Abstract
Colorectal cancer (CRC) is the second leading cause of cancer-related deaths. It is estimated that about half the cases of CRC occurring today are preventable. Recent studies showed that human gut microbiota and their collective metabolic outputs play important roles in CRC. However, the mechanisms by which human gut microbial metabolites interact with host genetics in contributing CRC remain largely unknown. We hypothesize that computational approaches that integrate and analyze vast amounts of publicly available biomedical data have great potential in better understanding how human gut microbial metabolites are mechanistically involved in CRC. Leveraging vast amount of publicly available data, we developed a computational algorithm to predict human gut microbial metabolites for CRC. We validated the prediction algorithm by showing that previously known CRC-associated gut microbial metabolites ranked highly (mean ranking: top 10.52%; median ranking: 6.29%; p-value: 3.85E-16). Moreover, we identified new gut microbial metabolites likely associated with CRC. Through computational analysis, we propose potential roles for tartaric acid, the top one ranked metabolite, in CRC etiology. In summary, our data-driven computation-based study generated a large amount of associations that could serve as a starting point for further experiments to refute or validate these microbial metabolite associations in CRC cancer.
Similar content being viewed by others
Introduction
Colorectal cancers are the second leading cause of cancer-related deaths in in the United States and the third most common cancer in men and in women1. In the United States alone, an estimated 135,430 men and women will be diagnosed with CRC in the year 2017 and 50,260 will die from this disease2. It is estimated that forty-five percent of CRC are preventable by modifiable environmental factors such as food, nutrition, lifestyle, and physical activity, among others3,4.
Human gut microbiota, the collection of microorganisms that live in the human digestive tracts, play central roles in human health and diseases, by metabolizing nutrients and food components and by controlling the immune response of the human body5,6,7,8. Growing evidence suggests that gut microbiota and their metabolites not only influence carcinogenesis and tumor progression, but also influence the efficacy of anticancer therapies9,10,11. Human microbiome (the collective genomes of the microbiota) studies have revealed that gut dysbiosis (an imbalance in the intestinal bacteria) is associated with the increased incidence of CRC11,12,13,14.
Undigested dietary components that reach the large intestine are fermented by microbiota to produce a variety of metabolites and nutrients. It has become increasingly clear that the collective metabolic outputs of gut microbiota strongly influence cancer susceptibility and progression5,15,16. For example, recent studies have shown that the short-chain fatty acid (SCFA) butyrate, one of the most abundant metabolites of gut microbiota in the fermentation of fiber, has a role in the suppression of inflammation and colorectal cancer17.
Currently, the mechanisms by which gut microbial metabolites interact with host genetics in promoting or protecting against CRC remain unknown. Computational approaches have been widely used in drug development18,19,20,21,22,23,24 and disease mechanism understanding25,26,27. We have recently developed a hypothesis-driven systems approach to understand how trimethylamine N-oxide (TMAO), a gut microbial metabolite of dietary meat and fat, is mechanistically involved in CRC28. Here, we present a data-driven computational approach to estimate which microbial metabolites might affect CRC. The identification of human gut microbial metabolites and the understanding of their role as key mediators through which bacteria might promote or protect against CRC is important. Taken together, characterizing these microbial metabolites will likely enhance our understanding of the complex gene-environment interactions in carcinogenesis, and add up to new possibilities for CRC diagnosis, prevention, and treatment.
Data and Methods
Data
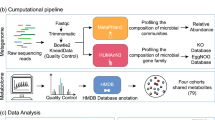
We used large amounts of publicly available data, including human metabolome, disease genetics, chemical genetics, signaling pathways, and mouse genome-wide mutation phenotypes for both the prediction and functional characterization of gut microbial metabolites for CRC (Fig. 1).
Data resources of CRC-associated genes
We used three complementary disease genetics resources to obtain CRC-associated genes. We obtained (1) 32 CRC-associated genes from the Catalog of Published Genome-Wide Association Studies (GWAS), a comprehensive collection of all published GWAS studies29; (2) 38 CRC-associated genes from the Online Mendelian Inheritance in Man (OMIM), a comprehensive collection of human genes and genetic phenotypes for Mendelian disorders30; and (3) 31 genes that are significantly mutated in colorectal cancer patients from the Cancer Genome Atlas (TCGA), a comprehensive cancer database and contains genetic and clinical data for 283 colorectal patients31. We used these three complementary and independent disease genetics resources to demonstrate the robustness of the algorithms and our findings.
The Human Metabolome Database (HMDB)
HMDB is a comprehensive database of small molecule metabolites found in the human body32. Currently, HMDB contains 52,658 metabolites, including 172 metabolites originated in human gut microbiota.
Data resource of metabolite-associated genes
We obtained metabolite/chemical-associated genes from STITCH (Search Tool for Interactions of Chemicals). STITCH contains chemical-gene association data for > 300,000 small molecules and 2.6 million proteins from 1,133 organisms33. We used chemical-gene associations found in human body, which include 1,466,636 chemical-gene pairs, 259,171 chemicals, and 15,620 human genes.
Genetic pathway data
We used gene-pathway association data from the Molecular Signatures Database (MSigDB) to construct molecular profiles for CRC and metabolites and to study the interplaying pathways underlying top identified microbial metabolites and CRC. MSigDB contains 10,295 annotated pathways and gene sets34.
Genome-wide mutational phenotypes in experimental mouse models
Recently, the Mouse Genome Database (MGD) has made available large amounts of phenotypic descriptions of systematic gene knockouts in mouse models35. We have recently shown that these strong causal gene-phenotype annotations (278,553 gene-phenotype associations for 41,905 mutant alleles and 10,744 phenotypes) have great potential for virtual phenotypic screening for drug discovery21,22,23. In this study, we used gene-phenotype associations from MGD to assess the functional effects of top ranked microbial metabolites on CRC-related phenotypes.
Metabolome-wide prediction of gut microbial metabolites for CRC
The experimental flowchart is summarized in Fig. 2 and described in details in subsequent sections.
Construct molecular profiles for diseases
We identified CRC-associated genes from the three disease genetics databases: the GWAS catalog, OMIM, and TCGA. Pathways associated with each gene were obtained from MSigDB. For each pathway, we assessed its probability of being associated with the given set of CRC-associated genes as compared to its probability associated with the same number of randomly selected genes. The random process is repeated 1000 times and a t-test was used to assess the statistical significance. As an example, the pathway “colorectal cancer” is associated with 7 out of 31 (29.0%) CRC genes from TCGA, which represents a significant 39-fold enrichment as compared to the random expectation of 0.7%. The molecular profile for CRC consists of a list of significantly enriched pathways. Three molecular profiles were built for CRC using genes from three complementary disease genetics resources.
Build molecular profiles for chemicals/metabolites
Similarly, we built one molecular profile for each of the 259,170 chemicals/metabolites from the STITCH database. For example, the molecular profile for butyric acid, a human gut microbial metabolite, consists of 609 pathways.
Prioritize metabolites for CRC
Metabolites were prioritized based on how their molecular profiles are similar to CRC-specific molecular profile. We implemented three commonly used set similarity measures: overlap, Jaccard coefficient, and cosine similarity36. Overlap is defined as the intersection of disease profile set and metabolite profile set. Jaccard coefficient of two sets is defined as the size of the intersection divided by the size of the union. The cosine similarity is defined as the Euclidean dot product of two sets. The output is a ranked list of 259,170 chemicals/metabolites for CRC prioritized based on their profile similarities with CRC.
Evaluation using known CRC-associated metabolites
We evaluated the prioritization algorithm using 32 known CRC-associated metabolites extracted from a recent review paper15. Recall, mean and median rankings were used for performance measures. Significance was calculated by comparing actual rankings to random expectation (based on random expectation, a metabolite shall have an average ranking of 50%). We also examined the number of known metabolites at 10 decile rankings. A good prioritization algorithm shall enrich true positives among top-ranked entities. We calculated the number of known metabolites at each decile and plotted decile enrichment distribution.
Evaluate the rankings of all human microbial metabolites for CRC
We investigated whether human gut microbial metabolites in general are highly related to CRC in terms of molecular relevance. We examined the rankings of all 172 microbial metabolites among prioritized chemicals. Recall, mean and median rankings were calculated and decile ranking was plotted.
Functional characterization of top ranked novel microbial metabolite in CRC
Identify common pathways between novel metabolite and CRC
We identified common genetic pathways that are significantly enriched for the novel metabolite and CRC. We then developed an algorithm to further prioritize these common pathways. The ranking of each common pathway is a balance measure of rankings from the disease (CRC) and from the metabolite. A pathway ranks highly if and only if it ranks highly for both the metabolite and the disease. The ranking of a common pathway is defined as: ranking_combined = 2*(ranking_d * ranking_m)/(ranking_d + ranking_m), where ranking_d is the ranking score of a pathway for CRC; and ranking_m is the ranking score of the same pathway for the metabolite.
Functional characterization of phenotypic effects of the novel metabolite on CRC
We obtained metabolite-associated genes from STITCH and then mapped genes to their corresponding mouse gene homologs (e.g., SMAD4 = > 18Wsu70e) using human-mouse homolog mapping data from MGD35. The mapped mouse genes were then linked to their corresponding mutational phenotypes in mouse models (e.g., SMAD4 = > increased intestinal adenoma incidence, TP53 = > colon polyps) using gene-phenotype association data from MGD. For each mapped phenotype, we assessed its probability of being associated with the given set of metabolite-associated genes as compared to its probability associated with the same number of randomly selected genes. The random process is repeated 1000 times and a t-test was used to assess the statistical significance. Similarly, we built a CRC-specific phenotype profile using data from disease genetics databases. CRC- and metabolite-specific phenotype profiles were intersected. Shared phenotypes were prioritized as described for prioritizing shared pathways between the metabolite and CRC. A phenotype (e.g., colon polyps) ranked highly if and only if it ranks high for both CRC and the novel metabolite.
Data availability
Results
Known CRC-associated microbial metabolites ranked highly
The algorithm found 26 of 32 known metabolites (recall: 0.813) and the recall is determined by the coverage of the STITCH database. The not-perfect recall indicates that although STITCH is currently the most comprehensive chemical genetics database of 1,466,636 chemical-gene pairs for 259,171 chemicals/metabolites, the coverage for gut microbial metabolites is not perfect. Known CRC metabolites ranked significantly high as compared to random expectation (Table 1). As an example, when CRC-associated genes from the GWAS catalog were used to build disease-specific molecular profile, known CRC-associated microbial metabolites on average ranked at top 10.52% among 259,170 chemicals/metabolites, which is significantly higher than random expectation (P-value: 3.85E-6). These findings are consistent when CRC-associated genes from three complementary disease genetics data resources were used (Table 1).
The decile rankings further demonstrate that the ranking algorithm effectively enriched known CRC-associated metabolites at top. For example, 14 of 26 known metabolites are ranked at the first decile (top 10%) (Fig. 3).
We then examined which categories of microbial metabolites ranked highly for CRC. We classified known metabolites into six categories15. Short chain fatty acids (SCFAs) are known to be involved in colon health15,17. Our study indeed shows that SCFAs ranked highest (top 3.86%) among all known CRC-associated metabolites. The results are consistent when three independent disease genetics databases were used to obtain CRC-associated genes (Table 2).
Human gut microbial metabolites in general ranked highly for CRC
Microbial metabolites in general are highly associated with CRC based on molecular convergence. The algorithm found 131 of 172 (recall: 0.761) metabolites originated in gut microbiota (as determined by HMDB database)32. These 131 microbial metabolites ranked consistently highly when CRC-associated genes from three complementary databases were used (Table 3). As an example, microbial metabolites on average ranked at top 14.43% (P-value: 2.27E-57) among 259,170 prioritized chemicals/metabolites.
The decile rankings show that the majority of gut microbial metabolites were ranked at first decile (top 10%) (Fig. 4). For example, when CRC-associated genes from the GWAS catalog were used to build disease-specific molecular profile, 64 of 131 gut microbial metabolites were ranked at the first decile.
The top 20 ranked microbial metabolites are shown in Table 4. Seven of these top 20 metabolites are known CRC-associated metabolites. Tartaric acid ranked at top 3, immediately following butyric acid, a well-known microbial metabolite associated with CRC and colon health. Trimethylamine n-oxide (TMAO) also ranked highly (top 13). Previous studies showed that TMAO is both mechanistically and clinically associated with increased risk of CRC28,37.
Tartaric acid may be both genetically and functionally involved in CRC
Tartaric acid is the top one ranked microbial metabolite that is not included in the list of 32 known CRC-associated metabolites. Tartaric acid is a phytochemical found abundantly in nuts, apricots, apples, sunflower, avocado, grapes, among others38. Tartaric acid is associated with 305 genes (based on the STITCH database). These genes are significantly associated with 611 pathways, demonstrating that tartaric acid may participate in many biological functions. Many of the top ranked pathways are related to immune functions, including Cytokines and Inflammatory Response, IL12-mediated signaling events, and Downstream signaling in naïve CD8 + T cells. Among 117 pathways significantly associated for CRC, 64 (54.7%) pathways are also associated with tartaric acid, demonstrating that tartaric acid may be mechanistically involved in CRC etiology. The top 20 pathways shared by both CRC and tartaric acid are shown in Table 5. Many of these top common pathways are directly involved in CRC, including Regulation of nuclear SMAD2/3 signaling, β-catenin pathway, WNT pathway, TGF-beta signaling pathway, and Colorectal cancer.
To estimate the possible effects of tartaric acid on CRC, we identified mouse mutational phenotypes that are significantly associated with both CRC and tartaric acid. A total of 2441 mouse mutational phenotypes are significantly associated with tartaric acid, and 600 phenotypes are significantly associated with CRC. Among the 600 CRC-associated phenotypes, 267 (45%) phenotypes are also associated with tartaric acid. The top 20 shared phenotypes are shown in Table 6. Both top 1 and 2 ranked phenotypes are directly related to digestive system.
Discussion
More than half the cases of cancer, including CRC, occurring today are preventable and about one-third of the cases can be attributed to modifiable environmental factors such as food, nutrition, lifestyle, and physical activity, among others4. The susceptibility, initiation, and progression of CRC and many other cancers is primarily driven by gene–environment interactions. Human gut microbiota are important modifiable environmental factors that are part of the ecosystem of our bodies. Functional studies in germ-free mouse models of cancer have demonstrated that microbiota can affect cancer susceptibility and progression in various organs, including colon, however, the mechanisms by which gut microbial metabolites are involved in cancer remain unknown.
In this study, we presented a data-driven systems approach to identify and estimate gut microbial metabolites playing a role in CRC. Our approach is a data-driven computational estimation, which can be applied to different traits and diseases. In this study, we focused on CRC because of the vast availability of known CRC-associated gut microbial metabolites. Our data-driven computational method to estimate associations can take a disease name or a list of disease-associated genes as input, and the output will be a ranked list of microbial metabolites (along with shared molecular signatures and functional phenotypes) for the input disease. For clarity, this ‘in silico’ study is not functional microbiome study. Instead, it largely complements existing microbiome studies by identifying microbial metabolites within vast amounts of existing database information of diseases, genes, pathways, functional phenotypes, and metabolome.
However, our study holds several limitations that warrant further discussion. First, the relationships among microbes, their metabolites, and hosts are complex, non-linear and bi-directional39. For example, recent studies showed that gut microbial metabolites are involved in CRC etiology by altering host epigenome37. Our study focused on the database-dependent interactions between microbial metabolites and CRC genetics of the host. Indeed, we lack the necessary data in order to computationally model the effects of metabolites on microbe populations, the host genetics on microbe variations or epigenetic effects on host genomes. The goal of this study was to provide estimates of associations between human gut microbial metabolites and CRC, which in turn may inform the identification of responsible microbe composition in cancer etiology.
Second, host genetics can affect gut microbiota composition and metabolic outputs in response to environmental factors40,41. A person’s genetic make-up can influence his/her response to environmental stressors, gut microbiota population, and microbiome-genome interactions. As personal genetic and genomics information becomes increasingly available, a patient-focused understanding of environment-microbiome-genome-cancer interactions is possible by linking personal genome to metabolite-gene-pathway-disease connections as identified in this study.
Third, among the 41,806 small molecule metabolites available in HMDB, only 172 metabolites (~0.4%) originate in gut microbiota. The field of microbiome research is a fast-moving target with an increasing number of microbial metabolites being identified. The computational algorithms we developed have built-in flexibility and capability to incorporate new data as it becomes available.
Lastly, our current study is pure ‘in-silico’. Our goal was to generate estimates of associations data/hypotheses that may be tested to refute or validate our suggestions of gut microbial metabolites role in CRC. We anticipate that both the data-driven computational methods developed and the associations generated in this study will likely stimulate further studies of microbiome-gene interactions in cancer etiology. Taken together, validating identified metabolite-CRC associations in animal models or humans are needed in order to translate the findings into cancer diagnosis, prevention, and treatment.
References
Centers for Disease Control and Prevention. Colorectal cancer statistics. https://www.cdc.gov/cancer/colorectal/statistics/ (accessed in 2017).
American Cancer Society. Key Statistics for Colorectal Cancer. https://www.cancer.org/cancer/colon-rectal-cancer/about/key-statistics.html (accessed in 2017).
American Institute for Cancer Research. What you need to know about preventing Colorectal Cancer. http://www.aicr.org/reduce-your-cancer-risk/cancer-site/cancersite_colon_rectum.html (accessed in 2017).
Colditz, G. A., Wolin, K. Y., & Gehlert, S. Applying what we know to accelerate cancer prevention. Science translational medicine. 4, 127, 127rv4–127rv4 (2012).
Nicholson, J. K. et al. Host-gut microbiota metabolic interactions. Science. 1223813 (2012).
Quigley, E. M. Gut bacteria in health and disease. Gastroenterology & hepatology. 9(9), 560 (2013).
Lloyd-Price, J., Abu-Ali, G. & Huttenhower, C. The healthy human microbiome. Genome medicine. 8(1), 51 (2016).
Tremaroli, V. & Bäckhed, F. Functional interactions between the gut microbiota and host metabolism. Nature. 489(7415), 242 (2012).
Zitvogel, L. et al. Cancer and the gut microbiota: an unexpected link. Science translational medicine. 7(271), 271ps1–271ps1 (2015).
Thomas, R. M. & Jobin, C. The microbiome and cancer: is the ‘oncobiome’ mirage real? Trends in cancer. 1(1), 24–35 (2015).
Schwabe, R. F. & Jobin, C. The microbiome and cancer. Nature Reviews Cancer. 13(11), 800 (2013).
Kostic, A. D. et al. Genomic analysis identifies association of Fusobacterium with colorectal carcinoma. Genome research. 22(2), 292–298 (2012).
Sears, C. L. & Garrett, W. S. Microbes, microbiota, and colon cancer. Cell host & microbe. 15(3), 317–328 (2014).
Garrett, W. S. Cancer and the microbiota. Science. 348(6230), 80–86 (2015).
Louis, P., Hold, G. L. & Flint, H. J. The gut microbiota, bacterial metabolites and colorectal cancer. Nature Reviews Microbiology. 12(10), 661 (2014).
Gill, S. R. et al. Metagenomic analysis of the human distal gut microbiome. Science. 312(5778), 1355–1359 (2006).
Donohoe, D. R. et al. The microbiome and butyrate regulate energy metabolism and autophagy in the mammalian colon. Cell metabolism. 13(5), 517–526 (2011).
Sliwoski, G., Kothiwale, S., Meiler, J. & Lowe, E. W. Computational methods in drug discovery. Pharmacological reviews. 66(1), 334–395 (2014).
Li, J. et al. A survey of current trends in computational drug repositioning. Briefings in bioinformatics. 17(1), 2–12 (2015).
Xu, R. & Wang, Q. PhenoPredict: A disease phenome-wide drug repositioning approach towards schizophrenia drug discovery. Journal of biomedical informatics. 56, 348–355 (2015).
Nagaraj, A. B. et al. Using a novel computational drug-repositioning approach (DrugPredict) to rapidly identify potent drug candidates for cancer treatment. Oncogene. 37(3), 403 (2018).
Chen, Y., Gao, Z., Wang, B. & Xu, R. Towards precision medicine-based therapies for glioblastoma: interrogating human disease genomics and mouse phenotypes. BMC genomics. 17(7), 516 (2016).
Chen, Y. & Xu, R. Drug repurposing for glioblastoma based on molecular subtypes. Journal of biomedical informatics. 64, 131–138 (2016).
Xu, R. & Wang, Q. A genomics-based systems approach towards drug repositioning for rheumatoid arthritis. BMC genomics. 17(7), 518 (2016).
Moreau, Y. & Tranchevent, L. C. Computational tools for prioritizing candidate genes: boosting disease gene discovery. Nature Reviews Genetics. 13(8), 523 (2012).
Chen, Y., Li, L., Zhang, G. Q. & Xu, R. Phenome-driven disease genetics prediction toward drug discovery. Bioinformatics. 31(12), i276–i283 (2015).
Chen, Y. & Xu, R. Context-sensitive network-based disease genetics prediction and its implications in drug discovery. Bioinformatics. 33(7), 1031–1039 (2017).
Xu, R., Wang, Q. & Li, L. A genome-wide systems analysis reveals strong link between colorectal cancer and trimethylamine N-oxide (TMAO), a gut microbial metabolite of dietary meat and fat. BMC genomics. 16(7), S4 (2015).
Welter, D. et al. The NHGRI GWAS Catalog, a curated resource of SNP-trait associations. Nucleic acids research. 42(D1), D1001–D1006 (2013).
Hamosh, A., Scott, A. F., Amberger, J. S., Bocchini, C. A. & McKusick, V. A. Online Mendelian Inheritance in Man (OMIM), a knowledgebase of human genes and genetic disorders. Nucleic acids research. 33(suppl_1), D514–D517 (2005).
Weinstein, J. N. et al. Cancer Genome Atlas Research Network. The cancer genome atlas pan-cancer analysis project. Nature genetics. 45(10), 1113 (2013).
Wishart, D. S. et al. HMDB 3.0—the human metabolome database in 2013. Nucleic acids research. 41(D1), D801–D807 (2012).
Kuhn, M. et al. STITCH 4: integration of protein–chemical interactions with user data. Nucleic acids research. 42(D1), D401–D407 (2013).
Liberzon, A. et al. Molecular signatures database (MSigDB) 3.0. Bioinformatics. 27(12), 1739–1740 (2011).
Eppig, J. T., Blake, J. A., Bult, C. J., Kadin, J. A. & Richardson, J. E. & Mouse Genome Database Group. The Mouse Genome Database (MGD): facilitating mouse as a model for human biology and disease. Nucleic acids research. 43(D1), D726–D736 (2014).
Han, J., Pei, J. & Kamber, M. Data mining: concepts and techniques. Elsevier (2011).
Paul, B. et al. Influences of diet and the gut microbiome on epigenetic modulation in cancer and other diseases. Clinical epigenetics. 7(1), 112 (2015).
HealthBlog. Phytochemicals in Foods – 9 health benefits of tartaric acid. http://kylenorton.healthblogs.org/2012/03/31/phytochemicals-in-foods-9-health-benefits-of-tartaric-acid/ (accessed in 2017).
Buffie, C. G. et al. Precision microbiome reconstitution restores bile acid mediated resistance to Clostridium difficile. Nature. 517(7533), 205 (2015).
Ussar, S. et al. Interactions between gut microbiota, host genetics and diet modulate the predisposition to obesity and metabolic syndrome. Cell metabolism. 22(3), 516–530 (2015).
Benson, A. K. et al. Individuality in gut microbiota composition is a complex polygenic trait shaped by multiple environmental and host genetic factors. Proceedings of the National Academy of Sciences. 107(44), 18933–18938 (2010).
Acknowledgements
R.X. was supported by the Eunice Kennedy Shriver National Institute Of Child Health & Human Development of the National Institutes of Health under the NIH Director’s New Innovator Award (DP2HD084068, Xu), Research Scholar Grant (RSG-16-049-01 – MPC, Xu) from the American Cancer Society, 2015 Landon Foundation-AACR INNOVATOR Award for Cancer Prevention Research (Grant Number 15-20-27-XU), Mary Kay Foundation Grant (057-15, Xu), and Pfizer Investigator-Initiated Research Grant (WI206753, Xu). Li was supported by NIH/NCI (U01 CA181770, Li) and NIH/NCI (R03 CA212558, Xu).
Author information
Authors and Affiliations
Contributions
Q.W. have jointly conceived, designed and implemented the algorithms and wrote the manuscript. Li Li has participated in manuscript preparation. R.X. have jointly conceived, designed and implemented the algorithms and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, Q., Li, L. & Xu, R. A systems biology approach to predict and characterize human gut microbial metabolites in colorectal cancer. Sci Rep 8, 6225 (2018). https://doi.org/10.1038/s41598-018-24315-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-24315-0
This article is cited by
-
Gut–microbiota–microglia–brain interactions in Alzheimer’s disease: knowledge-based, multi-dimensional characterization
Alzheimer's Research & Therapy (2021)
-
Drug repurposing for opioid use disorders: integration of computational prediction, clinical corroboration, and mechanism of action analyses
Molecular Psychiatry (2021)
-
Automatic extraction, prioritization and analysis of gut microbial metabolites from biomedical literature
Scientific Reports (2020)
-
Challenges in the construction of knowledge bases for human microbiome-disease associations
Microbiome (2019)
-
Data-driven multiple-level analysis of gut-microbiome-immune-joint interactions in rheumatoid arthritis
BMC Genomics (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.