Abstract
The Cretaceous greenhouse climate was accompanied by major changes in Earth’s hydrological cycle, but seasonally resolved hydroclimatic reconstructions for this anomalously warm period are rare. We measured the δ18O and CO2 clumped isotope Δ47 of the seasonal growth bands in carbonate shells of the mollusc Villorita cyprinoides (Black Clam) growing in the Cochin estuary, in southern India. These tandem records accurately reconstruct seasonal changes in sea surface temperature (SST) and seawater δ18O, allowing us to document freshwater discharge into the estuary, and make inferences about rainfall amount. The same analytical approach was applied to well-preserved fossil remains of the Cretaceous (Early Maastrichtian) mollusc Phygraea (Phygraea) vesicularis from the nearby Kallankuruchchi Formation in the Cauvery Basin of southern India. The palaeoenvironmental record shows that, unlike present-day India, where summer rainfall predominates, most rainfall in Cretaceous India occurred in winter. During the Early Maastrichtian, the Indian plate was positioned at ~30°S latitude, where present-day rainfall and storm activity is also concentrated in winter. The good match of the Cretaceous climate and present-day climate at ~30°S suggests that the large-scale atmospheric circulation and seasonal hydroclimate patterns were similar to, although probably more intense than, those at present.
Similar content being viewed by others
Introduction
One consequence of global warming is predicted to be an increase in the frequency and intensity of extreme weather events, including large storms1. Palaeoclimate records of the size and frequency of storms during past periods of sustained high global temperatures are essential for testing this prediction, but such datasets are rare and difficult to extract from the geological record. In present-day India, tropical storms and cyclones are associated with increased atmospheric convection during the summer monsoon. This activity brings large amounts of fresh water to the sub-continent and increases river discharge, reducing the salinity of coastal waters. Seasonal variation in temperature and rainfall are important factors that are likely to be affected by rising levels of atmospheric CO22. There is strong evidence for a shift in the seasonal distribution and inter-annual variability of precipitation worldwide, particularly at higher latitudes, as atmospheric pCO2 has increased over the last century1.
The Cretaceous was a period of global greenhouse conditions when atmospheric pCO2 was at least three times the present level3. This period saw one of the largest-known increases in sea level and global temperature, factors which may have been responsible for major changes in the hydrological cycle4. A record of expansion of the Hadley circulation and seasonal variations in wind strength is preserved in high latitude sedimentary deposits5. Palaeoclimate modelling indicates elevated humidity and rainfall at mid-latitudes of the southern hemisphere, although identifying the source of the moisture for regional precipitation remains problematic6. Proxy records of temperature and rainfall seasonality during the Cretaceous potentially can provide insight into the conditions to be expected in the future should the modern trend in global warming continue.
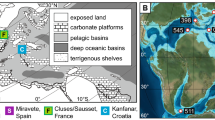
During the Late Cretaceous, the Indian plate was located about 30° south of the equator. A history of Cretaceous sedimentation at the south-eastern margin of the Indian continent is well preserved in the Cauvery Basin (Fig. 1), an elongated pericratonic rift basin extending from south-eastern India into western Sri Lanka7,8,9. Sandstones of Campanian age are overlain by Early Maastrichtian limestone of the Kallankuruchchi Formation, which contains shells of molluscs such as Phygrea sp. and Gryphaea sp.10,11. It has been argued, based on the presence of hummocky cross stratification, that the sediments containing the shells were transported from a nearby shelf region and re-deposited as a result of storm activity10.
Location of the study areas. (A) Map of India showing the location of the modern Cochin estuary (yellow star) and Late Cretaceous Cauvery Basin (red star). (B) Geological map of the Cauvery Basin showing the sampling location and lithostratigraphy of the Late Cretaceous succession. Maps were drawn using CorelDRAW Graphics Suite (2017) Education License, Graphic design software, https://www.coreldraw.com/.
Some well-preserved shells with original mother-of-pearl nacreous layers have alternating dark and light growth bands, reflecting growth under relatively quiet and turbulent sea conditions, respectively. These growth bands record seasonal changes in the isotopic composition of the sea water, reflecting changes in the discharge of freshwater from local rivers12 (Suppl. Fig. S3). In combination, the δ18O and CO2 clumped isotope (Δ47) signatures in the carbonate growth bands carry information about fluctuations in both the water temperature and freshwater input into the shallow estuary13 in which the molluscs grew.
The contribution of freshwater to an estuarine system is governed by rainfall amount in the local catchment. The salinity and δ18O of estuarine water, therefore, is expected to covary with precipitation. In the first part of our study, we show that measurements of δ18O and Δ47 in seasonal growth bands in modern shells of the Black Clam (Villorita cyprinoides; growth rate ~0.75 mm/month, Suppl. Fig. S1a) accurately record shifts in the δ18O of estuarine water that directly reflects measured seasonal variation in local freshwater discharge. Estimates of seasonal freshwater discharge obtained from the isotopic records (Suppl. Fig. S4) are closely correlated with the measured cumulative discharge from the major regional river feeding the estuary (the Periyar River) for the year 2008–200914. Seasonal changes in rainfall measured at the Wellington Island meteorological station, near Cochin, also correlate strongly with the river discharge data15. Based on this result, we then apply the same analytical approach to well-preserved mollusc shells from the Early Maastrichtian sedimentary succession in the nearby Cauvery Basin. The new palaeoenvironmental record provides insight into rainfall seasonality and freshwater discharge in southern India during the hothouse conditions that prevailed in the Late Cretaceous.
Results
Reconstruction of freshwater discharge
Studies of the V. cyprinoides from the coastal region of Cochin, Kerala (Fig. 1), show that the growth rate of their shells approaches a minimum of 0.25 mm/month during cooler drier months, when the salinity of estuarine water is at a maximum, and peaks at >1.3 mm/month during warmer wetter months, when freshwater discharge lowers salinity16,17.
The shell isotopic data reported here are from the first ~2.5 years of the life of a Black Clam collected live from Thevara, ~11 km upstream from the mouth of the Cochin estuary. The sclerochronological uncertainty of the record is about two weeks18. The δ18O and Δ47 measured across the growth bands of the clam shell capture a seasonal pattern (Fig. 2, Table 1). Rainfall in the region is prolonged due to the influence of two monsoon systems: the summer Southwest (SW) Monsoon from June to September, and the winter Northeast (NE) Monsoon from October to December. The δ18O of the water in the Cochin estuary close to the shell collection site ranged between −3.5‰VSMOW during the entire period encompassing both the monsoons (June–December) and −0.5‰VSMOW during the dry season (January–May)19. The corresponding carbonate growth bands had an average δ18O of −4.5‰VPDB for the period of rain and −2.3‰VPDB for the drier months. The record of daily rainfall at Cochin was obtained from the ‘World Weather’ TuTiempo Network (http://en.tutiempo.net) (Suppl. Information 2). It shows that rainfall, and hence freshwater input into the estuary, peaked in the period May through September, which corresponds to the period of thunderstorm activity during the pre-monsoon months (March–May) and increased rainfall during the monsoon (May–September) months.
δ18O in V. cyprinoides, reconstructed temperatures and instrumental records for the Cochin estuary. (A) Temperature estimates (red circles) derived from clumped isotope thermometry (CDES scale) compared with observed temperatures45 (grey shading denotes amplitude of signal). δ18O values for V. cyprinoides measured by continuous flow IRMS at 1 mm resolution (grey squares) are also plotted. Slower shell growth during cooler periods reduced the temporal resolution for the temperature estimates. (B) Observed daily rainfall (grey bars, http://www.tutiempo.net/) compared with the reconstructed percentage contribution of freshwater to the Cochin estuary (blue squares) and cumulative river discharge (purple diamonds, discussed in detail in Section S4 of the Suppl. Information 1). The freshwater contribution was calculated by subtracting the influence of temperature from the shell δ18O record, and applying a mixing model with end-member δ18O values for freshwater and seawater (green circles denote calculated δ18O water) (see text for details).
The analysis of CO2 clumped isotopes (CO2 isotopologues) in aragonite growth bands of modern V. cyprinoides and bio-calcite of Cretaceous Phygraea (Phygrea) vesicularis makes it possible to reconstruct seasonal ambient water temperatures with an uncertainty of ±2 °C20,21. Seasonal temperatures calculated from clumped isotope ratios (Δ47) measured on the aragonite growth increments in the V. cyprinoides shell ranged between 20.4 and 32.5 °C (Fig. 2), with a mean value of 28 °C. These temperatures are close to the directly-measured temperatures of the estuarine water (Suppl. Fig. S5). As seen in Suppl. Fig. S5, a poor correlation (R2 of 0.36, P = 0.04) is seen with the measured temperature. We suspect the factors like uncertainty in the determination of water temperature and the difference in the surface and bottom water temperature could have influenced the correlation. Monthly mean temperatures for the year 2009–2010 approached a minimum of 26 ± 1 °C during the winter months (December-February), whereas the regional maximum of 31 ± 1 °C was recorded during the summer months of March and April22 (Fig. 2, Table 2).
The δ18O of precipitated carbonate (mollusc shell) depends on both the isotopic composition and temperature of the water. Once the temperature has been determined independently by CO2 clumped isotopes (Suppl. Fig. S2), the δ18O of the water can be calculated. High rainfall during the monsoon months coincides with water δ18O values as low as −3.5‰VSMOW, whereas during non-monsoon months, when the water is more saline, δ18O approached −0.4‰VSMOW. This seasonal change in the δ18O of water in the Cochin estuary can be modelled as mixing between two components: rainwater runoff and seawater. The average δ18O of rainwater collected in the region in different seasons (−3.5‰VSMOW)23,24 is used as the rainwater end-member and the δ18O of the Arabian Sea (0.9‰VSMOW)25 is used for seawater.
Cretaceous climate reconstruction using Phygraea (Phygraea) vesicularis
The concentrations of atmospheric greenhouse gases were significantly higher in the Cretaceous than they are today, therefore the global temperature was higher than it is now. The average equatorial sea surface temperature is estimated to have been ~31 °C26, compared to ~27 °C at present. This higher temperature is likely to have induced changes in the hydrological cycle. During the Early Maastrichtian, the Indian plate was positioned further south at mid-latitudes (~30°S) relative to its present position in the northern hemisphere. The tropical to sub-tropical climate favoured the formation of coal and limestone, and the higher concentrations of greenhouse gases led to enhanced terrestrial productivity27.
A fossilized shell of the mollusc Phygraea (Phygraea) vesicularis, with well-preserved distinguishable carbonate growth bands (Suppl. Fig. S6), was recovered from Late Cretaceous (Early Maastrichtian) shell-bearing strata near the village of Ottakoil, Tamil Nadu, India. Siliciclastic sediments (Ottakoil Formation) immediately above the fossiliferous carbonate layer and a conformable off-lap of much younger fluvial sand deposits (Kallamedu Formation) represent a regressive marine sequence. An overlying conglomerate bed has structures indicative of deposition in a shallow marine environment.
Oxygen isotope analysis of the carbonate in the growth bands of P. vesicularis revealed a record of seasonal changes in seawater temperature and salinity. The δ18O and Δ47 of the shell carbonate, and a Ce anomaly in the adjacent Kallankurichchi limestone are indicators of evaporative, anoxic environmental conditions at the time of deposition28.
The studied P. vesicularis shell had distinct prismatic and nacreous layers, no gross differences in Fe and Mn concentrations as measured by electron microprobe (Suppl. Information 3), and a lack of cathodoluminescence, indicating good preservation of its original composition29,30. Electron Backscattered Diffraction (EBSD) images of its growth structures, texture, crystal size and crystal orientation showed features similar to those characteristics of seasonal growth in modern day estuarine oysters (Suppl. information 1 Section S7, Figs S7, S8), consistent with there having been little or no diagenetic alteration28. The shell also had a large and systematic range of δ18O (>3‰, Fig. 3) as measured by both gas-source isotope ratio mass spectrometry (GIRMS) and Sensitive High Resolution Ion Micro Probe (SHRIMP).
Reconstructed Early Maastrichtian temperature and seawater δ18O for the Cauvery Basin. (A) δ18O of Phygraea vesicularis shell 1 measured by dual inlet IRMS (squares) and shell 2 (diamonds) measured by continuous flow IRMS (1 mm resolution). Shell 2 was also analysed by SHRIMP with a 25 µm spot size and 150 µm spacing (circles). (B) Seasonal temperatures for shell 1 reconstructed using clumped isotope thermometry (CDES scale). (C) Calculated δ18O of water using IRMS shell δ18Odata in (A, B) and the empirical δ18O-temperature relationship for molluscan calcite of Dettman et al., 1999 (ref.12). Tan shading indicates cooler/wetter winter seasons.
The analysed section of shell recorded two cycles of δ18O, reflecting repetition of the seasonal change in temperature and salinity (Fig. 3). The temporal variation in δ18O followed a sinusoidal pattern, with higher δ18O in summer and lower δ18O in winter. The GIRMS measurements of aliquots of calcite micro-drilled from the shell showed a seasonal change in δ18O of ~3‰ (−1.5 to −4.6‰) with a mean value of −2.7‰ (n = 21). The individual SHRIMP microanalyses (n = 125) had a larger range (~6‰). The differences in δ18O between adjacent 25 µm spots were particularly large for the periods of summer growth (up to 3‰), much larger than can be explained by analytical uncertainty (±~0.3‰) or temperature fluctuations (Table 3). This variability coincided with changes in the crystal structure of the shell (Suppl. Fig. S6).The coarsely crystalline growth bands, mostly denoting summer growth, were more heterogeneous than the finely crystalline bands formed during winter. The Δ47 determined from the clumped isotope analyses ranged from 0.66 to 0.73‰, corresponding to a temperature range of 21 to 37 °C, with a mean value of 30 °C. This temperature range accounts for about 3.5‰ of the variation in the measured δ18O. All remaining variation (~2.5‰) is due to changes in the δ18O of the water in which the mollusc grew.
Discussion
The well-preserved P. vesicularis shell recovered from Late Cretaceous sediments of the Cauvery Basin is an ideal specimen with which to determine the past rainfall seasonality pattern on the Indian coast when the subcontinent was positioned at latitude ~30°S. The observed variations in Cretaceous seawater δ18O and temperature are similar to those found in a modern coral-based proxy record from the Dampier Archipelago27, Western Australia (20.5°S), which shows the strong seasonal effect of evaporation on salinity. Coupled analyses of Sr/Ca and δ18O in the coral growth bands indicate that sea surface salinity during the period 1988–1994, as calculated from the residual δ18O after the temperature component was subtracted, was highest when strong evaporation in summer caused salinity (and water δ18O) and to increase. In contrast, reduced evaporation and increased rainfall in winter cause a decrease in seawater δ18O and salinity27.
The seasonal range in Early Maastrichtian water temperature in the Cauvery Basin was 21–37 °C, so the −1.6 to −3.1‰ range in δ18O measured in the P. vesicularis shell growth bands translates to a range in water δ18O from −0.5 to 1.7‰VSMOW (Table 3). The seasonal change in freshwater contribution to the Cretaceous estuary can be calculated assuming the average δ18O of Early Maastrichtian rainfall to be about −6‰VSMOW (inferred from the δ18O of contemporaneous palaeosol carbonates7) and seawater in the evaporating enclosed basin to have a δ18O of ~2‰ (as in the modern Red Sea—global seawater δ18O database; Craig, 1966). Based on this two end-member mixing model, the seasonal change in freshwater contribution to the Cretaceous estuary ranged from 3 to 31% between summer and winter (Table 3).
Further, water δ18O and temperature are strongly correlated, with the lowest temperatures associated with the lowest water δ18O values, suggesting a climatological control on the amount of runoff to the estuary. Temperature and the δ18O of precipitation are directly correlated with latitude7 whereby the δ18O of Late Cretaceous precipitation was lower while India was located in the southern hemisphere mid-latitudes. This conclusion is consistent with a Late Cretaceous palaeoclimate reconstruction from further south at Seymor Island31, where maximum freshwater discharge occurred during winter, suggesting warmer winters than those currently experienced in coastal regions in southern mid-latitudes.
The sedimentary record of the Kallankuruchchi Formation provides a further opportunity to probe the Late Cretaceous temperature and hydrological seasonality. Previous studies have interpreted sedimentary structures in the Kallankuruchchi Formation (cross bedding, cut and fill, hummocky cross stratification) as indicating deposition during major storm events. Evidence of storm deposition is also found in other contemporaneous strata in the Cauvery basin32. The weight of evidence is that, unlike the modern day, Cretaceous southern India experienced the bulk of its rainfall in winter, whereas reduced rainfall and runoff in summer increased both the salinity and temperature of the estuarine water, resulting in deposition of thick piles of evaporite and carbonate sediments in the stratigraphic succession.
The climatic conditions experienced in southern India at ~30°S latitude during the Late Cretaceous were evidently similar to those of the modern-day coastal plain of Western Australia around the same latitude, where seasonal storms and cyclones impact on the carbonate platform33. Our finding suggests that large-scale atmospheric circulation and seasonal hydroclimate patterns at mid-latitudes during the Cretaceous global warming interval were not substantially different from the present-day. The result is relevant for climate models designed to simulate the extent to which elevated atmospheric CO2 levels, and the accompanying global warming, might alter the Hadley circulation and mid-latitude storms in the future.
Methods
Background
The Cochin Backwaters at Kerala, India (9°40′ N–10°08′ N, 76°11′ E–76°25′ E) is populated by several species of mollusc, the most widespread being Villorita, known for its edible value. The area occupies the northern part of the Vembanad-Kol wetlands, and covers ~255 km2, extending from Alleppey to Cochin before merging with the Arabian Sea via two permanent openings. The region has a modern-day tropical climate with two main rainy seasons. Most of the rainfall (~70%) occurs during the SW monsoon from June to September. Much of the remaining rainfall (~15%) occurs during the NE monsoon from October to November, while the December-to-May pre-monsoon period has sporadic rainfall accounting for the remaining 15%. The surface water temperature in the estuary reaches a maximum of 32 °C in April and drops to as low as 24 °C in August.
Sample collection
Several specimens of Villorita cyprinoides were harvested live from the southern extremity of the Cochin Backwaters on 20 January 2010 for the present investigation. The growth rate of V. cyprinoides varies over a year and is characterised by growth increments of aragonite separated by laminar bands. The average growth rate for the species is ~8.3 mm/year16, determined by monitoring a population of several individuals in situ within a cage experiment. For the present study, the incremental growth bands were drilled at a spatial resolution of 1 mm for δ18O and clumped isotope analysis.
The Cauvery Basin, at the southern tip of peninsular India (Fig. 1), hosts a complete Cretaceous sedimentary sequence of shallow marine to estuarine deposits. The sequence consists of the Uttatur, Trichinopoly and Ariyalur rock groups, representing Early, Middle and Late Cretaceous successions, respectively. Specimens of Phygraea (Phygraea) vesicularis were collected from an exposure in the Kallankuruchchi Formation (Ariyalur group), well exposed near the PNR mine of Dalmia Cement Limited (11°7′11′′N 79°7′59′′E)34.
Recovery of carbonate from mollusc growth bands
In preparation for δ18Oand clumped isotope analysis, internal soft body parts were discarded from V. cyprinoides and the outer carbonate shells were treated with H2O2 for complete removal of organic debris, and then air dried for sectioning and drilling. Shells were dissected along the growth axis (Suppl. Figs S1a, S1b) using a section cutter and sampled along individual growth bands at 1 mm resolution using a battery operated micro mill. The recovered powder was analysed by XRD, showing aragonite to be the primary mineral phase.
The shell for the Cretaceous P. vesicularis was processed in a similar manner, sectioned along the growth axis and sampled at 1 mm and 2 mm resolution for δ18O and clumped isotope analysis. Powder analysed by XRD suggested calcite as the primary mineral in the Cretaceous specimen. The same section was polished for in situ analysis of δ18O at a spatial resolution of 25 μm by Sensitive High Resolution Ion Microprobe (SHRIMP).
Conventional δ18O measurements
The δ18O (and δ13C) of the shell samples were measured at high-resolution using a Thermo Finnigan MAT 253 isotope ratio mass spectrometer (IRMS) coupled with a Gas bench II in continuous flow mode. About 100 μg of carbonate powder was reacted with 1 ml of H3PO4 using the boat method described elsewhere35 and the overall δ18O reproducibility for NBS-19 calcite was ±0.08‰. Water samples collected from the Cochin estuary were analysed for δ18O following the CO2-water equilibration method, where 100 μl of water was equilibrated with the CO2+He mixture for more than 18 hours (as described previously)36. The over-all δ18O reproducibility of replicate analyses of water standards was ±0.06‰.
Larger shell sample weights (~5–10 mg) required for the preparation of CO2 gas for clumped isotope measurements were obtained by combining powders from two or more growth bands (averaging ~2–3 months of growth). δ18O (and δ13C) in the larger samples were measured on the MAT 253 IRMS in dual inlet mode along with measurements of mass-47 isotopologues of CO2 for clumped isotope analysis. The overall δ13C, δ18O and Δ47 reproducibility for NBS-19 calcite value of ±0.04, 0.05 and 0.01‰ respectively.
Measurements of Δ47 in shell growth bands
Δ47 analyses were performed using the dual inlet peripherals on the Thermo MAT 253 IRMS following the preparation steps of CO2 cleaning through use of an external GC setup37. The experimental procedure for sample preparation for clumped isotope analysis used the sealed vessel method of 38,39. All carbonate samples were prepared in the experimental setup designed at the Indian Institute of Science, Bangalore. For each analysis, ~5–10 mg of carbonate powder was reacted with 1 ml of H3PO4 in a sealed reaction vessel. The reaction vessel was evacuated on a gas-extraction line to a pressure of 10−4 mbar using a combination of turbomolecular and roughing pumps. The stopcock in the evacuated vessel was then closed and the vessel kept in a water bath maintained at a constant temperature of 25 ± 0.1 °C. The reaction of carbonate with H3PO4 commenced by a simple transfer of acid from the arm of the reaction vessel to the compartment with carbonate powder.
The CO2 generated during the reaction was cleaned using a cryogenic extraction protocol to remove contaminants responsible for isobaric interferences37. Purification steps involved removal of water vapour and other contaminants by a combination of liquid nitrogen trap and a dry ice and ethanol slush trap. The CO2, once extracted onto a cold finger, was entrained with a He stream through a capillary column (PoraPLOT Q, 25 m × 0.32 mm i.d.; Varian Inc., Palo Alto, CA, USA) and held at −10 °C for gas chromatographic separation of CO2 from other mixtures of trace hydrocarbon and halocarbon. Eventually, the purified CO2 sample was taken into a glass cold finger and analysed using the MAT 253 IRMS dual inlet system.
The MAT 253 IRMS was configured to analyse mass-47 isotopologues of CO2 by simultaneously measuring mass 47, 48 and 49 (measured with 1012 Ω resistors). Masses 48 and 49 were monitored in order to ensure that there were no isobaric interferences due to the presence of contaminants. These measurements were done in dual inlet mode with a source pressure sufficient to maintain the CO2 mass-44 ion beam intensity at a voltage of 10–12 V. Each analysis involved 60 measurement cycles of the sample CO2 and reference CO2 (six acquisition lines with 10 cycles each, with a signal integration time of 8 s per measurement). The reference CO2 gas (Linde CO2) had δ13C and δ18O values of −4.41‰ (VPDB) and 24.59‰ (VSMOW), based on repeat analyses of the NBS-19 carbonate standard.
The Δ47 analyses were standardized using in-run measurements of NBS-19, heated gas and in-house MAR J1 calcite (generated from Carrara marble)39. Calibration of MARJ1, which was run more frequently during the course of our measurements, was done by adopting two published values for the other two reference materials (NBS-19 and heated CO2 at 1000 °C). The Δ47 value of 0.392‰ for NBS-19, and 0.026‰ for heated CO2, were adopted to relate the heated gas scale to the CDES scale22. The MAR J1 Carrara marble was assigned a value of 0.395‰ for scale conversion purposes. The long-term reproducibility of MAR J1 over the period 2010–2012 (n = 59) yielded a Δ47 value of 0.343 ± 0.01 (1σ) on the heated gas scale. All the shell carbonate samples were analysed during that period.
In order to convert the Δ47 values to the heated gas scale, a large number of CO2 samples were treated at 1000 °C for 2 hours upon recovery of the analysed samples. The CO2 samples used for high temperature treatment were obtained upon transferring the sample CO2 to an ultrapure synthetic quartz tube (6 mm o.d.), which was evacuated and sealed. The sealed tube containing the CO2 sample was heated in a muffle furnace at 1000 °C for >2 hours and then quickly quenched to room temperature. The difference between the Δ47 values measured in the sample CO2 and randomized CO2 generated upon heating at 1000 °C allowed the definition of the Δ47-HG value in the heated gas scale20. All data are reported at the 25 °C reaction temperature and therefore no additional correction for the reaction temperature fractionation factor was applied. For the period of analysis, heated CO2 yielded Δ47 values (n = 66) of −1.47 ± 0.06‰ (1σ). The values on the heated gas scale are converted to the absolute CDES scale by using the proposed equation22 relating the absolute value of Carrara marble and heated gas and is given here as:
where the subscripts ‘NBS_HG’, ‘NBS_WG’ and ‘HG_WG’ represent, respectively, the Δ47 value of the NBS-19 with respect to heated CO2, the NBS-19 value with respect to our Linde reference CO2 used here as working gas and the heated CO2 value with respect to Linde reference CO2 (working gas). The values for δ18O (and δ13C) in the carbonate samples were reported on the VPDB scale, while Δ47 values were expressed following the carbon dioxide equilibrium scale (CDES) scale22.
For deriving the δ18O of water from measurements of δ18O in carbonates, the carbonate δ18O values were converted to the VSMOW scale using the following equation40:
Furthermore, the water δ18O values for modern V. cyprinoides samples were deduced using the growth band δ18O, Δ47-based temperature and the relationship proposed for shell aragonite41:
where δ18O of aragonite is in VPDB and δ18O water is in VSMOW. Similarly, for Late Cretaceous P. vesicularis samples, water δ18O was deduced using the relationship for calcite42:
where the calcite-water fractionation factor (α) is defined as:
SHRIMP δ18O measurements
A glass-mounted polished thin section of the Phygraea vesicularis shell was cut to size and cast with grains of NBS-18 and NBS-19 reference calcite in a disc of Struers Epofix epoxy resin 35 mm diameter. The disc was lightly polished with 3 µm and 1 µm diamond paste to expose the standards, degreased, photographed at high magnification, washed with petroleum spirit, warm detergent solution and Millipore water, dried in a 60 °C vacuum oven for 24 hours and coated with 12 nm of high purity Al before being loaded into the Australian National University SHRIMP II for O isotopic analysis.
The analytical procedure was based on that described by Ickert et al.43 and Long et al.44. In brief, a ~3 nA primary ion beam of ~15 kV Cs+ was focused to a probe ~25 µm in diameter, and secondary ions of O−(16O ≈ 1.9 × 109 c/s) were extracted at ~10 kV for isotopic analysis by dual Faraday cup multiple collection (current mode, 1011 Ω resistors). Charge build-up on the sample surface was neutralised using a focused ~600 eV electron beam. Each analysis consisted of a 90 s pre-burn during which electrometer baselines were measured, ~2 min of ion focusing and 12 × 10 s measurements of 18O/16O. Internal precision of each spot analysis was ≤0.15‰ (s.e.). Corrections for electron induced secondary ion emission (EISIE) were ~0.1‰. δ18O was calculated relative to analyses of several fragments of NBS-19 (δ18OVPDB = −2.2‰) distributed throughout the 20-hour analytical session. Reproducibility of the analyses of NBS-19 over the whole session was 0.21‰ (s.d., n = 27).
Change history
03 July 2018
A correction to this article has been published and is linked from the HTML and PDF versions of this paper. The error has not been fixed in the paper.
References
Feng, X., Porporato, A. & Rodriguez-Iturbe, I. Changes in rainfall seasonality in the tropics. Nat. Clim. Change 3, 811–815 (2013).
Pachauri, R. K. et al. Climate change 2014: synthesis report. Contribution of Working Groups I, II and III to the fifth assessment report of the Intergovernmental Panel on Climate Change. (IPCC, 2014).
Barclay, R. S. & Wing, S. L. Improving the Ginkgo CO2 barometer: Implications for the early Cenozoic atmosphere. Earth and Planetary Science Letters. 439, 158–171 (2016).
Miller, K. G., Barrera, E., Olsson, R. K., Sugarman, P. J. & Savin, S. M. Does ice drive early Maastrichtian eustasy? Geology. 27, 783–786 (1999).
Hasegawa, H. et al. Drastic shrinking of the Hadley circulation during the mid-Cretaceous Super Greenhouse. Climate of the Past. 8, 1323–1337 (2012).
Floegel, S. & Wagner, T. Insolation-control on the Late Cretaceous hydrological cycle and tropical African climate—global climate modelling linked to marine climate records. Palaeogeography, Palaeoclimatology, Palaeoecology. 235, 288–304 (2006).
Ghosh, P. et al. Tracking the migration of the Indian continent using the carbonate clumped isotope technique on Phanerozoic soil carbonates Scientific Reports. 6 (2016).
Sastri, V., Venkatachala, B. & Narayanan, V. The evolution of the east coast of India. Palaeogeography, Palaeoclimatology, Palaeoecology. 36, 23–54 (1981).
Chari, M. V. N., Sahu, J. N., Banerjee, B., Zutshi, P. L. & Chandra, K. Evolution of the Cauvery Basin, India from subsidence modeling. Marine and Petroleum Geology. 12, 667–675 (1995).
Rao, L. R. Recent contributions to our knowledge of the cretaceous rocks of South India. Proceedings of the Indian Academy of Sciences-Section B 44(4), 185–245 (1956).
Madhavaraju, J. et al. Carbon, oxygen and strontium isotopic signatures in Maastrichtian-Danian limestones of the Cauvery Basin, South India. Geosciences Journal. 19, 237–256 (2015).
Dettman, D. L., Reische, A. K. & Lohmann, K. C. Controls on the stable isotope composition of seasonal growth bands in aragonitic fresh-water bivalves (unionidae). Geochimica et Cosmochimica Acta. 63, 1049–1057 (1999).
Banerjee, Y., Ghosh, P., Bhushan, R. & Rahul, P., Strong sea forcing and warmer winter during solar minima ~2765 yr BP recorded in the growth bands of Crassostrea sp. from the confluence of river Ganges, Eastern India., Quaternary International. in press (2017).
Jacob, B., Revichandran, C. & Kumar, N. Salt intrusion study in Cochin estuary-using empirical models. Indian Jour. Of Marine Science. 42(3), 304–313 (2013).
Vinita, J. et al. Salinity response to seasonal runoff in a complex estuarine system (Cochin Estuary, west coast of India). Journal of Coastal Research. 31(4), 869–878 (2015).
Arun, A. U. Gametogenic cycle in Villorita cyprinoides and the influence of salinity. Aquaculture, Aquarium, Conservation & Legislation-International Journal of the Bioflux Society (AACL Bioflux). 2 (2009).
Arun, A. U. An assessment on the influence of salinity in the growth of black clam (Villorita cyprinoides) in cage in Cochin Estuary with a special emphasis on the impact of Thanneermukkom salinity barrier. Aquaculture, Aquarium, Conservation & Legislation-International. Journal of the Bioflux Society (AACL Bioflux). 2, 319–330 (2009).
Schone, B. R., Tanabe, K., Dettman, D. L. & Sato, S. Environmental controls on shell growth rates and delta O-18 of the shallow-marine bivalve mollusk Phacosoma japonicum in Japan. Marine Biology. 142, 473–485 (2003).
Kaushal, R., Ghosh, P. & Geilmann, H. Fingerprinting environmental conditions and related stress using stable isotopic composition of rice (Oryza sativa L.) grain organic matter. Ecological Indicators. 61, 941–951 (2016).
Eiler, J. M. & Schauble, E. 18O13C16O in Earth’s atmosphere. Geochimica et Cosmochimica Acta. 68, 4767–4777 (2004).
Schauble, E. A., Ghosh, P. & Eiler, J. M. Preferential formation of C-13-O-18 bonds in carbonate minerals, estimated using first-principles lattice dynamics. Geochimica et Cosmochimica Acta. 70, 2510–2529 (2006).
Dennis, K. J., Affek, H. P., Passey, B. H., Schrag, D. P. & Eiler, J. M. Defining an absolute reference frame for ‘clumped’ isotope studies of CO2. Geochimica et Cosmochimica Acta. 75, 7117–7131 (2011).
Unnikrishnan, A. S. & Shankar, D. Are sea-level-rise trends along the coasts of the north Indian Ocean consistent with global estimates? Global and Planetary Change. 57, 301–307 (2007).
Lekshmy, P. R., Midhun, M., Ramesh, R. & Jani, R. A. O-18 depletion in monsoon rain relates to large scale organized convection rather than the amount of rainfall. Scientific Reports. 4 (2014).
Singh, A., Jani, R. A. & Ramesh, R. Spatiotemporal variations of the delta O-18-salinity relation in the northern Indian Ocean. Deep-Sea Research Part I-Oceanographic Research Papers. 57, 1422–1431 (2010).
Zakharov, Y. D. et al. Cretaceous climate oscillations in the southern palaeolatitudes: New stable isotope evidence from India and Madagascar. Cretaceous Research. 32, 623–645 (2011).
Gagan, M. K. et al. New views of tropical paleoclimates from corals. Quaternary Science Reviews. 19, 45–64 (2000).
Nagendra, R., Sathiyamoorthy, P. & Reddy, A. N. In STRATI 2013: First International Congress on Stratigraphy At the Cutting Edge of Stratigraphy (eds Rogério Rocha, João Pais, Carlos José Kullberg, & Stanley Finney) 547–551 (Springer International Publishing, 2014).
Jacob, D. E. et al. Nanostructure, composition and mechanisms of bivalve shell growth. Geochimica et Cosmochimica Acta. 72, 5401–5415 (2008).
Pérez-Huerta, A., Cuif, J. P., Dauphin, Y. & Cusack, M. Crystallography of calcite in pearls. European Journal of Mineralogy 26(4), 507–516 (2014).
Petersen, S. V., Dutton, A. & Lohmann, K. C. End-Cretaceous extinction in Antarctica linked to both Deccan volcanism and meteorite impact via climate change. Nature Communications 7 (2016).
Ramkumar, M. A. Storm event during the Maastrichtian in the Cauvery basin, south India. Geoloski anali Balkanskoga poluostrva 67, 35–40 (2006).
Berry, P. & Marsh, L. History of investigation and description of the physical environment. Faunal Surveys of The Rowley Shoals, Scott Reef and Seringapatam Reef North-West Australia: Records of the Western Australian Museum, Supplement 25, 1–19 (1986).
Ayyasami, K. Role of oysters in biostratigraphy: A case study from the Cretaceous of the Ariyalur area, southern India. Geosciences Journal. 10, 237–247 (2006).
Rangarajan, R. High Resolution Reconstruction of Rainfall Using Stable Isotopes in Growth Bands of Terrestrial Gastropod Doctor of Philosophy (Ph.D.) thesis, Indian Institute of Science (2014).
Rangarajan, R. & Ghosh, P. Role of water contamination within the GC column of a GasBench II peripheral on the reproducibility of O-18/O-16 ratios in water samples. Isotopes in Environmental and Health Studies. 47, 498–511 (2011).
Ghosh, P. et al. (13)C-(18)O bonds in carbonate minerals: A new kind of paleothermometer. Geochimica et Cosmochimica Acta. 70, 1439–1456 (2006).
McCrea, J. M. On the isotopic chemistry of carbonates and a paleotemperature scale. The Journal of Chemical Physics 18(6), 849–857 (1950).
Rangarajan, R., Ghosh, P. & Naggs, F. Seasonal variability of rainfall recorded in growth bands of the giant African land snail Lissachatina fulica (Bowdich) from India. Chemical Geology. 357, 223–230 (2013).
Coplen, T. B., Kendall, C. & Hopple, J. Comparison of stable isotope reference samples. Nature 302(5905), 236 (1983).
Grossman, E. L. & Ku, T. L. Oxygen and carbon isotope fractionation in biogenic aragonite: temperature effects. Chemical Geology (Isotope Geoscience Section) 59, 59–74 (1986).
Kim, S. T. & O’Neil, J. R. Equilibrium and nonequilibrium oxygen isotope effects in synthetic carbonates. Geochimica et Cosmochimica Acta. 61, 3461–3475 (1997).
Ickert, R. B. et al. Determining high precision, in situ, oxygen isotope ratios with a SHRIMP II: Analyses of MPI-DING silicate-glass reference materials and zircon from contrasting granites. Chemical Geology. 257(1), 114–128 (2008).
Long, K. et al. Fish otolith geochemistry, environmental conditions and human occupation at Lake Mungo, Australia. Quaternary Science Reviews. 88, 82–95 (2014).
Geetha, P., Thasneem, P. & Nandan, S., Macrobenthos and Its Relation to Ecosystem Dynamics in the Cochin Estuary, in Lake 2010: Wetlands, Biodiversity and Climate Changes (IISc, Bangalore, 2010), 1–12 (2010).
Author information
Authors and Affiliations
Contributions
P.G. designed the project. P.K. collected the modern sample and Y.B. collected the Cretaceous shell. P.K. and Y.B. made clumped isotope and stable isotope measurements on the modern and Cretaceous shells, respectively. P.G., P.K. and Y.B. interpreted data and wrote the manuscript. I.W. made SHRIMP measurements on the Cretaceous shell and helped write the manuscript. M.K.G. provided the modern analogue coral record for Western Australia and helped write the manuscript. A.C., S.S., Y.B. and P.G. carried out the EBSD study on the Cretaceous shell and interpreted the data.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ghosh, P., Prasanna, K., Banerjee, Y. et al. Rainfall seasonality on the Indian subcontinent during the Cretaceous greenhouse. Sci Rep 8, 8482 (2018). https://doi.org/10.1038/s41598-018-26272-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-26272-0
This article is cited by
-
Oxygen Isotopic Studies of a Species of Pitar (Hyphantosoma) from Quilon Formation, Kerala, Southwest India: Inferences on Seasonality during the Miocene (late Burdigalian)
Journal of the Geological Society of India (2022)
-
Clumped isotope analysis of lacustrine endogenic carbonates and implications for paleo-temperature reconstruction: A case study from Dali Lake
Science China Earth Sciences (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.