Abstract
Andropogon gayanus Kunth. is a large African tussock grass invading Australia’s tropical savannas. Invasion results in more intense fires which increases the mortality rate of adult woody plants. Invasion may also affect community structure by altering the recruitment potential of woody plants. We investigated the effects of A. gayanus invasion on ground-level microclimate, and the carbon assimilation potential and recruitment potential of two Eucalyptus species. We compared microclimatic variables from the early wet-season and into the mid-dry season to coincide with the period of growth of A. gayanus. We assessed Eucalyptus recruitment by monitoring seedling establishment, growth and survival of experimentally sown seed, and estimating seedling density resulting from natural recruitment. A. gayanus invasion was associated with increased grass canopy height, biomass and cover. Following invasion, the understorey microclimate had significantly reduced levels of photon flux density, increased air temperatures and vapour pressure deficit. The conditions were less favourable for woody seedling with aboveground biomass of seedlings reduced by 26% in invaded plots. We estimated that invasion reduced daily carbon assimilation of woody seedlings by ~30% and reduced survivorship of Eucalyptus seedlings. Therefore, A. gayanus invasion reduces recruitment potential, contributing to the transformation of savanna to a grassland ecosystem.
Similar content being viewed by others
Introduction
Invasive alien grasses threaten the structure and function of many of the world’s tropical savanna ecosystems1,2,3,4. Ecological impacts include change in the plant community composition and structure, changes in nutrient cycling and altered fire regimes4,5,6,7,8. Invasive plants can affect regeneration of native plants by suppressing seed germination and altering the ground layer so that seedling establishment and survival are reduced9,10,11. The alien C4 perennial grass Andropogon gayanus (gamba grass) is invading large areas of the tropical savanna ecosystems of northern Australia2,12. A. gayanus has higher rates of stomatal conductance, assimilation and water use; a longer daily assimilation period and growing season; and higher photosynthetic nitrogen use efficiency than the native grass species13. Coupled with fire tolerance in a highly flammable ecosystem, these factors confer a significant competitive advantage over native grass species. The rapid invasion and high impact of this species is reflected by its status as one of Australia’s 32 Weeds of National Significance14.
A. gayanus differs structurally from Australia’s native savanna grass species as it forms taller tussocks (typically 2–5 m c.f. 0.5–3 m for native grasses) with a larger basal area (up to 70 cm diameter c.f. up to 20 cm for native grasses). Early invasion is characterised by a change in the structure of the ground layer as A. gayanus tussocks displace the native species15. As invasion proceeds, a near complete replacement of the native understorey species occurs, resulting in significantly increased fuel loads and fires that are up to five times more intense than native grass fires8,16. This can ultimately lead to a reduction in the cover and density of the overstorey trees12,17. This is a significant change in this tropical savanna ecosystem which is characterised by frequent fire, generally occurring at intervals of 1–3 years18.
In addition to altering the community structure via increased tree and shrub mortality, A. gayanus invasion may alter the recruitment potential of woody species. Recruitment of woody seedlings depends on the supply of water, light and nutrients relative to the demand for these resources from herbaceous competitors19. Grass species vary in their level of resource use and changes in grass composition can affect the availability of resources to woody seedlings20,21. Changes in resource availability and stand structure affect microclimatic variables at ground level, such as the solar radiation regime (both quantity and spectral composition of radiation), soil and air temperature, humidity and wind dynamics22,23,24. Ground-level microclimate has a strong influence on seedling recruitment and changes caused by exotic species have affected the germination, growth and survival of woody seedlings in a range of ecosystems20,25,26,27,28.
The aim of this study was to investigate whether A. gayanus invasion affects: (1) ground-level microclimate; and (2) the establishment, growth and survival of woody seedlings. Our study site contained ‘invaded’ plots, which were at the early stage of invasion with a dense A. gayanus understorey but with a relatively intact native woody species overstorey12,15, and ‘native grass’ plots which did not contain A. gayanus in the grass layer. We hypothesised that dense A. gayanus invasion would alter both soil and near-ground air temperature and humidity and thus vapour pressure deficit (VPD), reduce incident radiation at ground level and reduce soil moisture during the late wet season when the grass canopy reaches maturity. If changes in microclimate are detected, we would expect to also detect a reduction in recruitment, growth rate, and survival of Eucalyptus seedlings in invaded and native grass dominated plots.
Results
Vegetation structure
The invasion by A. gayanus resulted in marked change in the structure of the savanna ground layer, particular by the mid-wet season. In November, at the beginning of the wet season, when ground-layer species are germinating and establishing prior to their main growth period29,30, the grass cover (10–15%) did not vary significantly between native and invaded savanna (Table 1). By February, grass cover was significantly higher in the invaded plots (30%) compared to the native grass plots (15%) and this continued throughout the wet season and into the mid-dry season (Table 1). The grass layer was significantly taller in invaded plots throughout the wet season (Table 1) and by the mid-dry season (July), mean grass height was nearly an order of magnitude higher in the invaded plots (2.1 m compared to 0.39 m native grass; Table 1). The invaded plots had a significantly lower forb cover in February (~20% c.f. ~40% in native plots) and April (~9% c.f. ~25% in native plots), by which time forb species had begun to senesce and covered less than 5% of the quadrats by July (Table 1). The sites used in this study were in the early stage of invasion, defined by the alternative state in which the overstory remains intact but the native understory is significantly displaced by A. gayanus9,15.
Microclimate
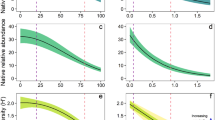
The change in ground layer structure was correlated with change in a range of microclimate characteristics. As grass cover and height increased during the wet season, the percentage of light reaching ground level (photon flux density (PFD)) declined from 70% in November to 32.6% in April in the invaded plots, compared to a decline of 68.5% to 60.6% over the same time period in the native plots (Table 2); the decline was strongly correlated with grass cover (Fig. 1). PFD at ground level did not vary significantly between grass types in November but was significantly lower in the invaded plots compared to native grass plots in February and April (Table 2). The daytime mean (i.e. between 0900 and 1700 hr) air temperature within the grass layer (daytime mean Tair) showed a similar pattern of change in light transmission. In November, there was no significant difference in daytime mean Tair between grass types, however from the mid-wet season to mid-dry season, the daytime mean Tair within invaded plots was 0.8 °C to 1.5 °C higher than native grass plots (Table 2). Similarly, the mean Tair at midday was significantly higher at all times of the year in invaded plots, increasing from a mean difference of 0.8 °C in November to 2.4 °C by April (Table 2). By July, midday temperatures had decreased by 1.9 °C. Mean Tair at midday correlated with grass cover (Fig. 2a). The nocturnal (i.e. 1900–0700 hr) Tair were not significantly different between invaded and native plots for most of the year with the only significant difference occurring in November (invaded plots 27.34 ± 0.2 °C c.f. gamba plots 26.94 ± 0.22 °C). We present the typical diurnal patterns of PFD and Tair for the two grass types in February, the peak growing season (Fig. 3). These patterns demonstrate the low radiation incident at 10 cm in invaded plots, with Tair elevated by a minimum of 1 °C between 11 to 2 pm, and a maximum difference of 2 °C at solar noon (1300hrs local time).
Typical diurnal patterns of (a) PFD and (b) Tair beneath gamba and native grass canopies. Data are composite means using diurnal observations from all sites. On panel (b), the solid line is the optimal leaf temperature for Eucalyptus seedling photosynthesis28.
The vapour pressure deficit (VPD) in the grass layer followed a similar trend to Tair, with daytime VPD being significantly higher in invaded plots compared to native plots during November, April and July (Table 2). Midday VPD was also significantly higher in invaded plots for these 3 months. Nocturnal mean VPD was significantly lower in invaded plots during November but did not differ significantly for the remainder of the wet and dry season.
We estimated the potential woody seedling assimilation (Apot) based on previously established relationships between Eucalyptus seedling photosynthesis and light and temperature31. We used these functions to estimate the impacts of grass type on Apot using diurnal PFD and Tair data for each grass type (Fig. 3). The daily integral of PFD beneath each canopy was 31% lower under gamba (11.4 ± 0.6 mol m−2 d−1) compared to native grass canopy (17.6 ± 2.6 mol m−2 d−1), which translated into a 27% reduction in Apot (1-Way ANOVA, df = 1, F = 13.4, P < 0.01). At the end of the growing season, a similar and significant decline in biomass of E. miniata seedlings in invaded plots was also observed (24%, Table 3). Supra-optimal temperatures for photosynthesis beneath gamba canopies were evident. Figure 3b provides mean diurnal curves from the plot pairs with the optimal temperature for leaf-scale photosynthesis in E. tetrodonta seedlings (~33 °C31) also plotted. Beneath gamba canopies, supra-optimal temperatures occurred for 5 hours of the day and Tair was up to 6 °C above the optimal temperature for photosynthesis. In native sites, temperatures were 3 °C above the optimal. Estimating Apot using these temperatures resulted in a lower Apot (1-WayANOVA, df = 1, F = 8.51, P < 0.01) within invaded sites, although this effect on Apot was not as large when driven by reduced radiation, with mean Apot only 4% lower in invaded plots.
We found that volumetric soil moisture θv was consistently higher in invaded plots compared to native grass plots (Fig. 4) in November (F1,11 = 4.76, P < 0.05) and February (F1,11 = 26.33, P < 0.001). By April, measurements still showed a strong trend towards being higher in invaded plots (F1,3 = 9.83, P = 0.052), although the difference was reduced by May (Fig. 4).
Seedling recruitment
There was no significant difference in seedling emergence between treatments for either E. miniata or E. tetrodonta. Mean emergence for E. miniata was higher in invaded plots with 44% of seeds emerging compared to 38% in native plots. Mean emergence of E. tetrodonta was 11% for both treatments. The ‘control’ (no seed augmentation) plots showed that no natural recruitment occurred in either plot type. Overall emergence of E. tetrodonta was significantly lower than E. miniata (F1,2 = 72.0, P < 0.05). Similarly, there was little variation in predation rates between treatments, except in December, with lower rates observed in invaded plots (F1,2 = 25.0, P < 0.05).
Despite differences in microclimate, there was no significant difference in height or number of leaves per seedling between grass treatments for either tree species. However, there was a significant difference in the above-ground biomass for E. miniata seedlings; mean above-ground biomass was 0.165 g dry weight in invaded plots compared to 0.223 g in native plots (F1,14 = 17.36, P < 0.001). Above-ground biomass for E. miniata in May was negatively associated with mean grass cover of monthly measurements recorded from December to May (r2 = −0.60, P < 0.01; df = 18). Despite differences in above-ground biomass, there was no significant difference in mean seedling lignotuber weight in invaded or native plots.
Seedling survival to the early dry season (May) varied significantly between grass treatment types (F1,21 = 4.60, P = 0.044) for both tree species with a reduction of 11% survival in invaded plots. Survival of E. miniata seedlings during the wet season was negatively correlated with mean grass cover over this period (r2 = −0.53, P < 0.05). Also, the number of E. miniata seedlings that died between late April and late May was negatively correlated with seedling above-ground biomass (r2 = −0.55, P < 0.05) although it was poorly correlated with lignotuber biomass. Natural E. miniata seedling recruitment was significantly lower in areas invaded by gamba (F1,76 = 5.21, P < 0.05) with a mean of 0.52 seedlings per tree in invaded plots compared to 2.14 seedlings per tree in native grass plots.
Discussion
The maintenance of tree populations in savanna ecosystems requires suitable microenvironment for germination and growth of juveniles, either re-sprouts or seedlings30,32. The ground-layer microenvironment of savanna plots invaded by A. gayanus in our study site differed significantly from the microenvironment in native grass dominated plots despite its the relatively recent invasion6. Invaded plots had a dense grass cover that was almost twice that of native grass areas in the wet season, with an increase in grass layer height of up to 2 m in the late wet/early dry season. Consequently, there was a 30% reduction in radiation levels incident at ground level relative to uninvaded savanna. Reduced radiation load is likely to reduce the growth potential of many savanna woody seedlings. For example, Eucalyptus species growing in open forests and woodlands are light demanding species33, and for saplings of E. tetrodonta and E. miniata, light saturation for photosynthesis is typically high at approximately 1500 μmol m−2 s−1 31. Similarly, saplings of Terminalia ferdinandiana Exell., a common deciduous small tree of these open-forest savannas, is light saturated at 800 μmol m−2 s−1 during the wet season34. Elsewhere, invasive grasses have been shown to reduce biomass production of woody seedlings; Cabin et al.25 showed that Pennisetum setaceums invasion of Hawaiian tropical dry forests reduced ground level PFD by 55%, which caused a 31% reduction in the photosynthetic rates of woody seedlings compared to seedlings growing in uninvaded areas.
Reduced PFD within plant canopies generally results in reduced Tair23, which is likely to minimise carbon loss via reduced rates of autotrophic respiration. However, in this study, the A. gayanus canopy was associated with an increase in Tair of 2.4 °C relative to adjacent native plots. This increase is likely to be due to a combination of factors, namely, increased grass density and biomass heat storage coupled with reduced within-canopy mixing of air. During the early wet season (November to January), when the grass canopies were not fully developed, the differences between invaded and native plots were minimal. As structural differences in grass canopies increased with growth through the wet season, there were changes in the diurnal patterns of Tair. Daytime Tair became increasingly hotter in invaded plots compared to native plots, due to increased heat capture and storage by the developing A. gayanus grass cover and biomass. The increased Tair and VPD is likely to have significant negative impacts on growth potential of woody seedlings, by reducing their assimilation rates when the optimum temperature for photosynthesis is exceeded via increased autotrophic respiration35. The optimal temperature for assimilation for E. tetrodonta is ~33 °C (and assimilation declines rapidly above 35 °C31,34). Mean mid-day Tair within the grass layer of the invaded plots exceeded 35 °C for all measurement periods during the study and was particularly high during April (39.6 °C, Table 2). This suggests that for Eucalyptus seedlings, within-canopy Tair would be almost 10 °C above the optimal temperature for photosynthesis. During the growing season Apot of Eucalyptus seedlings was 30% lower in invaded plots given the elevated temperatures and reduced transmitted radiation, similar to the observed fractional reduction in seedling biomass growing under gamba canopies (Table 3). Woody species of these savannas also regenerate via basal sprouts after disturbance such as insect damage and fire32, and the saplings would also experience altered microclimate conditions.
The impacts of VPD on seedling assimilation and stomatal conductance may be offset by the observed elevated surface soil θv within invaded plots, which was consistently higher compared to adjacent native plots. Higher soil moisture levels may have resulted from reduced evaporation rates due to the reduced radiation load and shaded soil surfaces and reduced turbulent mixing. Alternatively, this surface moisture may have resulted from hydraulic lift, with wetter sub-soils and drier surface soils providing the required water potential gradient. Hydraulic lift is not commonly reported in grass species, but C4 grasses such as Aristida stricta are capable of this phenomenon36. Elevated θv in A. gayanus plots occurred at the beginning of the wet season which could increase the likelihood of seedling emergence37 and may also provide additional nutrients in the top 10 cm of soil via increased rates of mineralisation38,39, potentially favouring A. gayanus growth given this species’ ability to assimilate both nitrate and ammonium as nitrogen sources6.
A. gayanus invasion did not negatively impact establishment of artificially sown Eucalyptus seed, but it did reduce survival of seedlings over the nine month dry-wet-dry seasonal monitoring period. The lack of impact on seedling establishment was not surprising given that seed fall and seedling recruitment occurs during the early wet season when differences in microclimate between grass types was minimal. In addition, there was no difference in seed predation rates between grass types. Ants are a major seed predator in the Australian savannas and a significant factor limiting the recruitment of woody species in northern Australian savannas40, however the ant community and composition is not significantly affected by A. gayanus despite the substantial changes in habitat structure resulting from invasion15. The major impact is therefore the significant reduction in Eucalyptus seedling biomass and survival, which was correlated with grass type and microclimate. Higher than optimal temperatures, VPD and light regime limited seedling photosynthesis beneath the canopy of A. gayanus reduces assimilation potential and lowered fitness13. This physiological impact has been detected in moderately invaded ( < 50% cover) sites16. Dense invasion can result in A. gayanus canopy approaching 100% cover12, which would further limit radiation transmission with increased temperatures and VPD likely.
In addition to unfavourable microclimatic effects, woody seedling recruitment in A. gayanus plots will be reduced over the longer-term by the shift in fire regime associated with invasion, with increased fire frequency and severity commonly observed in gamba patches7. Increased intensity and leaf scorch height increase heat damage to reproductive structures41, and also reduce flowering in years following fires30, possibly due to the diversion of resources to canopy maintenance at the expense of ovule development42, factors that would reduce potential seed fall, further limiting woody regeneration potential. Reduced seed input and seedling survival are likely to both be major contributors to the substantially lower number of naturally recruited seedlings in invaded plots compared to native grass plots.
In summary, this study has shown that even at a relatively early stage of invasion, the microclimate beneath A. gayanus canopies is characterised by high temperatures and light-limiting radiation. This altered microclimate will influence a range of process including seedling emergence, growth and survival. We have shown that the seedling growth and survival of a dominant woody overstorey species is reduced in the altered conditions. The density of established seedlings in invaded patches is significantly reduced when compared to the density in native grass patches. This is likely to be due to both reduced seed supply, due to the impacts of fire on seed production, and the direct impacts on seedling survivorship. These processes will result in savanna of low floristic diversity dominated by A. gayanus as opposed to the floristically rich natural savanna of this region that consists of four phenological guilds of woody species43 and a range of annual and perennial C4 grasses.
Methods
Site description
Our study was conducted at the Mary River National Park (12°38′S, 131°45′E), ~100 km southeast of Darwin, Northern Territory, Australia. Sites were located within open-forest savanna (sensu sensu44), with an average canopy cover of 50–60% and canopy height of 15–20 m12. Overstorey vegetation was dominated by Eucalyptus miniata A. Cunn ex Schauer and E. tetrodonta F. Muell. and the mid-storey consisting of deciduous and semi-deciduous small trees and shrubs. These two eucalypt species represent over 80% of the leaf area index and biomass of the NT’s mesic savanna45. The dominant native grasses are C4 grasses Alloteropsis semialata (R. Br.) A. Hitchc and Eriachne triseta Nees ex Steud46. This and similar savanna types occupy almost 200,000 km2 across northern Australia47. The climate is characterised by monsoonal rains and high humidity in the wet season (October-April) and virtually no rain with high rates of evaporation throughout the dry season (May-September). The mean annual rainfall for Mary River National Park is ~1570 mm48. Monthly mean maximum temperature increases significantly in September, peaking at approximately 37 °C in October/November, before stabilising to 33–34 °C for the remainder of the wet season, and reaching a minimum of 31 °C in the mid-dry season48. Experimental sites were on deeply weathered and partly laterised, late Tertiary sediments of the Koolpinyah surface and associated course sandy Quaternary alluvium. Soils were of the Kandosol order (after49 and varied between deep, gravel-free red kandosols and moderately deep red and red-yellow kandosols with some gravel.
Sampling regime
Our study used a randomised block design at savanna sites described by Rossiter-Rachor et al.50. Four blocks were established (hereafter referred to as plot-pairs), with each plot-pair consisting of an area dominated by native grass (hereafter referred to as ‘native grass’ plots), and an adjacent (~50 m distant) A. gayanus dominated area (hereafter referred to as ‘invaded’ plots). Plot-pairs were located up to 600 m apart, and each plot was 50 × 50 m in size. All plot-pairs were burnt in May as part of the Park’s management burning program, prior to commencement of the experiment, and consequently there was very low (<10%) grass cover until the wet-season rains began in October, just prior to the initial measurements. Plot-pairs remained unburned for the duration of the data collection period.
Vegetation and microclimate
We measured the vegetation structure and microclimate within experimental plots on four occasions during a full wet-dry cycle: November (pre-wet season), the following February (mid-wet), April (early-dry) and July (mid-dry season). Grass cover was characterised by randomly placing 15, 1 m2 quadrats within each plot (i.e. n = 30 quadrats per plot-pair) and estimating the percent cover of grasses (live and dead), forbs, re-shoots, litter and bare soil. Mean grass tussock height was also recorded based on five random measures per quadrat.
We assessed differences in near-surface microclimate by estimating canopy transmission of photon flux density (PFD) to the soil surface, and within-understorey canopy air temperature (Tair) and relative humidity (RH%) to estimate within-understorey canopy vapour pressure deficit (VPD). Canopy transmission was estimated by comparing incoming PFD received at the soil surface beneath each grass canopy type to incoming PFD measured in a large canopy gap. Incoming PFD was measured using a quantum sensor (LI-190SB, Licor, USA) mounted at 1.5 m above ground in the canopy gap where there was no vegetation to obscure the sky to within 52° of the zenith. Two additional quantum sensors were used to measure PFD in the invaded and native grass plots. We quantified sensor bias prior to measurements by placing sensors side by side and simultaneously logging PFD for four hours. From this comparison, a regression equation was derived and used to remove sensor bias. PFD sensors were then located 10 cm above ground, with measurements made every 1 m along three replicate 20 m transects within each plot-pairs at each of the four sites. Measurements from both sensors were simultaneously logged every 1 minute and converted into a percentage of incoming radiation reaching ground level. To reduce variation resulting from sun angle, measurements were made between 1030 and 1330 hours local time, with one plot-pair being measured per day. To further minimise bias due to sun angle, measurements alternated between treatment plots. Measurements were taken under direct and diffuse radiation conditions, however, measurements were not taken if conditions changed from direct to diffuse or vice versa during a measurement run.
We measured air temperature and humidity using shielded and ventilated temperature and relative humidity probes (CS 500, Campbell Scientific, Nebraska, USA) with measurements logged (CR10X, Campbell Scientific, Nebraska USA). Measurements were taken over a four-day period with a plot-pair measured for 22 hours per day (0900 to 0700 h the following day). For each grass canopy type, we installed a CS500 probe at 40 cm above the ground, supplemented by a further four shielded thermocouples (type T, copper-constantan) randomly located within a 20-m radius within the plot. Plot-pairs were measured simultaneously, with the sensor array located at four random locations within each treatment plot. VPD was calculated from Tair and RH% measures (after51. All sensors were logged using 30 minute averages, with sensor channels on the logger scanned every 10 seconds. To avoid biased sampling, consecutive days with contrasting meteorological conditions were avoided. Sensor bias was checked prior to fieldwork. Data summaries were prepared using the 30 minute means for each sensor, which were further averaged over the following local time periods: daytime mean (0900–1900 h); nocturnal mean (1900–0700 h); midday mean (1100–1500 h). The maximum values of each sensor were also logged. In February, Tair and PFD beneath the grass canopies was also measured simultaneously across all plot-pairs diurnally (0800–1730h), with mean values logged every 5 minutes. Sensors were mounted as described above within each canopy type.
Volumetric soil moisture (θv) was measured in November, February, and April using a hand-held TDR probe (Theta Probe, ML2, Delta-T Devices, Cambridge, UK) with the probe inserted vertically into the soil to a depth of 6 cm. Measurements were taken every three metres along two ×24 m transects randomly located within each plot type. The mean value of each transect was used for statistical analysis. Measurements for all plot-pairs were collected on the same day.
Seedling photosynthetic potential
We used the PFD and Tair data collected from each grass type to estimate the potential woody seedling assimilation (Apot) based on previously established relationships between Eucalyptus seedling photosynthesis and light and temperature31. In our scenario, Apot represents the assimilation potential of a regenerating seedling for the temperature and light regime quantified for both native and gamba grass canopies. Prior et al.31 examined leaf scale photosynthetic properties of E. tetrodonta saplings at similar savanna sites in the Darwin region and developed relationships between leaf scale photosynthesis (A) and PFD and leaf temperature. Our temperature measure was air temperature and we assumed Tair and leaf temperature were similar during the wet season when leaf water stress and resultant elevated leaf temperatures would be at a minimum. These relationships were used to estimate Apot during peak growing season conditions (February) using diurnal PFD and Tair data collected from each plot-pair at each site. Instantaneous values of Apot were estimated and these were summed to give Apot as g C per m2 d−1 for seedlings within each grass canopy type.
Seedling establishment and survivorship
To investigate the effects of A. gayanus invasion on woody recruitment, we conducted a seed augmentation experiment using E. miniata and E. tetrodonta seed. Seed fall for these species occurs from September to December52 and seeds have high viability, but no innate dormancy and germinate quickly30. Seed of both species were collected near the study sites. This experiment was undertaken within the three plot-pairs with the highest cover of A. gayanus. Within each plot-pair, nine permanent 1 × 1 m quadrats were randomly located; three were sown with 250 E. miniata seeds, three with 250 E. tetrodonta seeds and three were “control” quadrats with no seed added. Quadrats were constructed using 5 cm steel mesh wrapped in flywire (after30 to stop seeds being washed out of the quadrats by the monsoonal rains that can result in significant runoff. In December, during the time of natural seed fall, seed (250 seed of either E. miniata or E. tetrodonta) was mixed with a handful of dry sand and sprinkled evenly across the quadrats, resulting in three replicate quadrats per species in each plot combination. The control quadrats enabled assessment of natural recruitment rates. Seedlings were counted fortnightly during the wet season from mid-January to mid-April, with a final count taking place in the dry season in mid-July. To determine whether differences in seedling recruitment between treatments could have been caused by seed predation, experiments took place in mid-November and mid-December. Seed depots consisted of ten E. miniata seeds placed in a 3–4–3 grid, with 10 cm spacing. This grid was repeated three times within a plot. Seeds were placed on a sprinkling of white sand to aid relocation. Seeds were counted daily for 2 days after30.
Seedling growth was assessed by tagging 20 randomly selected individuals per quadrat (or less if fewer than 20 individuals emerged) with numbered skewers. The height and number of true leaves were recorded in March for each tagged individual. Height was measured to the apical meristem and the presence of true leaves was confirmed if the pair of leaves had separated. Each seedling was re-measured four months later (May) to determine wet-season seedling growth rate. Growth of E. miniata seedlings was further quantified by measuring aboveground biomass and lignotuber biomass of five randomly selected seedlings quadrat, harvested in May and dried at 70 °C for 48 h (n = 90). To test for responses to light quantity, specific leaf area (SLA, leaf area/leaf dry weight) was calculated by measuring the length and width of a leaf at the third node from the apical meristem from all tagged seedlings to give an average leaf area per quadrat. Leaf weight was calculated by weighing the dried leaves from the harvested seedlings. Finally, to investigate any response to light quality (red:far-red ratio), internode length was compared by measuring the internode at the midpoint of each tagged seedling’s stem, and the foliage:stem weight ratio of dried seedlings was calculated to assess whether there was a difference in above-ground mass allocation. These data were not taken for E. tetrodonta because there were insufficient seedlings to undertake both harvest and survival monitoring. Seed predation and seedling survival were converted to proportion relative to the initial population.
Ground cover was recorded monthly between January and May, and the following July with % cover of grass, forbs, litter, woody re-shoots and bare ground recorded along with mean grass tussock height. Overstorey tree cover was recorded at the time of seed sowing and also at both seedling-growth measurement dates.
Natural seeding recruitment
Natural seedling recruitment was assessed at seven random sites. At each site, six E. miniata trees surrounded by native grass, and six surrounded by A. gayanus were chosen. Trees were mature and at least 30 cm DBH and for each tree a circular plot was established with a radius of five (area = 78.5 m2) and seedling density within the plot measured. This is the zone of highest seed fall for these dominant Eucalyptus species52. Seedlings were identified as less than one year old based on the development of their lignotuber, which requires twelve to eighteen months to develop.
Statistical analysis
The differences between the microclimate, environmental variables and Apot in invaded and native grass plots at each sampling period were tested using two-factor mixed-model ANOVA’s, with factors grass canopy (native, gamba; fixed) and plot-pair (random). Seedling survival data was analysed with a three-factor ANOVA, with the factors including plot-pair (fixed), grass type (fixed) and species (fixed). Seedling biomass and seed predation data were analysed using a two-factor fixed analysis. Effect of gamba cover on natural seedling recruitment was analysed using a one-way ANOVA. Data that did not meet assumptions were either log-transformed, or arc-sine transformed for proportional data (seedling and seed predation data) to meet the assumptions of normality. Post-hoc pooling of treatment data was done if the plot-pair by treatment interaction was non-significant with P > 0.2553. Pearson correlation coefficients were used to investigate relationships between microclimatic factors, seedling recruitment and environmental variables.
References
Gorgone-Barbosa, E. et al. How can an invasive grass affect fire behavior in a tropical savanna? A community and individual plant level approach. Biological Invasions 17, 423–431 (2015).
Hutley, L. B. & Setterfield, S. A. in Encyclopedia of Ecology (eds Jørgensen S.E. & Fath B.) (Elsevier, 2008).
Klink, C. A. & Machado, R. B. Conservation of the Brazilian cerrado. Conservation Biology 19, 707–713 (2005).
Pyšek, P. et al. A global assessment of invasive plant impacts on resident species, communities and ecosystems: the interaction of impact measures, invading species’ traits and environment. Global Change Biology 18, 1725–1737 (2012).
Hoffmann, W. A., Schroeder, W. & Jackson, R. B. Positive feedbacks of fire, climate, and vegetation and the conversion of tropical savanna. Geophysical Research Letters 29, 9-1–9-4 (2002).
Rossiter-Rachor, N. A. et al. Invasive Andropogon gayanus (gamba grass) is an ecosystem transformer of nitrogen relations in Australian savanna. Ecological Applications 19, 1546–1560, https://doi.org/10.1890/08-0265.1 (2009).
Setterfield, S. A. et al. Adding fuel to the fire: The impacts of non-native grass invasion on fire management at a regional scale. Plos One 8, https://doi.org/10.1371/journal.pone.0059144 (2013).
Setterfield, S. A., Rossiter-Rachor, N. A., Hutley, L. B., Douglas, M. M. & Williams, R. J. Turning up the heat: the impacts of Andropogon gayanus (gamba grass) invasion on fire behaviour in northern Australian savannas. Diversity and Distributions 16, 854–861, https://doi.org/10.1111/j.1472-4642.2010.00688.x (2010).
Ens, E.-J. & French, K. Exotic woody invader limits the recruitment of three indigenous plant species. Biological Conservation 141, 590–595 (2008).
Minden, V., Jacobi, J., Porembski, S. & Boehmer, H. Effects of invasive alien kahili ginger (Hedychium gardnerianum) on native plant species regeneration in a Hawaiian rainforest. Applied Vegetation Science 13, 5–14 (2010).
Stinson, K. A. et al. Invasive Plant Suppresses the Growth of Native Tree Seedlings by Disrupting Belowground Mutualisms. PLoS biology 4, e140 (2006).
Brooks, K. J., Setterfield, S. A. & Douglas, M. M. Exotic grass invasions: Applying a conceptual framework to the dynamics of degradation and restoration in Australia’s tropical savannas. Restoration Ecology 18, 188–197, https://doi.org/10.1111/j.1526-100X.2008.00470.x (2010).
Ens, E., Hutley, L. B., Rossiter-Rachor, N. A., Douglas, M. M. & Setterfield, S. A. Resource-use efficiency explains grassy weed invasion in a low-resource savanna in north Australia. Frontiers in plant science 6, 560, https://doi.org/10.3389/fpls.2015.00560 (2015).
NRM Ministerial Council. Weeds of National Significance Gamba Grass (Andropogon gayanus) Strategic Plan. (Australian Weeds Committee, Canberra, 2012).
Parr, C. L., Ryan, B. J. & Setterfield, S. A. Habitat Complexity and Invasive Species: The Impacts of Gamba Grass (Andropogon gayanus) on Invertebrates in an Australian Tropical Savanna. Biotropica 42, 688–696, https://doi.org/10.1111/j.1744-7429.2010.00637.x (2010).
Rossiter, N. A., Setterfield, S. A., Douglas, M. M. & Hutley, L. B. Testing the grass-fire cycle: alien grass invasion in the tropical savannas of northern Australia. Diversity and Distributions 9, 169–176, https://doi.org/10.1046/j.1472-4642.2003.00020.x (2003).
Levick, S. R. et al. Monitoring the distribution and dynamics of an invasive grass in tropical savanna using airborne LiDAR. Remote Sensing 7, 5117–5132 (2015).
Andersen, A. N. et al. Fire frequency and biodiversity conservation in Australian tropical savannas: implications from the Kapalga fire experiment. Austral Ecology 30, 155–167, https://doi.org/10.1111/j.1442-9993.2005.01441.x (2005).
Davis, M. A., Wrage, K. J. & Reich, P. B. Competition between tree seedlings and herbaceous vegetation: support for a theory of resource supply and demand. Journal of Ecology 86, 652–661 (1998).
Denslow, J. S., Uowolo, A. L. & Hughes, R. F. Limitations to seedling establishment in a mesic Hawaiian forest. Oecologia 148, 118–128 (2006).
Scholes, R. & Archer, S. Tree-grass interactions in savannas. Annual Review of Ecology and Systematics 28, 517–544 (1997).
Hunter, A. & Aarssen, L. Plants helping plants: new evidence indicates that beneficence is important in vegetation. Bioscience 38, 34–40 (1988).
Jones, H. G. Plants and Microclimate: A Quantitative Approach to Environmental Plant Physiology, 2nd edn. (Cambridge, 1992).
Kozlowski, T. T., Pallardy, S. G. & Kramer, P. J. The Physiological Ecology of Woody Plants. (Academic Press, 1991).
Cabin, R. J. et al. Effects of long-term ungulate exclusion and recent alien species control on the preservation and restoration of a Hawaiian tropical dry forest. Conservation Biology 14, 439–453 (2000).
Eliason, S. A. & Allen, E. B. Exotic grass competition in suppressing native shrubland re-establishment. Restoration Ecology 5, 245–255 (1997).
Gordon, D. R. & Rice, K. J. Competitive suppression of Quercus douglasii (Fagaceae) seedling emergence and growth. American Journal of Botany 87, 986–994 (2000).
Standish, R. J., Robertson, A. W. & Williams, P. A. The impact of an invasive weed Tradescantia fluminensis on native forest regeneration. Journal of Applied Ecology 38, 1253–1263 (2001).
Scott, K. A., Setterfield, S. A., Andersen, A. N. & Douglas, M. M. Correlates of grass-species composition in a savanna woodland in northern Australia. Australian Journal of Botany 57, 10–17, https://doi.org/10.1071/bt08120 (2009).
Setterfield, S. A. Seedling establishment in an Australian tropical savanna: effects of seed supply, soil disturbance and fire. Journal of Applied Ecology 39, 949–959, https://doi.org/10.1046/j.1365-2664.2002.00772.x (2002).
Prior, L., Eamus, D. & Duff, G. Seasonal and diurnal patterns of carbon assimilation, stomatal conductance and leaf water potential in Eucalyptus tetrodonta saplings in a wet–dry savanna in northern Australia. Australian journal of botany 45, 241–258 (1997).
Russell-Smith, J. & Setterfield, S. A. Monsoon rain forest seedling dynamics, northern Australia: contrasts with regeneration in eucalypt-dominated savannas. Journal of Biogeography 33, 1597–1614, https://doi.org/10.1111/j.1365-2699.01527.x (2006).
Eamus, D., Myers, B., Duff, G. & Williams, D. Seasonal changes in photosynthesis of eight savanna tree species. Tree Physiology 19, 665–671 (1999).
Prior, L., Eamus, D. & Duff, G. Seasonal trends in carbon assimilation, stomatal conductance, pre-dawn leaf water potential and growth in Terminalia ferdinandiana, a deciduous tree of northern Australian savannas. Australian Journal of Botany 45, 53–69 (1997).
Larcher, W. Physiological Plant Ecology, 4th edn. (Springer-Verlag, 2003).
Espeleta, J., West, J. & Donovan, L. Species-specific patterns of hydraulic lift in co-occurring adult trees and grasses in a sandhill community. Oecologia 138, 341–349 (2004).
Bell, D. T. Turner Review No. 1. The process of germination in Australian species. Australian Journal of Botany 47, 475–517 (1999).
Mack, M. C. Effects of exotic grass invasion on ecosystem nitrogen dynamics in a Hawaiian woodland. PhD thesis, University of California, Berkeley (1998).
Theodose, T. A. & Martin, J. Microclimate and substrate quality controls on nitrogen mineralization in a New England high salt marsh. Plant Ecology 167, 213–221 (2003).
Setterfield, S. A. Factors Affecting Seedling Regeneration of Woody Species in a Northern Australian Tropical Savanna. PhD Dissertation. (Northern Territory University, 1997).
Setterfield, S. A. The impact of experimental fire regimes on seed production in two tropical eucalypt species in northern Australia. Australian Journal of Ecology 22, 279–287, https://doi.org/10.1111/j.1442-9993.1997.tb00673.x (1997).
Reekie, E. & Bazzaz, F. Reproductive effort in plants. 3. Effect of reproduction on vegetative activity. American Naturalist 129, 907–919 (1987).
Williams, R., Myers, B., Muller, W., Duff, G. & Eamus, D. Leaf phenology of woody species in a north Australian tropical savanna. Ecology 78, 2542–2558 (1997).
Specht, R. In Vegetation Classification in Australia (eds Gillison A. N. & Anderson D. J.) 10–21 (CSIRO Publishing, Canberra, 1981).
O’Grady, A., Eamus, D. & Hutley, L. Transpiration increases during the dry season: patterns of tree water use in eucalypt open-forests of northern Australia. Tree Physiology 19, 591–597 (1999).
Rossiter-Rachor, N. et al. Invasive Andropogon gayanus (Gamba grass) alters litter decomposition and nitrogen fluxes in an Australian tropical savanna. Scientific Reports 7, 11705, https://doi.org/10.1038/s41598-017-08893-z (2017).
Fox, I. D., Neldner, V. J., Wilson, G. W. & Bannink, P. J. The Vegetation of the Australian Tropical Savannas. (Environment Protection Agency, Brisbane, Australia, 2001).
Bureau Of Meteorology. Climate data online. Australian Government Publishing Service, Canberra, Australia. URL: http://www.bom.gov.au/climate/data/ (last accessed April 2017) (2017).
Isbell, R. F. Australian Soil Classification. (CSIRO Publishing, 2002).
Rossiter-Rachor, N. A., Setterfield, S. A., Douglas, M. M., Hutley, L. B. & Cook, G. D. Andropogon gayanus (Gamba grass) invasion increases fire-mediated nitrogen losses in the tropical savannas of northern Australia. Ecosystems 11, 77–88, https://doi.org/10.1007/s10021-007-9108-x (2008).
Monteith, J. L. & Unsworth, M. H. Principles of Environmental Physics, 2nd edn. (Edward Arnold, London, 1990).
Setterfield, S. A. & Williams, R. J. Patterns of flowering and seed production in Eucalyptus miniata and E. -tetrodonta in a tropical savanna woodland, northern Australia. Australian Journal of Botany 44, 107–122, https://doi.org/10.1071/bt9960107 (1996).
Quinn, G. P. & Keough, M. J. Experimental Design and Data Analysis for Biologists. (Cambridge University Press, Cambridge, 2002).
Acknowledgements
The authors are most grateful to the NT Parks and Wildlife Service (Mary River Ranger Station) for providing access to the study site, accommodation and advice. We thank Keith McGuiness for advice on statistical design and analysis and Jane Barratt for lab and field assistance, and several volunteers for field assistance.
Author information
Authors and Affiliations
Contributions
Author contributions: L.H., S.S. and M.D. conceived the research. All authors contributed to data collection, analysis and writing of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Setterfield, S.A., Clifton, P.J., Hutley, L.B. et al. Exotic grass invasion alters microsite conditions limiting woody recruitment potential in an Australian savanna. Sci Rep 8, 6628 (2018). https://doi.org/10.1038/s41598-018-24704-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-24704-5
This article is cited by
-
Effects of native and invasive grasses on the survival and growth of tree seedlings in a neotropical savanna
Biological Invasions (2023)
-
The role of microsite sunlight environment on growth, architecture, and resource allocation in dominant Acacia tree seedlings, in Serengeti, East Africa
Plant Ecology (2020)
-
Andropogon gayanus Kunth invasion in the Cerrado: from seed production to seedling establishment along roadsides
Biological Invasions (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.