Abstract
Coral reefs harbor diverse assemblages of organisms yet the majority of this diversity is hidden within the three dimensional structure of the reef and neglected using standard visual surveys. This study uses Autonomous Reef Monitoring Structures (ARMS) and amplicon sequencing methodologies, targeting mitochondrial cytochrome oxidase I and 18S rRNA genes, to investigate changes in the cryptic reef biodiversity. ARMS, deployed at 11 sites across a near- to off-shore gradient in the Red Sea were dominated by Porifera (sessile fraction), Arthropoda and Annelida (mobile fractions). The two primer sets detected different taxa lists, but patterns in community composition and structure were similar. While the microhabitat of the ARMS deployment affected the community structure, a clear cross-shelf gradient was observed for all fractions investigated. The partitioning of beta-diversity revealed that replacement (i.e. the substitution of species) made the highest contribution with richness playing a smaller role. Hence, different reef habitats across the shelf are relevant to regional diversity, as they harbor different communities, a result with clear implications for the design of Marine Protected Areas. ARMS can be vital tools to assess biodiversity patterns in the generally neglected but species-rich cryptic benthos, providing invaluable information for the management and conservation of hard-bottomed habitats over local and global scales.
Similar content being viewed by others

Introduction
Understanding how diversity is partitioned along natural and anthropogenic gradients within ecosystems is a central topic in ecology and a key goal for conservation1,2. Cross-shelf habitats present some of the sharpest gradients in physical and chemical conditions (e.g. salinity, temperature and nutrients) in marine systems3,4,5. Near-shore areas are under the influence of terrestrial systems while outer regions are more exposed to oceanic currents and wave action3,4,5,6. These environmental differences, in turn, affect the distribution of marine organisms as a function of taxon-specific physiological requirements and life history traits7. For example, Ellis and co-authors8 indicated that nutrient availability across the shelf gradient in the southern Red Sea was associated with patterns of macro-algal cover (higher in nutrient enriched near-shore sites) and scleractinian coral diversity (higher at off-shore sites with lower levels of nutrients). Further in the central Red Sea distinct gradients across the shelf have been detected in physio-chemical parameters (e.g. chlorophyll and sedimentation)9 as well as differences in the coral community with Acriporoa and Pocillopora being comparatively rare in near-shore reefs10.
On coral reefs, various studies have reported cross-shelf differences in population size, biomass, recruitment rates and species composition among fish11,12,13, corals10,14, sponges15,16, molluscs17, crustaceans18 and siphonophores19. However, studies assessing multiple taxonomic groups have received less attention and have predominantly focused on more conspicuous taxa5,7,8,20,21,22,23,24. As a result, there is still a poor understanding of community-wide changes both in terms of the composition and structure across inshore-offshore gradients, especially in highly diverse tropical ecosystems such as coral reefs. This is particularly true for the small and highly diverse cryptic invertebrates that exist within the reef matrix7,25,26,27,28,29. This neglect is due to the fact that many of these species are either undescribed or very difficult to identify.
Despite being overlooked in most of the coral reef research, benthic cryptic fauna include a variety of sessile and mobile taxa30,31,32,33,34,35 inhabiting hidden spaces that can account for up to two thirds of the reef’s volume36. Little is understood about the life histories and habitat preferences of these organisms beyond size-related generalizations37. Some studies have investigated the community composition patterns at very small spatial scales and found that crevices within reefs are often dominated by coralline algae near sunlit entrances and by filter feeders in posterior sections38,39. Species inhabiting these cryptic spaces, especially Porifera, act as a sink for dissolved organic matter, thus preventing energy and nutrient losses into the open ocean40. Therefore, a better understanding of the distributional patterns of the crypto-benthic fauna may reveal insights into biogeochemical cycles within the reef system.
In recent decades, standardized sampling units have been proposed as a means to quantify the diversity and composition of benthic assemblages in a consistent manner across space and time. In this study, we use Autonomous Reef Monitoring Structures (ARMS) which are designed to partially mimic the 3D structure of a reef41,42. The structures enable the study of both the sessile components growing on the plates as well as the mobile organisms that settle on or move into the spaces. The communities collected in ARMS are typically analyzed using some form of genetic barcoding (of individual organisms) (e.g.)43 or high-throughput amplicon sequencing (i.e. metabarcoding of communities) (e.g.)42. Studies utilizing ARMS have revealed much about the hidden diversity of the Indo-Pacific and Caribbean43, as well as using high throughput amplicon sequencing (i.e. metabarcoding) to assess the sessile and mobile fractions on the US Atlantic coast42 and the Red Sea44,45.
Cryptic organisms include a diverse selection of ecologically important groups such as suspension feeders46,47, predators48, herbivores49 and detritivores50. Because of their smaller sizes, they also have larger population sizes and faster generation times51. Their response to environmental changes may therefore differ from those of the better-studied fish and corals. The cryptic fauna represents a large proportion of the reef diversity, and thus to best conserve biodiversity of the reefs across spatial scales, an understanding of how the cryptic fauna responds to environmental changes is vital33. To achieve this, an assessment of not only the local species assemblages (alpha diversity) but also the inter-site differences (beta diversity) is required2. While the total number of species is an important element in conservation, the change in species across space strongly influences the optimal spatial arrangement of conservation areas26.
Besides a mere quantification of the differentiation of biological communities, beta diversity can be partitioned into different components allowing for a more comprehensive understanding of the mechanisms driving diversity patterns52. For example, Legendre52 partitioned beta diversity into species replacement and richness difference. Species replacement refers to the substitution of species along an environmental/temporal gradient due to environmental filtering, competition, and historical events53. On the other hand, species richness refers to the number of species contained within each community, which may reflect the diversity of niches available at different locations52. A difference in the relative contribution of these two components to total beta diversity suggests that the structure of species assemblages is being controlled by different mechanisms54. Understanding the contribution of these partitions is vital to linking dissimilarities amongst communities to ecological processes55 and subsequently to informing conservation and management decisions54.
Here, we assess the composition and structure of the cryptobenthic communities that colonized ARMS at eight reefs (11 sites) in the central Red Sea region, an area of high diversity and endemism56. We use metabarcoding sequencing of two gene regions that differ in their level of taxonomic coverage and resolution to better reflect the diversity of life forms on the ARMS57 as well as standard barcoding of the largest organisms. For metabarcoding, we used highly versatile PCR primers available for the slowly evolving 18S rRNA to target the whole eukaryotic domain and general primers optimized for metazoans for the hypervariable mitochondrial Cytochrome C Oxidase Subunit I (mtCOI) with general primers optimized for metazoans.
Based on trends in coral reef fauna observed within the region8 and globally10,11,14,15 we hypothesized that there would be substantial changes in the community structure and composition across the shelf gradient. Also we expected the near-shore reefs to be more depauperate in nature (i.e. lower species richness) due to a stronger influence from human activities and higher levels of sedimentation. We also hypothesized that the different faunal components would respond similarly to changes in environmental conditions because of the sharp cross-shelf environmental gradients. Finally, we also expected patterns of alpha and beta diversity obtained from the two independent target genes to be correlated despite targeting different fractions of the communities.
Overall, our analysis provides the first comprehensive assessment of the biodiversity changes across shelf gradients in cryptic reef organisms by combining information on alpha and beta diversity (partitioned into replacement and richness). Our analysis illustrates how standardized sampling approach combined with molecular techniques (i.e. PCR-based analysis of two marker genes) can provide a basis for the comparative assessment of cryptic benthic organisms in hard bottomed substrates and help understanding how anthropogenic pressures affect this functionally important component of benthic communities.
Methods
Environmental data
Satellite data were downloaded from the NASA Oceancolor website (https://oceancolor.gsfc.nasa.gov/ downloaded 14th February 2017). Monthly averages of chlorophyll (MODIS A) and sea surface temperature (SST) (MODIS A) at a 4 km resolution were retrieved. The data value for each reef site was based on the value associated with the nearest appropriate grid point. Due to the spatial resolution of the data, some sites were assigned to the same grid point and thus had the same environmental data.
Sampling strategy and laboratory processing
Triplicate ARMS were deployed on 11 reef sites near Thuwal (Lat: 22.30N; Lon 39.12E) in the central Red Sea for two years at approximately 10 m depth (Fig. 1 and Supplementary Table S1). The reefs were characterized as either near-shore (n = 5), mid-shelf (n = 4) or off-shore (n = 2), according to the distance to the shore (near-shore, <5 km; mid-shelf, 5–25 km; off-shore, >25 km) (Fig. 2). ARMS were deployed in two separate batches. A first deployment of four sets of three ARMS was deployed in February 2013. They were positioned on hard coral framework (later referred to as reef). A second deployment of 7 sets of three ARMS was conducted in May/June 2013. They were positioned on loose coral rubble (later referred to as rubble). The times of deployment and retrieval for each, as well as the depth for each reef site, are detailed in Supplementary Table S1. Overall, deployment, retrieval, and laboratory processing of the units, as well as the extraction of the DNA, followed the procedures previously described42. ARMS were retrieved so as to retain all organisms on or inside the units. These organisms were subsequently sorted into various size fractions for analysis. For the largest samples (>2000 μm) DNA was extracted for individual barcoding using the DNeasy kit (Qiagen) instead of the AutoGeneprep 965 (AutoGen). The smaller fractions (Sessile, 106–500 μm and 500–2000 μm) were processed for metabarcoding as described by42.
Chlorophyll a concentrations (mean of composite monthly averages over the time frame of the study) for the study regions derived from NASA’s Oceancolor website (https://oceancolor.gsfc.nasa.gov/) derived from the MODIS A satellites at a 4 km resolution.
DNA amplification and sequencing
Two primer sets were used to amplify eukaryotic sequences from the various fractions of the ARMS units (see Supplementary Table S2 for sequences). A 313 bp fragment of the mitochondrial COI gene was targeted using versatile PCR primers58 and PCR conditions previously described42. The v4 region of the 18S rRNA gene was amplified using primers designed by59 and using the PCR conditions described by45. Triplicate PCR products were merged, cleaned of primer dimers using streptavidin magnetic beads and normalized, before a second round of PCR amplification was undertaken using the Illumina 16S metagenomic sequencing library preparation protocol. An Illumina MiSeq sequencing platform (v3 chemistry) was used with the sequences (2 × 300 bp) being generated at the King Abdullah University of Science and Technology (KAUST) Bioscience Core Laboratory (BCL). Raw reads were deposited at Figshare with the following doi’s: https://doi.org/10.6084/m9.figshare.5559106.v1, https://doi.org/10.6084/m9.figshare.5558113, https://doi.org/10.6084/m9.figshare.5549365, https://doi.org/10.6084/m9.figshare.5552782, https://doi.org/10.6084/m9.figshare.5552932.
Extracted DNA for the barcoding samples was amplified using the mitochondrial COI gene primers jgLCO and jgHCO (Supplementary Table S2) and the PCR mix as described by60 with the addition of 0.2 μL Bovine Serum Albumin (BSA) (20 mg/mL) and using the PCR thermal cycling conditions detailed previously42. For specimens for which amplification could not be obtained using the jgLCO/jgHCO primer combination, PCRs were repeated using the dgLCO/dgHCO primer set with the conditions as described by61. Primers were cleaned and sequenced on either a Sanger ABI 3730 capillary platform (KAUST BCL) or a 3730xl DNA Analyzer (Applied Biosystems) at the Smithsonian’s National Museum of Natural History Laboratory of Analytical Biology. Sequences were deposited in the NCBI database with the accession numbers: MH338260-MH339541.
Bioinformatics
Metabarcoding
We followed the bioinformatics protocol detailed by62. Briefly, raw reads from the sequencing machine were unzipped and denoised using the program BFC63. Contigs were formed between the forward and reverse reads using usearch (v8.1)64 with fastq_merge_maxee set to 1.0. Fastq files were converted to fasta files using mothur65. These were subsequently quality filtered and the forward and reverse primers removed using trim.seqs in mothur (maxambig = 0; maxhomop = 8; pdiffs = 0). Dereplication of the sequences was undertaken with the trie function in QIIME66. Sequences of the 18S gene were aligned in mothur while COI sequences were aligned using MACSE to remove reads with stop codons58. Reads were then pre-clustered (using the pre.cluster script with diffs = 3) and singletons, which are likely to be sequencing errors, were removed (split.abund with a cut off of 1). The remaining sequences were checked for chimeras (reference based using usearch) and clustered using CROP67 (COI: −l 3 and −u 4; 18S: using −s).
Reference sequences of the OTUs were taxonomically classified to the species level using Blast68 at the 97% similarity level against the BOLD69 and Midori70 databases for the COI gene and the PR271 and Silva 12872 databases for 18S rDNA. Those reference sequences which did meet the designated cutoff were subsequently classified using the Statistical Assignment Program73 or rdp74 for the COI gene and 18S rDNA datasets, respectively.
Barcoding
Forward and reverse reads were combined into contigs using Geneious (Biomatters) with sequences trimmed if there was a greater than 5% chance of a base error. Sequences were also discarded if a stop codon was present in the translated sequence or the sequence had three ambiguous (N) bases resulting in the wrong amino acid translation. Sequences were clustered into OTUs using CROP (−l 3 and −u 4).
Diversity statistics
Alpha diversity statistics were calculated using the R (R development core team) package phyloseq75 and species accumulation curves were produced with the specaccum function in vegan76. Samples were sub-sampled to an even depth for comparisons (34,000 and 19,750 reads per sample for COI and 18S datasets, respectively). Alpha diversity statistics of the metabarcoding fraction were tested for significant differences (Kruskal-Wallis) in terms of two factors: shelf (three levels: near-, mid-shelf and off-shore) and fractions (three levels: sessile, 106–500 μm and 500–2000 μm). For the shelf factor analyses, only ARMS which contained all metabarcoding fractions were used (32 ARMS for COI and 26 ARMS for the 18S rDNA datasets. Differences in number of ARMS analyzed was due to some fractions not meeting thresholds for subsampling). Tests for multiple comparisons were undertaken with the Dunn test using the method proposed by77 implemented in the package FSA78. Due to the unbalanced nature of the sampling design, comparisons of unique OTUs were undertaken by selecting combinations of two reefs from each shelf position (all possible combinations were tested) and then calculating the average number of unique OTUs. The taxonomic composition of each sample was summarized in phyloseq with taxa merged at the phylum level and filtered so those groups representing on average less than 0.5% of the community were removed. Composition plots were produced using the package ggplot79. Similarly, PCoA plots were produced using ggplot with the ordination function of phyloseq on square root transformed data for both Jaccard and Bray Curtis dissimlarity matrices. Differences in the Jaccard and Bray Curtis values within and between reefs were tested using Kruskal Wallis. Using the PRIMER v6 package80 with the PERMANOVA + add-on81 a three-way PERMANOVA was undertaken for the metabarcoding fraction. This assessed the significance of the factors “Shelf Position” (orthogonal three levels, near-shore, mid-shelf and off-shore), “Fraction” (orthogonal three levels, Sessile, 106–500 μm and 500–2000 μm) and “Placement” (orthogonal two levels, Reef and Rubble). The statistical significance was tested using 9999 permutations of the residuals with a significance level of α = 0.05 under a reduced model. Significant effects were further investigated through a series of pairwise comparisons. The fractionated beta diversity, using the Jaccard measure, was calculated using the package BAT82 within the R framework. Total beta diversity (βtotal) was partitioned into its two components, richness (βrich) and replacement (βrepl), as described by83. The decomposition of the Jaccard dissimilarity was performed for the combined ARMS (all metabarcoded fractions combined) and each fauna component separately (Sessile, 106–500 μm, 500–2000 μm and >2000 μm). Triangle plots were used to provide a two-dimensional illustration of the relationships among the two components of beta-diversity in analysis (replacement, richness difference) and the Jaccard similarity index84. Each vertex of the equilateral triangle corresponds to one of the coefficients and the sum of them equals 1. Each dot represents a pair of samples and its position is proportional to the respective coefficient values84. The density of the dots illustrates the structure of the data in terms of faunal similarity, replacement and richness difference. The higher the values associated with each of the vertices, the more relevant the corresponding coefficient is. Plots were created for: (1) between all samples (all pairwise comparisons possible); (2) between samples belonging to the same sites (within-site variation); (3) between samples belonging to different sites (among-site variation); (4) between samples belonging to the same shelf position (within-shelf variation); and (5) between samples belonging to different shelf positions (among-shelf variation). Local Contributions to Beta Diversity (LCBD) as described by85, i.e., the degree of uniqueness of the sites in terms of species richness, was calculated in the adespatial package in R86. Total diversity was calculated on a Hellinger transformed community matrix. Beta diversity was partitioned computing the sums of squares of a sampling unit as a proportion of the total diversity85. Linear distances were calculated between pairs of reefs and plotted against similarity (defined as 1 – Jaccard/Bray Curtis dissimilarity).
To assess the correlation in dissimilarity patterns between the different components of the fauna (sessile and mobile fractions of the metabarcoding), comparative (Mantel-type) tests were produced based on the Jaccard dissimilarity matrices using the vegan package in R. Similar tests were undertaken to assess the patterns produced in the metabarcoded datasets using the two different primer sets. To assess the differences in the number of OTUs per taxon both primers sets were subsampled to 19750 reads for the purpose of the comparison.
Data accessibility
Sequences from the barcoding are deposited in the NCBI database under the following accession: MH338260-MH339541. The raw sequences from the metabarcoding are deposited in Figshare at the following: https://doi.org/10.6084/m9.figshare.5559106.v1, https://doi.org/10.6084/m9.figshare.5558113, https://doi.org/10.6084/m9.figshare.5549365, https://doi.org/10.6084/m9.figshare.5552782, https://doi.org/10.6084/m9.figshare.5552932.
Results
Environmental variability
SST varied between 28 °C (January and March) and 34 °C (August) with no differences observed among reefs (Supplementary Figure S1 and Supplementary Table S3).
Chlorophyll a data showed a more variable pattern based on the reef category. In general, the lowest values were observed for the two off-shore reefs (Abu Madafi and Shib Nazar). The near-shore reef East Fsar had the highest values throughout the two-year period. The two other near-shore reefs, Abu Shosha and KAEC, had low values of chlorophyll a (the data for KAEC was, however, sparse), while the mid-shelf reefs showed intermediate concentrations of chlorophyll a (Fig. 1 and Supplementary Table S3).
Biodiversity patterns
Alpha-diversity
After sub-sampling at an even depth (34,000 and 19,750 reads per sample for COI and 18S datasets, respectively), a total of 5420 OTUs and 3830 OTUs were obtained with the COI and 18S primers, respectively. Metabarcoding of the combined biological fractions of ARMS showed an average of 660.46 ± 151.50 OTUs for COI and 750.03 ± 106.97 OTUs for 18S (Supplementary Table S4).
Combining the whole dataset, a total of 1023 OTUs (26.7%) were shared amongst the different size fractions in the 18S dataset, whereas 872 OTUs (16.0%) were shared in the COI dataset. Statistically significant differences were observed in alpha-diversity among the size fractions (chi-squared = 45.358, P < 0.001 for 18S; chi-squared = 44.978, p < 0.001 for COI). For both primer sets, the 106–500 μm size fraction was the most diverse (18S: 522.18 ± 127.82; COI: 456.46 ± 113.03) and had the highest number of unique OTUs (Table 1). This fraction also made the largest contribution to total biodiversity (Fig. 3). Sessile and 500–2000 μm size fractions contributed to a lesser extent and similarly to the total biodiversity (Fig. 3). The diversity was under-sampled, as the rarefaction curves did not reach a plateau (Fig. 3).
In terms of shelf position, little variability in alpha-diversity (i.e. number of OTUs) was detected for either the 18S or COI data (chi-squared: 0.735 and 5.776; p = 0.69 and 0.55 for the 18S and COI, respectively) (Fig. 4 and Table 1). Off-shore reefs had the most distinctive OTUs (472 and 530 unique OTUs for the COI and 18S datasets, respectively) for both primer sets. However, as a proportion of observed OTUs, unique OTUs accounted for a higher percentage in the near-shore (22.3% compared with 20.7% Off-Shore) for the COI dataset. For the 18S and the COI primers, respectively, 28.7% and 15.0% of the OTUs were shared amongst all shelf positions. The near-shore and off-shore reefs shared fewer OTUs than the near-shore and mid-shelf reefs. There was no significant correlation between the number of OTUs and chlorophyll a values (F = 0.4255, p = 0.5197). In terms of differences in OTUs between the two placements, pairwise comparisons revealed no significant difference in OTU number within a reef position.
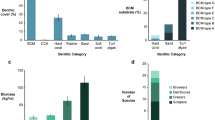
Differences in average number of OTUs per ARMS among size fractions (Sessile, 106–500 μm, 500–2000 μm) and shelf positions (Near-shore, Mid-shore, Off-shore). The average for the shelf positions was taken from those ARMS that had all size fractions present. The median is shown by the solid line across the box whilst the mean is indicated by the black diamond. Small dots represent outliers. Letters denote those categories, which are based on the DunnTest. n is the number of samples per shelf or size fraction.
In total, there were 287 OTUs for the >2000 μm (barcoded) fraction. Only 22 (7.7%) of these were shared among all areas, with the mid-shelf reefs having the highest proportion of exclusive OTUs (34.4%). The near- and off-shore reefs shared the fewest OTUs. On average, there were 20.85 ± 8.64 barcoded OTUs per ARMS and no clear pattern across the shelf gradient was detected (Fig. 4).
Community composition and structure
In the COI dataset, a high proportion (~42%) of the OTUs could not be assigned to any taxonomic group; however, these were generally low abundance OTUs, as only 18.4% of reads were unassigned. Arthropoda contributed the highest number of assigned OTUs in all fractions. However, Porifera accounted for a higher proportion of reads (25.8%) than Arthropoda in the sessile fraction. In the mobile fraction, Annelida had a small number of assigned OTUs although they accounted for a high proportion of reads (20.0%). Similarly, the species richness (number of OTUs) of Chordata was low but in various reefs (especially East Fsar and Al Fahal) they made substantial contributions to the total number of reads (Fig. 5 and Supplementary Figure S2).
In the 18S dataset, Arthropoda were not as dominant in terms of either OTUs or reads, compared to the COI dataset. Arthropoda were almost absent from the sessile fraction (3.0% in terms of reads) and only had an average of 15.6% of the reads (compared to 28.2% in the COI) in the 106–500 μm fraction. In the sessile fraction, Porifera was the dominant group (29.7% of reads), and Dinophyta accounted for ~15% of reads. Annelida accounted for a high proportion of reads (28.7%) of the mobile fractions, as did Mollusca (11.7% and 20.2% for the 106–500 μm and 500–2000 μm size fractions, respectively). Some groups, such as Cercozoa and Rhodophyta, had relatively high numbers of OTUs but these did not account for a high proportion of reads (Figs 5 and S2).
In general the ARMS placed on the reef framework had a higher proportion of Chordata except in Shib Nazar, which was dominated by Cnidaria. Al Fahal, which had ARMS placed in both microhabitats (reef and rubble), interestingly had higher proportions of Chordata in both sets of ARMS. In contrast the ARMS positioned in rubble had a higher proportion of Annelida especially in the COI dataset. In the sessile fraction of the 18S rDNA dataset Dinophyta were more prevalent in the rubble ARMS.
In the barcoding of the >2000 μm fraction, members of the Arthropoda were the dominant taxa at all reefs with the exception of Shib Nazar (for both OTU counts and abundance) (Fig. 5). Other taxa contributing substantially to the composition of the >2000 μm fraction included Mollusca across all stations, and Chordata especially in Al Wasel North and at the two offshore reefs (Shib Nazar and Abu Madafi).
As expected based on primer versatility, differences were noticed in the taxonomic composition of the community using the two target genes (Supplementary Table S5). For instance, the 18S rRNA gene dataset revealed Dinophyta to be a substantial contributor to the sessile fraction. However, it was absent from the COI data, which reflects the broader taxonomic range of the ribosomal unit. There were also differences in the proportion of metazoan reads recovered. Echinodermata and Sipuncula were observed in the COI dataset at average proportions greater than 0.5% but were negligible in the 18S rRNA gene dataset (on average <0.25%). On the other hand, Platyhelminthes accounted for a minor component in the mobile fractions in the 18S dataset, but were absent from the COI data. Despite these differences in detection efficiency, Bray Curtis and Jaccard distance matrices built with the markers were not significantly different based on Mantel tests (r = 0.412; p < 0.001 for Bray-Curtis and r = 0.578; p < 0.001 for Jaccard).
Beta-diversity
Within and between reef comparisons indicated that within reef similarity was higher than between reef similarity (p < 0.001). PCoA analysis indicated that both the structure and composition of the ARMS communities differed across the shelf (Figs 6 and S3). For both the Bray Curtis and Jaccard analysis significant interactions were observed between the factors, suggesting inconsistent variation in the trends (Supplementary Table S6). Pairwise comparisons showed that in general the community differed significantly in terms of shelf position and fraction (Supplementary Table S6). As patterns are similar, the COI data are shown with the 18S rRNA gene data being provided in supplementary data (Figs 6 and S3 and Supplementary Table S6). Further, there was a split between the different microhabitat placements. This pattern was observed regardless of the primer set or the molecular approach used (i.e. metabarcoding or barcoding).
PCoA analysis illustrating dissimilarities in community composition based on Jaccard and Bray Curtis dissimilarity matrices of COI. Analysis was undertaken on the full ARMS unit as well as the different fractions (Sessile, 106–500 μm and 500–2000 μm and >2000 μm). Points are coloured according to shelf position.
Considering all the fractions and units, and for both primers, pairwise comparisons between samples indicated that between 28 and 46% of species were shared (Figs 7 and S4: Total: All). Higher variability was observed for the 106–500 μm size fraction, whereas the barcoded fraction showed the lowest similarity in species composition (i.e., the highest beta-diversity) (Figs 7 and S4). However, the mean values of similarity were rather consistent for the metabarcoding fractions. Slightly lower similarity was detected in the mobile assemblages compared to the sessile assemblages. In addition, similarity values dropped with the increase in spatial scale (from within-reef comparisons to cross-shelf comparisons) (Figs 7 and S4). Nevertheless, mean similarity values within sites for each one of the assemblages assessed using metabarcoding techniques (sessile: 0.38; 106–500 μm: 0.37; 500–2000 μm: 0.33) indicate considerable changes in the species composition at the smaller spatial scale (i.e., the reef). For the largest mobile size fraction (>2000 μm), the maximum average similarity detected at the scale of the reef was 0.22 (i.e., 78% dissimilarity), indicating high heterogeneity in the species composition at the finer spatial resolution (<10 m).
Ternary plots of similarity [1-D (Dissimilarity obtained using Jaccard)] and the partitions of beta diversity (replacement and richness) for the full ARMS community and the various fractions obtained from the COI metabarcoding and barcoding. Ternary plots are shown for the total experiment as well as within and among sites and within and among shelf positions. Numbers in brackets on the axis labels represent the mean value.
Partitioning of the beta-diversity into its replacement and richness components showed that most of the beta-diversity was accounted for by replacement with only a minor contribution from richness. When considering all the possible combinations (i.e. Total:Metabarcoding All), mean values of pairwise similarity indicated that 72% of the species were observed in a single ARMS unit. On average the majority (56%) of the beta-diversity was due to changes in the identity of the community (replacement). Only 15% of OTUs were unique to the richest ARMS sample. The contribution of richness is slightly higher for the >2000 assemblage yet still low, ranging from 18% for within-sites comparison to 24% between-shelf position (Figs 7 and S4).
In the near-shore sites, the ratio replacement:richness was highest and decreased toward the off-shore, indicating richness had more of an impact on beta diversity in the offshore stations than the near or mid-shelf reefs (Supplementary Table S7).
Mantel tests on the correlation between the sessile fraction and the various size fractions of the motile fauna showed significant positive correlations of all possible combinations (p < 0.001). However, there was a decline in R value with increasing size fraction, from 0.5886 (106–500 μm) to 0.5265 (500–2000 μm) to 0.4251 (>2000 μm).
The contribution of each reef to the overall beta-diversity of reef cryptic benthic assemblages, assessed using LCBD indices, indicated that only a few replicates had a significant contribution to the overall beta-diversity (Fig. 8A). Reefs did not contribute evenly and no clear patterns were detected according to their position on the shelf. LCBD ranged from 0.021 for the near-shore reef Abu Shosha to 0.046 for the offshore reef Shib Nazar (Fig. 8B). Five out of 11 reefs sites showed contributions above the average.
Local Contribution to Beta Diversity (LCBD) values. (A) The individual replicates. Values are depicted for the COI metabarcoding of the full ARMS and the various fractions. The size of the circle is proportional to the LCBD value and red indicates a significant difference. (B) The average LCBD for the full ARMS per reef. The dotted line in (B) indicates the value of LCBD if all reefs contributed equally.
There was a negative relationship between the linear distance between reefs and similarities in community composition (Supplementary Table S8) regardless of primer, dissimilarity equation or size fraction analyzed. However, variability amongst the data was high with low R2 values being observed.
Discussion
The use of ARMS combined with molecular methods on a cross-shelf gradient in the central Red Sea has revealed important insight into the hidden majority of biodiversity in coral reef habitats. We identified thousands of OTUs across our sites. Most sites had sessile communities dominated by representatives of Porifera while Arthropoda and Annelida were important components of the mobile fraction. Distinct cross-shelf gradients were observed for both species composition and structure, with replacement contributing the most to beta-diversity. Similar trends in community patterns were observed for both the COI and 18S genes.
Traditional transect surveys undertaken in the Red Sea reveal between 1–100 coral species per site87,88,89 with 54–70 fish species being present88. In the current study the extent of the hidden majority of cryptic fauna was observed with on average, 660 and 750 OTUs per ARMS and a total of 5420 OTUs and 3830 OTUs for the COI and 18S metabarcoding datasets, respectively. Of the totals, 2042 and 1245 OTUs, respectively, were invertebrates giving a greater or similar number of OTUs to a recent attempt to create an species inventory (albeit incomplete) of non-coral invertebrates in the whole of Red Sea90. While it is acknowledged that metabarcoding studies can over estimate the diversity present in a ecosystem91,92, the high alpha and gamma (i.e. total number of OTUs) diversity observed in this study within a single region confirms: (i) the overwhelming diversity of cryptic species present in the reef that has been overlooked44,45, and (ii) the importance of supplementing traditional survey methods (i.e. visual surveys) with DNA-based surveys in order to broaden the taxonomic scope of reef monitoring programs. ARMS sampling allows for the quantification of the diversity of organisms that depend on the three-dimensional structure present in reef habitats, especially species living hidden among and on coral rubble.
Porifera was the most prominent sessile component on the ARMS in agreement with other ARMS in the central Red Sea45, but in contrast to that reported for the Gulf of Aqaba where algal groups had higher contributions44. The presence of Porifera on the ARMS is expected due to their high abundance and diversity in cryptic spaces of the reef38,39. These sponges most likely represent an important habitat for vagile species recorded in this study. The use of the 18S rRNA gene allowed for the identification of dinoflagellates, primarily the symbiotic genus Symbiodinium, to be a substantial component of the sessile fraction. In agreement with the other ARMS studies in the Red Sea, Annelida and Arthropoda played major roles in the composition and structure of the mobile community45.
While the overall patterns were similar between faunal components, there was an inverse relationship between organism size and community similarity, with the largest and most vagile assemblages being the most dissimilar at the scale of our study. Furthermore, the partitioning of beta diversity indicates that replacement increased with size. Indeed, Mantel test comparisons between the sessile component and the various size fractions indicated a decreasing relationship with an increase in the size fraction. This suggests that the structuring factors appear to be different for the largest organisms assessed. While this may be related to the different methodological approach used for this fraction (i.e., barcoding versus metabarcoding, which samples organisms in the gut or growing on surfaces as well) it is likely that the main differences are related to responses to different biological (such as mobility and dispersal behavior, predation) or environmental parameters93,94.
Cross-shelf patterns
Benthic communities are structured at multiple scales depending on global-, regional- (currents, affecting dispersal patterns) and local- (such as biological interactions, niche availability) scale processes93,95. Dispersal patterns of organisms within a reef system are affected by the larval behavior of the dispersing organisms and the prevailing currents in the region96. A significant negative relationship between linear distance and similarity was shown in the current study, although a low R2 value suggests that distance per se is unlikely to play a driving role in community patterns at the scales studied. This would be in agreement with previous studies, which showed that changes in sponge communities were not accounted for by distance97,98. However, linear distance does not necessarily accurately reflect complexity of connectivity potential between sites99. Prevailing currents often have complex patterns and specific formations, such as boundary currents, may affect dispersal across a shelf100. Cross-shelf patterns have previously been reported for large sessile organisms within coral reefs (e.g.)6,14,21,101,102 and algal biofilms9. In our study region, cross-shelf shifts have been documented in fishes88, in scleractinians10 and algal biofilms9. However, the responses of cryptic fauna to this gradient has previously not received much attention.
Regardless of the primer used or the size fraction examined, differences in both the composition and structure of the ARMS community were documented across the shelf, as hypothesized. Previous studies in other regions of the world have suggested that environmental factors such as water quality6,103,104, sedimentation101,105,106 and/or nutrient enrichment107,108,109 influence the ecology of the benthic assemblages of coral reefs. Indeed elevated near-shore nutrient concentrations have been found to be a factor in structuring benthic communities in the southern Red Sea8. While assessments of these factors were out of the scope of the current study, satellite data indicated that off-shore reefs were associated with the lowest levels of chlorophyll a. Values at the near-shore reefs were variable (high for East Fsar but low for Abu Shoosha) with mid-shelf reefs having values intermediate between East Fsar and the off-shore reefs. Differences in chlorophyll a concentrations are likely to indicate differences in nutrient availability on the reefs, as well as variations in the concentrations of organic matter within the water column. Alterations in the concentrations of organic matter are likely to have an effect on various benthic organisms such as those that are predominantly filter feeders or detritivores. The collection of in-situ measurements for a variety of environmental variables during the period of the submersion of the ARMS units at each reef would allow for a better characterization of the environmental factors affecting the reef community.
To date, only a few studies have explicitly measured the partitioning of beta-diversity in reef environments110,111,112. Overall, in the present study, it was observed that the contribution of sites to the overall diversity generally increased towards the off-shore with Abu Madafi, in particular, having a unique species composition. This is further exemplified by the fact that the off-shore position had on average the highest number of unique OTUs, although in the COI dataset this could partially be explained by the higher number of OTUs observed.
Compartmentalizing the beta diversity suggested, regardless of the primer used, that species replacement was the predominant component of beta diversity and richness differences only made a limited contribution. The contribution of richness to the overall biodiversity was highest within the off-shore sites, maybe resulting from the higher number of observed OTUs in Abu Madafi compared to Shib Nazar. The high contribution of replacement (i.e. substitution of species) suggests that the structural complexity of the reefs produces a wide variety of microhabitats that can be colonized by a myriad of organisms, which has been observed for other habitats in the Red Sea113. Also, it could indicate that dispersal of the organisms between reef sites is limited by processes such as coastal boundary layers100, as it is proposed that high connectivity results in homogenization of the community114. The variability in niches allows for the potential co-existence of species with the same ecological roles/functions, which would promote the resilience of the reefs (high levels of redundancy). This is exemplified by the high variability in beta-diversity observed between reefs sharing the same position on the shelf as well as within sites. A range of factors including light, sediment, wave energy, substrate stability and biological interactions115 influence the diverse nature of the cryptic environment. Microzonation of cavity dwellers (coelobites) has been previously shown in coral rubble116 and across a single reef117. High variation in species composition of coral-dwelling invertebrates has been shown among different coral families118. This suggests that there is a degree of host specificity within the coral invertebrate community118, which may reduce competition and allow for greater resource partitioning119, which promotes replacement.
Differences between ARMS sets
Although there were clear cross-shelf effects in the dataset, there was also a difference in the composition and structure between the two microhabitats where ARMS were placed. This difference was observed for all fractions of the dataset. While this could have just been due to the broader spatial scale of assessed reefs for those ARMS placed on rubble, the fact that the two microhabitats on Al Fahal S differed suggests other factors affected the community composition. While the current study is unable to determine the causes of these differences there are two possibilities: small discrepancies in the microenvironment of the deployment habitat between the two ARMS sets as well as differences in the timing of deployment and retrieval of the two sets. Although all ARMS were positioned on the exposed side of the reef, other aspects of positioning within the reef structure were different, particularly whether the ARMS were positioned on the coral framework or on coral rubble. Thus, factors such as the input of sediment or the local community pool, for instance, could have affected the composition of the benthic community. Also it cannot be ruled out that temporal factors could impact the community present on the ARMS. The use of molecular methods allows for the detection of eggs and larvae, which may not be visible in morphological studies. While little is known about the spawning periods of benthic fauna, with the exception of some coral and echinoderms120,121, colonization of ARMS plates by eggs or larvae and the predators consuming them could have an impact on the community assemblages.
Primer comparisons
There is a well-recognized trade-off between taxonomic resolution and amplification efficiency among metabarcoding markers. Recently, Leray and Knowlton57 recommended targeting the slowly evolving 18S rRNA gene region to get a broad overview of the eukaryotic domain followed by the amplification of a lineage-specific hypervariable marker such as COI for higher taxonomic resolution among metazoans. Previous studies characterizing marine diversity from ARMS or other habitats have typically targeted either the 18S rRNA gene45 or the COI gene42, but never both. Here, as expected, we showed differences between the two primer sets in the taxa, which we could annotate taxonomically. This could be either due to differences in amplification efficiency or reference sequences being missing from the appropriate databases and thus amplified sequences not being assigned the correct taxonomic group42. For example, in the current dataset, Alveolata were not observed at all in the COI dataset despite accounting for large numbers of OTUs in the 18S rRNA gene data. On the other hand there was a substantial increase in the number of observed Arthropoda OTUs in the COI dataset compared to the 18S rDNA likely linked to the bias in the COI reference libraries towards this taxon. Despite these differences in both the taxonomy and the observed number of OTUs, comparative Mantel tests (Spearman’s rank) showed that the diversity patterns obtained from both primer sets were not significantly different. This suggests that whilst using two primer sets might be useful for obtaining a better understanding of the taxa present in the community in agreement with previous studies59,122,123, the use of a single primer may be adequate for detecting biodiversity patterns as these broad scale patterns appear to be independent of marker choice122,124.
Conclusion
We observed that across the shelf and biological components, the underlying processes shaping cryptic biodiversity are consistent and driven by spatial turnover (or replacement). The high replacement observed within reefs may suggest low levels of connectivity and a high heterogeneity in niches present at the scale of the reef. This combined with the possibility of a high degree of host specialization among coral-dwelling invertebrates118 promotes the coexistence of a myriad of species and the high rates of replacement observed.
Conservation efforts are often limited by funding and appropriate selection of protected areas is vital. In this case, patterns of beta-diversity can have an impact on the debate. The current study revealed the prominence of replacement processes in the partitioning of beta-diversity for the cryptic organisms. Thus, to best conserve the diversity within the reefs and to minimize species loss, a network of reefs must be protected rather than just the most species-rich reef 2,125. The present study reinforces the importance of taking into account beta-diversity patterns to better understand the ecological changes across environmental gradients in reef systems, as had been suggested before for other habitats2,26,126 and for many of our study reefs88. The incorporation of beta-diversity analysis at multiple spatial scales in ecosystem-based management approaches is recommended in order to attain the long-term conservation of the reef biodiversity and the overall ecosystem health115.
Finally, we showed that the two primer sets amplified different fractions of the cryptobenthic community, but the overall diversity patterns were similar. Currently, one of the major limiting factors in metabarcoding studies is the incomplete nature of reference databases, and the future development of these databases will likely dictate the accuracy of taxonomic assignments and improve ecological interpretations.
With marine habitats under severe pressure it is vitally important for conservation that changes in biodiversity patterns can be detected using standardized methodologies. In this study we demonstrated the ability to detect changes in the composition and structure of the cryptic fauna, a vital component of reef diversity across a cross-shelf gradient.
References
Legendre, P., Borcard, D. & Peres-Neto, P. R. Analyzing beta diversity: partitioning the spatial variation of community composition data. Ecological Monographs 75, 435–450 (2005).
Socolar, J. B., Gilroy, J. J., Kunin, W. E. & Edwards, D. P. How should beta-diversity inform biodiversity conservation? Trends in Ecology and Evolution 31, 67–80, https://doi.org/10.1016/j.tree.2015.11.005 (2016).
Cleary, D. et al. Variation in the composition of corals, fishes, sponges, echinoderms, ascidians, molluscs, foraminifera and macroalgae across a pronounced in-to-offshore environmental gradient in the Jakarta Bay–Thousand Islands coral reef complex. Marine Pollution Bulletin 110, 701–717 (2016).
Cleary, D. F. et al. Relating variation in species composition to environmental variables: a multi-taxon study in an Indonesian coral reef complex. Aquatic Sciences-Research Across Boundaries 70, 419–431 (2008).
Cleary, D. F. R. et al. Variation in the diversity and composition of benthic taxa as a function of distance offshore, depth and exposure in the Spermonde Archipelago, Indonesia. Estuarine, Coastal and Shelf Science 65, 557–570, https://doi.org/10.1016/j.ecss.2005.06.025 (2005).
Fabricius, K. E. Effects of terrestrial runoff on the ecology of corals and coral reefs: review and synthesis. Marine Pollution Bulletin 50, 125–146, https://doi.org/10.1016/j.marpolbul.2004.11.028 (2005).
Becking, L. E. et al. Beta diversity of tropical marine benthic assemblages in the Spermonde Archipelago, Indonesia. Marine Ecology 27, 76–88, https://doi.org/10.1111/j.1439-0485.2005.00051.x (2006).
Ellis, J. et al. Cross shelf benthic biodiversity patterns in the Southern Red Sea. Scientific Reports 7 (2017).
Roik, A. et al. Year-long monitoring of physico-chemical and biological variables provide a comparative baseline of coral reef functioning in the central Red Sea. Plos One 11, e0163939 (2016).
Roik, A., Roder, C., Röthig, T. & Voolstra, C. R. Spatial and seasonal reef calcification in corals and calcareous crusts in the central Red Sea. Coral Reefs 35, 681–693, https://doi.org/10.1007/s00338-015-1383-y (2016).
Williams, D. M. & Hatcher, A. I. Structure of fish communities on outer slopes of inshore, mid-shelf and outer shelf reefs of the Great Barrier Reef. Marine Ecology Progress Series 10, 239–250 (1983).
Hoey, A., Brandl, S. & Bellwood, D. Diet and cross-shelf distribution of rabbitfishes (f. Siganidae) on the northern Great Barrier Reef: implications for ecosystem function. Coral Reefs 32, 973–984 (2013).
Hernández-Miranda, E., Palma, A. & Ojeda, F. Larval fish assemblages in nearshore coastal waters off central Chile: temporal and spatial patterns. Estuarine, Coastal and Shelf Science 56, 1075–1092 (2003).
Done, T. J. Patterns in the distribution of coral communities across the central Great Barrier Reef. Coral Reefs 1, 95–107 (1982).
Wilkinson, C. R. & Cheshire, A. C. Patterns in the distribution of sponge populations across the central Great Barrier Reef. Coral Reefs 8, 127–134 (1989).
Bannister, R. J., Battershill, C. N. & de Nys, R. Demographic variability and long-term change in a coral reef sponge along a cross-shelf gradient of the Great Barrier Reef. Marine and Freshwater Research 61, 389–396, https://doi.org/10.1071/mf09067 (2010).
Harrison, M. A. & Smith, S. D. Cross-shelf variation in the structure of molluscan assemblages on shallow, rocky reefs in subtropical, eastern Australia. Marine Biodiversity 42, 203–216 (2012).
Preston, N. P. & Doherty, P. J. Cross-shelf patterns in the community structure of coral-dwelling Crustacea in the central region of the Great Barrier Reef. II. Cryptofauna. Marine Ecology Progress Series, 27–38 (1994).
Thibault-Botha, D., Lutjeharms, J. R. E. & Gibbons, M. J. Siphonophore assemblages along the east coast of South Africa; mesoscale distribution and temporal variations. Journal of Plankton Research 26, 1115–1128, https://doi.org/10.1093/plankt/fbh104 (2004).
Adjeroud, M. Factors influencing spatial patterns on coral reefs around Moorea, French Polynesia. Marine Ecology Progress Series 159, 105–119 (1997).
Wismer, S., Hoey, A. S. & Bellwood, D. R. Cross-shelf benthic community structure on the Great Barrier Reef: relationships between macroalgal cover and herbivore biomass. Marine Ecology Progress Series 376, 45–54 (2009).
Kramer, M., Bellwood, D. & Bellwood, O. Large-scale spatial variation in epilithic algal matrix cryptofaunal assemblages on the Great Barrier Reef. Marine Biology 161, 2183–2190 (2014).
Smith, T. et al. Assessing coral reef health across onshore to offshore stress gradients in the US Virgin Islands. Marine Pollution Bulletin 56, 1983–1991 (2008).
Polónia, A. R. M. et al. Habitat and water quality variables as predictors of community composition in an Indonesian coral reef: a multi-taxon study in the Spermonde Archipelago. Science of the Total Environment 537, 139–151 (2015).
Connell, J. H. et al. A long-term study of competition and diversity of corals. Ecological Monographs 74, 179–210 (2004).
McKnight, M. W. et al. Putting beta-diversity on the map: broad-scale congruence and coincidence in the extremes. Plos Biol 5, e272, https://doi.org/10.1371/journal.pbio.0050272 (2007).
Arias-González, J. E., Legendre, P. & Rodríguez-Zaragoza, F. A. Scaling up beta diversity on Caribbean coral reefs. Journal of Experimental Marine Biology and Ecology 366, 28–36, https://doi.org/10.1016/j.jembe.2008.07.035 (2008).
Mellin, C., Bradshaw, C., Fordham, D. & Caley, M. Strong but opposing β-diversity–stability relationships in coral reef fish communities. Proceedings of the Royal Society of London B: Biological Sciences 281, 20131993 (2014).
Takada, Y., Abe, O. & Shibuno, T. Colonization patterns of mobile cryptic animals into interstices of coral rubble. Marine Ecology Progress Series 343, 35–44 (2007).
Dennis, C. & Aldhous, P. Biodiversity: a tragedy with many players. Nature 430, 396–398 (2004).
Milne, R. & Griffiths, C. Invertebrate biodiversity associated with algal turfs on a coral-dominated reef. Marine Biodiversity 44, 181–188, https://doi.org/10.1007/s12526-013-0199-7 (2014).
Bouchet, P., Lozouet, P., Maestrati, P. & Heros, V. Assessing the magnitude of species richness in tropical marine environments: exceptionally high numbers of molluscs at a New Caledonia site. Biological Journal of the Linnean Society 75, 421–436 (2002).
Enochs, I. C., Toth, L. T., Brandtneris, V. W., Afflerbach, J. C. & Manzello, D. P. Environmental determinants of motile cryptofauna on an eastern Pacific coral reef. Marine Ecology Progress Series 438, 105–118 (2011).
Kramer, M., Bellwood, D. & Bellwood, O. Benthic Crustacea on coral reefs: a quantitative survey. Marine Ecology Progress Series 511, 105–116 (2014).
Kramer, M., Bellwood, D. & Bellwood, O. Cryptofauna of the epilithic algal matrix on an inshore coral reef, Great Barrier Reef. Coral Reefs 31, 1007–1015 (2012).
Glynn, P. W. & Enochs, I. C. In Coral Reefs: An Ecosystem in Transition (eds Zvy Dubinsky & Noga Stambler) 273–325 (Springer Netherlands, 2011).
Reaka-Kudla, M. L. In Caribbean Marine Biodiversity: The Known and the Unknown (eds Miloslavich, P. & Klein, E.) 259–276 (2005).
Richter, C., Wunsch, M., Rasheed, M., Kötter, I. & Badran, M. I. Endoscopic exploration of Red Sea coral reefs reveals dense populations of cavity-dwelling sponges. Nature 413, 725–730 (2001).
Wunsch, M., Al-Moghrabi, S. & Kötter, I. In Proceedings of the 9th International Coral Reef Conference Vol. 1 (eds Moosa, M. K. et al.) 595–600 (Bali, Indonesia, 2000).
de Goeij, J. M. et al. Surviving in a marine desert: the sponge loop retains resources within coral reefs. Science 342, 108–110 (2013).
Zimmerman, T. L. & Martin, J. W. Artificial reef matrix structures (ARMS): an inexpensive and effective method for collecting coral reef-associated invertebrates. Gulf and Caribbean Research 16, 59–64 (2004).
Leray, M. & Knowlton, N. DNA barcoding and metabarcoding of standardized samples reveal patterns of marine benthic diversity. Proc Natl Acad Sci USA 112, 2076–2081, https://doi.org/10.1073/pnas.1424997112 (2015).
Plaisance, L., Caley, M. J., Brainard, R. E. & Knowlton, N. The diversity of coral reefs: What are we missing? Plos One 6, e25026, https://doi.org/10.1371/journal.pone.0025026 (2011).
Al-Rshaidat, M. M. et al. Deep COI sequencing of standardized benthic samples unveils overlooked diversity of Jordanian coral reefs in the northern Red Sea. Genome 59, 724–737, https://doi.org/10.1139/gen-2015-0208 (2016).
Pearman, J. K., Anlauf, H., Irigoien, X. & Carvalho, S. Please mind the gap - Visual census and cryptic biodiversity assessment at central Red Sea coral reefs. Mar Environ Res 118, 20–30, https://doi.org/10.1016/j.marenvres.2016.04.011 (2016).
Richter, C. & Wunsch, M. Cavity-dwelling suspension feeders in coral reefs—a new link in reef trophodynamics. Marine Ecology Progress Series, 105–116 (1999).
Scheffers, S. R., Van Soest, R. W., Nieuwland, G. & Bak, R. P. Coral reef framework cavities: Is functional similarity reflected in composition of the cryptic macrofaunal community? Atoll Research Bulletin 583 (2010).
Reaka, M. L. Adult-juvenile interactions in benthic reef crustaceans. Bulletin of Marine Science 41, 108–134 (1987).
Coen, L. D. Herbivory by Caribbean majid crabs: feeding ecology and plant susceptibility. Journal of Experimental Marine Biology and Ecology 122, 257–276 (1988).
Rothans, T. & Miller, A. A link between biologically imported particulate organic nutrients and the detritus food web in reef communities. Marine Biology 110, 145–150 (1991).
Finlay, B. J. Global dispersal of free-living microbial eukaryote species. Science 296, 1061–1063 (2002).
Legendre, P. Interpreting the replacement and richness difference components of beta diversity. Global Ecology and Biogeography 23, 1324–1334, https://doi.org/10.1111/geb.12207 (2014).
Leprieur, F. et al. Partitioning global patterns of freshwater fish beta diversity reveals contrasting signatures of past climate changes. Ecology Letters 14, 325–334, https://doi.org/10.1111/j.1461-0248.2011.01589.x (2011).
Loiseau, N. et al. Multi‐component β‐diversity approach reveals conservation dilemma between species and functions of coral reef fishes. Journal of Biogeography 44, 537–547 (2017).
Baselga, A. & Orme, C. D. L. betapart: an R package for the study of beta diversity. Methods in Ecology and Evolution 3, 808–812 (2012).
Roberts, C. M. et al. Marine biodiversity hotspots and conservation priorities for tropical reefs. Science 295, 1280–1284, https://doi.org/10.1126/science.1067728 (2002).
Leray, M. & Knowlton, N. Censusing marine eukaryotic diversity in the twenty-first century. Phil. Trans. R. Soc. B 371, 20150331 (2016).
Leray, M. et al. A new versatile primer set targeting a short fragment of the mitochondrial COI region for metabarcoding metazoan diversity: application for characterizing coral reef fish gut contents. Frontiers in Zoology 10, 34, https://doi.org/10.1186/1742-9994-10-34 (2013).
Stoeck, T. et al. Multiple marker parallel tag environmental DNA sequencing reveals a highly complex eukaryotic community in marine anoxic water. Molecular Ecology 19(Suppl 1), 21–31, https://doi.org/10.1111/j.1365-294X.2009.04480.x (2010).
Geller, J., Meyer, C., Parker, M. & Hawk, H. Redesign of PCR primers for mitochondrial cytochrome c oxidase subunit I for marine invertebrates and application in all‐taxa biotic surveys. Molecular Ecology Resources 13, 851–861 (2013).
Meyer, C. P. Molecular systematics of cowries (Gastropoda: Cypraeidae) and diversification patterns in the tropics. Biological Journal of the Linnean Society 79, 401–459 (2003).
Leray, M. & Knowlton, N. Random sampling causes the low reproducibility of rare eukaryotic OTUs in Illumina COI metabarcoding. PeerJ 5, e3006 (2017).
Li, H. BFC: correcting Illumina sequencing errors. Bioinformatics 31, 2885–2887, https://doi.org/10.1093/bioinformatics/btv290 (2015).
Edgar, R. C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26, 2460–2461, https://doi.org/10.1093/bioinformatics/btq461 (2010).
Schloss, P. D. et al. Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Applied and Environmental Microbiology 75, 7537–7541, https://doi.org/10.1128/aem.01541-09 (2009).
Caporaso, J. G. et al. QIIME allows analysis of highthroughput community sequencing data. Nature Methods 7, 335–336 (2010).
Hao, X., Jiang, R. & Chen, T. Clustering 16S rRNA for OTU prediction: a method of unsupervised Bayesian clustering. Bioinformatics 27, 611–618, https://doi.org/10.1093/bioinformatics/btq725 (2011).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic Local Alignment Search Tool. Journal of Molecular Biology 215, 403–410, https://doi.org/10.1016/s0022-2836(05)80360-2 (1990).
Ratnasingham, S. & Hebert, P. D. N. BOLD: The Barcode of Life Data System (http://www.barcodinglife.org). Molecular Ecology Notes 7, 355–364, https://doi.org/10.1111/j.1471-8286.2006.01678.x (2007).
Machida, R. J., Leray, M., Ho, S. L. & Knowlton, N. Metazoan mitochondrial gene sequence reference datasets for taxonomic assignment of environmental samples. Scientific Data 4, 170027, https://doi.org/10.1038/sdata.2017.27 (2017).
Guillou, L. et al. The Protist Ribosomal Reference database (PR2): a catalog of unicellular eukaryote Small Sub-Unit rRNA sequences with curated taxonomy. Nucleic Acids Research 41, D597–D604, https://doi.org/10.1093/nar/gks1160 (2013).
Pruesse, E. et al. SILVA: a comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Research 35, 7188–7196, https://doi.org/10.1093/nar/gkm864 (2007).
Munch, K., Boomsma, W., Huelsenbeck, J. P., Willerslev, E. & Nielsen, R. Statistical assignment of DNA sequences using Bayesian phylogenetics. Systematic Biololgy 57, 750–757, https://doi.org/10.1080/10635150802422316 (2008).
Wang, Q., Garrity, G. M., Tiedje, J. M. & Cole, J. R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Applied and Environmental Microbiology 73, 5261–5267 (2007).
McMurdie, P. J. & Holmes, S. phyloseq: An R package for reproducible interactive analysis and graphics of microbiome census data. Plos One 8, e61217, https://doi.org/10.1371/journal.pone.0061217 (2013).
Oksanen, J. et al. The vegan package. Community ecology package 10, 631–637 (2007).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. Journal of the Royal Statistical Society. Series B (Methodological), 289–300 (1995).
Ogle, D. FSA: fisheries stock analysis. R package version 0.2–8 (2012).
Wickham, H. ggplot2: elegant graphics for data analysis. (Springer, 2016).
Clarke, K. R. & Gorley, R. N. PRIMER V6: user manual-tutorial. (Plymouth Marine Laboratory, 2006).
Anderson, M., Gorley, R. N. & Clarke, R. K. Permanova + for Primer: Guide to Software and Statisticl Methods (Primer-E Limited, 2008).
Cardoso, P., Rigal, F. & Carvalho, J. C. BAT–Biodiversity Assessment Tools, an R package for the measurement and estimation of alpha and beta taxon, phylogenetic and functional diversity. Methods in Ecology and Evolution 6, 232–236 (2015).
Carvalho, J. C., Cardoso, P. & Gomes, P. Determining the relative roles of species replacement and species richness differences in generating beta-diversity patterns. Global Ecology and Biogeography 21, 760–771, https://doi.org/10.1111/j.1466-8238.2011.00694.x (2012).
Podani, J. & Schmera, D. A new conceptual and methodological framework for exploring and explaining pattern in presence–absence data. Oikos 120, 1625–1638 (2011).
Legendre, P. & De Caceres, M. Beta diversity as the variance of community data: dissimilarity coefficients and partitioning. Ecology Letters 16, 951–963, https://doi.org/10.1111/ele.12141 (2013).
Dray, S. et al. adespatial: Multivariate multiscale spatial analysis. R package version 0.0-3, https://cran. r-project. org/package = adespatial (2016).
DeVantier, L., Turak, E., Al-Shaikh, K. & De’ath, G. Coral communities of the central-northern Saudi Arabian Red Sea. Fauna of Arabia 18, 23–66 (2000).
Khalil, M. T., Bouwmeester, J. & Berumen, M. L. Spatial variation in coral reef fish and benthic communities in the central Saudi Arabian Red Sea. PeerJ 5, e3410 (2017).
Riegl, B. M., Bruckner, A. W., Rowlands, G. P., Purkis, S. J. & Renaud, P. Red Sea coral reef trajectories over 2 decades suggest increasing community homogenization and decline in coral size. Plos One 7, e38396, https://doi.org/10.1371/journal.pone.0038396 (2012).
DiBattista, J. D. et al. A review of contemporary patterns of endemism for shallow water reef fauna in the Red Sea. Journal of Biogeography 43, 423–439 (2016).
Flynn, J. M., Brown, E. A., Chain, F. J., MacIsaac, H. J. & Cristescu, M. E. Toward accurate molecular identification of species in complex environmental samples: testing the performance of sequence filtering and clustering methods. Ecology and evolution 5, 2252–2266 (2015).
Brown, E. A., Chain, F. J., Crease, T. J., MacIsaac, H. J. & Cristescu, M. E. Divergence thresholds and divergent biodiversity estimates: can metabarcoding reliably describe zooplankton communities? Ecology and Evolution 5, 2234–2251 (2015).
Witman, J. D., Etter, R. J. & Smith, F. The relationship between regional and local species diversity in marine benthic communities: a global perspective. Proceedings of the National Academy of Sciences of the United States of America 101, 15664–15669 (2004).
Albertelli, G. et al. Differential responses of bacteria, meiofauna and macrofauna in a shelf area (Ligurian Sea, NW Mediterranean): role of food availability. Journal of Sea Research 42, 11–26 (1999).
Palmer, M. A., Allan, J. D. & Butman, C. A. Dispersal as a regional process affecting the local dynamics of marine and stream benthic invertebrates. Trends in Ecology & Evolution 11, 322–326 (1996).
Cowen, R. K. & Sponaugle, S. Larval dispersal and marine population connectivity. Annual Review of Marine Science 1, 443–466 (2009).
Pandolfi, J. M. et al. Global trajectories of the long-term decline of coral reef ecosystems. Science 301, 955–958 (2003).
de Voogd, N. J., Cleary, D. F. R., Hoeksema, B. W., Noor, A. & van Soest, R. W. M. Sponge beta diveristy in the Spermonde Archipelago, SW Sulawesi, Indonesia. Marine Ecology Progress Series 309, 131–142 (2006).
Almany, G. R. et al. Larval fish dispersal in a coral-reef seascape. Nature Ecology & Evolution 1, 0148 (2017).
Largier, J. L. Considerations in Estimating Larval Dispersal Distances from Oceanographic Data. Ecological Applications 13, S71–S89 (2003).
Fabricius, K. & De’Ath, G. Environmental factors associated with the spatial distribution of crustose coralline algae on the Great Barrier Reef. Coral Reefs 19, 303–309 (2001).
Cleary, D. F. R. & de Voogd, N. J. Environmental associations of sponges in the Spermonde Archipelago, Indonesia. Journal of the Marine Biological Association of the UK 87, 1669–1676, https://doi.org/10.1017/s0025315407052770 (2007).
Anthony, K. R. & Fabricius, K. E. Shifting roles of heterotrophy and autotrophy in coral energetics under varying turbidity. Journal of Experimental Marine Biology and Ecology 252, 221–253 (2000).
Cooper, T. F., Uthicke, S., Humphrey, C. & Fabricius, K. E. Gradients in water column nutrients, sediment parameters, irradiance and coral reef development in the Whitsunday Region, central Great Barrier Reef. Estuarine, Coastal and Shelf Science 74, 458–470, https://doi.org/10.1016/j.ecss.2007.05.020 (2007).
Rogers, C. S. Responses of coral reefs and reef organisms to sedimentation. Marine Ecology Progress Series 62, 185–202 (1990).
Van Woesik, R., Tomascik, T. & Blake, S. Coral assemblages and physico-chemical characteristics of the Whitsunday Islands: evidence of recent community changes. Marine and Freshwater Research 50, 427–440 (1999).
Chazottes, V., Le Campion-Alsumard, T., Peyrot-Clausade, M. & Cuet, P. The effects of eutrophication-related alterations to coral reef communities on agents and rates of bioerosion (Reunion Island, Indian Ocean). Coral Reefs 21, 375–390 (2002).
Loya, Y. In Coral Health and disease 1–34 (Springer, 2004).
Uthicke, S. & Altenrath, C. Water column nutrients control growth and C: N ratios of symbiont‐bearing benthic foraminifera on the Great Barrier Reef, Australia. Limnology and Oceanography 55, 1681–1696 (2010).
Balata, D., Piazzi, L. & Benedetti-Cecchi, L. Sediment disturbance and loss of beta diversity on subtidal rocky reefs. Ecology 88, 2455–2461 (2007).
Belmaker, J., Ziv, Y., Shashar, N. & Connolly, S. R. Regional variation in the hierarchical partitioning of diversity in coral-dwelling fishes. Ecology 89, 2829–2840 (2008).
Lamy, T., Legendre, P., Chancerelle, Y., Siu, G. & Claudet, J. Understanding the spatio-temporal response of coral reef fish communities to natural disturbances: Insights from beta-diversity decomposition. Plos One 10, e0138696, https://doi.org/10.1371/journal.pone.0138696 (2015).
Alsaffar, Z., Cúrdia, J., Borja, A., Irigoien, X. & Carvalho, S. Consistent variability in beta-diversity patterns contrasts with changes in alpha-diversity along an onshore to offshore environmental gradient: the case of Red Sea soft-bottom macrobenthos. Marine Biodiversity, 1–16 (2017).
Limberger, R. & Wickham, S. A. Transitory versus persistent effects of connectivity in environmentally homogeneous metacommunities. Plos One 7, e44555, https://doi.org/10.1371/journal.pone.0044555 (2012).
Rodríguez-Zaragoza, F. A. et al. Additive partitioning of reef fish diversity variation: a promising marine biodiversity management tool. Biodiversity and Conservation 20, 1655–1675, https://doi.org/10.1007/s10531-011-0053-9 (2011).
Choi, D. R. & Ginsburg, R. N. Distribution of coelobites (cavity-dwellers) in coral rubble across the Florida reef tract. Coral Reefs 2, 165–172 (1983).
Gischler, E. & Ginsburg, R. N. Cavity dwellers (coelobites) under coral rubble in southern Belize barrier and atoll reefs. Bulletin of Marine Science 58, 570–589 (1996).
Stella, J. S., Jones, G. P. & Pratchett, M. S. Variation in the structure of epifaunal invertebrate assemblages among coral hosts. Coral Reefs 29, 957–973, https://doi.org/10.1007/s00338-010-0648-8 (2010).
Miller, J. R. Biodiversity conservation and the extinction of experience. Trends in Ecology & Evolution 20, 430–434 (2005).
Bouwmeester, J. et al. Multi-species spawning synchrony within scleractinian coral assemblages in the Red Sea. Coral Reefs 34, 65–77, https://doi.org/10.1007/s00338-014-1214-6) (2015).
Bouwmeester, J., Gatins, R., Giles, E. C., Sinclair-Taylor, T. H. & Berumen, M. L. Spawning of coral reef invertebrates and a second spawning season for scleractinian corals in the central Red Sea. Invertebrate Biology 135, 273–284, https://doi.org/10.1111/ivb.12129 (2016).
Clarke, L. J., Beard, J. M., Swadling, K. M. & Deagle, B. E. Effect of marker choice and thermal cycling protocol on zooplankton DNA metabarcoding studies. Ecology and Evolution 7, 873–883 (2017).
Cowart, D. A. et al. Metabarcoding is powerful yet still blind: a comparative analysis of morphological and molecular surveys of seagrass communities. Plos One 10, e0117562, https://doi.org/10.1371/journal.pone.0117562 (2015).
Drummond, A. J. et al. Evaluating a multigene environmental DNA approach for biodiversity assessment. GigaScience 4, 46 (2015).
Baselga, A. Partitioning the turnover and nestedness components of beta diversity. Global Ecology and Biogeography 19, 134–143 (2010).
Condit, R. et al. Beta-diversity in tropical forest trees. Science 295, 666–669 (2002).
Acknowledgements
We are grateful to the personnel from the King Abdullah University of Science and Technology (KAUST) Coastal and Marine Resources Core Lab for logistical support as well as to the crew of RV Thuwal. We thank Jeffrey Hunt, Matthew Kweskin and the staff of the Laboratory of Analytical Biology at the Smithsonian National Museum of Natural History (Washington DC) who helped with logistics. The authors would also like to thank Holger Anlauf and Riaan van der Merwe who helped with the field work and sample processing along with Richard Payumo and Saskia Kürten who helped with the sample processing. We would also like to thank Ute Langner for the production of the map figures. Figure 2 was produced by Xavier Pita, scientific illustrator at King Abdullah University of Science and Technology (KAUST). Finally we would like to thank the editor and reviewers for their contribution in improving this manuscript. This work was supported by funding to SC from Saudi Aramco within the framework of the Saudi Aramco – KAUST Center for Marine Environmental Observations, KAUST baseline funding to MLB as well as the Sant Chair of the Smithsonian National Museum of Natural History.
Author information
Authors and Affiliations
Contributions
S.C., N.K., M.L.B. and M.L. designed the experiment while J.K.P., S.C., M.L., R.V. and R.M. undertook the laboratory work with J.K.P., M.L., R.V. and R.M. undertaking the data analysis. J.K.P. and S.C. wrote the paper while all authors critiqued various drafts of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pearman, J.K., Leray, M., Villalobos, R. et al. Cross-shelf investigation of coral reef cryptic benthic organisms reveals diversity patterns of the hidden majority. Sci Rep 8, 8090 (2018). https://doi.org/10.1038/s41598-018-26332-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-26332-5
This article is cited by
-
New ecological and phylogenetic insights in the boring barnacle Berndtia Utinomi, 1950 (Acrothoracica: Lithoglyptidae) reveal higher diversity, new hosts, and range extension to the Western Indian Ocean
Marine Biodiversity (2023)
-
Use of Autonomous Reef Monitoring Structures to Monitor Changes in the Marine Environment in Jeju, South Korea: A Brief Review
Ocean Science Journal (2023)
-
Blood meal identification reveals extremely broad host range and host-bias in a temporary ectoparasite of coral reef fishes
Oecologia (2023)
-
Inter-annual variability patterns of reef cryptobiota in the central Red Sea across a shelf gradient
Scientific Reports (2022)
-
Microbial predators form a new supergroup of eukaryotes
Nature (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.