Abstract
We evaluated five non-mutually exclusive hypotheses driving the biogeographic regions of anuran species in the Amazonia. We overlaid extent-of-occurrence maps for anurans 50 × 50 km cells to generate a presence–absence matrix. This matrix was subjected to a cluster analysis to identify the pattern and number of biogeographic regions for the dataset. Then, we used multinomial logistic regression models and deviance partitioning to explore the relative importance of contemporary and historical climate variables, topographic complexity, riverine barriers and vegetation structure in explaining the biogeographic regions identified. We found seven biogeographic regions for anurans in the Amazonia. The major rivers in the Amazonia made the largest contribution to explaining the variability in anuran biogeographic regions, followed by climate variables and topography. The barrier effect seems to be strong for some rivers, such as the Amazon and Madeira, but other Amazonia rivers appear to not be effective barriers. Furthermore, climate and topographical variables provide an environmental gradient driving the species richness and anuran range-size distributions. Therefore, our results provide a spatially explicit framework that could be used to address conservation and management issues of anuran diversity for the largest tropical forests in the world.
Similar content being viewed by others
Introduction
The spatial patterns of species distributions express many ecological and evolutionary processes and are linked to a complex and historically contingent setting. Since the 19th century, studies have divided large geographic extents into regions of similar faunistic or floristic composition1,2,3,4. This approach, called biogeographical regionalization, has helped us understanding whether the processes influencing species distributions are determined by shared evolutionary histories (i.e. speciation, extinction and distribution), past or current climatic oscillations (i.e. precipitation and temperature gradients) and/or physical barriers (i.e. mountains and oceans) that limit species dispersal between areas2,4,5,6,7,8. For example, Holt et al.2 identified 20 distinct zoogeographic regions by combining data on the distributions and phylogenetic relationships of vertebrate species and found that spatial turnover in phylogenetic composition is higher in the Southern than in the Northern Hemisphere. Furthermore, global biogeographical regionalization has been used to evaluate international conservation priorities based on the degradation of natural habitats and ecosystems as a result of human activities3,9. Although large-scale global patterns are relatively well established2,4,9, intracontinental regionalization patterns are still scarce for some Neotropical areas, representing an opportunity for new insights about the processes influencing species distributions8,10.
The Amazonia encompasses more than 6 million km2 across eight countries in South America and is one of the most critical natural environments both in regulation climate and sustaining biodiversity at global scale11,12. Currently, Amazonia is threatened by several anthropic pressures, such as dam constructions, deforestation, and fire that will cascade onto the patterns of species distribution of the largest and most species-rich tropical forest in the world. Although, previous studies have delimited biogeographical regions for mammals and birds in the Amazonia, their results are not consensual. For example, Wallace1, considering primate ranges, identified four regions in the Amazonia. Haffer13, Cracraft14 and Silva et al.15, considering bird ranges, identified six, seven and eight regions in the Amazonia respectively. Thus, uncertainty about biogeographical regionalization in the Amazonia remains open to debate and different hypotheses have been proposed to explain the pattern of species distributions in the Amazonia1,16,17,18,19. Among competing hypotheses, the riverine barrier hypothesis states that the major rivers of Amazonia act as geographic barriers to gene flow, promoting the genetic divergence of populations and, therefore, speciation1. The Pleistocene refuge hypothesis states that during the Pleistocene, decreases in temperature and humidity in the Amazonia Basin left relatively small ‘islands’ of tropical rainforests surrounded by xeric habitats, isolating populations and changing distribution patterns4,16. The orogenic hypothesis states that the uplift of the Andes in Neogene and its effect on regional climate has had a substantial impact on the landscape evolution in the Amazonia18. Therefore, the pattern of species distribution in Amazonia will not be explained entirely by any single simple model, but it depends on the combination of more realistic, complex scenarios19,20,21.
Biogeographical units are hierarchically arranged, and no single biogeographic framework is optimal for all taxa2,3,6,9. To date, no study has evaluated the importance of multiple scenarios shaping present-day patterns of amphibian species composition along the Amazonia. Amphibians are the most threatened vertebrate group22, with Amazonia harboring the highest species richness in the world23. Moreover, patterns of amphibian species richness distribution are not randomly distributed throughout the Amazonia23,24. Because amphibian species are normally separated into more regions than other vertebrate groups due to their small-ranges25 and physiological constraints6,26, we believe that the Amazonia will present more than the eight regions previously proposed for birds14,15. Here, we performed a regionalization scheme for the current original extent of the Amazonia in order to explore how anurans are distributed throughout this complex and biodiverse biome. Our goal is to determine the biogeographical regions for anuran species in the Amazonia evaluating five non-mutually exclusive hypotheses:
-
i)
Contemporary Climate hypothesis – present-day climate variables are key environmental determinants of anuran composition because they act as environmental filters influencing which species can inhabit specific areas26,27. Under this hypothesis is expected that areas with different climate gradients would harbor distinct species compositions due to specific physiological requirements or life history traits;
-
ii)
Pleistocene Climate Variation hypothesis – while current patterns of amphibian distributions in Europe28 and Brazilian Atlantic Forest29 were shaped by climate changes in the past, there is still no evidence that amphibian distributions in Amazonia has been influenced by Pleistocene climate variation30. However, Amazonia presents a large spatial extent, and stable climatically areas since the Pleistocene were not randomly distributed in the space15. Under this hypothesis is expected that areas that maintained similar climatic conditions, but are far apart from each other, would harbor distinct species compositions due to dissimilar rates of speciation, extinction and colonization that delimited different regional species pools31,32 along the Amazonia;
-
iii)
Topography hypothesis – areas with larger ranges in elevation increase the speciation rate and endemism18,33. Under this hypothesis is expected that these areas would harbor small-ranged species with historically limited dispersal capabilities due to physical barriers and/or physiological constraints;
-
iv)
Vegetation Structure hypothesis – the concept of habitat templets argues that habitat provides the templet on which evolution forges animal life-history strategies. Based on this idea, previous studies have found that floristic structure has a strong correlation with the biogeographic regions of amphibians identified in Europe6 and the Brazilian Atlantic Forest8,10. Under this hypothesis is expected that the biographical regions of anurans would be recognized because of the vegetation distribution within the Amazonia;
-
v)
Riverine Barrier hypothesis – the major rivers of Amazonia act as geographic barriers to the dispersal of organisms and hamper gene flow between populations, increasing speciation rates1,12,34. Under this hypothesis is expected that some anuran species could not traverse the major rivers in the Amazonia, thus creating different species compositions between opposite banks of the major rivers of Amazonia.
Results
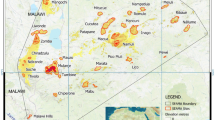
We identified seven biogeographic regions in the Amazonia based on anuran species composition with explained dissimilarity values of 92% and a mean silhouette width of 0.33 (Fig. 1, Table 1). From the seven biogeographic regions observed, three biogeographical regions (BR1, BR2, and BR3) are delimited to the north of the Amazon River, three (BR5, BR6, and BR7) are delimited to the south of the Amazon River and one (BR4) is delimited in the western portion of Amazonia (Fig. 1). The grids in BR4 contain the highest values of species richness, while the grids in BR3, BR6, and BR7 contain the lowest values (Fig. 2A). Based on the range size of species distributions, we observed that BR4 contains anuran species with restricted range sizes while the grids in the BR1 and BR7 contain anuran species with wide range sizes (Fig. 2B).
Dendrogram and Amazonia map depicting the regionalization of anuran dissimilarity into seven biogeographical regions (BR) based on recluster.region algorithm68. Colors used in dendrogram and map are identical. Black lines in the map represent the ten major rivers in Amazonia. Map generated using ESRI ArcMap 9.2. https://www.esri.com.
Gradients of (A) anuran species richness, and (B) mean range-size of anuran species occurring in each grid of the Amazonia. Black lines are delimiting the seven biogeographical regions of anuran identified in Fig. 1. Maps generated using ESRI ArcMap 9.2. https://www.esri.com/.
Among all models of predictor variables, the model without VEGE.PC1 and TOPO.PC2 was the best one for explaining the cluster patterns (∆ AICc > 6.4; Table 2). This model explains 80% of the cluster patterns (Table 2). The partitions of deviance indicated that the independent effect of riverine barriers accounted for 38% of the variability in the anuran biogeographic regions, followed by climate variables with 16% and topography with 3% (Fig. 3). Vegetation structure has a weak association with anuran biogeographic regions in the Amazonia (Fig. 3).
Discussion
This is the first study showing that multiple factors shape anuran biogeographical regions in Amazonia. We found that the major rivers in Amazonia strongly contributed to explaining the variability in anuran biogeographic regions, followed by climate and topography variables. We identified seven biogeographic regions that partially overlap with the eight areas of endemism previously proposed for terrestrial vertebrates in Amazonia14,15. To the north of the Amazon River, we found that part of BR1, BR2, and BR3 are nested in the area corresponding to Guiana, a biogeographic unit identified by Cracraft14. BR4 and BR5 are partially congruent with the areas of Napo and Inambari, identified by Cracraft14, respectively. However, BR6 and BR7 in the southern Amazon River differ from spatial arrangements of the Rondonia, Tapajós, Xingu, and Belém areas of Cracraft14 and Silva et al.15. There is no single biogeographic solution that is optimal for all taxa1,2,4,7,14,15. For example, Rueda et al.6 found substantial variation in the number of regions considering different taxonomic groups in Europe. Naka20, redefined the boundaries of the Guiana region14 for Amazonia birds using different quantitative methods. Therefore, regionalization patterns depend on the taxonomic group of interest or the clustering methods used to delineate biogeographic units7,35. Because previous studies in Amazonia were performed with the spatial distributions of primate1 and bird14,15,20 species, our results provide new information about the factors associated with the spatial patterns of anuran species distribution in Amazonia.
A cluster analysis based on amphibian distribution recognized the boundaries of Amazonia as one of the four biogeographic regions in South America27. We scaled down the analysis and described the effects of riverine barriers, climatic and topographic variables acting inside Amazonia. Our results showed that the Amazon River separates the biogeographical regions in the north (BR1, BR2, and BR3) from those in the south (BR5, BR6 and BR7), while the Madeira river separates the southeastern biogeographical regions (BR6 and BR7) from that in the southwest (BR5). A longstanding debate exists as to whether the riverine barrier hypothesis has played an important role in shaping the present-day species distribution patterns in Amazonia1,14,19,21,33,34,36. Wallace1 defined distinct areas based on primate species composition that were separated by the Amazon, Solimões, Negro, and Madeira rivers. Recently, Dias-Terceiro et al.37 and Moraes et al.34 showed that the Madeira and Tapajós River respectively are barriers to some amphibian lineages in western and eastern Amazonia. In contrast, Gascon et al.33 did not find a relationship between amphibian species composition and the banks of the Juruá River. Taken together, these results indicate that rivers contribute unequally to the observed patterns of amphibian distribution in the Amazonia. Oliveira et al.21 found similar results to bird distributions and showed that some bird species with low dispersal ability were limited by all major Amazonia rivers, while many other species can apparently cross some rivers. Thus, the barrier effect might be strong for some rivers, such as the Amazon and Madeira, but others rivers might not be an effective barrier. We still lack a consensus on why different rivers are barriers to some species of mammals, birds, and amphibians but not others. To improve our understanding, we must consider life-history traits, dispersal ability, and phylogenetic relationships that are undoubtedly important factors related to the patterns of species distributions21,34. However, considering that over 2,000 new species of plants and vertebrates having been described since 199912, several of these information are currently lacking for most of species in the Amazonia.
Climate and topographic variables explained the second and third highest percentages of variance in the distribution of biogeographic regions, respectively. This result agrees with previous studies that defined biogeographic regions for amphibians in South America27, Europe6, the Atlantic Forest8 and at a global scale4. Amazonia has a well-defined climate gradient, with southeastern areas presenting warmer and more seasonal climate than northwestern areas11,18. This pattern is associated with orography of the northwestern areas, which contain the highest elevations in the Amazonia. We found that most small-ranged anuran species inhabit biogeographic regions with high elevations and humidity. Mountains affect species richness by fostering the diversification of unique lineages and as natural barriers to species with limited dispersal ability38,39. The distribution of amphibian species richness is usually associated with physiological constraints that reflects differences in tolerance to precipitation and temperature38,40,41,42. For example, Da Silva et al.26 found that humidity-related variables are key environmental factors related to both the richness of reproductive modes and anuran phylogenetic diversity in the Brazilian Atlantic Forest. Variation in the climatic and orographic variables seem to influence speciation, extinction, and dispersal rates of anuran species throughout Amazonia43,44. Therefore, different from Ficetola et al.4 who found that continental drift, climate differences, and mountain chains interact to determine the boundaries of biogeographic regions at global scale, we highlight an important role for climatic and orographic variables shaping anuran distributions at intermediate scale.
Previous studies have found that vegetation structure is an important factor related to biogeographical regions for amphibians6,8. In contrast, we found that vegetation types have a weak association with biogeographical regions. According to Charity et al.12, moist forest is the dominant vegetation type in the Amazonia, covering nearly 80 percent of the biome; other forest types include flooded and swamp forests (3.9 per cent), deciduous forest (1.4 per cent), savannah (6.8 per cent) and others (1.1. per cent). At broad scales, this homogenization of vegetation decreases the importance of vegetation structure in explaining the distribution of biogeographical regions. However, this is not the case when considering finer scales. For example, Gascon et al.33 found that flooded versus upland forest is an important predictor of community similarity in species composition of amphibians at the Juruá River. Islands of savannah of varying size occurring within the Amazonia biome are home to unique flora and fauna, including numerous endemics. Nonetheless, Amazonia savannahs are little known, highly threatened, and under-protected45. Thus, vegetation structure might be important for the distribution of biodiversity and conservation purposes when evaluating the biogegraphical units at finer resolutions.
For the first time, BR1 appears as a biogeographic region in the central part of Amazonia. One possible explanation for the identification of BR1 is that it is a biogeographical transition zone, representing geographical areas of species overlap, with a gradient of replacement and partial segregation between anuran species from neighboring biogeographic regions creating a distinct species composition46,47. Biogeographical transition zone is an area where historical and ecological changes allow both the mixture and the co-occurrence of species from two or more biogegraphical regions46. For example, the boundaries of BR1 are in contact with those of six biogeographic regions. If BR1 shares some anuran species with each of the six neighboring biogeographic regions, its identification as a biogeographic transition zone is valid. However, our knowledge of biodiveristy distribution is far from complete, and the geographical distribution of species already described is also fragmentary (i.e. Wallacean shortfall48). We are aware that the accuracy of amphibian range maps is not without criticism, mainly in megadiverse tropical regions, such as Amazonia49. Thus, the identification of BR1 could also be an artefact of the limitation in the knowledge about anuran distribution49. For example, Naka20 found a single area of endemism for 85 avian species in the Guiana shield that coincides with part of our BR1 and BR2 boundaries. This area of endemism is congruent with the Amazon River to the south, the lower Negro river to the south-west, and the Branco river to the west20. The remaining part of BR1 are congruent with the area of Imeri identified by Cracraft14. This area of endemism is congruent with the Negro River to the north-east and the Japurá river to the south-west14. Therefore, future studies with more accurate information on anuran distribution in Amazonia will be able to answer whether BR1 is a valid biogeographic region or an artifact of limited current datasets.
Biogeographical regionalization provides a framework for addressing evolutionary and ecological processes that underlie present-day distributions and several studies have used them as templates to test areas of endemism, historical relationship among areas, delimit regional species pools, and investigate macroecological patterns5,7,9,31,47. Understanding the occurrence of different species in particular geographical areas permit the identification of patterns that can be the starting point in conservation biogeography50,51. For example, the frog-killing fungus Batrachochytrium dendrobatidis, has been linked to extirpations and extinctions of amphibian species in several continents52 and one of the main hypotheses explaining this decline is the side effects of climate change53,54. Becker et al.55 found an increase in Batrachochytrium dendrobatidis positive samples in the southwestern Amazonia, coinciding with reported amphibian declines in neighboring high elevation sites on Andean slopes of Peru. Considering that the pathogen thrives in cool, moist environments in high-elevation tropical rainforests, our results indicate that anuran species occurring in BR4 would be the most susceptible to Batrachochytrium dendrobatidis expansion and anuran species populations in this region should be careful monitored.
Currently, the integrity of the Amazonia is under pressure from dam constructions, deforestation, climate change and unsustainable economic activities12,56,57. For example, large dam constructions could not only block movements that connect anuran populations, but also result in the loss of terrestrial habitats by flooding indigenous lands and conservation units that are protecting several endemic and undescribed species56,58. Based on the predictions of Latrubesse et al.57, if the planned dams are constructed in Amazon basin, BR4 and BR5 will be the most impacted biogeographic regions. These regions harbor the highest anuran species richness, with most species showing a restricted range-size distribution. Furthermore, future projections indicated that agricultural expansion and climate variability will change regional precipitation patterns in Amazonia11,59,60. Sorribas et al.60 projected a decrease in river discharges for eastern basins, and decrease in inundation in central and lower Amazonia. These projections are worrisome because most of these changes will occur with replacement of tropical forest by seasonal forest and tropical savanna59. The likelihood of “savannization” of parts of Amazonia could favor the invasion of these altered areas by anuran species from the Cerrado that are more resistant to desiccation and have more generalized reproductive mode61. Taken together, these actions could threat the integrity of the ecosystem, and alter the patterns of species distribution.
Methods
Species distribution data
We downloaded range maps for all species of anurans recorded in the Amazonia region from the IUCN version 2015.262. Then, we overlaid the range maps into grid cells at 50 × 50 km to generate a presence–absence matrix and determine the number of species by grid cell. We considered the extent of the Amazonia region based on the Cracraft14 delimitation and subsequently modified by Silva et al.15. We excluded all species from other biomes (e.g. Cerrado) with marginal occurrences inside the Amazonia region. In the end, a total of 577 anuran species were considered for the regionalization process (see Appendix S1 in Supporting Information). We standardized the nomenclature of anuran species following the Amphibian Species of the World (Frost)63.
We are aware that biogeographical inferences are affected by incomplete taxonomic and distributional knowledge7,64. Although the IUCN anuran maps might include either over- or underpredictions mainly in megadiverse tropical regions49, range maps have been used to investigate amphibian regionalization across a range of spatial scales4,6,8. Furthermore, from a macroecological perspective, range maps have performed very well at resolutions greater than 50 × 50 km65. However, to understand the effects of anuran species that were described recently or whose range size distribution is underpredicted, we also analyzed three other datasets excluding from the presence–absence matrix the small-ranged species that occurred in only one (501 species remained in the matrix), two (440 species) and three (418 species) grid cells. Biogeographical regions delimited using the 577 anuran species and the three datasets excluding small-ranged species were similar. Therefore, we will present only the results considering the 577 anuran species (see Appendix S2 in Supporting Information for a discussion about the results).
Clustering procedures
We used the recluster.region algorithm66,67 available in the recluster R package68 to identify the biogeographic regions in Amazonia with distinct anuran species compositions. This algorithm calculates the dissimilarity of species compositions between each pair of grid cells using the Simpson index (βsim), which is not affected by variations in species richness:
where component a comprises the total number of species shared by two grids; component b comprises the total number of species that occur in the neighboring grids but not in the focal one; and component c comprises the total number of species that occur in the focal grid but not in the neighboring one. This index is a desirable choice for regionalization because species replacement is largely influenced by vicariance and endemism phenomena7. Then, we used Ward hierarchical clustering to convert dissimilarity matrices into bifurcated dendrograms69. This method performs better in a simulation for recognizing regionalization patterns than other hierarchical clustering methods commonly used for biogeographical analyses67. According to Dapporto et al.66, due to a high frequency of ties and zero values produced by beta-diversity turnover indices, the topology and bootstrap support of dendrograms are affected by the order of areas in the original presence–absence matrix. To avoid these problems, the recluster.region algorithm produces n trees (n = 50 by default) by randomly reordering the areas in the original dissimilarity matrix. Next, the function cuts these trees at different k1 − kn levels (i.e. the number of regions to be identified), producing n matrices of areas x cluster membership67. We delimited the maximum number of regions at 50 clusters. Lastly, to identify the number of regions, the function provides the explained dissimilarity2 and the mean silhouette width70 for all the clustering solutions. The explained dissimilarity is represented by the ratio between the sums of the mean dissimilarities among members of different clusters and the sum of all dissimilarities in the matrix. This method maximizes the between-cluster variation relative to the within-cluster variation. According to Holt et al.2, clusters that reach the threshold value of 90% are an appropriate choice for establishing a suitable tree cut. The mean silhouette width measures the strength of any of the partitions of objects from a dissimilarity matrix. This index ranges between −1 and +1, with negative values indicating that cells are probably located in incorrect clusters70. Here, we identify biogeographic regions based on the number of clusters that considerably improved the explained dissimilarity and the mean silhouette width together. For that, we first found the number of cluster that reach the threshold value of 90% proposed by Holt et al.,2, then we delimited the cluster number when the mean silhouette value stopped increasing.
Predictor variables
To test the potential correlates in the anuran cluster patterns, we obtained current and historical climate data, topographic data, riverine barriers and vegetation structure, which are detailed below:
Current climate variables – the selected climate variables were: i) average annual maximum temperature (AMAXTE); ii) average annual minimum temperature (AMINTE); iii) temperature seasonality (TESE); iv) annual precipitation (APRE); v) precipitation range (PRER); and vi) precipitation seasonality (PRSE). These variables were chosen because they describe a central tendency as well as the variation in the descriptors representing physiological limits or dispersal barriers for anurans6,8,25. These data were downloaded from the WorldClim database at a resolution of 5′ arc-minutes71.
Pleistocene climate variables – we downloaded the values of annual precipitation and annual mean temperature from three models of the Last Glacial Maximum (LGM; CCSM4, MIROC-ESM, MPI-ESM-P) available from the WorldClim database (http://www.worldclim.org/downscaling). Following Moura et al.10 we calculated two historical difference in climate variables: i) historical difference in annual precipitation (HDP) was calculated by the difference between current and LGM annual precipitation; and ii) historical difference in annual mean temperature (HDT) was calculated by the difference between current and LGM annual mean temperature. These two measures indicate the historical variation in water availability and energy input respectively. In order to couple with the variations among the circulation models, we averaged the grid cell values among them prior to the calculation of historical difference10.
Topographic variables – for each grid cell, we calculated six measures of topographic heterogeneity based on elevation data (~1 × 1 km resolution) available at https://lta.cr.usgs.gov/GTOPO30. These measures were: i) maximum elevation (TOPOMAX); ii) minimum elevation (TOPOMIN); iii) elevational standard deviation (TOPOSTD); iv) slope range (SLOPERAN); v) slope standard deviation (SLOPESTD); and vi) aspect standard deviation (ASPECTSTD).
Riverine barrier – we categorized the grid cells into different regions based on the banks of the largest rivers in the Amazonia in terms of water discharge72 and preview studies14,17: i) Amazon (mean annual discharge − 209000 m3/s), ii) Orinoco (35000 m3/s), iii) Madeira (32000 m3/s), iv) Negro (28400 m3/s), v) Japurá (18600 m3/s), vi) Tapajós (13500 m3/s), vii) Purus (11000 m3/s), viii) Xingu (9700 m3/s), ix) Uacayali (9544 m3/s), x) Putumayo (8760 m3/s), xi)Tocantins (8440 m3/s) and xii) Rio Branco (1462 m3/s) (Fig. 4). These data were downloaded from the database of USGS at https://www.sciencebase.gov/catalog/item/56814fc2e4b0a04ef492213e.
Distribution of predictor variables used to evaluate the anuran biogeographical regions in the Amazonia. Current climate variables - first axis of principal components analyses (PCA) with precipitation and temperature variables (AMAXTE, AMINTE, TESE, APRE, PRER and PRSE); Historical difference precipitation (HDP) - difference between current and Last Glacial Maximum (LGM) annual precipitation; Historical difference temperature (HDT) - difference between current and LGM annual mean temperature; Topographic variables - first axis of PCA with elevation and slope variables (TOPOMAX, TOPOMIN, TOPOSTD, SLOPERAN, SLOPESTD and ASPECTSTD); Vegetation structure – Amazonia ecoregions based on the classification of Olson et al.9; and Riverine barriers - classification of grids based on the banks of ten major rivers in Amazonia. Maps generated using ESRI ArcMap 9.2. https://www.esri.com/.
Vegetation structure – we used the classification of Olson et al.9 to determine the percentage of vegetation type covering each grid (Fig. 4). The main vegetation types observed were moist forest, dry forest, varzea, mangrove and montane.
Statistical analysis
Correlates of biogeographical regions
To reduce the dimensionality and number of correlations between variables in our database, we performed three separate principal components analyses (PCA), a first one with the set of current climate variables (AMAXTE, AMINTE, TESE, APRE, PRER and PRSE), a second one with the set of topographic variables (TOPOMAX, TOPOMIN, TOPOSTD, SLOPERAN, SLOPESTD and ASPECTSTD) and a final one with the percentage of each vegetation type. Therefore, for the subsequent analysis, we used nine variables: i) the first two axes from the current climate variables (CURE.PC1 and CURE.PC2), ii) the first two axes from the topographic variables (TOPO.PC1 and TOPO.PC2), iii) the first two axes from the vegetation structure (VEGE.PC1 and VEGE.PC2), iv) two Pleistocene climate variations (HDP and HDT), and v) the classification of grids based on the banks of eight major rivers. We also evaluated the correlation between original environmental variables and the first two axes of the three PCAs using significance tests of Pearson correlation coefficients (see Appendix S3 in Supporting Information).
We used multinomial logistic regression models to investigate the influence of predictor variables in explaining the anuran biogeographic regions8,10. To determine the optimal model related to biogeographical regions, we started with a full model containing all explanatory variables. Then we generated sub-model sets from the full model using the dredge function implemented in the MuMIn package73. We used Akaike’s information criterion corrected for small sample sizes (AICc74) to determine the optimal model. The AICc is calculated for each model from its log-likelihood and the number of parameters, and the model with the lowest AICc is judged to be the best of the candidate models74. Furthermore, to evaluate model selection uncertainty, we used Akaike weights (ὠ), which express the likelihood of each model given the data and the set of candidate models. Finally, we used variation partitioning analysis75 to partition the total percentage of variation into unique contributions of the sets of predictors of the best model.
All analyses were performed with R 3.2.3 software76.
Data accessibility statement
All data were gathered on public databases that are available on-line.
References
Wallace, A. R. On the monkeys of the Amazon. Proc. Zool. Soc. Lond. 20, 107–110 (1852).
Holt, B. G. et al. An update of Wallace’s zoogeographic regions of the world. Science 339, 74–78, https://doi.org/10.1126/science.1228282 (2013).
Whittaker, R. J., Riddle, B. R., Hawkins, B. A. & Ladle, R. J. The geographical distribution of life and the problem of regionalization: 100 years after Alfred Russel Wallace. J. Biogeogr. 40, 2209–2214, https://doi.org/10.1111/jbi.12235 (2013).
Ficetola, G. F., Mazel, F., & Thuiller, W. Global determinants of zoogeographical boundaries. Nat. Ecol. Evol. 1; https://doi.org/10.1038/s41559-017-0089 (2017).
Mackey, B. G., Berry, S. L. & Brown, T. Reconciling approaches to biogeographical regionalization: a systematic and generic framework examined with a case study of the Australian continent. J. Biogeogr. 35, 213–229, https://doi.org/10.1111/j.1365-2699.2007.01822.x (2008).
Rueda, M., Rodriguez, M. A. & Hawkins, B. A. Towards a biogeographic regionalization of the European biota. J. Biogeogr. 37, 2067–2076, https://doi.org/10.1111/j.1365-2699.2010.02388.x (2010).
Kreft, H. & Jetz, W. A framework for delineating biogeographical regions based on species distribution. J. Biogeogr. 37, 2029–2053, https://doi.org/10.1111/j.1365-2699.2010.02375.x (2010).
Vasconcelos, T. S., Prado, V. H. M., da Silva, F. R. & Haddad, C. F. B. Biogeographic distribution patterns and their correlates in the diverse frog fauna of the Atlantic Forest Hotspot. PlosOne 9, 1–9, https://doi.org/10.1371/journal.pone.0104130 (2014).
Olson, D. M. et al. Terrestrial ecoregions of the world: a new map of life on Earth. BioScience 51, 933–938, https://doi.org/10.1641/0006-3568(2001)051[0933:TEOTWA]2.0.CO;2 (2001).
Moura, M. R., Argôlo, A. J. & Costa, H. C. Historical and contemporary correlates of snake biogeographical subregions in the Atlantic Forest hotspot. J. Biogeogr. 44, 640–650, https://doi.org/10.1111/jbi.12900 (2017).
Davidson, E. A. et al. The Amazon basin in transition. Nature 481, 321–328, https://doi.org/10.1038/nature10717 (2012).
Charity, S., Dudley, N., Oliveira, D. & Stolton, S. Living amazon report 2016: A regional approach to conservation in the Amazon. WWF Living Amazon Initiative, Brasília and Quito (2016).
Haffer, J. Distribution of Amazon birds. Bonn. Zool. Bull. 29, 38–78 (1978).
Cracraft, J. Historical biogeography and patterns of differentiation within the South American avifauna: areas of endemism. Ornithol. Monogr. 36, 49–84 (1985).
Silva, J. M. C., Novaes, F. C. & Oren, D. C. Differentiation of Xiphocolaptes (Dendrocolaptidae) across the river Xingu, Brazilian Amazonia: recognition of a new phylogenetic species and biogeographic implications. Bull. Br. Orn. Club 122, 185–194 (2002).
Haffer, J. Speciation in Amazonian forest birds. Science 165, 131–137 (1969).
Haffer, J. Hypotheses to explain the origin of species in Amazonia. Braz. J. Biol. 68, 917–947 (2008).
Hoorn, C. et al. Amazonia through time: Andean uplift, climate change, landscape evolution, and biodiversity. Science 330, 927–931, https://doi.org/10.1126/science.1194585 (2010).
Smith, B. T. et al. The drivers of tropical speciation. Nature 515, 406–409, https://doi.org/10.1038/nature13687L3 (2014).
Naka, L. N. Avian distribution patterns in the Guiana Shield: implications for the delimitation of Amazonian areas of endemism. J. Biogeogr. 38, 681–696, https://doi.org/10.1111/j.1365-2699.2010.02443.x (2011).
Oliveira, U., Vasconcelos, M. F., & Santos, A. J. Biogeography of Amazon birds: rivers limit species composition, but not areas of endemism. Sci. Rep. 7, https://doi.org/10.1038/s41598-017-03098-w (2017).
Catenazzi, A. State of the Wold’s amphibians. Annu. Rev. Environ. Resour. 40, 91–119, https://doi.org/10.1146/annurev-environ-102014-021358 (2015).
Jenkins, C. N., Pimm, S. L. & Joppa, L. N. Global patterns of terrestrial vertebrate diversity and conservation. Proc. Natl. Acad. Sci. USA 110, E2602–E2061, https://doi.org/10.1073/pnas.1302251110 (2013).
Azevedo-Ramos, C. & Galatti, U. Patterns of amphibian diversity in Brazilian Amazonia: conservation implications. Biol. Conserv. 103, 103–111, https://doi.org/10.1016/S0006-3207(01)00129-X (2002).
da Silva, F. R., Almeida-Neto, M., Prado, V. H. M., Haddad, C. F. B. & Rossa-Feres, D. C. Humidity levels drive reproductive modes and phylogenetic diversity of amphibians in the Brazilian Atlantic Forest. J. Biogeogr. 39, 1720–1732, https://doi.org/10.1111/j.1365-2699.2012.02726.x (2012).
Pimm, S. L. et al. The biodiversity of species and their rates of extinction, distribution and protection. Science 344, 1–10, https://doi.org/10.1126/science.1246752 (2014).
Vasconcelos, T. S., Rodríguez, M. A. & Hawkins, B. A. Biogeographic distribution patterns of South American amphibians: a regionalization based on cluster analysis. Nat. Conservacao 9, 67–72, https://doi.org/10.4322/natcon.2011.008 (2011).
Araújo, M. B. et al. Quaternary climate changes explain diversity among reptiles and amphibians. Ecography 31, 8–15, https://doi.org/10.1111/j.2007.0906-7590.05318.x (2008).
Carnaval, A. C., Hickerson, M. J., Haddad, C. F. B., Rodrigues, M. T. & Moritz, C. Stability predicts genetic diversity in the Brazilian Atlantic Forest hotspot. Science 323, 785–789, https://doi.org/10.1126/science.1166955 (2009).
Antonelli, A. et al. In Amazonia, landscape and species evolution: a look into the past (eds Hoorn, C. & Wesselingh, E. P.) 386–404 (Wiley-Blackwell, 2010).
Cornell, H. V. & Harrison, S. P. What are species pools and when are they important? Annu. Rev. Ecol. Evol. Syst. 45, 45–67, https://doi.org/10.1146/annurev-ecolsys-120213-091759 (2014).
Carstensen, D. W., Lessard, J.-P., Holt, B. G., Borregaard, M. K. & Rahbek, C. Introducing the biogeographic species pool. Ecography 36, 1–9, https://doi.org/10.1111/j.1600-0587.2013.00329.x (2013).
Gascon, C. et al. Riverine barriers and the geographic distribution of Amazonian species. Proc. Natl. Acad. Sci. USA 97, 13672–13677, https://doi.org/10.1073/pnas.230136397 (2000).
Moraes, L. J. C. L., Pavan, D., Barros, M. C. & Ribas, C. C. The combined influence of riverine barriers and flooding gradients on biogeographical patterns for amphibians and squamates in south-eastern Amazonia. J. Biogeogr. 43, 2113–2124, https://doi.org/10.1111/jbi.12756 (2016).
Bloomfield, N. J., Knerr, N. & Encinas-Viso, F. A comparison of network and clustering methods to detect biogeographical regions. Ecography 41, 1–10, https://doi.org/10.1111/ecog.02596 (2018).
Silva, J. M. C., Rylands, A. B. & Fonseca, G. A. B. The fate of the Amazonian areas of endemism. Conserv. Biol. 19, 689–694, https://doi.org/10.1111/j.1523-1739.2005.00705.x (2005).
Dias-Terceiro, R. G. et al. A matter of scale: historical and environmental factors structure anuran assemblages from the Upper Madeira River, Amazonia. Biotropica 47, 259–266, https://doi.org/10.1111/btp.12197 (2015).
Kozak, K. H. & Wiens, J. J. Does niche conservatism promote speciation? A case study in North American salamanders. Evolution 60, 2604–2621, https://doi.org/10.1554/06-334.1 (2006).
Kozak, K. H. & Wiens, J. J. Climatic zonation drives latitudinal variation in speciation mechanisms. Proc. R. Soc. B 274, 2995–3003, https://doi.org/10.1098/rspb.2007.1106 (2007).
Buckley, L. B. & Jetz, W. Environmental and historical constraints on global patterns of amphibian richness. Proc. R. Soc. B 274, 1167–1173, https://doi.org/10.1098/rspb.2006.0436 (2007).
Qian, H., Wang, X., Wang, S. & Li, Y. Environmental determinants of amphibian and reptile species richness in China. Ecography 30, 471–482, https://doi.org/10.1111/j.0906-7590.2007.05025.x (2007).
Vasconcelos, T. S., Santos, T. G., Haddad, C. F. B. & Rossa-Feres, D. C. Climatic variables and altitude as predictors of anuran species richness and number of reproductive modes in Brazil. J. Trop. Ecol. 26, 423–432, https://doi.org/10.1017/S0266467410000167 (2010).
Ricklefs, R. E. Community diversity: relative roles of local and regional processes. Science 235, 167–171 (1987).
Wiens, J. J., Graham, C. H., Moen, D. S., Smith, S. A. & Reeder, T. W. Evolutionary and ecological causes of the latitudinal diversity gradient in hylid frogs: treefrogs trees unearth the roots of high tropical diversity. Am. Nat. 168, 579–596, https://doi.org/10.1111/j.1558-5646.2009.00610.x (2006).
Carvalho, W. D. & Mustin, K. The highly threatened and little known Amazonian savannahs. Nat. Ecol. Evol. 1, https://doi.org/10.1038/s41559-017-0100 (2017).
Ferro, I. & Morrone, J. J. Biogeographical transition zones: a search for conceptual synthesis. Biol. J. Linnean Soc. 113, 1–12, https://doi.org/10.1111/bij.12333 (2014).
Morrone, J. J. The spectre of biogeographical regionalization. J. Biogeogr, https://doi.org/10.1111/jbi.13135 (2018).
Hortal, J. et al. Seven shortfalls that beset large-scale knowledge on biodiversity. Annu. Rev. Ecol. Evol. Syst. 46, 523–549, https://doi.org/10.1146/annurev-ecolsys-112414-054400 (2015).
Ficetola, G. F. et al. An evaluation of the robustness of global amphibian range maps. J. Biogeogr. 41, 211–221, https://doi.org/10.1111/jbi.12206 (2014).
Whittaker, R. J., Araújo, M. B., Jepson, P., Ladle, R. J., Watson, J. E. M. & Willis, K. J. Conservation biogeography: assessment and prospect. Diversity Distrib. 11, 3–23, https://doi.org/10.1111/j.1366-9516.2005.00143.x (2005).
Luna-Vega, I., Morrone, J. J., & Escalante, T. In Biogeography (eds Gailis M. & Kalnins, S.) 229–240 (Nova-Science Publishers, 2010).
Fisher, M. C., Garner, T. W. & Walker, S. F. Global emergence of Batrachochytrium dendrobatidis and amphibian chytridiomycosis in space, time, and host. Annu. Rev. Microbiol. 63, 291–310, https://doi.org/10.1146/annurev.micro.091208.073435 (2009).
Pounds, J. A. et al. Widespread amphibian extinctions from epidemic disease driven by global warming. Nature 439, 161–167, https://doi.org/10.1038/nature04246 (2006).
Rohr, J. R. & Raffel, T. R. Linking global climate and temperature variability to widespread amphibian declines putatively caused by disease. Proc. Natl. Acad. Sci. USA 107, 8269–8274, https://doi.org/10.1073/pnas.0912883107 (2010).
Becker, C. G., Rodriguez, D., Lambertini, C., Toledo, L. F. & Haddad, C. F. B. Historical dynamics of Batrachochytrium dendrobatidis in Amazonia. Ecography 39, 954–960, https://doi.org/10.1111/ecog.02055 (2016).
Winemiller, K. O. et al. Balancing hydropower and biodiversity in the Amazon, Congo, and Mekong. Science 351, 128–129, https://doi.org/10.1126/science.aac7082 (2016).
Latrubesse, E. M. et al. Damming the rivers of the Amazon basin. Nature 546, 363–369, https://doi.org/10.1038/nature22333 (2017).
Fearnside, P. M. Amazon dams and waterways: Brazil’s Tapajós basin plans. Ambio 44, 426–439, https://doi.org/10.1007/s13280-015-0642-z (2015).
Nobre, C. A. et al. Land-use and climate change risks in the Amazon and the need of a novel sustainable development paradigm. Proc. Natl. Acad. Sci. USA 113, 10759–10768, https://doi.org/10.1073/pnas.1605516113 (2016).
Sorribas, M. V. et al. Projections of climate change effects on discharge and inundation in the Amazon basin. Clim. Change 136, 555–570, https://doi.org/10.1007/s10584-016-1640-2 (2016).
Haddad, C. F. B. & Prado, C. P. A. Reproductive modes in frogs and their unexpected diversity in the Atlantic Forest of Brazil. Bioscience 55, 207–217, https://doi.org/10.1641/0006-3568(2005)055[0207:RMIFAT]2.0.CO;2 (2005).
IUCN IUCN red list of threatened species, ver. 2015.2. http://www.iucnredlist.org/technical-documents/spatial-data (2015)
Frost, D. R. Amphibian Species of the World: an Online Reference. – Version 6.0, http://research.amnh.org/herpetology/amphibia/index.html (2016).
Vale, M. M., Marques, T. L., Cohn-Haft, M. & Vieira, M. V. Misuse of bird digital distribution maps creates reversed spatial diversity patterns in the Amazon. Biotropica 49, 636–642, https://doi.org/10.1111/btp.12460 (2017).
Hawkins, B. A., Rueda, M. & Rodríguez, M. Á. What do range maps and surveys tell us about diversity patterns? Folia Geobot. 43, 345–355, https://doi.org/10.1007/s12224-008-9007-8 (2008).
Dapporto, L. et al. recluster: an unbiased clustering procedure for beta-diversity turnover. Ecography 36, 1070–1075, https://doi.org/10.1111/j.1600-0587.2013.00444.x (2013).
Dapporto, L., Ciolli, G., Dennis, R. L. H., Fox, R. & Shreeve, T. G. A new procedure for extrapolating turnover regionalization at mid-small spatial scales, tested on British butterflies. Methods in Ecol. Evol. 6, 1287–1297, https://doi.org/10.1111/2041-210X.12415 (2015).
Dapporto, L. et al. recluster: Ordination methods for the analysis of beta-diversity Indices. R package version 2.8, https://CRAN.R-project.org/package = recluster (2015).
Legendre, P., & Legendre, L. Numerical ecology. Elsevier, Cambridge and Oxford (2012).
Bocard, D., Francois, G. & Legendre, P. Numerical Ecology with R. Springer-Verlag, New York. (2011).
Hijmans, R. J., Cameron, S. E., Parra, J. L., Jones, P. G. & Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 25, 1965–1978, https://doi.org/10.1002/joc.1276 (2005).
Latrubesse, E. M., Stevaux, J. C. & Sinha, R. Tropical rivers. Geomorphology 70, 187–206, https://doi.org/10.1016/j.geomorph.2005.02.005 (2005).
Barton, K. MuMIn: multi-model inference. R package version 1.40, http://CRAN.R-project.org/package=MuMIn (2017).
Burnham, K. P. & Anderson, D. R. Model selection and Multimodel inference. Springer-Verlag (2002).
Bocard, D., Legendre, P. & Drapeau, P. Partialling out the spatial component of ecological variation. Ecology 73, 1045–1055, https://doi.org/10.2307/1940179 (1992).
R Development Core Team R: A language and environment for statistical computing. – R Foundation for Statistical Computing, Vienna, Austria, http://www.Rproject.org (2015).
Acknowledgements
MBCG thanks Coordenação de Aperfeiçoamento de Pessoal do Nível Superior (CAPES) for master fellowship. FRdaS thanks São Paulo Research Foundation (FAPESP, 2013/50714-0) for financial support.
Author information
Authors and Affiliations
Contributions
F.R.S. conceived the idea; M.B.C.G. gathered the data; M.B.C.G. and F.R.S analysed the data and led the writing.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Godinho, M.B.d.C., da Silva, F.R. The influence of riverine barriers, climate, and topography on the biogeographic regionalization of Amazonian anurans. Sci Rep 8, 3427 (2018). https://doi.org/10.1038/s41598-018-21879-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-21879-9
This article is cited by
-
Alfred Russel Wallace’s legacy: an interdisciplinary conception of evolution in space and time
npj Biodiversity (2023)
-
Fine scale population genetic structure of Euphlyctis karaavali (Amphibia: Anura) using newly developed microsatellite markers
Biologia (2023)
-
Historical biogeography highlights the role of Miocene landscape changes on the diversification of a clade of Amazonian tree frogs
Organisms Diversity & Evolution (2023)
-
Geographic Drivers of Genetic and Plumage Color Diversity in the Blue-Crowned Manakin
Evolutionary Biology (2023)
-
Phylogenetic relationships, population demography, and species delimitation of the Alouatta belzebul species complex (Atelidae: Alouattinae)
Primates (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.