Abstract
Carnivorous fish is thought to be high-glucose intolerance. But the reasons were still unclear. The aim of the present study is to investigate the effects of high level of dietary carbohydrate on the survival, growth and immune responses of Paralichthys olivaceus, and the underlying molecular mechanism related to the immune and glucose metabolism. P. olivaceus were fed with 8%, 16% and 24% of dietary carbohydrate for 10 weeks, respectively. After that, a glucose tolerance test (GTT) was conducted. Results showed that excessive (24%) dietary carbohydrate significantly decreased the growth and glucose tolerance ability according to the GTT. It significantly increased hepatic NADPH oxidase activity and malondialdehyde content and serum contents of IL-6 and advanced glycation end products. The expressions of glucose transport-relevant genes in liver and the content of related hormones in serum were analyzed. In conclusion, it was confirmed that IL-6 increased the expression of suppressor of cytokine signaling 3 (SOCS3) and regulated the downstream targets of PI3K-AKT mediated signal transduction, and then downregulated the glucose transporter 2 activity in liver of P. olivaceus fed diet with excessive carbohydrate level. It was suggested that SOCS3 served as a bridge between immune response and glucose metabolism in P. olivaceus.
Similar content being viewed by others
Introduction
Carbohydrate is an excellent energy source for vertebrates, but cannot be fully and efficiently used by fish, especially for carnivorous fish species. Most carnivorous fish have been considered as glucose intorlerance and often display a prolonged postprandial hyperglycemia after oral or injected glucose loading and intake of carbohydrate-enriched diets1,2,3. In addition, fish showed high mortality, reduced growth performance, low nutrient utilization efficiencies and poor physiological functions, when administrated with excessive carbohydrate levels4,5,6,7,8. Many reasons were speculated to explain high-glucose intolerance in carnivorous fish, including a higher sensitivity of insulin to amino acids rather than glucose, inefficiencies in peripheral glucose utilization and absorption, inadequacies in homeostatic glucose regulation and imbalances of endogenous versus exogenous glucose sources9,10,11,12,13. However, there was no clearly conclusion at present.
Pronounced changes in glucose metabolism in fish could be induced by dietary carbohydrate levels. These changes included activities of the key enzymes involved in glucose metabolism, such as pyruvate kinase and glucokinase14,15. In previous study, genes of most of these enzymes have been cloned and characterized in fish. Meanwhile, the regulation of these enzymes at the molecular level by dietary carbohydrate manipulation has been reported16,17,18,19,20. However, most of the previous studies focused on the glycolysis and gluconeogenesis. Little research was reported on the molecular regulation of the upstream pathways.
Previous studies showed that the carnivorous fed high level of dietary carbohydrate could have suppressed immunity21,22. However, only some apparent parameters were analyzed, such as growth performance, mortality and activity of the immune related enzymes23,24. In mammals, it was confirmed that glucose metabolism was related to the immune response of animals, such as anti-oxidative capacity and inflammation25,26,27,28. Production of H2O2 has been shown to regulate insulin release to modulate proximal and distal insulin signaling25,29. At the same time, impaired immunity including ROS and inflammation could also affect inslulin signal pathway through activating numerous intracellular serine kinases and negative regulators of cytokine signaling, such as c-Jun N-terminal kinase (JNK) and suppressor of cytokine signaling (SOCS)30,31,32. Recent years, human diabetes was even regarded as a chronic inflammatory response33. So, it is worth to investigate if the high-glucose intolerance in fish is linked to the impaired immunity.
Japanese flounder (Paralichthys olivaceus) is a typical marine carnivorous fish species. After a 45-day feeing trial with different dietary carbohydrate sources, the previous study found that the Japanese flounder utilized dextrin more efficiently than glucose, and dextrin was a better source of energy than lipid. In that study, however, only the apparent parameters including growth performance, feed utilization and body compositions were analyzed34. The aim of the present study is to investigate the effects of high level of dietary carbohydrate on the survival, growth and immune responses of P. olivaceus, and the underlying molecular mechanism related to the immune and glucose metabolism.
Results
Survival and growth performance
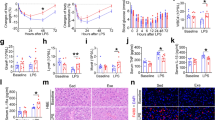
There were no significant differences in survival rate (SR, about 95%) among all the treatments (Fig. 1). With the increase of dietary carbohydrate levels, specific growth rate (SGR) significantly increased in the C16 group, and then decreased in the C24 group (p-value = 0.002). The lowest value of SGR was observed in the C8 group.
Serum parameters after feeding trial
Data of the serum parameters are shown in Table 1. Compared with the C8 and C16 group, contents of total protein (TP), albumin (Alb), globulin (Glo), and advanced glycationend products (AGEs) in serum reached the significant highest values in the group of C24 (TP, p-value < 0.001; Alb, p-value = 0.024; Glo, p-value < 0.001; AGEs, p-value < 0.001). There were no significant differences in the content of tumor necrosis factor (TNF-α) and fasting serum insulin (FINS), activities of aspartate transaminase (AST) in serum among all the groups. The highest concentration of interleukin 6 (IL-6) in serum was found in C24 group, which was significantly higher than that in C16 group (p-value = 0.015). The activities of glutamic-pyruvic transaminase (ALT) and alkaline phosphatase (ALP) in serum significantly increased with dietary carbohydrate levels (ALT, p-value = 0.025; ALP, p-value = 0.045). There was no significant difference in the content of leptin between the group of C16 and C24. Both of them were significantly higher than that in C8 group (P < 0.001). There were no significant differences in concentration of adiponectin in serum among all the three groups.
Analysis of GTT
The fasting serum glucose (FSG) of the three groups were 1.15 ± 0.17, 1.25 ± 0.21 and 1.35 ± 0.12 mmol/L, respectively. In addition, there were no significant differences among all the three groups (Table 2). At the beginning, glucose injection induced a notable increase of the serum glucose level in all the groups, and then resulted in a treatment (C8, C16 and C24) × time (sampling time points after injection) interaction (Fig. 2a). During the test, serum glucose concentrations in all groups were significant higher than the FSG. At the 48th hour after injection, the serum glucose concentration in all the groups returned to the basal level.
Response to glucose tolerance test of Japanese flounder after the 10-week feeding trial. (a) Serum glucose concentrations (b) Serum insulin concentrations. Values at 0 h are presented as mean ± S.D., n = 3 (8 fish/replicate) and the other values represented the means ± S.D., n = 3 (3 fish/replicate). * represents significant difference between C24 and C8 (P < 0.05); ** represents significant difference among all groups, (P < 0.05).
The parameters of fitting equation (equation (3)) are shown in Table 3. Coefficients of all parameters were with 95% confidence bounds. The values of R square in all equations were over 0.84. According to the fitted equation, the max serum glucose concentration (Cmax) of serum peaked at 7.35 ± 0.90, 7.65 ± 0.40, 3.21 ± 0.20 hour with the Cmax at 10.67 ± 0.35, 11.85 ± 0.32, 14.02 ± 0.13. No significant differences were found in areas under the curve of glucose during the 48 h of GTT (AUC48) among all the groups (Table 2). The highest value of clearance rate during the 48 h of GTT (CR48) was found in C16 group (2.38 ± 0.23%/h), and the lowest one was in C24 group (0.27 ± 0.25%/h). Compared with that in C24 group, the time return to basal serum glucose (Tbasal) was significantly shorter in C16 group (33.32 ± 1.61) (p-value = 0.000) and C8 group (37.22 ± 1.17) (p-value = 0.002).
In the regard to the three groups, there were no significant differences in the serum insulin concentrations among all the sampling time points after injection of glucose. And there was no clear trend on the serum insulin concentrations changing with the sampling times (Fig. 2b).
Histological structure and oxidation resistance of liver
Hepatocytes from group C8 and C16 showed normal histology (Fig. 3). In C24 group, some hepatic cell outlines were indistinguishable and some nuclei were dissolved. Obvious hepatocytes swelling, nucleus polarization and lipid vacuolization were observed in C24 group.
The value of total anti-oxidative capacities (T-AOC) in liver in C24 group was significantly lower than those in C8 and C16 groups (Table 4) (p-value = 0.004). The C8 group had a significant higher superoxide dismutase (SOD) activity in liver than C24 group (p-value = 0.007). The C24 group had a significant higher malondialdehyde (MDA) in liver than C8 and C16 groups (p-value = 0.002). C16 group had a significant lower NADPH oxidase (NOX) activity in liver than C24 (p-value = 0.040), and there was no significant difference between C8 and C16.
Gene and protein expression in liver
The gene expressions of glucose utilization-relevant genes are shown in Fig. 4a. The mRNA levels of insulin receptor substrate 2 (IRS2), c-Jun N-terminal kinase 1 (JNK1) and protein kinase B 1 (AKT1) in liver had no significant differences among all the three groups. Gene expressions of the insulin receptor substrate 1 (IRS1) and glucokinase (GK) in C24 group was significantly lower than that in C16 group (IRS1, p-value = 0.039; GK, p-value = 0.003). Compared with C8, the mRNA level of SOCS3 had the significant highest value in C24 group (p-value = 0.014), and the significant lowest value in C16 group (p-value = 0.001)). The mRNA level of phosphoinositide 3-kinase (PI3K), protein kinase B 2 (AKT2), glucose transporter 2 (GLUT2) and phosphofructokinase (PFK) had the significant highest value in C16 group (PI3K, p-value = 0.014; AKT2, p-value = 0.002; GLUT2, p-value = 0.008; PFK, p-value = 0.001), but no differences were found between C8 and C24. The mRNA levels of pyruvate kinase (PK) had the significantly higher and lower value in C16 and C24, respectively (p-value < 0.001). At the same time, phosphorylation of AKT (P-AKT) level in C16 was significantly higher than that in C24 (p-value = 0.009) and the total of AKT (T-AKT) in C16 was significantly higher than other two groups (T-AKT, p-value = 0.032) (Fig. 4b–d).
Gene and protein levels in liver of Japanese flounder after the 10-week feeding trial. (a) Expressions levels of glucose metabolism related genes were normalized to β-actin levels and expressed as relative expression values to those in C8 group. (b) The relative protein abundances of T-AKT and P-AKT in livers were expressed as relative expression values to those in C8 group. (c,d) The bars represent the mean ± S.D., n = 3 (8 fish/replicate). Values with different letters mean significantly different (P < 0.05). Unprocessed original scans of blots are shown in Supplementary Fig. 1.
Discussion
Based on the growth rate, the optimal levels of dietary carbohydrate for the carnivorous species, such as gilthead sea bream, giant croaker, Chinese long snout catfish, were determined less than 20%15,35,36. Instead, compared to that in the carnivorous species, the optimal levels for herbivorous species and omnivore species, such as gibel carp, white sea bream and grass carp, can be higher than 30%, even to 50%37,38,39. Excessive level of dietary carbohydrate could result in negative effects. In the present study, dietary carbohydrate supplementation at 24% led to reduced growth compared with 16% of dietary carbohydrate. Liver is a main metabolic organ. According to the histological analysis, the liver cell of Japanese flounder fed with 24% of dietary carbohydrate potentially indicated the impaired fuction of liver. In addtion, more oxidative damage in the liver of Japanese flounder was found in group with 24% of dietary carbohydrate compared with the other two groups. Based on these data, it was suggested 24% of dietary glucose concentration was overdose for Japanese flounder.
Impaired immunity in fish by overdose of dietary carbohydrate was observed in previous studies. It was reported that high level of dietary carbohydrate reduced plasma IgM and lysozyme levels in European whitefish40. The blood haemoglobin was negatively correlated with dietary carbohydrate levels in Atlantic salmon by long-term feeding of a high carbohydrate diet (20–30%)24. After the 10-week feeding trial, in the present study, the concentration of TP, Alb, Glo, AGEs and IL-6, the activities of ALP and ALT in serum and the content of MDA in liver significantly increased, while the activity of T-AOC and SOD in liver of Japanese flounder significantly decreased by the excessive dietary carbohydrate inclusion at 24%. This suggested that excessive dietary carbohydrate level could increase the oxidative stress and inflammation in Japanese flounder.
A fact was found that the dietary carbohydate intake affected the glucose tolerance in fish, while it seemed to have a poor ability to take care of excess glucose41,42. In mammals, fasting glucose concentration and fasting insulin concentration were used in calculating insulin sensitivity or resistance, in order to evaluate the glucose utilization ability. The relationship between blood glucose concentration in fish and dietary carbohydrate level was inconclusive. Hemre et al. found that blood glucose level in Atlantic salmon was correlated with dietary carbohydrate level43, while some studies reported serum glucose did not differ between the groups fed different dietary carbohydrate levels24,44. Insulin is a major pancreatic endocrine hormone, which regulates blood glucose levels and the underlying metabolic pathways in higher vertebrates. In the present study, FSG was not significantly affected by dietary levels. Meanwhile, the levels of circulating insulin kept the same level in Japanese flounder fed different levels of dietary carbohydrate in this study. This result was in accordance with an earlier study in rainbow trout, in which it was found that the insulin content in plasma of trout fed with the carbohydrate-enriched diet was not significantly increased when compared with that with carbohydrate-free diet45.
In fact, apart from FSG and in FINS, GTT is a more accurate way to evaluate the glucose utilization ability, where AUC of glucose was used to estimate the content of individual loaded glucose, CR and Tbasal were used to judge the speed of glucose disappearance. A lower CR and longer Tbasal with a large AUC were regared as impaired glucose tolerance46,47,48. GTT is required when humans have high fasting glucose levels but do not meet the diagnostic criteria for diabetes. GTT have been conducted in many fish species and is useful for evaluating glucose tolerance, such as rainbow trout46, channel catfish49 and white sturgeon50. In the present study, the mathematical modeling was used to evaluate the ability of glucose utilization during GTT in Japanese flounder after a 10-week feeding trial with different ditary carbohydrate levels. Consistent with the increased SGR, although no significant differences in AUCs were found, the fish fed with 16% of dietary carbohydrate had a significant higher CR and lower Tbasal. According to the data on growth and GTT, it was suggested that the ability of glucose utilization of Japanese flounder was improved when fed with 16% of dietary carbohydrate. And the feeding adapting was helpful for glucose tolerance.
Ability of glucose utilization is related to glucosesensing. Rainbow trout fed a diet with 20% of carbohydrate showed a higher postprandial glucose than that fed carbohydrate-free diet. This showed a positive response to the dietary carbohydrate based on the major components of glucosensing system including glucose transporters (GLUTs)51. Glucose metabolism depends on the intake of glucose. Insulin-induced PI3K-AKT signaling contributes to the regulation of GLUT2-mediated glucose uptake in liver. PI3K and AKT, which were two major nodes downstream of insulin receptor substrate (IRS), have been implicated in many of the metabolic actions of insulin, such as glucose transport. The rate-limiting enzymes in the glycolysis pathways, such as GK, PFK and PK, played key roles in glucose metabolism. Results in the present study showed that the upregulation of PI3K, AKT2, GLUT2, PK and PFK transcription and activies of the total AKT and AKT phosphorylation were found in C16 group. These could be helpful to explain why the Japanes flounder fed with 16% of dietary carbohydrate had better glucose utilization ability as shown by the data on growth and GTT.
Moreover, many studies in mammals have found that leptin and adiponectin could improve the glucose transport52,53,54, eventhough the detail was not clear. Fish definitely do have these hormones with some functional similarities and many other distinct features55. In the present study, there were no significant differences in concentration of adiponectin in serum among all the three groups. However, the concentrations of leptin in groups of C16 and C24 were significantly higher than that in C8 group. It was suggested that an increasing level of leptin not adiponectin improved the glucose transport in Japanese flounder. In the previous study in zebrafish, it was also found that leptin signaling regulates glucose homeostasis56. In the present study, however, Japanese flounder fed with higher dietary carbohydrate (24%) had a longer Tbasal with lower CR, a dereasesd activety of PI3K-AKT and glycolysis pathways, a downregulated GLUT2 transcription than those fed with 16% of dietary carbohydrate. This result implied that excessive dietary carbohydrate inhibited the ability of glucose transportation and glucose catabolism, which was not associated with the content of serum insulin, leptin and adiponectin.
Previous studies suggested many reasons to explain the high-glucose intolerance in fish, and many issues remained under debate. A growing number of studies indicated that the oxidative stress influences the insulin signal pathway in mammals57,58. Although these findings were reported in muscle cell, it is expected that they also occur in hepatocytes because the primary metabolic abnormalities in these cells also induce ROS production, thus affecting glucose utilization. AGEs accumulate under high-glucose conditions and affect ROS producing by NADPH oxidase59,60,61,62. In the present study, it was implied that ROS level increased in organism with a significant up-regulation in AGEs and NADPH oxidase in Japanese flounder fed diet with excessive carbohydrate level (24%). Houstis et al. pointed out that increases in ROS levels precede the onset of insulin resistance (IR) and might be causally linked to it63. It is still not clear exactly how oxidative stress causes insulin resistance or metabolic dysfunction in mammals. The possible mechanism suggested is that ROS could inhibit IRS to regulate the insulin signal pathway by c-Jun N-terminal kinase (JNK)31,32. JNK family members are encoded by three genetic loci, JNK1-3, JNK1 isoform is the major contributor to insulin resistance64. Activation of JNK1 has been shown to directly phosporylate IRS-1 at inhibitory sites that prevent recruitment of this protein to the activated insulin receptor32. In the present study, no significant difference in JNK1 gene expression was found although it was suggested that Japanese flounder fed with 24% of dietary carbohydrate was under oxidative stress. Another fact was found that inflammation can be induced by ROS through the induction of pro-inflammatory cytokines (e.g., TNF-α and IL-6), which are main key factors to affect glucose utilization via inducing liver insulin resistance65,66,67,68,69. TNF-α could also induce JNK to inhibit IRS70. In the present study, the serum TNF-α and hepatic JNK1 gene expressions were not significantly affected by dietary carbohydrate levels, whereas IL-6 significanlty increased in C24 group compared with C16 group. These results indicated that TNF-α was not associated with the direct effect of carbohydrate. In fact, IL-6 could induce suppressor of cytokine signaling 3 (SOCS3) expression to affect insulin signaling71. SOCS3 is highly expressed in the liver and plays an important role in regulating IR72. The overexpression of SOCS3 can reduce insulin secretion by inhibiting preproinsulin gene transcription73. Furthermore, overexpression of SOCS3 decreased the hepatic expressions of IRS1, IRS2 and PI3K, and resulted in glucose intolerance and decrease glucose uptake74,75. In the present study, the observed differences in the gene expression levels of SOCS3 and IRS1 between Japanese flounder fed 24% and 16% carbohydrate. The downregulation gene expressions of downstream PI3K, AKT2, GLUT2, GK, PFK and PK, and total and phosphorylation of AKT level were found in C24 compared with C16. It was indicated that the high level of SOCS3 level of Japanese flounder fed with 24% of dietary carbohydrate inhibited the glucose uptake through decreasing IRS1 gene expression and the downstream PI3K-AKT signal way and thus affected glucose utilization. In addition, energy sensing was another factor could affect glucose utilization, and AMPK was an important energy sensor. In present study, the increasing digestable energy may result in the change of AMPK activity. The activation of AMPK was found to induce glucose uptake in trout myotubes76. In terms of nutritional regulation, high glucose intake was found to reduce the phosphorylation of AMPK in the liver of fish77,78. Moreover, the activity of AMPK was associated with imflammation. The upregulation of AMPK decreased the mRNA expression of proinflammatory79. Based on above researches, it can be speculate that the activity of AMPK decreasd and thus further promoted the inflammation. These data partly explained why the fish fed with 24% of dietary carbohydrate had lower ability of glucose utilization than those fed with 16% of dietary carbohydrate.
In conclusion, dietary carbohydrate is necessary for the growth and immune response of Japanese flounder. Diet with 16% of carbohydrate significantly improved the growth, glucose utilization and immunity of fish. And the feeding adapting was helpful for glucose tolerance. Excessive level (24%) of dietary carbohydrate caused oxidative stress, inflammation and influenced glucose transport. Based on the present data, it was suggested that SOCS3 served as a bridge between immune response and glucose metabolism. IL-6 increased the gene expression of SOCS3 and thus regulated the downstream targets of PI3K-AKT mediated signal transduction that underlie the downregulation of GLUT2 activity, glucose uptake and metabolism in liver. This hypothesis was illustrated in Fig. 5.
Materials and Methods
Experimental diet
Diet formulation and its proximate composition are given in Table 5. Three isonitrogenous and isolipidic diets are formulated with graded levels of carbohydrate (8%, 16% and 24%, respectively). They were named as C8, C16 and C24, respectively. All ingredients were finely ground, well mixed, and dry extruded in a laboratory pellet mill (EL220, Shangdong Haiyang, China). The diameters of the diet particles were 3 mm and 5 mm die. The particles were dried in a forced air oven at 50 °C for 8 h and stored in a refrigerator (−20 °C) until used.
Animals and feeding trial
Japanese flounders were obtained from Haiyang, Shandong Province, China. They (initial body weight: 7.14 ± 0.10 g) were randomly distributed into 9 tanks (3, 000 L) in a flow-through water system supplied with sand filtered seawater at a flow rate of 2.5 L/min. Three tanks were used for one treatment (150 fish/tank). The fish were fed to apparent satiation twice daily (8:00 and 18:00) for 10 weeks. During the feeding trial, the dissolved oxygen content was approximately 7.4 mg/L. Water temperature naturally ranged from 21 to 24 °C. The photoperiod was maintained at intervals of 13-h light: 11-h dark. All procedures performed in study were in strict accordance with the recommendations in the Guide for the Use of Experimental Animals of Ocean University of China. The protocols for animal care and handing used in this study were approved by the Institutional Animal Care and Use Committee of Ocean University of China.
Sampling
At the end of the 10 weeks feeding trial, all the fish were fasted for 24 h after the last feeding and anaesthetized with MS-222 (Sigma, 50 mg L−1 water) before sampling. Fish in each tank were weighed and counted to determine SGR and SR. Eigth fish per tank were sampled to collect blood from coccygeal vertebra vein. The serum was separated and stored at −80 °C until analysis80,81. After that, these fish were dissected to collect liver. And the livers were quick frozen using liquid nitrogen and stored at −80 °C for subsequently analysis.
Glucose tolerance test
At the end of the feeding trial, the serum of fish starved for 24 h was used to measure FSG and FINS concentration, which were setted as a baseline for AUC of glucose calculation and for comparison with insulin content after GTT, respectively. Another 30 anaesthetized fish per tank were used for GTT according to the method of López-Olmeda82. Anaesthetized fish per tank were intraperitoneal injected with glucose at a concentration of 600 mg per kg fish. After that, the blood was taken at 1, 3, 6, 9, 12, 18, 24 and 48 h after injection, respectively, to analyze serum glucose and insulin concentrations. Three fish per tank were used for collecting the serum at each sampling time point. The AUC of glucose, CR, Cmax, Tbasal and the time reach the max serum glucose concentration (Tmax) were calculated.
Serum and liver parameters
The concentrations of glucose, TP, Alb and Glo, and the activities of ALP, ALT and AST in serum were analyzed by the automatic biochemical analyzer (AU5400, Beckman). The concentration of insulin, leptin, adiponectin, IL-6, AGEs and TNF-α in serum were determined using a double antibody sandwich enzyme-linked immunosorbent assay. Kits for insulin, leptin, IL-6 and TNF-α were purchased from Cusabio, China. Kits for adiponectin and AGEs were purchased from Mybiosource, USA. All these kits were developed using antigenic regions completely conserved in fish83,84,85,86. The protein concentration, T-AOC, MDA, and SOD activity in liver were measured by commercial kits (Nanjing Jiancheng Bioengineering Institute, China)87,88,89. And the activity of NOX in liver was detected according to Li et al.89.
Histological analysis
Liver samples were fixed in 10% neutral buffered formalin for 24 h and then transferred to 70% ethanol. Samples were then dehydrated through graded levels of ethanol (70%, 80%, 85%, 90%, 95%, 100%) using a tissue processor, cleared and embedded in paraffin wax (PPDT-12C1, Ceike, China). Transverse sections (~7 μm) were prepared using a rotary microtome (RM2235, Leica, China), mounted onto slides and processed for staining (Thermo Gemini A2, USA) with hematoxylin and eosin (H&E, Nanjing Jiancheng Bioengineering Institute, China). Slides were viewed under light microscopy and photographed with a digital camera (Olympus, DP72, Nikon, Japan).
Real time PCR
Total RNA was extracted from 0.1 g of liver tissue using Trizol Reagent (Invitrogen, USA). It was then quantified and the purity was assessed by spectrophotometry. The 260:280 ratios were 1.8 ~ 2.0. Complementary DNA (cDNA) was synthesized from 1 mg of total RNA using PrimeScript® RT reagent Kit with gDNA Eraser (Takara, Japan). Expressions of the selected genes were analyzed by the real-time PCR, which was performed with an ABI 7500 instrument (Applied Biosystems) using SYBR Green PCR (Takara). The Reaction mixtures (SYBR® Premix Ex TaqTM II (2×) 10.0 μl, Forward Primer (10 μM) 0.8 μl, Reverse Primer (10 μM) 0.8 μl, DNA template 2.0 μl and ddH2O 6.4 μl) were programmed 30 s at 95 °C, followed by 40 cycles of 5 s at 95 °C, 30 s at 58 °C, 30 s at 72 °C, and finally 15 s at 95 °C, 1 min at 60 °C and 15 s at 95 °C. For each mRNA, gene expression was corrected by β-ACTIN in each sample. The data were analyzed by the ΔΔCt method, and the primers used are shown in Table 6.
Western blot analysis
Tissues were homogenized with glass Tenbroeck tissue grinders (Kimble Chase, USA) on ice and lysed with 50 mM Tris·HCl, 150 mM NaCl, 0.5% NP-40, 0.1% SDS, and 1 mM EDTA, pH 7.5, with protease and phosphatase inhibitor cocktails (Roche, Switzerland) at 4 °C for 1 h and cleared by centrifugation at 12,000 g for 20 min. Protein concentrations were determined with a BCA protein assay kit (Beyotime Biotechnology, China) using bovine serum albumin as standard. Protein samples (30 μg protein per lane) were separated by SDS-PAGE and transferred to 0.45 μm PVDF membrane (Millipore, USA) for Western analysis. The membrane was blocked with 5% nonfat milk in TBST buffer (20 mM Tris·HCl, 500 mM NaCl, 0.1% Tween 20) for 1 h at room temperature and incubated with primary antibody overnight at 4 °C before horseradish peroxidase (HRP)-conjugated secondary antibodies were added and visualized using ECL reagents (GoodHere, China). The following antibodies were used: antibodies against protein kinase B (AKT, 9272), phospho-AKT (Ser473, 9271) and β-tubulin (2146) were purchased from Cell Signaling Technology. All these antibodies were developed using antigenic regions completely conserved in fish, and could be used in scophthalmidae turbot90. The Western bands were quantified using NIH Image 1.63 software.
Calculation and statistical analysis
The following parameters were calculated as:
The PROC NLIN was used to fit exponential curves for glucose concentration during 48 hours GTT, using the following equation performed by Metlab2017:
where F (t) was the metabolite concentration at time t (hour); the method was modified based on Pires et al.91. CR (%/h), Cmax (mmol/L), Tbasal (h) and Tmax (h) were calculated as the actual equation. CR was calculated as: clearance rate
where [ta] was the concentration of glucose at time a (ta), and [tb] was the concentration of glucose at time b (tb). The AUC was calculated using the trapezoidal method and actual concentration values, after discounting the baseline concentration (FSG, calculated by averaging value before injection)87.
All statistical evaluations were analyzed using the software SPSS 19.0. The growth parameters, immune parameters and gene expression were analyzed using one-way analysis of variance (ANOVA) followed by Tukey’s multiple range tests. The effects of time and diets and their interactions were analyzed by two-way ANOVA. Tukey’s multiple range tests were used to examine treatment differences among the interactions. When the interaction was significant, the results were further analyzed using one-way ANOVA and Tukey’s multiple range test. In case unequal variance was determined by Levene’s test, the data were square root-transformed before statistical analysis. Differences were regarded as significant when P < 0.05. All data were expressed as means ± standard deviations.
Data availability
The datasets generated and analysed during the current study are vailable from the corresponding author on reasonable request.
References
Polakof, S., Panserat, S., Soengas, J. L. & Moon, T. W. Glucose metabolism in fish, a review. Journal of Comparative Physiology B. 182, 1015–1045 (2012).
Kamalam, B. S., Medale, F. & Panserat, S. Utilisation of dietary carbohydrates in farmed fishes: New insights on influencing factors, biological limitations and future strategies. Aquaculture. 467, 3–27 (2017).
Stone, D. A. J. Dietary carbohydrate utilization by fish. Rev. Fish. Sci. 11, 337–370 (2003).
Hilton, J. W. & Atkinson, J. L. Response of rainbow trout (Salmo gairdneri) to increased levels of available carbohydrate in practical trout diets. Br J Nutr. 47, 597–607 (1982).
Li, X. F., Wang, Y., Liu, W. B., Jiang, G. Z. & Zhu, J. Effects of dietary carbohydrate/lipid ratios on growth performance, body composition and glucose metabolism of fingerling blunt snout bream Megalobrama amblycephala. Aquacult Nutr. 19, 701–708 (2013).
Ren, M., Ai, Q., Mai, K., Ma, H. & Wang, X. Effect of dietary carbohydrate level on growth performance, body composition, apparent digestibility coefficient and digestive enzyme activities of juvenile cobia, Rachycentron canadum L. Aquaclture Research. 42, 1467–1475 (2011).
Wang, J. et al. Effects of different dietary carbohydrate levels on growth, feed utilization and body composition of juvenile grouper Epinephelus akaara. Aquaculture. 459, 143–147 (2016).
Furuichi, M. & Yone, Y. Change of blood sugar and plasma insulin levels of fishes [carp, cyprinus carpio, red sea bream, pagrus major, yellowtail, seriola quinqueradiata] in glucose tolerance test. Nippon Suisan Gakk. 47, 761–764 (1981).
Austreng, E., Risa, S., Edwards, D. J. & Hvidsten, H. Carbohydrate in rainbow trout diets. II. Influence of carbohydrate levels on chemical composition and feed utilization of fish from different families. Aquaculture. 11, 39–50 (1977).
Harpaz, S. & Uni, Z. Activity of intestinal mucosal brush border membrane enzymes in relation to the feeding habits of three aquaculture fish species. Comp Biochem Physiol A Mol Integr Physiol. 124, 155–60 (1999).
Hemre, G., Mommsen, T. P. & Krogdahl, A. Carbohydrates in fish nutrition, effects on growth, glucose metabolism and hepatic enzymes. Aquacult Nutr. 8, 175–194 (2002).
Enes, P., Panserat, S., Kaushik, S. & Oliva-Teles, A. Nutritional Regulation of Hepatic Glucose Metabolism in Fish. Fish Physiol Biochem. 35, 519–539 (2009).
Kirchner, S., Panserat, S., Lim, P. L., Kaushik, S. & Ferraris, R. P. The role of hepatic, renal and intestinal gluconeogenic enzymes in glucose homeostasis of juvenile rainbow trout. J. Comp Physiol B 178, 429–438 (2008).
Meton, I. et al. Effect of diet composition and ration size on key enzyme activities of glycolysis-gluconeogenesis, the pentose phosphate pathway and amino acid metabolism in liver of gilthead sea bream (Sparus aurata). Br J Nutr. 82, 223–232 (1999).
Caseras, A. et al. Nutritional regulation of glucose-6-phosphatase gene expression in liver of the gilthead sea bream (Sparus aurata). Br J Nutr. 88, 607–614 (2002).
Blin, C. et al. Teleost liver hexokinase- and glucokinase-like enzymes, partial cDNA cloning and phylogenetic studies in rainbow trout (Oncorhynchus mykiss), common carp (Cyprinus carpio) and gilthead seabream (Sparus aurata)+. Fish Physiol Biochem. 21, 93–102 (1999).
Leung, L. Y. & Woo, N. Y. S. Effects of growth hormone, insulin-like growth factor I, triiodothyronine, thyroxine, and cortisol on gene expression of carbohydrate metabolic enzymes in sea bream hepatocytes. Comp Biochem Physiol A Mol Integr Physiol. 157, 272–282 (2010).
Panserat, S. et al. Molecular cloning, tissue distribution and sequence analysis of complete glucokinase cDNAs from gilthead seabream (Sparus aurata), rainbow trout (Oncorhynchus mykiss) and common carp (Cyprinus carpio). Biochim Biophys Acta. 1474, 61–69 (2000).
Panserat, S. et al. Glucokinase is highly induced and glucose-6-phosphatase poorly repressed in liver of rainbow trout (Oncorhynchus mykiss) by a single meal with glucose. Comp Biochem Physiol B Biochem Mol Biol. 128, 275–283 (2001).
Panserat, S., Plagnes-Juan, E. & Kaushik, S. Gluconeogenic enzyme gene expression is decreased by dietary carbohydrates in common carp (Cyprinus carpio) and gilthead seabream (Sparus aurata). Biochim Biophys Acta. 1579, 35–42 (2002).
Pieper, A. & Pfeffer, E. Studies on the comparative efficiency of utilization of gross energy from some carbohydrates, proteins and fats by rainbow trout (Salmo gairdneri, R.). Aquaculture. 20, 323–332 (1980).
Pickering, A. D. & Pottinger, T. G. Stress responses and disease resistance in salmonid fish, Effects of chronic elevation of plasma cortisol. Fish Physiol Biochem. 7, 253–258 (1989).
Page, G. I., Hayworth, K. M., Wade, R. R., Harris, A. M. & Bureau, D. P. Non-specific immunity parameters and the formation of advanced glycosylation end products (AGE) in rainbow trout, Oncorhynchus mykiss (Walbaum), fed high levels of dietary carbohydrates. Aquaclture Research. 30, 287–297 (1999).
Waagbo, R., Glette, J., Sandnes, K. & Hemre, G. I. Influence of dietary carbohydrate on blood chemistry, immunity and disease resistance in Atlantic salmon, Salmo salar L. J Fish Dis. 17, 245–258 (1994).
Goldstein, B. J., Mahadev, K., Wu, X., Zhu, L. & Motoshima, H. Role of insulin-induced reactive oxygen species in the insulin signaling pathway. Antioxid Redox Signal. 7, 1021–1031 (2005).
Bokov, A., Chaudhuri, A. & Richardson, A. The role of oxidative damage and stress in aging. Mech Ageing Dev. 125, 811–826 (2004).
Cai, D. et al. Local and systemic insulin resistance resulting from hepatic activation of IKK-beta and NF-kappaB. Nat Med. 11, 183–190 (2005).
Quan, Y. et al. High glucose stimulates TNF-α and MCP-1 expression in rat microglia via ROS and NF-KB pathways. Acta Pharmacol Sin. 32, 188–193 (2011).
Pi, J. et al. Reactive oxygen species as a signal in glucose-stimulated insulin secretion. Diabetes. 56, 1783–1791 (2007).
Rui, L., Yuan, M., Frantz, D., Shoelson, S. & White, M. F. SOCS-1 and SOCS-3 block insulin signaling by ubiquitin-mediated degradation of IRS1 and IRS2. J Biol Chem. 277, 42394–42398 (2002).
Hotamisligil, G. K. S. Inflammation and metabolic disorders. Nature. 444, 860–867 (2006).
Aguirre, V., Uchida, T., Yenush, L., Davis, R. & White, M. F. The c-Jun NH2-terminal Kinase Promotes Insulin Resistance during Association with Insulin Receptor Substrate-1 and Phosphorylation of Ser307. J Biol Chem. 275, 9047–9054 (2000).
Fernández-Real, J. M. & Pickup, J. C. Innate immunity, insulin resistance and type 2 diabetes. Trends in Endocrinology & Metabolism. 19, 10–16 (2008).
Lee, S., Kim, K. & Lall, S. P. Utilization of glucose, maltose, dextrin and cellulose by juvenile flounder (Paralichthys olivaceus). Aquaculture. 221, 427–438 (2003).
Tan, Q., Xie, S., Zhu, X., Lei, W. & Yang, Y. Effect of dietary carbohydrate‐to‐lipid ratios on growth and feed utilization in Chinese longsnout catfish (Leiocassis longirostris Günther). J Appl Ichthyol. 23, 605–610 (2007).
Fernández, F. et al. Effects of diets with distinct protein-to-carbohydrate ratios on nutrient digestibility, growth performance, body composition and liver intermediary enzyme activities in gilthead sea bream (Sparus aurata, L.) fingerlings. J Exp Mar Biol Ecol. 343, 1–10 (2007).
Sa, R., Pousao-Ferreira, P. & Oliva-Teles, A. Growth performance and metabolic utilization of diets with different protein,carbohydrate ratios by white sea bream (Diplodus sargus, L.) juveniles. Aquaclture Research. 38, 100–105 (2007).
NRC. Nutrient requirements of fish and Shrimp. (ed. National Academic Press) (Washington 2011).
Li, X. et al. Carbohydrate utilization by herbivorous and omnivorous freshwater fish species, a comparative study on gibel carp (Carassius auratus gibelio. var CAS III) and grass carp (Ctenopharyngodon idellus). Aquaclture Research. 47, 128–139 (2016).
Vielma, J., Koskela, J., Ruohonen, K., Jokinen, I. & Kettunen, J. Optimal diet composition for European whitefish (Coregonus lavaretus), carbohydrate stress and immune parameter responses. Aquaculture. 225, 3–16 (2003).
Mazur, C. N., Higgs, D. A., Plisetskaya, E. & March, B. E. Utilization of dietary starch and glucose tolerance in juvenile Chinook salmon (Oncorhynchus tshawytscha) of different strains in seawater. Fish Physiol Biochem. 10, 303–313 (1992).
Hilton, J. W., Plisetskaya, E. M. & Leatherland, J. F. Does oral 3,5,3′-triiodo-l-thyronine affect dietary glucose utilization and plasma insulin levels in rainbow trout (Salmo gairdneri)? Fish Physiol Biochem. 4, 113–120 (1987).
Hemre, G. I., WaagbØ, R., Hjeltnes, B. & Aksnes, A. Effect of gelatinized wheat and maize in diets for large Atlantic salmon (Salmo salar L.) on glycogen retention, plasma glucose and fish health. Aquacult Nutr. 2, 33–39 (1996).
Hung, S. S., Fynn-Aikins, F. K., Lutes, P. B. & Xu, R. P. Ability of juvenile white sturgeon (Acipenser transmontanus) to utilize different carbohydrate sources. The Journal of nutrition. 119, 727 (1989).
Capilla, E. et al. Muscle insulin binding and plasma levels in relation to liver glucokinase activity, glucose metabolism and dietary carbohydrates in rainbow trout. Regul Peptides. 110, 123–132 (2003).
Harmon, J. S., Eilertson, C. D., Sheridan, M. A. & Plisetskaya, E. M. Insulin suppression is associated with hypersomatostatinemia and hyperglucagonemia in glucose-injected rainbow trout. Am J Physiol. 261, R609–R613 (1991).
Legate, N. J., Bonen, A. & Moon, T. W. Glucose tolerance and peripheral glucose utilization in rainbow trout (Oncorhynchus mykiss), American eel (Anguilla rostrata), and black bullhead catfish (Ameiurus melas). Gen Comp Endocrinol. 122, 48–59 (2001).
Wilson, R. P. & Poe, W. E. Apparent inability of channel catfish to utilize dietary mono- and disaccharides as energy sources. J Nutr. 117, 280–285 (1987).
Ottolenghi, C., Puviani, A. C., Ricci, D., Brighenti, L. & Morsiani, E. The effect of high temperature on blood glucose level in two teleost fish (Ictalurus melas and Ictalurus punctatus). Comparative Biochemistry and Physiology Part A: Physiology. 111, 229–235 (1995).
Gisbert, E., Sainz, R. D. & Hung, S. S. O. Glycemic responses in white sturgeon after oral administration of graded doses of D-glucose. Aquaculture. 224, 301–312 (2003).
Polakof, S., Panserat, S., Plagnes-Juan, E. & Soengas, J. L. Altered dietary carbohydrates significantly affect gene expression of the major glucosensing components in Brockmann bodies and hypothalamus of rainbow trout. AJP: Regulatory, Integrative and Comparative Physiology. 295, R1077–R1088 (2008).
Fruebis, J. et al. Proteolytic cleavage product of 30-kDa adipocyte complement-related protein increases fatty acid oxidation in muscle and causes weight loss in mice. Proc Natl Acad Sci USA 98, 2005–2010 (2001).
Barzilai, N. et al. Leptin selectively decreases visceral adiposity and enhances insulin action. J Clin Invest. 100, 3105–3110 (1997).
Berg, A. H., Combs, T. P., Du, X., Brownlee, M. & Scherer, P. E. The adipocyte-secreted protein Acrp30 enhances hepatic insulin action. Nat Med. 7, 947–953 (2001).
Moon, T. W. Hormones and fish hepatocyte metabolism, “the good, the bad and the ugly!”. Comp Biochem Physiol B Biochem Mol Biol. 139, 335–345 (2004).
Michel, M., Page-McCaw, P. S., Chen, W. & Cone, R. D. Leptin signaling regulates glucose homeostasis, but not adipostasis, in the zebrafish. Proceedings of the National Academy of Sciences. 113, 3084–3089 (2016).
Maddux, B. A. et al. Protection against oxidative stress-induced insulin resistance in rat L6 muscle cells by mircomolar concentrations of alpha-lipoic acid. Diabetes. 50, 404–410 (2001).
Gardner, C. D. et al. Hydrogen peroxide inhibits insulin signaling in vascular smooth muscle cells. Exp Biol Med (Maywood). 228, 836–842 (2003).
Moore, T. C. et al. The role of advanced glycation end products in retinal microvascular leukostasis. Invest Ophthalmol Vis Sci. 44, 4457–4464 (2003).
Coughlan, M. T. et al. RAGE-induced cytosolic ROS promote mitochondrial superoxide generation in diabetes. J Am Soc Nephrol. 20, 742–752 (2009).
Wautier, M. P. et al. Activation of NADPH oxidase by AGE links oxidant stress to altered gene expression via RAGE. Am J Physiol Endocrinol Metab. 280, E685–E694 (2001).
Brownlee, M. Glycation products and the pathogenesis of diabetic complications. Diabetes Care. 15, 1835–1843 (1992).
Houstis, N., Rosen, E. D. & Lander, E. S. Reactive oxygen species have a causal role in multiple forms of insulin resistance. Nature. 440, 944–948 (2006).
Hirosumi, J. et al. A central role for JNK in obesity and insulin resistance. Nature. 420, 333–336 (2002).
Codoner-Franch, P. & Alonso-Iglesias, E. Resistin, insulin resistance to malignancy. Clin Chim Acta. 438, 46–54 (2015).
Cairns, R. A., Harris, I. S. & Mak, T. W. Regulation of cancer cell metabolism. Nat Rev Cancer. 11, 85–95 (2011).
Park, J. et al. Increase in glucose-6-phosphate dehydrogenase in adipocytes stimulates oxidative stress and inflammatory signals. Diabetes. 55, 2939–2949 (2006).
Rahman, I., Gilmour, P. S., Jimenez, L. A. & MacNee, W. Oxidative stress and TNF-alpha induce histone acetylation and NF-kappaB/AP-1 activation in alveolar epithelial cells, potential mechanism in gene transcription in lung inflammation. Mol Cell Biochem. 234–235, 239–248 (2002).
Muse, E. D., Lam, T. K., Scherer, P. E. & Rossetti, L. Hypothalamic resistin induces hepatic insulin resistance. J Clin Invest. 117, 1670–1678 (2007).
Kamata, H. et al. Reactive oxygen species promote TNF alpha-induced death and sustained JNK activation by inhibiting MAP kinase phosphatases. Cell. 120, 649–661 (2005).
Kim, J. H., Kim, J. E., Liu, H. Y., Cao, W. & Chen, J. Regulation of interleukin-6-induced hepatic insulin resistance by mammalian target of rapamycin through the STAT3-SOCS3 pathway. J Biol Chem. 283, 708–715 (2008).
Emanuelli, B. et al. SOCS-3 inhibits insulin signaling and is up-regulated in response to tumor necrosis factor-alpha in the adipose tissue of obese mice. J Biol Chem. 276, 47944–47949 (2001).
Laubner, K. et al. Inhibition of preproinsulin gene expression by leptin induction of suppressor of cytokine signaling 3 in pancreatic beta-cells. Diabetes. 54, 3410–3417 (2005).
Lebrun, P. & Van Obberghen, E. SOCS proteins causing trouble in insulin action. Acta Physiol (Oxf). 192, 29–36 (2008).
Michael, M. D. et al. Loss of insulin signaling in hepatocytes leads to severe insulin resistance and progressive hepatic dysfunction. Mol Cell. 6, 87–97 (2000).
Magnoni, L. J. et al. Effects of sustained swimming on the red and white muscle transcriptome of rainbow trout (Oncorhynchus mykiss) fed a carbohydrate-rich diet. Comp. Biochem. Physiol. Part A: Mol. Integr. Physiol. 166, 510–521 (2013).
Kamalam, B. S. et al. Regulation of metabolism by dietary carbohydrates in two lines of rainbow trout divergently selected for muscle fat content. J. Exp. Biol. 215, 2567–2578 (2012).
Xu, C. et al. Interactions between dietary carbohydrate and metformin: Implications on energy sensing, insulin signaling pathway, glycolipid metabolism and glucose tolerance in blunt snout bream Megalobrama amblycephala. Aquaculture. 483, 183–195 (2018).
Huang, B. P. et al. AMPK activation inhibits expression of proinflammatory mediators through downregulation of PI3K/p38 MAPK and NF-κB signaling in murine macrophages. DNA and Cell Biology. 34, 133–141 (2015).
Leung, L. Y. & Woo, N. Y. Influence of dietary carbohydrate level on endocrine status and hepatic carbohydrate metabolism in the marine fish Sparus sarba. Fish Physiol Biochem. 38, 543–554 (2012).
Saeidi, A. M., Adel, M., Caipang, C. & Dawood, M. Immunological responses and disease resistance of rainbow trout (Oncorhynchus mykiss) juveniles following dietary administration of stinging nettle (Urtica dioica). Fish Shellfish Immunol. 71, 230–238 (2017).
López-Olmeda, J. F., Egea-Álvarez, M. & Sánchez-Vázquez, F. J. Glucose tolerance in fish, is the daily feeding time important? Physiol Behav. 96, 631–636 (2009).
Babaei, S., Abedian Kenari, A., Hedayati, M. & Yazdani Sadati, M. A. Growth response, body composition, plasma metabolites, digestive and antioxidant enzymes activities of Siberian sturgeon (Acipenser baerii, Brandt, 1869) fed different dietary protein and carbohydrate, lipid ratio. Aquaclture Research. 48, 2642–2654 (2017).
Madera-Sandoval, R. L. et al. Fat-associated lymphoid cluster in Cyprinus carpio, Characterisation and its relation with peritoneal haemangiosarcoma. Fish Shellfish Immun. 44, 633–641 (2015).
Kousoulaki, K. et al. Metabolism, health and fillet nutritional quality in Atlantic salmon (Salmo salar) fed diets containing n-3-rich microalgae. J Nutr Sci. 4, e24 (2015).
Birhanu, B. T. et al. Immunomodulation of Lactobacillus pentosus PL11 against Edwardsiella tarda infection in the head kidney cells of the Japanese eel (Anguilla japonica). Fish Shellfish Immunol. 54, 466–472 (2016).
Zhang, X., Zhu, Y., Cai, L. & Wu, T. Effects of fasting on the meat quality and antioxidant defenses of market-size farmed large yellow croaker (Pseudosciaena crocea). Aquaculture. 280, 136–139 (2008).
Ding, Z., Li, W., Huang, J., Yi, B. & Xu, Y. Dietary alanyl-glutamine and vitamin E supplements could considerably promote the expression of GPx and PPARα genes, antioxidation, feed utilization, growth, and improve composition of juvenile cobia. Aquaculture. 470, 95–102 (2017).
Li, M., Wang, J., Song, S. & Li, C. Molecular characterization of a novel nitric oxide synthase gene from Portunus trituberculatus and the roles of NO/O2 −- generating and antioxidant systems in host immune responses to Hematodinium. Fish Shellfish Immun. 52, 263–277 (2016).
Bian, F. et al. Dietary gossypol suppressed postprandial TOR signaling and elevated ER stress pathways in turbot (Scophthalmus maximus L.). American Journal of Physiology Endocrinology And Metabolism. 312, E37–E47 (2017).
Pires, J. A. A., Souza, A. H. & Grummer, R. R. Induction of hyperlipidemia by intravenous infusion of tallow emulsion causes insulin resistance in Holstein cows. J Dairy Sci. 90, 2735–2744 (2007).
Acknowledgements
The authors thank the participants who contribute to this trial. This work was financially supported by National Natural Science Foundation of China (Grant No. 31572628), the National Basic Research Program of China (973 program, Grant No. 2014CB138600), and Key R & D Program of Shandong Province, China (2016CYJS04A01).
Author information
Authors and Affiliations
Contributions
K.D. completed the experiment and prepared the manuscript. M.P., J.L., M.Y., Z.G., Y.Z., G.L. and D.L. analyzed the samples. W.Z. designed the experiment and revised the manuscript. K.M. designed the experiment. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Deng, K., Pan, M., Liu, J. et al. Chronic stress of high dietary carbohydrate level causes inflammation and influences glucose transport through SOCS3 in Japanese flounder Paralichthys olivaceus. Sci Rep 8, 7415 (2018). https://doi.org/10.1038/s41598-018-25412-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-25412-w
This article is cited by
-
New insights on intestinal microorganisms and carbohydrate metabolism in fish
Aquaculture International (2024)
-
Effects of parental nutritional programming on the hatching parameters, metabolome, glucose metabolism-related gene expressions, and global DNA methylation in larvae of yellow catfish Tachysurus fulvidraco
Aquaculture International (2024)
-
The protective role of daidzein in intestinal health of turbot (Scophthalmus maximus L.) fed soybean meal-based diets
Scientific Reports (2021)
-
Dietary carbohydrates influence muscle texture of olive flounder Paralichthys olivaceus through impacting mitochondria function and metabolism of glycogen and protein
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.