could reduce unnecessary transfusion and postoperative complications. In addition, previous guidelines36 recommended that liver resection as an indication of albumin supplementation. Whether preventive albumin supplement could reduce postoperative complications remains controversial. However, it is recognized that component transfusion is beneficial to correct postoperative hypoalbuminemia, severe coagulation disorder and relieve refractory ascites.
The present study has some limitations. First, the single-center study could not reduce information bias and selection bias. Second, a retrospective study lacked the validation set and had the poor strength of argument. Third, influence of chemoembolization or anticancer drugs on peripheral lymphocyte count before liver resection cannot be ignored. At last, multi-center prospective studies are required to to evaluate the role of early postoperative CONUTS.
In conclusion, early postoperative CONUTS is a useful tool in assessing the postoperative nutritional status in HCC patients who underwent liver resection. Postoperative high CONUTS did not only influence postoperative complication III-V, but also increase the mortality. The predictive value should be further evaluated.
Ethical review
This study was approved by the Clinical Research Ethics Committee of the West China Hospital, Sichuan University. Written informed consent was obtained from all patients according to the policies of the committee. All the methods used in this study were carried out according to the approved guidelines.
References
The Burden of Primary Liver Cancer and Underlying Etiologies From 1990 to 2015 at the Global, Regional, and National Level: Results From the Global Burden of Disease Study 2015. JAMA Oncol 3, 1683–1691 (2017).
Petrick, J. L. et al. International trends in liver cancer incidence, overall and by histologic subtype, 1978–2007. Int J Cancer 139, 1534–1545 (2016).
Takayama, T. et al. Early hepatocellular carcinoma as an entity with a high rate of surgical cure. Hepatology 28, 1241–1246 (1998).
Okabayashi, T. et al. Effects of branched-chain amino acids-enriched nutrient support for patients undergoing liver resection for hepatocellular carcinoma. J Gastroenterol Hepatol 23, 1869–1873 (2008).
Vyhnánek, F., Duchác, V., Vyhnánková, I. & Skála, P. Postoperative nutritional support in liver surgery. Effects of specialized parenteral nutrition enriched with branched-chain amino acids following liver resections for colorectal carcinoma metastases. Rozhl Chir 87, 21–25 (2008).
Hsieh, C. E. et al. Comparative factor analysis of the effect of postoperative peripheral parenteral nutrition on recovery of right lobe liver donors. Exp Clin Transplant 13, 157–162 (2015).
Lin, Z. H. et al. Performance of the aspartate aminotransferase-to-platelet ratio index for the staging of hepatitis C-related fibrosis: an updated meta-analysis. Hepatology 53, 726–736 (2011).
Wang, H. et al. Immediate postoperative Fibrosis-4 predicts postoperative liver failure for patients with hepatocellular carcinoma undergoing curative surgery. Dig Liver Dis (2017).
Johnson, P. J. et al. Assessment of liver function in patients with hepatocellular carcinoma: a new evidence-based approach-the ALBI grade. J Clin Oncol 33, 550–558 (2015).
Ke, M. et al. Prognostic nutritional index predicts short-term outcomes after liver resection for hepatocellular carcinoma within the Milan criteria. Oncotarget 7, 81611–81620 (2016).
de Ulíbarri, J. I. et al. CONUT: a tool for controlling nutritional status. First validation in a hospital population. Nutr Hosp 20, 38–45 (2005).
Iseki, Y. et al. Impact of the Preoperative Controlling Nutritional Status (CONUT) Score on the Survival after Curative Surgery for Colorectal Cancer. PLoS One 10, e0132488 (2015).
Takagi, K. et al. Preoperative Controlling Nutritional Status (CONUT) Score for Assessment of Prognosis Following Hepatectomy for Hepatocellular Carcinoma. World J Surg 41, 2353–2360 (2017).
Toyokawa, T. et al. The pretreatment Controlling Nutritional Status (CONUT) score is an independent prognostic factor in patients with resectable thoracic esophageal squamous cell carcinoma: results from a retrospective study. BMC Cancer 16, 722 (2016).
EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol 56, 908–943 (2012).
Bruix, J. & Sherman, M. Management of hepatocellular carcinoma: an update. Hepatology 53, 1020–1022 (2011).
Fung, J. et al. Profiles of HBV DNA in a large population of Chinese patients with chronic hepatitis B: implications for antiviral therapy. J Hepatol 54, 195–200 (2011).
Wen, T. et al. Continuous normothermic hemihepatic vascular inflow occlusion over 60 min for hepatectomy in patients with cirrhosis caused by hepatitis B virus. Hepatol Res 37, 346–352 (2007).
Huang, Z., Zhang, P., Wang, H., Yan, L. & Wang, W. Comparing outcomes of two vascular inflow occlusion techniques and treatment without vascular occlusion during major hepatectomy in patients with Hepatitis B-related hepatocellular carcinoma. PLoS One 9, e107303 (2014).
Santambrogio, R. et al. Hepatic resection for hepatocellular carcinoma in patients with Child-Pugh’s A cirrhosis: is clinical evidence of portal hypertension a contraindication. HPB (Oxford) 15, 78–84 (2013).
Dindo, D., Demartines, N. & Clavien, P. A. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240, 205–213 (2004).
Rahbari, N. N. et al. Posthepatectomy liver failure: a definition and grading by the International Study Group of Liver Surgery (ISGLS). Surgery 149, 713–724 (2011).
Koch, M. et al. Bile leakage after hepatobiliary and pancreatic surgery: a definition and grading of severity by the International Study Group of Liver Surgery. Surgery 149, 680–688 (2011).
Paccagnella, A., Morassutti, I. & Rosti, G. Nutritional intervention for improving treatment tolerance in cancer patients. Curr Opin Oncol 23, 322–330 (2011).
Kubota, K. et al. Preoperative oral supplementation support in patients with esophageal cancer. J Nutr Health Aging 18, 437–440 (2014).
Roxburgh, C. S. & McMillan, D. C. Role of systemic inflammatory response in predicting survival in patients with primary operable cancer. Future Oncol 6, 149–163 (2010).
Zhang, X. et al. Postoperative Prognostic Nutritional Index Predicts Survival of Patients with Hepatocellular Carcinoma within Milan Criteria and Hypersplenism. J Gastrointest Surg 21, 1626–1634 (2017).
Wang, Q. et al. Preoperative total cholesterol predicts postoperative outcomes after partial hepatectomy in patients with chronic hepatitis B- or C-related hepatocellular carcinoma. Surgery 155, 263–270 (2014).
Ropponen, K. M., Eskelinen, M. J., Lipponen, P. K., Alhava, E. & Kosma, V. M. Prognostic value of tumour-infiltrating lymphocytes (TILs) in colorectal cancer. J Pathol 182, 318–324 (1997).
Dolcetti, R. et al. High prevalence of activated intraepithelial cytotoxic T lymphocytes and increased neoplastic cell apoptosis in colorectal carcinomas with microsatellite instability. Am J Pathol 154, 1805–1813 (1999).
Menges, T. et al. Changes in blood lymphocyte populations after multiple trauma: association with posttraumatic complications. Crit Care Med 27, 733–740 (1999).
Takagi, K. et al. Preoperative Controlling Nutritional Status Score Predicts Mortality after Hepatectomy for Hepatocellular Carcinoma. Dig Surg (2018).
Probst, P. et al. Prospective trial to evaluate the prognostic value of different nutritional assessment scores in pancreatic surgery (NURIMAS Pancreas). Br J Surg 104, 1053–1062 (2017).
Moreno, E. A., Davenport, D. L., Hundley, J. C., Daily, M. F. & Gedaly, R. Predictors of surgical site infection after liver resection: a multicentre analysis using National Surgical Quality Improvement Program data. HPB (Oxford) 14, 136–141 (2012).
Pessaux, P. et al. Identification and validation of risk factors for postoperative infectious complications following hepatectomy. J Gastrointest Surg 17, 1907–1916 (2013).
Vermeulen, L. C., Ratko, T. A., Erstad, B. L., Brecher, M. E. & Matuszewski, K. A. A paradigm for consensus. The University Hospital Consortium guidelines for the use of albumin, nonprotein colloid, and crystalloid solutions. Arch Intern Med 155, 373–379 (1995).
Acknowledgements
This work was supported by the Natural Science Foundation of China (Nos 81470037 and 81770653).
Author information
Authors and Affiliations
Contributions
L.L. and L.C. wrote the main manuscript text, Y.J.Y. and Y.L.N. designed the research, L.C. and L.L. collected the data, L.L., L.C. and Y.J.Y. analysed the data. W.H., W.T.F., W.W.T. and L.B. revised the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, L., Liu, C., Yang, J. et al. Early postoperative controlling nutritional status (CONUT) score is associated with complication III-V after hepatectomy in hepatocellular carcinoma: A retrospective cohort study of 1,334 patients. Sci Rep 8, 13406 (2018). https://doi.org/10.1038/s41598-018-31714-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-31714-w
This article is cited by
-
Associations of creatinine/cystatin C ratio and postoperative pulmonary complications in elderly patients undergoing off-pump coronary artery bypass surgery: a retrospective study
Scientific Reports (2021)
-
Geriatric nutritional risk index predicts prognosis in hepatocellular carcinoma after hepatectomy: a propensity score matching analysis
Scientific Reports (2021)
-
Impact of the controlling nutritional status score on severe postoperative complications of pancreaticoduodenectomy for pancreatic cancer
Langenbeck's Archives of Surgery (2021)
-
Controlling nutritional status score predicts postoperative complications after hip fracture surgery
BMC Geriatrics (2020)
-
Prognostic significance of the Controlling Nutritional Status (CONUT) score in predicting postoperative complications in patients with Crohn’s disease
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.