Abstract
The largest insects to have ever lived were the giant meganeurids of the Late Palaeozoic, ancient stem relatives of our modern dragonflies. With wingspans up to 71 cm, these iconic insects have been the subject of varied documentaries on Palaeozoic life, depicting them as patrolling for prey through coal swamp forests amid giant lycopsids, and cordaites. Such reconstructions are speculative as few definitive details of giant dragonfly biology are known. Most specimens of giant dragonflies are known from wings or isolated elements, but Meganeurites gracilipes preserves critical body structures, most notably those of the head. Here we show that it is unlikely it thrived in densely forested environments where its elongate wings would have become easily damaged. Instead, the species lived in more open habitats and possessed greatly enlarged compound eyes. These were dorsally hypertrophied, a specialization for long-distance vision above the animal in flight, a trait convergent with modern hawker dragonflies. Sturdy mandibles with acute teeth, strong spines on tibiae and tarsi, and a pronounced thoracic skewness are identical to those specializations used by dragonflies in capturing prey while in flight. The Palaeozoic Odonatoptera thus exhibited considerable morphological specializations associated with behaviours attributable to ‘hawkers’ or ‘perchers’ among extant Odonata.
Similar content being viewed by others
Introduction
Despite rampant speculation as to the biology of gigantic insects from the Late Palaeozoic, particularly assumptions regarding their presumed predatory feeding and hunting behaviours, the most pertinent details regarding morphology of Meganeuridae are essentially unknown. Owing to their relationship to modern dragonflies and damselflies (Odonata), many details of odonate biology have been extended to the stem superorder Odonatoptera. Most critically, the anatomical details of the meganeurid head — especially their mouthparts and compound eyes — are effectively unknown. Although Meganeura monyi is the iconic giant dragonfly, it is poorly preserved. Only two prior fossils have been reported preserving such portions of giant dragonfly anatomy, viz. Meganeurula selysii in which the head and thorax are unfortunately artefacts produced by over preparation (Fig. 1, Extended Data Fig. 1); these, Namurotypus sippeli where the head is exceptionally poorly preserved, and Erasipteroides valentini revealing little more than the separation of the compound eyes (Supplementary Information), have formed the basis for reconstructions1. However, a third fossil, the holotype of Meganeurites gracilipes from the well-known Gzhelian outcrop of Commentry (France), has well-preserved head structures which have been long-ignored and therefore never studied or considered in regard to the palaeobiology of these giants. The animal was preserved in fine-grained micaceous sandstone and compressed during fossilization (Supplementary Information). The local palaeoenvironment, approximately 300 Ma (Stephanian B/C), corresponds to a Pennsylvanian limnic biotope in an intramontane basin adjacent to moorland facies2.
Results
(see Supplementary Information for a detailed description of Meganeurites).
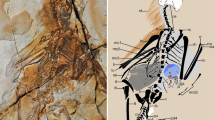
The holotype of M. gracilipes is fairly complete, certainly by the standards of most known material of meganeurids, with the head, thorax, and much of the legs, wings, and basal part of the abdomen complete, albeit compressed. The counter-part (MNHN.F.R53005) preserves the body in dorsal view (Figs 2 and 3), while the original print, preserving the ventral view of the insect, was lost long ago. The mandibles are prominently preserved and were strong, robust, and with large, sharply acute teeth, similar to those of extant Odonata (Fig. 2). The mandibular form and dentition demonstrates that M. gracilipes, and likely all species of the family Meganeuridae, were predators. The short antennae with a flagellum of Meganeurites probably had the same function in flight control as those of extant Odonata3. The positions of the three simple eyes (ocelli) on vertex are in the same positions as those of the extant dragonfly family Aeshnidae, supporting similar roles as the horizon detectors which contribute to the balance body control during fast flight maneuvers4.
Meganeurites gracilipes Handlirsch, 1919, holotype MNHN R53005, head and fore leg. (a) dorsal view; (b) reconstruction. f. frons, i1, i2 incisivum, lb. labrum, lat.oc. lateral ocellus, md. mandible, ml. molar plate, m.oc. median ocellus, mx. Maxilla, occ. occipital triangle, v. vertex (photograph Gaelle Doitteau, e-recolnat Project, MNHN, reconstruction M.P.). Scale bar, 10 mm.
As in Odonata, the thorax is slanted caudally, resulting in what is referred to as “thoracic skew”5, resulting in a slight oblique orientation to the dorsal surface of the thorax and thereby angling the plane of the wings relative to the longitudinal axis of the body. This skew was likely important for Meganeuridae, as it results in an anterior displacement of the legs in M. gracilipes, Meganeurula selysii (Figs 1 and 3, Extended Data Fig. 1), and Odonata. The more forward position of the legs makes grasping objects in front of the animal easier, as well as the manipulation of materials held in front of the head, such as a prey item. In addition, the presence of strong spines on the tibiae and tarsi, previously known for Meganeura monyi6,7, and present also in M. gracilipes, indicates that together the thorax and legs of Meganeurites functioned as a”flying trap” for the capture of prey, a morphological and behavioural suite identical to that of modern dragonflies and damselflies.
Unlike more basal, earlier-diverging Odonatoptera, Meganeurites has no trace of paranotal expansions (‘pronotal lobes’), or prothoracic winglets (Supplementary Information), and it is likely that such an absence is characteristic of all Meganeuridae. Accordingly, meganeurid flight was more similar to those of extant dragonflies than to those of any coeval “six-winged” Palaeodictyoptera8,9. Nevertheless, the absence of nodal flexion structure in Meganeuridae probably prevented them from achieving flight performances similar to those of the true Odonata, viz. with the capacity to twist and make abrupt, directional changes while in flight10. Accordingly, Meganeurites was more likely an open-space, ecotone, or riparian forest predator. Using modern odonates as an analogue, meganeurids would have been ‘hawkers’, patrolling above large rivers, ancient lakes, open forests, or even above the canopy11,12, rather than ‘perchers’, who fly in rapid, zig-zag formations through relatively dense forest environments, with liana-like foliage already present during the Ghezlian13,14. In the latter habitats, the large wingspan of M. gracilipes (ca. 320 mm) would have been a significant handicap to fly or even glide (Fig. 4). As is true for modern hawker dragonflies, excellent visual acuity is critical for catching large, flying preys. This would have been true for M. gracilipes. These large Meganeuridae were probably preying on large Palaeodictyoptera also present in the palaeobiota of Commentry.
Visual precision would also have been critical for Meganeurites to evade its own predators which were probably larger meganeurids, such as Meganeura monyi with its wingspan of ca. 700 mm, characteristic of the fauna at Commentry. Flying and gliding vertebrates appeared ca. 30 myr later. Similar preying behaviours are known for extant Anisoptera15. Meganeurites had enlarged compound eyes with broad dorsal portions, meeting medially for significant portion of their length, as in extant hawker dragonflies (Fig. 2, Extended Data Fig. 2). Among all fossil and extant Odonatoptera, only the Aeshnidae and some ‘libelluloids’ (Macromiidae, some ‘Corduliidae’, and Pantaliinae and Zyxommatinae among Libellulidae) have dorsally meeting compound eyes16,17,18, and are ‘hawkers’, “remaining in flight continuously throughout the day, and foraging in flight by swooping up to grab insects passing overhead”19. Owing to the hypertrophied development of the dorsal portion of the compound eyes and likely specialized ommatidia associated with such a condition, such dragonflies can more easily detect objects (prey or predators) against the blue sky20,21,22. We can infer from the eyes’ shape that the condition was similar for Meganeurites and that it would have also had excellent vision, consistent with the ‘hawker’ behaviour implied by the size of its wings. Extant Aeshnidae have specialized ommatidia on dorsal part of the eyes, but, unfortunately, the ommatidia are not preserved in the holotype of Meganeurites. Many of those extant taxa with large and broadly confluent compound eyes in dorsal view are crepuscular, such as Zyxommatinae and Tholymis (Libellulidae), Apomacromia (Corduliidae), Aeshna viridis, Limnetron (both Aeshnidae), or Gynacanthinae (Extended Data Fig. 2)16,23,24,25,26. This is not the case for all dragonflies with broadly confluent compound eyes, and some crepuscular gomphids have separated compound eyes. Accordingly, it is not possible to say with certainty that Meganeurites was similarly crepuscular, although this was most likely. The head of Meganeurites was narrower than the thorax (Figs 3 and 4), suggesting that it is possible that its vision was less optimal than that of modern Anisoptera with large compound eyes. The presence of dorsally adjoining eyes is a potential apomorphy of the Meganeuridae (or the subfamily Tupinae), to be verified through the discovery of new fossils spanning the diversity of this group. The widely separated compound eyes of the giant Erasipteroides valentini (Erasipteridae)27 led previous authors to consider erroneously that this was characteristic of all giant dragonflies28. On the contrary, the morphological disparity of the compound eyes among Palaeozoic meganeurids was apparently as important as it is today among modern dragonflies (Supplementary Information), suggesting a similar behavioural diversity among these early odonatopterans.
We performed two multivariate morphometric analyses on four fossil taxa and 21 extant Odonata in regards to the morphology of the head, forelegs, thorax, and wings (Supplementary Information). The considerably large sizes of the chosen Carboniferous and Jurassic Odonatoptera have a great influence on the results (Extended Data Fig. 4). Nevertheless, the observation along axes 2 and 3 of the first analysis (raw data) minimizes the effect of size and demonstrates that Meganeurites falls among the extant hawkers (Extended Data Fig. 5). We show that the false ‘head’ of Meganeurula selysii and the rather poor preservation of the head of Erasipteroides valentini place them well apart from all other taxa (Extended Data Figs 4–6).
During the Late Carboniferous, there simultaneously existed comparatively small, damselfly-like Odonatoptera29,30, probably living in densely forested environments, catching small prey along rivers or within the forests themselves. Later, during the Permian, the Meganeuridae also diversified into a range of taxa, spanning sizes from gigantic to species whose wingspans more closely approximate those of extant Anisoptera7, and confirming the co-existence of different life habits for these ancient flying predators. The discovery of evidence relating to the palaeobiology of the charismatic, iconic, and giant dragonflies also reveals a remarkable consistency in predatory biology, as well as a range of variants known still today, along the odonatopteran lineage over the course of at least 300 million years.
Methods
Material studied
The two specimens MNHN.F.R53005 and MNHN R52939 are deposited in the collection of Palaeontology, MNHN, Paris, France.
Observation and description
Photographs were taken with a Nikon D800 digital camera with AF-MicroNikkor 60 mm, using four optical fibers to improve the light. Original photographs were processed using the image-editing software Adobe Photoshop CS6. Standard wing venation nomenclature is followed for Odonatoptera31,32. Two Principal Component Analysis were performed to compare the morphology of the fossil and extant Odonatoptera (Supplementary Information).
Online Content
Additional Methods, Extended Data display items and Source Data are available in the online version of the paper; references unique to these sections appear only in the online paper.
Data availability statement
All data generated or analyzed during this study are included in this published article (and its Supplementary Information files).
References
Kukalová-Peck, J. Carboniferous protodonatoid dragonfly nymphs and the synapomorphies of Odonatoptera and Ephemeroptera (Insecta: Palaeoptera). Palaeodiversity 2, 169–198 (2009).
Schneider, J. W. Taxonomie, Biostratigraphie und Palökologie der Blattodea-Fauna aus dem Stefan von Commentry (Frankreich). Versuch einer Revision. Freiberger Forschungsh. (C) 384, 77–100 (1983).
Gewecke, M., Heinzel, H.-G. & Philippen, J. Role of antennae of the dragonfly Orthetrum cancellatum in flight control. Nature 249, 584–585 (1974).
Berry, R., van Kleef, J. & Stange, G. The mapping of visual space by dragonfly lateral ocelli. J. Comp. Physiol. (A) 193, 495–513 (2007).
Needham, J. G. & Anthony, M. H. The skewness of the thorax in the Odonata. J. N. Y. Entomol. Soc. 11(3), 117–125 (1903).
Brongniart, C. Sur un gigantesque Neurorthoptère, provenant des terrains houillers de Commentry (Allier). Compt. rend. hebdomadaires des séances Acad. Sci. 98, 832–833 (1884).
Nel, A.et al. Revision of Permo-Carboniferous griffenflies (Insecta: Odonatoptera: Meganisoptera) based upon new species and redescription of selected poorly known taxa from Eurasia. Palaeontographica (A) 289, 89–121 (2009).
Wootton, R. J. & Kukalová-Peck, J. Flight adaptations in Palaeozoic Palaeoptera (Insecta). Biol. Rev. 75, 129–167 (2000).
Prokop, J. et al. Paleozoic nymphal wing pads support dual model of insect wing origins. Curr. Biol. 27, 263–269 (2017).
Wootton, R. J., Kukalová-Peck, J., Newman, J. S. & Muzon, J. Smart engineering in the Mid-Carboniferous: how well could Paleozoic dragonflies fly? Science 282, 749–751 (1998).
DiMichele, W. A. & Hook, R. W. Paleozoic terrestrial ecosystems. In A. K. Behrensmeyer et al., eds., Terrestrial ecosystems through time. 205–325 (University of Chicago Press, 1992).
DiMichele, W. A. & Falcon-Lang, H. J. Fossil forests in growth position (T0 assemblages): origin, taphonomic biases and palaeoecological significance. J. Geol. Soc. 168, 585–605 (2011).
Krings, M., Kerp, H., Taylor, E. L. & Taylor, T. N. Reconstruction of Pseudomariopteris busquetii, a vine-like Late Carboniferous-Early Permian pteridosperm. Am. J. Bot. 88, 767–776 (2001).
Falcon-Lang, H. A calamitalean forest preserved in growth position in the Pennsylvanian coal measures of South Wales: Implications for palaeoecology, ontogeny and taphonomy. Rev. Palaeobot. Palynol. 214, 51–67 (2015).
Pritchard, G. The prey of adult dragonflies in Northern Alberta. The Canadian Entomologist 96, 821–825 (1964).
Watson, J. A. L. Apocordulia macrops, a new crepuscular gomphomacromiine dragonfly from South-East Australia (Odonata: Corduliidae). J. Austral. Entomol. Soc. 9(4), 287–292 (1980).
Bechly, G. Phylogenetic systematics of Odonata. - homepage on Internet site: https://bechly.lima-city.de/phylosys.htm (2016).
Carle, F. L., Karl, M., Kjer, K. M. & May, M. L. A molecular phylogeny and classification of Anisoptera (Odonata). Arthropod Syst. Phyl. 73, 281–301 (2015).
Olberg, R. M., Worthington, A. H., Fox, J. L., Bessette, C. E. & Loosemore, M. P. Prey size selection and distance estimation in foraging adult dragonflies. J. Comp. Physiol. (A) 191, 791–797 (2005).
Sherk, T. E. Development of the compound eyes of dragonflies (Odonata). III. Adult compound eyes. J. Exp. Zool. 203, 61–80 (1978).
Labhart, T. & Nilsson, D.-E. The dorsal eye of the dragonfly Sympetrum: specializations for prey detection against the blue sky. J. Comp. Physiol. 176, 437–453 (1995).
Sauseng, M., Pabst, M.-A. & Kral, K. The dragonfly Libellula quadrimaculata (Odonata: Libellulidae) makes optimal use of the dorsal fovea of the compound eyes during perching. Eur. J. Entomol. 100, 475–479 (2003).
Fraser, F. C. A handbook of the dragonflies of Australia with keys for the identification of all species. Proc. R. Zool. Soc. N. S. W. (pp. 1–67 (1960).
Pinhey, E. C. G. D. (Odonata) of CentralAfrica. Occ. Pap. Rhodes-Livingstone Mus. 14, 1–97 (1961).
Garrison, R. W., von Ellenrieder, N. & Louton, J. A. Dragonfly genera of the new world: an illustrated and annotated key to the Anisoptera, 368 pp. (The Johns Hopkins University Press, 2006).
Borkenstein, A., Schroeter, A. & Joedicke, R. Aeshna viridis is an early bird - matutinal matings in a crepuscular species (Odonata: Aeshnidae). Odonatologica 45(1–2), 37–56 (2016).
Brauckmann, C., Koch, L. & Kemper, M. Spinnentiere (Arachnida) und Insekten aus den Vorhalle-Schichten (Namurium B; Ober-Karbon) von Hagen-Vorhalle (West-Deutschland). Geol. Paläont. Westf., Westf. Mus. Naturk. 3, 1–132 (1985).
Zessin, W. Einige Aspekte zur Biologie paläozoischer Libellen (Odonatoptera). Entomol. Generalis 31(3), 261–278 (2008).
Jarzembowski, E. A. & Nel, A. The earliest damselfly-like insect and the origin of modern dragonflies (Insecta: Odonatoptera: Protozygoptera). Proc. Geol. Assoc. 113(2), 165–169 (2002).
Nel, A., Bechly, G., Prokop, J., Béthoux, O. & Fleck, G. Systematics and evolution of Palaeozoic and Mesozoic damselfly-like Odonatoptera of the ‘protozygopteran’ grade. J. Palaeont. 86(1), 81–104 (2012).
Riek, E. F. & Kukalová-Peck, J. A new interpretation of dragonfly wing venation based upon Early Carboniferous fossils from Argentina (Insecta: Odonatoidea) and basic characters states in pterygote wings. Can. J. Zool. 62, 1150–1166 (1984).
Bechly, G. Morphologische Untersuchungen am Flügelgeäder der rezenten Libellen und deren Stammgruppenvertreter (Insecta; Pterygota; Odonata), unter besonderer Berücksichtigung der Phylogenetischen Systematik und des Grundplanes der *Odonata. Petalura Spec. 2, 1–402 (1996).
Acknowledgements
We sincerely thank two anonymous referees for the very useful comments on the first version of the paper. This work benefited from a grant of the French ‘Agence Nationale de la Recherche’ via the program ‘Investissements d’avenir’ (ANR-11-INBS-0004-RECOLNAT). JP and MP gratefully acknowledge research support from the Grant Agency of the Czech Republic No. 18-03118 S. The work of MSE was supported by US National Science Foundation grant DEB-1144162.
Author information
Authors and Affiliations
Contributions
All authors prepared the manuscript; J.P., R.G., A.N. and M.P. prepared the figures; A.N. and R.G. designed the program.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nel, A., Prokop, J., Pecharová, M. et al. Palaeozoic giant dragonflies were hawker predators. Sci Rep 8, 12141 (2018). https://doi.org/10.1038/s41598-018-30629-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-30629-w
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.