Abstract
The International Commission on Radiological Protection has proposed an environmental assessment framework. This includes ionising radiation exposure assessment for different frog life-stages, but radiocaesium transfer parameters are unavailable. We collate data from the Fukushima Prefecture (contaminated by the Fukushima accident) and estimate radiocaesium concentration ratio (CRwo-water) values for tadpoles and adult frogs, presenting the largest available amphibian CRwo-water dataset. In total, 513 adult frogs and 2540 tadpoles were analysed in 62 and 59 composite samples respectively. Results suggest that equilibrium was reached between water and amphibian radiocaesium activity concentrations circa one-year after the accident. Radiocaesium transfer to tadpoles was higher than to adult frogs. Dose rates were estimated for different life-stages and species in both the aquatic and terrestrial environment. Estimated dose rates to adults and tadpoles were typically similar because external exposure dominated for both organisms; frogspawn dose rates were estimated to be orders of magnitude lower than other life-stages. For the two sites assessed, which were outside of the most contaminated areas of the Fukushima Prefecture, estimated dose rates were below those anticipated to present a risk to wildlife populations; it is likely that dose rates in more contaminated areas were in excess of some effects benchmark values.
Similar content being viewed by others
Introduction
The developing environmental protection framework of the International Commission for Radiological Protection (ICRP)1 is based around the concept of ‘Reference Animals and Plants’ (RAPs) with the RAPs being defined at the taxonomic level of family. The ICRP proposes that the exposure of different life-stages should be considered in environmental assessments. One of the ICRPs RAPs is the Reference Frog (defined as the Ranidae family) for which, the ICRP propose, spawn (mass of eggs), tadpoles and adult life-stages be considered in assessments.
To estimate exposure there is a need to quantify activity concentrations in organisms including different life-stages and this is a recommendation of the ICRP1. However, in ICRP2, there were insufficient data to recommend transfer parameter values for life-stages other than adults; data for adult frogs (Ranidae species) were only presented for four elements (Ca, Cr, Pb and Zn) in the freshwater ecosystem2. Similarly, neither the International Atomic Energy Agency (IAEA) wildlife transfer parameter handbook3 or the revised version of the commonly used model for wildlife dose assessment, the ERICA Tool4, present transfer parameter values for Cs and amphibians in the freshwater environment. A number of post-Fukushima studies in Japan have investigated radiocaesium concentrations in frog species e.g.5,6,7,8. However, to our knowledge transfer parameter values for frogs in the freshwater environment have not been published for studies conducted in Japan.
Limited data (for on two adults and three tadpoles) presented in IAEA9 suggests that the transfer of radiocaesium to tadpoles is approximately one order of magnitude higher than that to adults. Data from Fukushima reported by Watanabe et al.8 supported the suggestion that radiocaesium activity concentrations in tadpoles would be higher than those in adult frogs. However, Watanabe et al.8 did not report water activity concentrations and transfer parameter values could not be derived.
In this study, we have used open source monitoring data for the Fukushima Prefecture10 to calculate transfer parameter values for radiocesium for both tadpoles and adult frogs (‘frog’ is used here to describe any member of the order Anura which includes true frogs and toads). Data were collated from five river systems (Abukuma River (two sites, A-2 and B-3), Mano River, Niida River, Ota River and Uda River) and three lakes (Hayama Lake, Aimoto Lake, and Inawashiro Lake) (Fig. 1). The transfer parameter values presented in this paper are concentration ratios (CRwo-water), which relate the whole-organism activity concentration (Bq kg−1 fresh mass) to the activity concentration in water (Bq l−1). We also use the data to evaluate the exposure of different life-stages for two frog species and put these into context with international recommendations on the effects of radiation on amphibians.
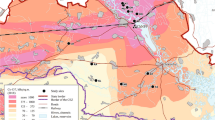
Frog and tadpole sampling sites in Fukushima (drawn from information presented by the Ministry of the Environment (http://www.env.go.jp/en/water/rmms/result_ao17-part.html)10 using Microsoft PowerPoint™ for Mac version 15.32).
Results
As an example of the collated data, Fig. 2 presents changes in 137Cs activity concentrations with time in water, sediments, adult frogs and tadpoles for Abukuma River and Inawashiro Lake (these were the two water bodies for which most data were available). The complete collated dataset is available as Supplementary Information. Data were available for: American bullfrog (Rana catesbeiana), Eastern-Japanese common toad (Bufo japonicus formosus), Forest Green frog (Rhacophorus arboreus), Japanese Brown frog (Rana japonica), Japanese tree frog (Hyla japonica), Kajika frog (Buergeria buergeri), Montane Brown frog (Rana ornativentris), Schlegel’s Green tree frog (Rhacophorus schlegelii), Tokyo Daruma pond frog (Rana porosa porosa), and Wrinkled frog (Glandirana rugosa)10 (Table 1). One measurement for each of adult frog and tadpole was reported as having a 137Cs activity concentration below detection limits (see Supplementary Information).
Concentration ratios estimated from the collated data for frogs collected in the Fukushima Prefecture are presented, by species and life-stage, in Table 2. For adults, 62 CRwo-water values were estimated, though individual CRwo-water values are based upon samples generally comprising of multiple individual frogs. For tadpoles, 59 CRwo-water data were calculated, but tadpole species was generally not given. Where tadpole species was specified, it was possible to calculate tadpole CRwo-water values for American bullfrog (n = 10), Kajika frog (n = 1) and Montane Brown frog (n = 1). Concentration ratio data generally have a lognormal distribution11. Graphical analysis of the tadpole and adult frog CRwo-water data also tended towards log-normal (Fig. 3) and hence data were logged here prior to statistical analysis. Given there was only one CRwo-water for each of adult frog and tadpole based on an organism activity concentration below detection limits, the ‘less than’ CRwo-water values have been used in the subsequent analyses.
CRwo-water values for 134Cs vs 137Cs
For a given element, CRwo-water values are generally assumed to be the same for all isotopes2,3,12,13, so CRwo-water values calculated for 137Cs would be applicable to 134Cs. This hypothesis was tested here using all adult frog and tadpole data for which we had both 134Cs and 137Cs data reported above the detection limit. Paired T-tests confirmed no significant difference in the transfer of the two Cs isotopes for both the adult frogs (p = 0.12; n = 53) and the tadpoles (p = 0.48; n = 55). Given the isotopic independence of transfer, only 137Cs derived CRwo-water values have been used in the subsequent data analysis presented here.
Temporal variation in CRwo-water values
Figure 4 shows estimated tadpole and adult frog CRwo-water values summarised by year. Only two adult frog CRwo-water values could be calculated for 2012 as water data were lacking for other samples. For 2016, data were only available for the first month of annual sampling. A regression analysis across all individual sampling times over the period for which data were available revealed no statistically significant time trend in either the adult (r2 = 0.08) or tadpole (r2 = 0.01) CRwo-water values. Therefore, it would appear that CRwo-water had reached an equilibrium by 2012/13 so hereafter we analyse the data ignoring year of sampling.
The data were insufficient to allow analysis of any seasonal trend without the confounding influence of site.
Comparison of CRwo_water for tadpole and adult frogs
When data for all species were considered, the geometric mean (GM) value for tadpole (3.5 × 103 l kg−1) was significantly higher than that for adult frog (5.8 × 102 l kg−1) (T-test, p < 0.0001) (Table 2). Where sufficient data were available, this trend was also consistent across the different water bodies. For each of Abukuma River, Akimoto Lake and Inawashiro Lake, tadpoles (n = 24, 9, 9 respectively) had a significantly higher CRwo-water value than adult frogs (n = 22, 11, 14 respectively) (T-test, p < 0.05).
Although species was not identified for most tadpole samples, where this information was provided a similar trend was seen with tadpoles having a higher CRwo-water than adults of the same species. For example, Fig. 5 presents data for American bullfrog for which the GM CRwo-water value was approximately ten times higher for tadpoles than for adults (T-test, p = 0.002).
CRwo-water values – species variation
Adult frog CRwo-water values for species with sufficiently high sample numbers (i.e. American bullfrog, Montane Brown frog, Tokyo Daruma pond frog and Wrinkled frogs) were compared using a General Linear Model. The Montane Brown frog was found to have a significantly higher CRwo-water value than the other three species (p < 0.05).
CRwo-water values – lake vs river
Data for radiocaesium transfer to adult frogs were available for both lakes and rivers, presenting an opportunity to test whether there was a significant difference in the transfer between these two types of freshwater system. A T-test showed no significant difference between the CRwo-water values for adult frogs in lakes (n = 26) and rivers (n = 36) (p = 0.19). However, for tadpoles the CRwo-water value for rivers (n = 41) was significantly higher than that for lakes (n = 18) (p = 0.012; Mann-Whitney Test).
Figure 2, suggests there may be more seasonality in the activity concentrations of river water compared to lake water and this may hence add to additional variability within CRwo-water values estimated for rivers. However, examination of the data (see Supplementary Information) shows no seasonal tend in CRwo-water values for Abukuma River (the river for which there are most data).
Estimated dose rates
Table 3 presents the estimated dose rates for the Montane Brown and Wrinkled frogs and their life-stages in Lake Akimoto in 2012 and 2016. Apart from estimates for the frogspawn life-stage, differences between the two species are minimal. As a consequence of the similarity in predictions for the two species at Lake Akimoto, dose rates for Uda River were only estimated for the Montane Brown frog (Table 4).
Discussion
The similarity in CRwo-water values for 134Cs and 137Cs provides further evidence to support the commonly used approach2,3,12,13 of treating isotopes of a given element as having the same CRwo-water value (if the isotopes can be assumed to be in equilibrium). The CRwo-water values reported here show considerable variability when summarised as the generic frog or tadpole level. However, this variation is typical of that seen for CRwo-water values of other organisms3. Whilst progress is being made in developing alternative approaches which account for the main cause of variability14,15 currently we are reliant of the CR approach and it is the parameter used in all environmental assessment models16 and proposed by international bodies2,3.
Although previous studies have suggested that tadpoles may have higher radiocaesium transfer than adults8,9, this suggestion was based on limited data. Our comparison of CRwo-water values between adult frogs and tadpoles is, to our knowledge, the first conclusive demonstration that there is a significant difference in radiocaesium transfer for these two life-stages. Tadpoles are generally thought to be herbivorous or detritivorous17, whereas adult frogs are carnivorous (Table 1). Whilst we may expect radiocaesium to biomagnify up aquatic foodchains18,19 (in common with terrestrial foodchains20,21), however, tadpoles feed solely in the aquatic environment whereas frogs mainly feed in the terrestrial environment often consuming insects (Table 1). Furthermore, there is some evidence that some tadpoles, including those of the American bullfrog, are carnivorous17.
The higher CRwo-water value for Montane Brown frog compared to other species may be due to differences in habitat utilisation; Montane Brown frogs frequent wet forest areas whereas the terrestrial habitats used by the other species are predominantly paddy fields or the area close to waterbodies (Table 1). Watanabe et al.8 previously reported that frog species living in forests had higher radiocaesium activity concentrations than those living in paddy fields. Kajika frogs live in similar areas as Montane Brown frogs22; the single sample of this species reported here also had a comparatively high CRwo-water value (Table 2).
Whilst our data suggest potential differences in the transfer of radiocaesium to different frog species, we acknowledge that this is based on relatively few measurements (though a comparatively large number of individual frogs which were bulked to ensure that radiocesium activity concentrations could be determined). The potential differences in transfer to different frog species need to be further substantiated before species-specific values could be recommended for use in assessments. Currently, we suggest that generic amphibian and tadpole CRwo-water values are used for screening assessments undertaken to establish that there will be no potential impact of ionising radiation under conservative assumptions23.
Our comparison of CRwo-water values between lakes and rivers identified a significantly higher transfer of radiocaesium to tadpoles in rivers, but no difference in the transfer to adult frogs. The reasons for this are currently unclear, although we note that comparatively few tadpole observations were available for lakes. Whilst international data collations13 of wildlife CRwo-water allow data to be categorised on the basis of lakes or flowing waters (i.e. river and streams) there has been little analyses of CRwo-water values for the two categories (to our knowledge this has been limited to a consideration of fish14).
There is no information to suggest how environmental factors may influence the transfer of radiocaesium to frogs in aquatic ecosystems. However, the K status of a waterbody may be expected to impact on Cs transfer to amphibians, as has been demonstrated for fish24,25. Whilst we do not have K data for the water bodies from which amphibians were sampled, NIRS26 presents data (n = 442) for major rivers throughout Japan. The 5th and 95th percentiles of these data show K concentrations in water vary by an order of magnitude and could therefore, based upon data for fish, explain some of the variation in the dataset presented here.
The aim of the dose rate assessment was to consider the dose received by different life-stages and also consider the impact of life history (by considering two species) on the dose received; it was not intended as a definitive assessment of dose and risk to frogs in the study are. Dose rates for the Montane Brown frog and the Wrinkled frog (Table 3) were similar for the adult and tadpole life-stages. This is because external dose rate from soil or sediment is predicted to dominate the total dose (≥90%). The dose rate to frogspawn was estimated to be approximately one order of magnitude higher for Montane Brown frog than Wrinkled frog. External dose was also estimated to dominate the total 137Cs dose to R. arvalis in a Swedish wetland27. Frogspawn was assumed to be in the water column and to receive no exposure to contaminated sediment, hence differences in the CRwo-water values assumed for the two species impact on the total dose rate. If frogspawn had been assumed to be on the sediment surface then the total dose rate would be similar to the other life-stages (i.e. two - three orders of magnitude higher); this demonstrates the importance of the occupancy assumption for this assessment. In 2012, predicted dose rates to the adult frogs in the terrestrial and aquatic ecosystems are similar for Lake Akimoto. However, in 2016 dose rates in the aquatic ecosystem are higher than those in the terrestrial ecosystem due to the increased sediment 137Cs activity concentrations in 2016 compared to 2012 (Table 5).
For Montane Brown frogs the period from egg to tadpole is reported to be circa 14 d and the tadpole life-stage to last about 120 d28; comparative values for the Wrinkled frog are cited as 5 d and 1 year respectively29. Therefore, from Table 3 it can be seen that the greatest contribution to lifetime dose is predicted to occur during the adult life stage; a similar conclusion was made for estimated 137Cs doses to R. arvalis in a Swedish wetland27. Furthermore, whilst the other life-stages may be more radiosensitive than adults1, their estimated absorbed dose rates are similar to, or considerably lower than, those for the adults.
Compared to Lake Akimoto, the predicted dose to adults at Uda River is higher in the terrestrial than aquatic environment (Tables 3 and 4). This is because the soil radiocaesium activity concentrations at this site are higher than those for sediment (Table 5), whereas at Lake Akimoto sediment activity concentrations are similar to, or higher than, those in soil.
Based on dose rates presented in Tables 3 and 4, if a four-month hibernation is assumed then this period (when it is assumed to be in sediments or underground) would result in a higher contribution to the annual dose of adult frogs than the remainder of the year.
For Lake Akimoto in 2012, 137Cs activity concentrations in water, sediment and soil were higher than 134Cs. However, the dose assessment demonstrated that for adults and tadpoles of both species 134Cs contributed a greater component of the total dose rate (c. 60%) than 137Cs in that year. This is due to the higher external dose conversion coefficient (relating Bq kg−1 in sediment to absorbed dose rate in µGy h−1) for 134Cs compared to 137Cs (Brown et al., 2016). For frogspawn, 137Cs dominates the total dose rate as a consequence of the low contribution of the external dose, higher activity concentrations of 137Cs compared to 134Cs in the environment and the higher internal dose conversion coefficient of 137Cs compared to 134Cs. Due to the shorter physical half-life of 134Cs (2.06 y) compared to 137Cs (30 y), at Uda River in 2013 the contributions of the two radiocaesium isotopes were similar and 137Cs contributed the most to doses for all life-stages in 2015/2016 at both sites (c. 70% of the estimated total dose rate).
Dose rates estimated for frogs at the two sites are considerably below the lower end of the Derived Consideration Reference Level (DCRL; ‘an order of magnitude dose rate band in which radiation induced effects might be expected to occur’) for amphibians of c. 40 µGy h−1 as suggested by ICRP1. Estimated dose rates are also at least an approximate order of magnitude below the generic 10 µGy h−1 no effects dose rate suggested by Andersson et al.30.
We acknowledge that there are some limitations to the dose assessment presented here. Whilst we used species-specific CRwo-water values for the adult and frogspawn life-stages, a single value was used for tadpole for both species. However, the impact of this is likely to be minimal given the dominance of external exposure which contributed ≥90% of the total dose under the scenarios considered. Consequently, a change in the tadpole CRwo-water by an order of magnitude (the approximate difference between the two species in CRwo-water values for the adult life-stage; Table 2) is not going to effect the overall dose estimate greatly. For the assessment of frogs in the terrestrial ecosystem, we assumed the same organism activity concentration as predicted for the aquatic system given we lacked specific data to estimate CRwo-soil values. Comparing the assumed wholebody activity concentrations to soil activity concentrations, predicted CRwo-soil values in the range E-3 to E-1 can be estimated. Brown et al.4 report a CRwo-soil value for amphibians of (4.6 ± 8.3)×10−1 (arithmetic mean value ± standard deviation; n = 139) from the latest version of the international Wildlife Transfer Database (WTD)13 which is a large compilation of CRwo values initially established to help prepare IAEA and ICRP publications2,3. Whilst the values estimated here for Fukushima frogs look reasonable compared to this mean value they are at the low end of the observed data in the WTD (the range in WTD entries, which are a mixture of means and individual data entries, is E-2 to E + 0). However, CRwo-soil values for a Ranidae species in Spain of E-3 to E-2 have recently been reported31,32. As noted in Materials and methods we used the ERICA Tool default amphibian geometry rather than creating specific geometries for the adult frog species considered as for the size range considered the actual geometry used has minimal influence on the dose rate estimated33,34.
In summary, the data presented here suggest that the transfer of radiocaesium to tadpoles is higher than that to adult frogs. This finding will be a useful input into the developing ICRP environmental protection framework as this considers various life-stages for the Reference Frog1,2, but lacks data to quantify transfer. Our CRwo-water values represent some of the first reported; currently the wildlife models and data compilations do not contain CRwo-water values for amphibians2,3,4,13. A CRwo-water value of around 250, based on two observations, was reported in IAEA9. For seven out of the eight species considered here, the geometric mean CRwo-water values were higher than this; the maximum values calculated here were about two orders of magnitude higher than that in IAEA9 (Table 2). Stark et al.27 reported CRwo-water values for Rana arvalis in a Swedish wetland of 2500–19100 that are more similar to those we have calculated here. Estimated absorbed doses rates for tadpoles and the adult lifestage were dominated by external exposure. The adult life-stage was estimated to receive the greatest contribution of total lifetime dose. Estimated doses rates were below suggested effects benchmark dose rates for the two sites assessed. However, on the basis of data presented by Matsushima et al.5, dose rates to adult frogs in the most contaminated parts of Fukushima Prefecture may have exceeded the 10 µGy h−1 no-effect dose rate30 and at least approached the lower-limit of the ICRP’s DCRL1. Therefore, the Fukushima impacted areas present a natural laboratory for studying ionising radiation effects on frog populations (and indeed other wildlife).
Materials and Method
We have extracted data from MOE (Ministry of the Environment)10 for the eight freshwater systems for samples collected between June 2012 and June 2016; the data included radiocaesium activity concentrations in water, sediment and biota. Corresponding soil data are also available from the terrestrial ecosystem adjacent to each site10; for rivers three to five soil cores were collected on each sampling occasion on either side of the river bank whilst for lakes five soil cores were collected around the sampling location35. The Ministry of the Environment use standard methods established by the Japanese Ministry of Education, Culture, Sports, Science and Technology (MEXT) (see MOE36). In brief, for sediments three replicate 5 cm cores were collected per site on each sampling occasion, these were dried and then homogenized and bulked into one sample prior to analyses. An approximately 20 l surface water sample was collected by bucket, if required this was filtered (1 µm) prior to concentration (by ammonium molybdophosphate coprecipitation37 or evaporation) for subsequent analyses. Biota samples were washed with de-ionised water, the gastrointestinal tract removed and homogenised for subsequent analyses. Due to the low mass of individual tadpoles and of many adult frogs, samples collected were often bulked for a given sampling site prior to analysis (see Table 2 and Supplementary Information). Eleven samples of adult frogs were a composite of two to five species. Three samples had no species information recorded. A total of 513 adult frogs and 2540 tadpoles were analysed in 62 and 59 composite samples respectively.
Gamma-emitting radionuclides were determined in all sample types using Ge-detectors.
Calculation CRwo-water values
The transfer parameter values presented in this paper are concentration ratios (CRwo-water), which relate the whole-organism activity concentration (Bq kg−1 fresh mass) to the activity concentration in water (Bq l−1). To calculate CRwo-water values, water data collected at the same sampling site as frog samples were used. Water data from the collection date closest to the biota sampling date were used to calculate CRwo-water values; if the biota sampling was between the water sampling dates, then the average water activity concentration was calculated. In all cases, the difference in sampling date between water and biota samples used to calculate a CRwo-water value was less than 1 month.
Caesium-137 and 134Cs activity concentration data were available for both adult frogs and tadpoles. However, 134Cs was undetectable in many of the water samples so only the 137Cs data have been used for calculating radiocaesium transfer parameter values. For 137Cs one measurement was under the detection limit for both frog and tadpole (both samples from Inawashiro Lake); the detection limit value was used for the calculation of CRwo-water.
Estimation of dose rates
Dose rates have been estimated for the Wrinkled frog and Montane Brown frog given they have somewhat different life-histories and are examples of a species with a comparatively low and high CRwo-water values respectively. Assessments have been conducted for Akimoto Lake and Uda River for both the first and last years that water activity concentration data were available. The CRwo-water for each species (Table 2) was used to estimate wholebody 134Cs and 137Cs activity concentrations; the overall tadpole CRwo-water value was used for both species given the lack of relevant species-specific data. No data were available for the transfer of Cs to frogspawn (eggs) for the sites considered here. Therefore, to allow a dose estimate for all life-stages we have assumed the CRwo-water for frogspawn was 0.09 times the CRwo-water for the adult of each species based upon data from the Chernobyl Exclusion Zone38 and a forest site in northeast England (Beresford & Barnett CEH pers comm.). Given the lack of any specific data, the wholebody radiocaesium activity concentrations for adult frogs were taken to be the same in the terrestrial environment as that predicted in the aquatic environment.
The ERICA Tool4 was used to estimate absorbed dose rates. When not hibernating it was assumed that the adult frog was on the soil surface in the terrestrial environment and at the sediment-water interface in the freshwater environment; tadpoles were modelled at the sediment-water interface and frogspawn in the water column. It was assumed that the Montane Brown frog hibernates in either soil or (aquatic) sediment whereas the Wrinkled frog hibernates in sediment only39,40. For adults of both species the default amphibian geometry from the ERICA Tool was used (this is the same as the Reference Frog geometry proposed in ICRP1); it has been demonstrated33,34 that there is minimal influence of size on dose within the size ranges of the frogs sampled here and the ERCIA default and hence there is no need to create organism in the Tool for each species. For frogspawn and tadpoles, new geometries were created in the ERICA Tool. The geometry presented for frogspawn mass in ICRP1 was used for this life-stage, and the tadpole geometry was based on information for the two species being assessed (assumed mass = 3 g, length = 6 cm, width and height = 1 cm).
Statistical analysis
All statistical analyses were performed using MINITAB version 17; the tests used are quoted in the text. MINITAB does not quote levels of significance below 0.0001, hence any p-values below this are quoted as p < 0.0001. By necessity statistical analyses had to be based upon the number of composite samples analysed rather than the number of individual animals analysed.
Data statement
All individual data (activity concentrations in different sample types, as extracted from MOE (http://www.env.go.jp/en/water/rmms/result_ao17-part.html)10, and estimated CRwo-water values) are available as supplementary information.
References
International Commission on Radiological Protection (ICRP). Environmental Protection: the Concept and Use of Reference Animals and Plants. ICRP Publication, 108, Ann. ICRP 38 (Elsevier, 2008).
International Commission on Radiological Protection (ICRP). Environmental Protection: Transfer Parameters for Reference Animals and Plants. ICRP Publication 114, Ann. ICRP 39 (Elsevier, 2009).
International Atomic Energy Agency (IAEA). Handbook of Parameter Values for the Prediction of Radionuclide Transfer to Wildlife. Technical Report Series No. 479 (IAEA, 2014).
Brown, J. E. et al. A new version of the ERICA Tool to facilitate impact assessments of radioactivity on wild plants and animals. J. Environ. Radioact. 153, 141–148, https://doi.org/10.1016/j.jenvrad.2015.12.011 (2016).
Matsushima, N., Ihara, S., Takase, M. & Horiguchi, T. Assessment of radiocesium contamination in frogs 18 months after the Fukushima Daiich nuclear disaster. Sci. Rep. 5, 9712, https://doi.org/10.1038/srep09712 (2015).
Sakai, M., Gomi, T., Nunokawa, M., Wakahara, T. & Onda, Y. Soil removal as a decontamination practice and radiocesium accumulation in tadpoles in rice paddies at Fukushima. Environ. Pollut. 187, 112–115 (2014).
Takahara, T. et al. Radiocesium accumulation in the anuran frog, Rana tagoi tagoi, in forest ecosystems after the Fukushima Nuclear Power Plant. Environ. Pollut. 199, 89–94 (2015).
Watanabe, I. et al. Radioactive Cs distribution of paddy field ecosystems in eastern part of Nihonmatsu, Fukushima, Japan. Nihon Hoshanou Jyosen Gakkaishi 3, 207–214 (2015).
International Atomic Energy Agency (IAEA). Handbook of Parameter Values for the Prediction of Radionuclide Transfer in Terrestrial and Freshwater Environments. Technical Report Series No. 472 (IAEA, 2010).
Ministry of the Environment (MOE). Radioactive Material Monitoring in the Water Environment in and around Fukushima Prefecture (Aquatic Organisms). http://www.env.go.jp/en/water/rmms/result_ao17-part.html (last accessed 17/07/2017), (2017).
Wood, M. D., Beresford, N. A., Howard, B. J. & Copplestone, D. Evaluating summarised radionuclide concentration ratio datasets for wildlife. J. Environ. Radioact. 126, 314–325, https://doi.org/10.1016/j.jenvrad.2013.07.022 (2013).
Beresford, N. A. et al. Derivation of transfer parameters for use within the ERICA Tool and the default concentration ratios for terrestrial biota. J. Environ. Radioact. 99, 1393–1407, https://doi.org/10.1016/j.jenvrad.2008.01.020 (2008).
Copplestone, D., Beresford, N. A., Brown, J. & Yankovich, T. An international database of radionuclide Concentration Ratios for wildlife: development and uses. J. Environ. Radioact. 126, 288–298, https://doi.org/10.1016/j.jenvrad.2013.05.007 (2013).
Beresford, N. A. et al. A new approach to predicting environmental transfer of radionuclides to wildlife taking account of inter-site variation using Residual Maximum Likelihood mixed-model regression: a demonstration for freshwater fish and caesium. Sci. Total Environ. 463-464, 284–292, https://doi.org/10.1016/j.scitotenv.2013.06.013 (2013).
Beresford, N. A. et al. Making the most of what we have: application of extrapolation approaches in radioecological wildlife transfer models. J. Environ. Radioact. 151, 373–386, https://doi.org/10.1016/j.jenvrad.2015.03.022 (2016).
Beresford, N. A. et al. Inter-comparison of models to estimate radionuclide activity concentrations in non-human biota. Radiat. Environ. Biophys. 47, 491–514, https://doi.org/10.1007/s00411-008-0186-82008 (2016).
Altig, R., Whiles, M. R. & Taylor, C. L. What do tadpoles really eat? Assessing the trophic status of an understudied and imperiled group of consumers in freshwater habitats. Freshwater Biology 52, 386–395, https://doi.org/10.1111/j.1365-2427.2006.01694.x (2007).
Rowan, D. J. & Rasmussen, J. B. Bioaccumulation of radiocaesium by fish: the influence of physicochemical factors and trophic structure. Canadian Journal of Fisheries and Aquatic Science 51, 2388–2410 (1994).
Smith, J. T., Kudelsky, A. V., Ryabov, I. N., Hadderingh, R. H. & Bulgakov, A. A. Application of potassium chloride to a Chernobyl – contaminated lake: modelling the dynamics of radiocaesium in an aquatic ecosystem and decontamination of fish. Sci Total Environ. 305, 217–227 (2003).
Pendleton, R. C. et al. Trophic level effect on the accumulation of caesium-137 in cougars feeding on mule deer. Nature 204, 707–709 (1964).
Lowe, V. P. W. & Horrill, A. D. Caesium concentration factors in wild herbivores and the fox (Vulpes vulpes L). Environ. Pollut. 70, 93–107 (1991).
Kaneko, Y. & Matsui, M. Buergeria buergeri. The IUCN Red List of Threatened Species 2017-1. https://doi.org/10.2305/IUCN.UK.2004.RLTS.T58782A11827604.en (2004). (last accessed on 01/08/2017).
Beresford, N. A. et al. Assessment of risk to wildlife from ionising radiation: can initial screening tiers be used with a high level of confidence? J. Radiol. Prot. 30, 265–284, https://doi.org/10.1088/0952-4746/30/2/S04 (2010).
John, E. P. III et al. Development and evaluation of a regression-based model to predict cesium concentration ratios for freshwater fish. J. Environ. Radioact. 34, 89–98 (2014).
Smith, J. T. et al. Radiocesium concentration factors of Chernobyl contaminated fish: a study of the influence of potassium and a “blind” testing of a previously developed model. J. Environ. Radioact. 48, 359–369 (2000).
National Institute of Radiological Science. Elemental concentrations in Japanese Rivers. NIRS-M200. http://www.nirs. qst.go.jp/publication/irregular/03.html (NIRS, 2007).
Stark, K. et al. Estimation of radiation doses from 137Cs to frogs in a wetland ecosystem. J. Environ. Radioact. 75, 1–14 (2004).
Kikuchi, Y. The Montane brown frog (Rana ornativentris). http://lifeinkochi.net/2011/08/the-montane-brown-frog-rana-ornativentris/ (last accessed on 17/07/2017) (2011).
Goris, R. C. & Maeda, N. Guide to the amphibians and reptiles of Japan. (Krieger Publishing Company, 2005).
Andersson, P. et al. Protection of the environment from ionising radiation in a regulatory context (PROTECT): proposed numerical benchmark values. J. Environ. Radioact. 100, 1100–1108, https://doi.org/10.1016/j.jenvrad.2009.05.010 (2009).
Guillén, J. et al. Transfer parameters for ICRP’s Reference Animals and Plants in a terrestrial Mediterranean ecosystem. J. Environ. Radioact. (in-press).
Guillén, J. et al. Elemental concentrations in representative species of the ICRP’s Reference Animals and Plants and associated soils in terrestrial Mediterranean ecosystems in Spain. NERC Environmental Information Data Centre; https://doi.org/10.5285/a1ab8c79-3426-43a4-ab42-6d1b218d1cc6 (2017).
Vives i Batlle, J. et al. Inter-comparison of absorbed dose rates for non-human biota. Radiat. Environ. Biophys. 46, 349–373, https://doi.org/10.1007/s00411-007-0124-1 (2007).
Vives i Batlle, J. et al. The estimation of absorbed dose rates for non-human biota: an extended intercomparison. Radiat. Environ. Biophys. 50, 231–251, https://doi.org/10.1007/s00411-010-0346-5 (2011).
Ministry of the Environment (MOE). FY2015 Radioactive material monitoring in the water environment in and around Fukushima Prefecture. (http://www.env.go.jp/en/water/rmms/attach/result_pw-fy2015/1-1.pdf (last accessed 01/11/2017) (2015).
Ministry of the Environment (MOE). Review of radioactive material survel methods etc. in water environment in FY 2012 (Work report). http://www.env.go.jp/jishin/monitoring/report_method-w_1303.pdf (in Japanese). (last accessed 25/07/2017). (2013).
Matsuda, H. T. & Abrão, A. New technique for synthesizing AMP: precipitation inside an ion-exchange resin and its application to separation of cesium from fission-products and to a 137mBa generator. IPEN-Pub-13. (Instituto de Pesquisas Energéticas e Nucleares, Available from: http://www.iaea.org/inis/collection/NCLCollectionStore/_Public/13/647/13647299.pdf (1980).
Beresford, N. A. et al. Radionuclide transfer to wildlife at a ‘Reference Site’ in the Chernobyl Exclusion Zone and resultant radiation exposures. J. Environ. Radioact. https://doi.org/10.1016/j.jenvrad.2018.02.007 (in-press, on-line).
Takao 599 Museum. Rana ornativentris (Montane Brown Frog). http://www.takao599museum.jp/treasures/others/%E3%82%A2%E3%82%AB%E3%82%AC%E3%82%A8%E3%83%AB%E7%A7%91-others/437/?lang=en (2014). (last accessed 18/07/2017).
CABI. Invasive species compendium. Dataseet: Rugosa rugosa (wrinkled frog). http://www.cabi.org/isc/datasheet/121512 (last accessed 18/07/2017) (2017)
Kusano, Y. Age and lifespan of amphibian. http://salamander.la.coocan.jp/salamander/shiryou/jumyou2.html (last accessed 25/07/2017) (2000).
Otagawa R Office. Bullfrog Rana catesbeiana. http://www.cgr.mlit.go.jp/ootagawa/Bio/amphi/index424.htm (last accessed 25/07/2017).
Ministry of Land, Infrastructure, Transport and Tourism Kyusyu Regional Development Bureau, Chikugo River Office. http://www.qsr.mlit.go.jp/chikugo/archives/ikimono/chikugo_kawa.html (last accessed 25/07/2017).
Acknowledgements
This collaboration was carried out under the framework of the IAEA’s MODARIA II programme. K. Tagami and S. Uchida were partially supported by the Agency for Natural Resources and Energy, the Ministry of Economy, Trade and Industry (METI), Japan. The inputs of N.A. Beresford and M.D. Wood were supported by the TREE (http://www.ceh.ac.uk/tree) project funded by the NERC, Environment Agency and Radioactive Waste Management Ltd. under the RATE programme.
Author information
Authors and Affiliations
Contributions
K.T. with assistance from S.U., collated all the base data, calculated CRwo-water values and drafted the initial manuscript. N.A.B. and M.D.W. conducted the dose assessments, statistical analyses, added relevant text to the manuscript and revised the paper in light of reviewer comments.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tagami, K., Uchida, S., Wood, M.D. et al. Radiocaesium transfer and radiation exposure of frogs in Fukushima Prefecture. Sci Rep 8, 10662 (2018). https://doi.org/10.1038/s41598-018-28866-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-28866-0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.