Abstract
The aim of this study was to assess the association between fasting triglyceride (FTG) levels and the prevalence of asymptomatic intracranial arterial stenosis (ICAS). The Asymptomatic Polyvascular Abnormalities Community (APAC) study is a sub-population of the Kailuan study which targeting on the epidemiology of asymptomatic polyvascular abnormalities in Chinese adults. A total number of 5345 participants, aged ≥40, and without history of stroke, transient ischemic attack, and coronary heart disease were enrolled in this study. Transcranial Doppler Ultrasonography was performed for the detection of ICAS presence. Out of 5345 participants, 698 subjects diagnosed ICAS (13.1%). In univariate analysis, the association between fasting TG (FTG) levels and asymptomatic ICAS didn’t reach statistical significance (OR: 0.99, 95% CI: 0.89–1.09; P = 0.79), the same conclusion was reached in multivariate analysis, after adjustment for age, sex (OR: 1.05, 95% CI: 0.95–1.17) and age, sex, current smoking status, hypertension, diabetes, body mass index, estimated glomerular filtration rate, total cholesterol, high-density lipoprotein cholesterol and low-density lipoprotein cholesterol (OR: 0.95, 95% CI: 0.84–1.06), respectively. FTG failed to show any statistical significance on ICAS presence in the APAC study, but the TG actually plays an important role in the progression of atherosclerosis as a biomarker.
Similar content being viewed by others
Introduction
Intracranial arterial stenosis (ICAS) is the result of atherosclerosis that affects large intracranial arteries1, which is a major cause of ischemic stroke worldwide2, especially in Asia3. It has been estimated that 33% to 84% of Asian ischemic stroke patients suffered from ICAS, both symptomatic and asymptomatic4. Since atherosclerotic lesions grow beneath the surface for several years until they become symptomatic, the early intervening treatment for asymptomatic phase, as a predictor for future stroke, is necessary and more effective.
Hyperlipidemia is significantly associated with the severity of ICAS5. Low-density lipoprotein cholesterol (LDL-C) and total cholesterol (TC) are well-established risk factors for atherosclerotic cardiovascular diseases (ASCVD, including ischemic stroke, myocardial infarction, ischemic heart disease). A number of large-scale epidemiological studies have shown that elevated triglyceride (TG) levels are substantial residual cardiovascular risk, even in patients received significant LDL-C lowering therapy with statins6,7,8.
Hypertriglyceridemia, defined as a TG level of ≥150 mg/dL (≥1.7 mmol/L), is brought to the forefront, act as a mediator of atherosclerosis with the growing epidemic of obesity, metabolic syndrome, type-2 diabetes mellitus9. Controversial results observed in studies between TG and ASCVD. Several studies demonstrated a univariate association between them, while after adjustment for other lipid parameters, particularly high-density lipoprotein cholesterol (HDL-C), the association is attenuated or non-significant10,11,12,13. But, it is worth noting that the reduction of TG pharmacologically by fibrate14, niacin15 or omega-3 fatty acids16 can lower the risk of ASCVD, acting as an effective preventive strategy. While recognizing this possibility, we therefore conducted this epidemiologic and observational study to investigate an association between FTG levels and asymptomatic ICAS in a Chinese community-based study population free of stroke, transient ischemic attack and coronary heart disease using samples from the Asymptomatic Polyvascular Abnormalities Community (APAC) study.
Results
Prevalence of ICAS
Out of the study population of 5345 participants, 698 of them were diagnosed of ICAS based on the TCD results, representing a prevalence of 13.1%. Among them, 6.4% (342/5345) had middle cerebral artery stenosis, 4.3% (230/5345) had anterior cerebral artery stenosis, 1.1% (58/5345) had posterior cerebral artery stenosis, 1.2% (66/5345) had vertebral artery stenosis and 1.7% (89/5345) had basilar artery stenosis.
Baseline characteristics
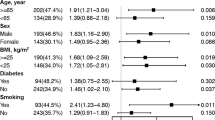
The baseline characteristics of all participants are presented in Table 1. As the number of each clinical TG level were in skewed distribution, medians were used for analysis, the median values were 93.0, 170.1, 270.7, respectively. There were significant differences for age, sex, current smoking status, hypertension, diabetes, body mass index (BMI), estimated glomerular filtration rate (e-GFR), TC, HDL-C and LDL-C (P < 0.05). Except for age and HDL-C whose absolute value decreased as the TG level increased, the rest of these variables shown the opposite tendency. Those with higher TG level were more likely to be men, smokers, suffering from hypertension and diabetes.
Correlation between baseline FTG levels and the prevalence of asymptomatic ICAS
In univariate analysis, the association between FTG levels and asymptomatic ICAS didn’t reach statistical significance (OR: 0.99, 95% CI: 0.89–1.09; P = 0.79). In the multivariate analysis, ICAS were included as dependent variable and the presence of other parameters included as independent variables. After adjustment for age and sex, there was still no evidence shown any correlation between FTG levels and the presence of asymptomatic ICAS (OR: 1.05, 95% CI: 0.95–1.17). Moreover, the result was remained insignificance after the adjustment of age, sex, current smoking status, hypertension, diabetes, BMI, e-GFR, TC, HDL-C and LDL-C (OR: 0.95, 95% CI: 0.84–1.06), additional detailed information were displayed in Table 2.
Sex and other selected risk factors were stratified for further analyses of the association between FTG levels and the prevalence of asymptomatic ICAS, and there was no significant association in each subgroup (Table 3).
Discussion
This study failed to show any significant relationship between FTG levels and the prevalence of asymptomatic ICAS. Several mechanisms can possibly explain the negative result.
First, increasing evidence show that it is TG-rich lipoproteins (TRLs), including chylomicrons, VLDL, and their remnants, rather than TG itself, contribute to the progression of atherosclerosis17. TRLs and their remnants are a cluster of particles which are heterogeneous in size, density as well as composition. There was a meaningful phenomenon that individuals with severe hypertriglyceridemia >100 mmol/L (88000 mg/dL) who suffered from chylomicronemia syndrome caused by lipoprotein deficiency was less likely to develop atherosclerosis than the mild-to-moderate hypertriglyceridemia, which could be explained by the fact that chylomicrons, the major part of TRLs in severe hypertriglyceridemia, were too large to enter the arterial endothelium18. While, the rest of TRLs are small enough to causing intimal cholesterol deposition directly. The TRLs also involved in a series of indirect pathways, including proinflammatory, proapoptotic, and procoagulant19, leading to atherosclerosis together. Furthermore, human genetic studies focusing on TRLs metabolism, supported its contribution to human ASCVD, including APOC3, APOA5, ANGPTL3 and many others20,21,22,23. Indicating that evaluating TG alone is limited, TG is merely a biomarker of TRLs and their remnants, the latter is atherogenic.
Our study discussing the FTG level, while people are in postprandial state for most of the day, leading to a hypothesis that postprandial TG (PTG) may play a more important role in the etiology of atherosclerosis than FTG. The Women’s Health Study, enrolled 20118 fasting and 6391 nonfasting participants, showed that only non-FTG levels were associated with incident cardiovascular events after adjustment for other risk factors, and the TG levels measured between 2 to 4 hours after a meal showed the strongest association24. Additionally, several other epidemiological studies have shown that the non-FTG levels is more closely associated with ASCVD than FTG25,26,27, which led to a change in the guideline. In the Endocrine Society Clinical Practice Guideline, it recommended the diagnose of hypertriglyceridemia on FTG, because the lack of a standard measurement on non-FTG levels28. The recently updated consensus statement by the European Atherosclerosis Society recommended that non-FTG can be used for risk evaluation and general screening29. And TG level has a high intra-individual variability which requires a more standardized method of measurement.
A majority of available literatures suggested that TG was a considerable risk factor for ASCVD, but when it comes to the relationship with possible ischemic stroke, an inconsistent result was obtained. A recent meta-analysis regarding to that, included 7 case-control studies revealed a significant odds ratio of 1.15 (95% CI, 1.08–1.21), but the significance was attributed to a single study only30, another systematic review also drew the similar conclusion31. The underlying mechanisms may attribute to the risk factors between them are different, the diameter of the vessel as coronary artery are larger than intracranial artery (ICA), as well as, ICA had greater antioxidant enzymes than other arteries of the same size32.
What’s more, a skewed distribution in TG levels can be seen in our study, the borderline-high TG group have a number of 735 (13.8%), the high TG group have a number of 954 (17.8%), the rest of the participants are all in the normal group, which may weaken the likelihood of positive result. Another limitation of our study is that when people with inadequate temporal window for TCD, we put them into the non-ICAS group. This may lead to an underestimation of ICAS prevalence. As a result of the observational design, residual confounding may be present because of remaining unknown, unmeasured, or inaccurately measured confounding factors. It is noteworthy that this is the first study investigating the association between FTG and the prevalence of ICAS. TG is a biomarker for atherosclerosis absolutely, but our study failed to show any expecting result for the reasons mentioned above.
In conclusion, FTG failed to show any statistical significance on ICAS presence in the APAC study, and the decades-long debate continues. But the TG level actually plays an important role in the progression of atherosclerosis as a biomarker, additional lifestyle intervention together with TG-lowering agents can reduce the residual risk. Further epidemiological studies are needed to investigate the relationship between them.
Subjects and Methods
Study Population
The APAC study is a community-based, prospective, long-term follow-up observational study targeting on the epidemiology of asymptomatic polyvascular abnormalities in Chinese adults. The study was sub-population of the Kailuan study which described previously33. A total of 5852 people stratified randomly by age and sex agreed to join the APAC study, and 5816 participants eventually completed the baseline data collection from June 2010 to June 2011. Among these 5816 participants, 376 did not meet the inclusion criteria: (1) aged 40 years or older; and (2) no history of stroke, transient ischemic attack, and coronary heart disease at baseline. Besides, 90 subjects with incomplete information on TG data, vascular examination results or under lipid-lowering treatment were further excluded. Finally, 5345 participants were available in this study, where 3212 participants are men.
The study was performed according to the guidelines from the Helsinki Declaration and was approved by the Ethics Committees of the Kailuan General Hospital and the Beijing Tiantan Hospital. Written informed consent was obtained from all participants. Subjects were also informed of abnormal findings and recommended treatment.
Measurement of Indicators
A questionnaire was used to obtain baseline information by trained investigators on enrolled participants, containing age, sex, smoking status, past medical history such as hypertension, diabetes, hyperlipidemia, and medications prescribed by physicians. The baseline data were further categorized by different parameters. Age was classified into “<60 years” and “≥60 years”. Smoking status was classified into “non-smoking” and “smoking” (current smoker who smokes at least one cigarette per day) by self-reported information.
Definition of hypertension, diabetes and hyperlipidemia
Hypertension was diagnosed if the subject had a history of hypertension, or a systolic blood pressure (SBP) ≥140 mmHg, or a diastolic blood pressure ≥90 mmHg, or currently taking antihypertensive medication. Diabetes mellitus was diagnosed if the subjects had a history of diabetes mellitus, or currently undergoing treatment with insulin or oral hypoglycemic, or the fasting blood glucose level was ≥7.0 mmol/L. Hyperlipidemia was diagnosed if the subject had a history of hyperlipidemia, or currently using cholesterol lowering agents, or the TC level ≥ 5.17 mmol/L or TG ≥ 1.7 mmol/L.
Physical examination
Body weight, height was measured and then BMI was calculated as body weight (kg) divided by the square of height (m2). For blood pressure, the participants should sit quietly for 5 minutes before the measurement, an average of two readings was used for the current study. If the two measurements differed by more than 5 mmHg, a third reading was taken, and the average of the three readings was used.
Biochemical Index
Blood samples were obtained from the antecubital vein after an overnight fasting in EDTA tubes and analyzed within 4 h of preparation using an auto-analyzer (Hitachi 747; Hitachi, Tokyo, Japan) at the central laboratory of the Kailuan hospital. Estimated GFR (eGFR) was calculated by using a modified 4-variable Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) formula with an adjusted coefficient of 1.1 for the Chinese population to estimate eGFR34. TC was measured using the endpoint test method. HDL-C and LDL-C levels were measured using a direct test method and TG were measured using the GPO method (inter-assay coefficient of variation: <10%; Mind Bioengineering Co. Ltd, Shanghai, China).
The following is the criteria for clinical diagnosis of elevated triglyceride levels under fasting conditions according to the Endocrine Society 2010 Guideline. Normal TG: <1.7 mmol/L, borderline-high TG: 1.7–2.3 mmol/L, high TG: 2.3–11.1 mmol/L, very high TG: 11.2–22.4 mmol/L and very severe ≥22.4 mmol/L28 (to convert triglyceride values to mg/dL multiply by 88.6). Accordingly, we classify the last 3 group together as ≥2.3 mmol/L in consideration of the limited number of participants.
Trans-cranial Doppler Ultrasonography (TCD) Examination
TCD, as a non-invasive, reliable and available method to diagnose ICAS35, was performed by two experienced neurologists using portable devices (EME Companion, Nicolet, Madison, WI, USA). The two neurologists were blinded to the baseline information of the participants. ICAS was defined according to the peak flow velocity criteria that was published and validated against MR angiography (MRA) and clinical outcomes36. In short, ICAS was defined by a peak systolic flowing velocity (Vp) >140 cm/s for the middle cerebral artery; >120 cm/s for the anterior cerebral artery and internal carotid siphon; and >100 cm/s for the posterior cerebral artery and vertebral-basilar. In addition to the above mentioned criteria, the patients’ age, the presence of turbulence sound or disturbance in echo frequency, and whether the abnormal velocity was segmental were also taken into account for ICAS diagnosis. Subjects without a good temporal window were considered without stenosis. ICAS was diagnosed if at least one of the studied arteries showed evidence of stenosis or occlusion.
Statistical analysis
We classified the participants into three groups according to clinical diagnosis of elevated triglyceride levels. All statistical analyses were performed using SAS software, version 9.1 (SAS Institute, Cary, North Carolina, USA). Continuous variables were compared using analysis of variance (ANOVA) and categorical variables were compared using chi-square tests. The age- and sex-adjusted or multivariate-adjusted odd ratios (OR) and 95% confidence interval (CI) were calculated using logistic regression model on the relationship between FTGlevels and ICAS. The multivariate-adjusted model were further adjusted for age, sex, current smoking status, hypertension, diabetes, BMI, e-GFR, TC, HDL-C and LDL-C levels. A trend test was applied to examine whether there was a dose-dependent relationship between the three clinical levels of FTG and ICAS prevalence. Categorical data were treated as continuous data using the median level of FTG levels in each group. Additionally, sex and other potential indicators were also evaluated in subgroups, to assess if there was any significant interaction between these variables and the relationship between FTG levels and ICAS prevalence. All statistical analyses were two-tailed, and a P value less than 0.05 was considered statistically significant.
Change history
17 May 2019
A correction to this article has been published and is linked from the HTML and PDF versions of this paper. The error has been fixed in the paper.
References
Battistella, V. & Elkind, M. Intracranial atherosclerotic disease. Eur J Neurol 21, 956–962 (2014).
Holmstedt, C. A., Turan, T. N. & Chimowitz, M. I. Atherosclerotic intracranial arterial stenosis: risk factors, diagnosis, and treatment. Lancet neurology 12, 1106–1114 (2013).
Suri, M. F. & Johnston, S. C. Epidemiology of intracranial stenosis. Journal of neuroimaging: official journal of the American Society of Neuroimaging 19(1), 11S–16S (2009).
Sung, Y. F. et al. Reversal of ophthalmic artery flow and stroke outcomes in Asian patients with acute ischemic stroke and unilateral severe cervical carotid stenosis. PLoS One 8, e80675 (2013).
Turan, T. N. et al. Risk factors associated with severity and location of intracranial arterial stenosis. Stroke 41, 1636–1640 (2010).
Fruchart, J.-C. et al. The Residual Risk Reduction Initiative: A Call to Action to Reduce Residual Vascular Risk in Patients with Dyslipidemia. The American journal of cardiology 102, 1K–34K (2008).
Reiner, Ž. Managing the residual cardiovascular disease risk associated with HDL-cholesterol and triglycerides in statin-treated patients: A clinical update. Nutrition, Metabolism and Cardiovascular Diseases 23, 799–807 (2013).
Sampson, U. K., Fazio, S. & Linton, M. F. Residual Cardiovascular Risk Despite Optimal LDL-Cholesterol Reduction with Statins: The Evidence, Etiology, and Therapeutic Challenges. Current atherosclerosis reports 14, 1–10 (2012).
Miller, M. et al. Triglycerides and cardiovascular disease: a scientific statement from the American Heart Association. Circulation 123, 2292–2333 (2011).
Nordestgaard, B. G., Benn, M., Schnohr, P. & Tybjærg-Hansen, A. Nonfasting triglycerides and risk of myocardial infarction, ischemic heart disease, and death in men and women. JAMA 298, 299–308 (2007).
Freiberg, J. J., Tybjærg-Hansen, A., Jensen, J. & Nordestgaard, B. G. Nonfasting triglycerides and risk of ischemic stroke in the general population. JAMA 300, 2142–2152 (2008).
Patel, A. et al. Serum triglycerides as a risk factor for cardiovascular diseases in the Asia-Pacific region. Circulation 110, 2678–2686 (2004).
Sarwar, N. et al. Triglycerides and the risk of coronary heart disease: 10,158 incident cases among 262,525 participants in 29 Western prospective studies. Circulation 115, 450–458 (2007).
Scott, R. et al. Effects of Fenofibrate Treatment on Cardiovascular Disease Risk in 9,795 Individuals With Type 2 Diabetes and Various Components of the Metabolic Syndrome: The Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) study. Diabetes care 32, 493–498 (2009).
Landray, M. J., Haynes, R. & Armitage, J. Niacin for reduction of cardiovascular risk. N Engl J Med 371, 1943–1944 (2014).
Yokoyama, M. et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): a randomised open-label, blinded endpoint analysis. The Lancet 369, 1090–1098 (2007).
Do, R. et al. Common variants associated with plasma triglycerides and risk for coronary artery disease. Nature genetics 45, 1345–1352 (2013).
J. Brunzell, S. Deeb Familial lipoprotein lipase deficiency, apoC-II deficiency, and hepatic lipase deficiency. (2001).
Rosenson, R. S., Davidson, M. H., Hirsh, B. J., Kathiresan, S. & Gaudet, D. Genetics and causality of triglyceride-rich lipoproteins in atherosclerotic cardiovascular disease. Journal of the American College of Cardiology 64, 2525 (2014).
Pollin, T. I. et al. A Null Mutation in Human APOC3 Confers a Favorable Plasma Lipid Profile and Apparent Cardioprotection. Science (New York, NY) 322, 1702–1705 (2008).
Romeo, S. et al. Rare loss-of-function mutations in ANGPTL family members contribute to plasma triglyceride levels in humans. The Journal of Clinical Investigation 119, 70–79 (2009).
Willer, C. J. et al. Discovery and Refinement of Loci Associated with Lipid Levels. Nature genetics 45, 1274–1283 (2013).
Peloso, G. M. et al. Association of Low-Frequency and Rare Coding-Sequence Variants with Blood Lipids and Coronary Heart Disease in 56,000 Whites and Blacks. American Journal of Human Genetics 94, 223–232 (2014).
Bansal, S. et al. Fasting compared with nonfasting triglycerides and risk of cardiovascular events in women. Digest of the World Core Medical Journals 298, 309–316 (2007).
Iso, H. et al. Nonfasting serum triglycerides and risk of ischemic heart disease among Japanese men and women. International Congress 153, 490 (2001).
Mora, S., Rifai, N., Buring, J. E. & Ridker, P. M. Fasting compared with nonfasting lipids and apolipoproteins for predicting incident cardiovascular events. Circulation 119, e384, author replye 385 (2009).
Nordestgaard, B. G., Benn, M., Schnohr, P. & Tybjærghansen, A. Nonfasting Triglycerides and Risk of Myocardial Infarction, Ischemic Heart Disease, and Death in Men and Women. Digest of the World Core Medical Journals 298, 299–308 (2007).
Berglund, L. et al. Evaluation and Treatment of Hypertriglyceridemia: An Endocrine Society Clinical Practice Guideline. Journal of Clinical Endocrinology & Metabolism 97, 2969–2989 (2012).
M. Authors/TaskForce et al. 2016 ESC/EAS Guidelines for the Management of Dyslipidaemias: The Task Force for the Management of Dyslipidaemias of the European Society of Cardiology (ESC) and European Atherosclerosis Society (EAS) Developed with the special contribution of the European Assocciation for Cardiovascular Prevention & Rehabilitation (EACPR). Atherosclerosis 253, 281–344 (2016).
Leonards, C. The role of fasting versus non-fasting triglycerides in ischemic stroke: a systematic review. Frontiers in Neurology 1 (2010).
Labreuche, J., Touboul, P. J. & Amarenco, P. Plasma triglyceride levels and risk of stroke and carotid atherosclerosis: a systematic review of the epidemiological studies. Atherosclerosis 203, 331–345 (2009).
D’Armiento, F. P. et al. Age-related effects on atherogenesis and scavenger enzymes of intracranial and extracranial arteries in men without classic risk factors for atherosclerosis. Stroke 32, 2472–2479 (2001).
Wu, J. et al. Association between non-high-density-lipoprotein-cholesterol levels and the prevalence of asymptomatic intracranial arterial stenosis. PLoS One 8, e65229 (2013).
Teo, B. W. et al. GFR estimating equations in a multiethnic Asian population. American Journal of Kidney Diseases the Official Journal of the National Kidney Foundation 58, 56–63 (2011).
Sharma, V. K., Wong, K. S. & Alexandrov, A. V. Transcranial Doppler. Frontiers of Neurology & Neuroscience 40, 124 (2016).
Gao, S., Lam, W. W., Chan, Y. L., Liu, J. Y. & Wong, K. S. Optimal values of flow velocity on transcranial Doppler in grading middle cerebral artery stenosis in comparison with magnetic resonance angiography. Journal of Neuroimaging 12, 213 (2002).
Acknowledgements
We appreciate all the participants and staffs for their contributions. This study is supported by the grants from the Ministry of Science and Technology and the Ministry of Health of the People’s Republic of China (No. 2008 BAI52B03), by a grant from the National Natural Science foundation of China (No. 81202279) and the 12th Five-Year Plan for “New Drugs innovation and Development” National Science and Technology Major Project: Clinical Resource Biobank Construction (2011ZX09307-001-06).
Author information
Authors and Affiliations
Contributions
J.W. and X.Z. conceived and designed this study, A.W. prepared the database and directed data analysis, J.W and Y.W. writing the paper. A.W., J.X. and X.Z. conducted the quality assurance and edited the paper. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wu, J., Wang, Y., Wang, A. et al. Association between fasting Triglyceride levels and the Prevalence of Asymptomatic Intracranial Arterial Stenosis in a Chinese Community-based Study. Sci Rep 8, 5744 (2018). https://doi.org/10.1038/s41598-018-24157-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-24157-w
This article is cited by
-
Dyslipidemia and its associated factors among community adults located in Shangcheng district, Zhejiang province
Scientific Reports (2024)
-
Prevalence and risk factors for dyslipidemia among South Indian adults: A community based-NCD study
International Journal of Diabetes in Developing Countries (2023)
-
Prevalence and risk factors for dyslipidemia among adults in rural and urban China: findings from the China National Stroke Screening and prevention project (CNSSPP)
BMC Public Health (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.