Abstract
The production of transgenic livestock is constrained due to the limited success of currently available methods for transgenesis. Testis mediated gene transfer (TMGT) is an emerging method that shows a high success rate in generating transgenic mice. In this study, we report a newly developed protocol for electroporation-aided TMGT to produce a transgenic goat. The injectable volume and concentration of the transgene were first standardized, and then electroporation conditions were optimized in vitro. In vivo experiments were performed by injecting a transgenic construct (pIRES2-EGFP; enhanced green fluorescent protein) into the testicular interstitium followed by electroporation. Immunohistochemistry, quantitative real-time PCR (qPCR) and western blotting analyses confirmed the successful transfer of the transgene into seminiferous tubules and testicular cells. Furthermore, chromosomal integration of the transgene and its expression in sperm were evaluated d60 and d120 post-electroporation. Our protocol neither altered the seminal characteristics nor the fertilization capacity of the sperm cells. In vitro fertilization using transgenic sperm generated fluorescent embryos. Finally, natural mating of a pre-founder buck produced a transgenic baby goat. The present study demonstrates the first successful report of an electroporation-aided TMGT method for gene transfer in goats.
Similar content being viewed by others
Introduction
The production of transgenic animals has predominantly been attempted by manipulating embryos, using a variety of techniques such as pronuclear microinjection, embryonic stem cell-mediated methods, and viral-mediated transfection1,2,3. Most of these methods, particularly pronuclear microinjection, are afflicted by poor efficiency, highly specialized laboratory techniques and skilled early embryonic manipulation1,2,4,5. During the last decade, the targeting of male germ cells has emerged as an alternative for transgenic animal production6,7. Generally, two strategies for gene transfer to male germ cells are employed; (1) sperm-mediated gene transfer (SMGT), and (2) testis-mediated gene transfer (TMGT). SMGT includes the direct transfer of genes into sperm cells, whereas TMGT involves in vivo introduction of foreign DNA into testicular germ cells to produce transgenic sperm cells. While SMGT appears to be a straightforward method, it suffers from poor repeatability and interspecies/intraspecies success variability7. On the other hand, the TMGT method, which involves surgical steps, presents a risk of infection and/or impotency if appropriate precautions are not taken. Nevertheless, TMGT allows for mass gene transfer by natural mating, exempting the use of cumbersome procedures such as in vitro fertilization and embryo transfer8. Furthermore, TMGT ensures a greater probability of stable integration of transgenes into the genome of the host animal8,9.
Several strategies, including viral, non-viral, physical and chemical methods, are employed in TMGT8,10. Owing to its higher efficiency, virus-mediated TMGT has become a preferred method, but it is limited by the harmful effects of inflammation11. Among non-viral methods, both lipofection-aided and electroporation-aided TMGT are considered to be easier and safer methods8,12. Further, in vivo electroporation is a safe method for gene transfer, as it does not cause adverse effects on testicular integrity or the fertilizing ability of spermatozoa13,14. Transgenic laboratory animals have been produced successfully using electroporation-aided TMGT8,15. However, the success rate of electroporation-aided TMGT depends on the sites of transgene injection, namely, the lumen of seminiferous tubules, rete testis or interstitium of the testis8,9,15. The direct injection of transgenes into the interstitium of the testis has shown a higher success rate than injection into the seminiferous tubules or rete testis, because the former site better facilitates the access of the transgene to undifferentiated spermatogonial germ cells8.
Available literature suggests an immense potential for electroporation-aided TMGT to produce transgenic laboratory animals8,15. Nevertheless, the enormous variation between species in the size, shape, and structures of testes necessitates the development of species-specific protocols. The goat is an ideal livestock species amenable to transgenesis. One specific application for goats is as mammary gland bioreactors. In addition, goats have smaller body size, a shorter gestation period, a higher prolificacy, and a relatively high content of protein in their milk16,17. The objective of the present study is to develop a protocol for electroporation-aided TMGT in goats.
Results
In vitro gene transfer and transgene expression in testis
First, we optimized the volume and concentration of the transgenic construct, linearized pIRES2-EGFP plasmid, by in vitro transfection of goat testis.
Injection volume
To optimize the injection volume, different volumes of phosphate-buffered saline (PBS) were injected into the testes. It was observed that the testis of pre-pubertal and adult bucks could accommodate a maximum of 1.0 and 1.5 ml of PBS, respectively. An increase in volume beyond the optimized level caused an apparent swelling of the testis.
DNA concentration
Under the optimum electroporation conditions, injecting the linearized plasmid at a concentration of 1 µg/µl resulted in a maximum EGFP expression in the seminiferous tubules (Fig. 1A) and spermatogonial stem cell (SSC) colonies (Fig. 1B). The EGFP protein expression was visible (green fluorescence) as early as day 3 (d3), and it lasted for more than three weeks in electroporated samples, suggesting non-episomal expression. In the PBS control group, no expression of EGFP was observed in the seminiferous tubules (Fig. 1C). In the absence of electroporation, there was no improvement in the efficiency of expression with increased plasmid concentration. However, the efficiency of expression was improved significantly when the plasmid concentration was increased from 0.1 to 1.0 µg/µl (Fig. 1D). However, the expression did not change significantly when the plasmid concentration was further increased to 1.5 µg/µl. For all the plasmid concentrations except 0.1 µg/µl, the non-electroporated samples showed ~five times lower (P < 0.001) efficiency in expressing EGFP (Supplementary Table S1).
EGFP expression in the goat testes transfected in vitro. EGFP expression was observed as green fluorescence in (A) cultured seminiferous tubules and (B) spermatogonial stem cell (SSC) colonies; (C) An absence of fluorescence was observed in cultured seminiferous tubules from the testis injected with only PBS (negative control). BF: Bright field, UV: Under UV light. Scale bar, 50 μm; (D) Graphical representation of transfection efficiency using different plasmid concentrations (0.1, 0.25, 0.5, 1.0 and 1.5 µg) in goat testis. After in vitro transfection, seminiferous tubules were isolated and cultured in 24-well plates. The X-axis denotes plasmid concentration and the Y-axis represents the number of wells showing EGFP expression in seminiferous tubules. P: PBS injection only, D: injection of plasmid DNA without electroporation and E + D: injection of plasmid DNA followed by electroporation. Data represent the mean ± s.e.m. for n = 3. Letters (a–d) above the bars indicate that these groups differ significantly. In the E + D, P < 0.05 (between 0.1 µg and 0.25 µg), P < 0.05 (between 0.25 µg and 0.5 µg) and P < 0.01 (between 0.5 µg and 1.0 µg). Between the D and E + D groups, ***P < 0.001.
Expression of EGFP in pre-pubertal goat testes after in vivo gene transfer
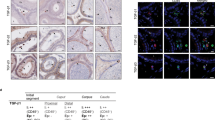
On d21 post-electroporation, microscopic examination of testes revealed EGFP expression in spermatogenic cells, Sertoli cells and other interstitial cells (Fig. 2A). Immunohistochemical (IHC) analysis of tissue sections of transfected testes showed localization of EGFP protein (brown color) in spermatogonial cells, adjacent to the basement membrane of seminiferous tubules (Fig. 2B). Seminiferous tubules showed clusters of green fluorescent cells (Fig. 2C). The EGFP expression was further confirmed by quantitative real-time PCR (qPCR) (Fig. 3A and B). Western blot analysis of transfected testes showed a 27 kDa EFGP protein (Fig. 3C).
Expression of EGFP in testicular cells on d21 after in vivo gene transfer. (A) Direct fluorescence microscopy of a testis cross-section showing green fluorescence in testicular cells; (B) IHC revealed the localization EGFP protein (in brown) in almost all the testicular cells including spermatogonial cells; (C) clusters of green fluorescent cells in seminiferous tubules; (D) IHC analysis of testis showing the location of SSCs in the basement membrane of seminiferous tubules. The α6 integrin antibody was used as a specific marker for SSCs. Scale bar, 50 μm.
EGFP expression in the testis of pre-pubertal bucks on d21 after in vivo gene transfer. (A) Gel electrophoresis of quantitative real-time PCR products from gene transferred testis revealed an EGFP-specific band of 104 bp. The top of the gel with the wells was cropped; N: no template control (NTC), G1-G3: testicular tissue from pre-pubertal goats with gene transferred testis, S1 & S2: cultured seminiferous tubules from in vitro transfected testes, P: positive control (pIRES2-EGFP plasmid), M: 100 bp molecular marker; (B) graphical representation of the relative expression of EGFP mRNA in testicular tissues from pre-pubertal bucks at d21 post-gene transfer. G1, G2 & G3 are pre-pubertal bucks #1, #2 & #3, respectively. Data represent the mean ± s.e.m. for n = 3. Here, ‘ns’ means statistically not significant; (C) an approx. 27 kDa protein band was observed by western blotting analysis, using total protein from testicular tissue of an in vivo electroporated goat. The marker lane was cut separately before incubating the membrane with the primary antibody. The wells are labeled 1: gene transferred testis of a pre-pubertal goat, 2: testis of a non-gene transferred goat, 3: protein marker (#1610373, Bio-Rad).
Effects of electroporation-mediated gene transfer on semen fertility parameters
The vital semen parameters, namely, progressive motility, viability, membrane integrity and acrosome integrity, did not vary significantly among the semen samples collected from the experimental bucks before and after electroporation (Fig. 4A). This result suggests the absence of any detrimental effects from electroporation-mediated gene transfer on the sperm quality.
Assessment of semen samples obtained from bucks with transfected testis. (A) The semen parameters showed no variation before and after gene transfer. Semen from three in vivo gene transferred bucks (n = 3) were assessed at different intervals. BE: before electroporation, MY: progressive motility, VY: viability, MI: membrane integrity, AI: acrosome integrity. Data represent the mean ± s.e.m.; (B) EGFP expression in the sperm from a gene transferred buck; (C) no expression of EGFP was observed in sperm from a wild buck. (a & c): Bright-field, (b & d): under UV light. Scale bar, 20 μm; (D) The graph shows qPCR analysis of sperm from in vivo gene transferred bucks at different intervals indicating the genomic integration of transgene. Data represent the mean ± s.e.m. for n = 3. Here, ‘ns’ means statistically not significant.
Integration and expression of the transgene in the sperm
Microscopic examinations of sperms on d60 showed a limited (0.83%) number of cells containing green fluorescent protein (Fig. 4B,C and Supplementary Table S2). However, we failed to observe EGFP transcripts in the semen by reverse transcription PCR (RT-PCR) (data not shown). qPCR analysis of semen samples from all three bucks revealed the presence of the EGFP gene until d120 post-electroporation, further confirming the chromosomal integration of the transgene into the sperm (Fig. 4D).
Transgenic embryo production using semen from an in vivo gene transferred buck
The effects of electroporation and foreign DNA on the fertilizing ability of the sperm was assessed using an in vitro fertilization (IVF) assay. There was no significant difference in the cleavage rate between the IVF embryos obtained using the semen from transfected (22.00 ± 1.30) and non-transfected (23.40 ± 1.07) bucks (Fig. 5A). Out of 110 embryos analyzed, three embryos (2.72%) showed transgenic expression of green fluorescence (Fig. 5B). RT-PCR analysis of these fluorescent embryos also confirmed the presence of EGFP mRNA (Fig. 5C).
Detection of EGFP expression in embryos produced in vitro using semen from in vivo gene transferred bucks. (A) In vitro produced embryos (pooled). Scale bar, 100 μm; (B) EGFP expression in the embryo (a) under bright-field and (b) UV light. Scale bar, 50 μm; (C) RT-PCR analysis revealed expression of EGFP mRNA in the fluorescent embryo. 1; normal embryo, 2; EGFP-expressing embryo, 3; 100 bp molecular marker. The gel picture was cropped, and the full-length gel is presented in Supplementary Figure 5C.
Production of a transgenic kid
The optimized procedure for transgenic baby goat production was assessed. A total of nine matings of three pre-founder bucks resulted in the birth of 13 kids (Supplementary Table S3). PCR analysis revealed the presence of pIRES2-EGFP in one kid, indicating that the transgene was integrated into the spermatogonial cells of the pre-founder male (Fig. 6A and B). This result was further confirmed using Southern blot analysis (Fig. 6C). However, neither fluorescence nor the EGFP transcript were detected in the blood or skin of the transgenic kid.
Generation of a transgenic kid from an in vivo gene transferred buck. A total of nine matings of three bucks carrying electroporated testis resulted in the birth of 13 kids. (A) Representative photograph of a transgenic baby goat. The kid showed no EGFP expression; (B) PCR analysis revealed the presence of the EGFP gene in the blood genomic DNA of the kid, indicating the integration of plasmid into the germline cells of the in vivo gene transferred buck. Lane 13–10 & lane 8–1: kids without transgene in their genome, 9: kid carrying the transgene, D: normal doe, P: positive control (pIRES2-EGFP plasmid), N: no-template control (NTC), M: 100 bp DNA ladder. The top of the gel containing the wells was cropped; (C) Southern blot analysis of genomic DNA from the transgenic kid, 1: genomic DNA from a wild goat, 2: DNA from the kid carrying the transgene, 3: DNA marker. The blot image was cropped, and the full-length blot is presented in Supplementary Figure 6C.
Discussion
Transgenic livestock animals have the potential to act as bioreactors for generating large quantities of biologically active proteins18,19. Nevertheless, most of the traditional methods for transgenic livestock production, particularly pronuclear microinjection, are constrained by limited success rates2,20. The lentiviral-mediated gene transfer method shows a comparatively higher efficiency but suffers from insertional mutagenesis21,22,23 and limitations in transgene-carrying capacity24,25. Recently, electroporation-mediated TMGT has emerged as an alternative method for transgenic animal production with a high success rate8, but it is restricted to laboratory animal species such as mice. In this study, we report for the first time a successful transgenesis method using electroporation-aided TMGT in goat.
Previous studies dealing with TMGT in laboratory animals investigated different concentrations and volumes of exogenous DNA with varying success rates8,14,15. In the present study, TMGT was employed for in vitro gene transfection of testes. First, we optimized the injection volume and the concentration of the transgenic vector. The results showed that the testes of a pre-pubertal and adult buck can accommodate injection volumes as high as 1.0 and 1.5 ml, respectively. The variation in the injection volume is possibly due to the change in testicular size with age and the onset of puberty26,27,28 (Supplementary Table S4). Using trypan blue solution as a marker dye9,14, we further confirmed that injection volumes of 1.0 and 1.5 ml are sufficient to cover the entire testicular area (Supplementary Figure S1). The results of in vitro transfection experiments further demonstrate that 1 µg/µl is an optimum plasmid concentration for gene transfer.
In vivo electroporation-mediated transfection studies of large mammals are very limited compared to those of laboratory animals. In human, a transdermal voltage of at least 50 V is necessary to cause a significant molecular transport across the skin, regardless of the fluorescent tracer used29. A range of 25 to 50 mV was shown to be the most efficient condition for electroporation-mediated transfection of SSCs in neonatal bovine testicular tissue in vitro30. However, in laboratory animals, the most favorable voltage ranges from 30 to 50 V, which guarantees minimal adverse effects on testicular integrity, a normal sperm quality, the maintenance of offspring production ability and sufficient transfection efficacy8,9,14. An increase in transfection efficiency by raising the voltage is accompanied by the adverse effect of testicular shrinking31. Further, we observed visible discomfort in the animal when we attempted a trial with a higher voltage (data not shown). Accordingly, in compliance with strict bio-ethical concerns and in the absence of any cited literature using a voltage higher than 50 mV, we employed the previously reported electroporation conditions (50 V and 50 ms) from our laboratory32.
It is now known that the site of injection greatly influences the success of TMGT. Previous studies have suggested that transgenic constructs can be injected into either the lumen of seminiferous tubules9, rete testis13 or in the interstitium8. Later, nonsurgical in vivo electroporation of testis was reported to be successful for gene transfer through the testicular interstitium33. In another study, researchers demonstrated the expression of GFP in the seminiferous tubules for more than two months when the transgenic construct was directly injected into the seminiferous tubules of mice, but they failed to produce a transgenic pup9. However, in the same year, another group reported the production of transgenic mice by intracytoplasmic sperm injection (ICSI) using fluorescent spermatozoa that were derived from the injection of a transgenic construct through the rete testis15. TMGT via direct injection into the rete testis in hamster, a non-murine species, also revealed the expression of the transgene in epididymal sperms on d60 post-electroporation14. In the current study, we injected the vector into the testicular interstitium and demonstrated the expression of EGFP in the seminiferous tubules, even on d21 after in vivo gene transfer, indicating a non-episomal expression of the transgene. IHC analysis further showed EGFP expression in the spermatogonial cells as well as other interstitial cells of the testis. The delivery of the transgene into the testicular interstitium in situ might have allowed for direct access to the undifferentiated spermatogonial cells, due to their location in the basement membrane of the seminiferous tubules8.
In the present study, we ascertained that neither electroporation nor the presence of foreign DNA affected the spermatogenic process and/or deteriorated the semen quality or the fertilizing ability of sperm. Except in hamster14, the effects of electroporation-mediated TMGT on semen quality have scarcely been reported. That study reported no adverse effects on the motility and viability of hamster sperm. The results of our study strengthen the previous findings, and we did not observe distinguishable changes in the vital parameters of semen from the experimental (i.e., with the electroporated testes) and control bucks. Moreover, embryos obtained by in vitro fertilization with the semen from experimental and control bucks showed similar cleavage rates. These results agree with literature14 stating that electroporation-mediated TMGT neither affects sperm function nor their fertilizing ability and subsequent embryonic development.
Our study assessed the integration of the transgene into the spermatogenic cells of pre-founder males. The most convincing direct evidence of genomic integration is the presence of fluorescent spermatozoa when examined under a fluorescence microscope. In a previous study performing TMGT in mice, the presence of clusters of fluorescent spermatozoa in the seminiferous tubules showed the expression of yellow fluorescent protein (YFP)15. The authors suggested that the absence of GFP expression in sperm was either due to potential toxicity from the GFP or a very low rate of chromosomal integration. In TMGT using hamsters, YFP fluorescence was detected in the mid-piece of approximately 10% of epididymal sperm14. Subsequently, Chandrashekran and co-workers showed GFP expression in porcine sperm by direct transfection with a pseudotyped lentiviral vector34. Other researchers observed fluorescent pig spermatozoa using GFP35 and Venus (yellow shifted variant of EGFP)36 constructs. The absence of Venus mRNA in the sperm, however, suggested the presence of Venus protein that had already been translated in pro-spermatogonial stages36. Using the CMV promoter, researchers demonstrated GFP expression in mice spermatogonia and round spermatids, but not in the spermatozoa37. We observed minimal EGFP expression under the CMV promoter in caprine spermatozoa, but we failed to observe EGFP mRNA in their semen. The green fluorescence in spermatozoa observed in the present study and in other work36 is likely due to the presence of EGFP protein that had already been translated in pre-prospermatogonial or spermatogonial stages. Contrary to earlier observations that mature spermatozoa lose most of their cytoplasm and become transcriptionally dormant38,39, recent studies have suggested the presence of remnant mRNAs in the mature spermatozoa of several mammals40,41.
Although our study demonstrated EGFP expression in less than 1% of sperm, the percentages of fluorescent embryos and transgenic kids were 2.72% and 7.69%, respectively. Further, RT-PCR and western blot analysis confirmed EGFP expression in different parts of the testis, and IHC demonstrated EGFP localization in spermatogonial cells near the basement membrane of seminiferous tubules. Finally, qPCR analysis confirmed the presence of the EGFP gene until d120 after electroporation. In goats, the spermatogenic cycle is approximately 47.7 days42. Therefore, the presence of EGFP in the sperm, even up to 120 d after electroporation, confirms the chromosomal integration of EGFP into SSCs and thus agrees with the literature9,14.
Based on the results of this study, for the first time we have demonstrated the production of fluorescent IVF goat embryos using electroporation-mediated TMGT. The cleavage rates of embryos obtained after IVF using semen from either experimental or control bucks were comparable in this study. In our earlier report, we observed 4.31% fluorescent embryos while using SMGT43. Previously, another group also produced transgenic embryos using intra-cytoplasmic sperm injection (ICSI) of fluorescent sperm in mice15. The smaller number of fluorescent embryos produced in this study might be due to several reasons. First, there might be fewer sperms carrying the transgene than non-transgenic ones. Second, oocytes may have been preferentially fertilized with non-transgenic sperm, since transgenic sperm may be less competitive in reaching the oocytes or in penetrating the egg coats44,45. The interaction of exogenous DNA with sperm cells is also known to activate endogenous nucleases, which in turn might degrade the foreign DNA and/or sperm chromosomal DNA46. However, such a possibility seems to be remote in TMGT, as the transgene integration should have actually taken place in the progenitors of the sperm cells, i.e., SSCs.
Electroporation-aided TMGT has been reported with low9,47 to high success8 rates in laboratory animals. In the present study, one transgenic kid was born using electroporation-aided TMGT. The ability of the pre-founder buck to sire a transgenic kid after d60 post-electroporation suggested the integration of the transgene into the spermatogonial cells. However, we failed to detect either in vivo fluorescence or EGFP transcripts in the kid. Other studies have also reported the absence of EGFP expression in transgenic offspring due to either a low copy number of the transgene or mosaicism12,48. Further, the expression of the reporter gene, as observed in pronuclear injection, might have been influenced by the highly likely event of random integration and concatemer formation by homologous recombination before integration into the host genome49,50,51. This limitation needs to be circumvented by using the recently developed gene editing technologies such as zinc finger nucleases (ZFNs)52, transcription activator-like effector nucleases (TALENs)53 or clustered regularly interspersed short palindromic repeat (CRISPR)/CRISPR-associated protein 9 (Cas9)54 to ensure targeted integration of transgenes. In the future, combination of any of the above technologies with TMGT could offer efficient gene transfer in large animals.
In conclusion, the present study is the first successful report of an electroporation-aided TMGT technique for in vivo transfection of spermatogenic cells in farm animals. The results suggest that the direct injection of a transgenic construct into the testicular interstitium followed by electroporation results in successful integration of a transgene into the genome of testicular cells. This method also allows for the integration of transgenes into spermatogonial cells without affecting the fertilizing ability of the spermatozoa. This electroporation-aided TMGT method, with the help of recent gene editing technologies, seems to be the most convenient and promising method for the generation of transgenic farm animals, including transgenic goats. Future studies are warranted to further optimize the procedure to recover more fluorescent spermatozoa to use for artificial insemination.
Methods
Experimental animals
The care and experimental use of animals were approved by the Institutional Animal Ethics Committee (IAEC) as well as the Institutional Biosafety Committee (IBC) of the Indian Veterinary Research Institute (IVRI), Uttar Pradesh, India. All the experiments were performed in accordance with the guidelines of the Review Committee on Genetic Manipulation (RCGM) under the Department of Biotechnology (DBT), India. Male goats maintained under identical management conditions at the experimental herd of our laboratory were included in the study. Animals were divided into two groups. The first group (n = 3) consisted of pre-pubertal male goats 3–4 of months of age and the second group (n = 3) included adult male goats approximately three years of age (bucks).
In vitro transfection of goat testis using electroporation
The plasmid containing the GFP coding sequence (pIRES2-EGFP), a kind gift from Dr. Subeer Majumdar (NII, New Delhi) was used as a transgenic construct. The plasmid was isolated using an endotoxin-free quanta maxi kit (Advanced Microdevices Pvt. Ltd., India) and digested with the restriction enzyme StuI (Fermentas, Canada). The linearized plasmid was purified by ethanol precipitation and finally dissolved in sterilized PBS (pH 7.4).
Prior to the in vivo gene transfer experiment, a pilot study was conducted using pre-pubertal and adult goat testes collected from an abattoir in Bareilly, UP. Testes were immediately transported to the laboratory in PBS on the ice. In the laboratory, testes were washed with PBS containing 50 µg/ml Gentamicin (Sigma, USA). First, to optimize the injection volume of the transgenic construct, different volumes (0.25 to 2.0 ml) of PBS were injected into the interstitium of testes using a 27- G needle (BD Biosciences®, USA) attached to a tuberculin disposable syringe. The maximum volume of PBS that a testis can accommodate without any swelling was considered to be the optimum volume. The optimum volume was also confirmed by the injection of a 0.4% solution of trypan blue in buffered isotonic salt solution (pH 7.3) into the testicular interstitium (Supplementary Figure S1). To optimize the concentration (µg/µl) of the transgenic construct, the optimized volume of linearized plasmid DNA, at concentrations varying from 0.1 to1.5 µg/µl, was injected into ten different sites of the testicular interstitial space of pre-pubertal and adult goat testes (Supplementary Table S5). After each injection, the needle was removed very slowly to avoid any leakage due to the backflow of the injected solution.
Immediately after each injection, the testis was held between a pair of caliper-type electrodes (ECM830, BTX, USA, Item#45-0102) and square electric pulses were applied using electric pulse generator (ECM830, BTX, USA). A total of eight pulses (i.e., four pulses in one direction followed by another four pulses in the reverse direction) were applied. Each pulse was 50 V for 50 ms with an inter-pulse interval of 1s. After the electroporation, seminiferous tubules and SSCs were isolated from the testes and were cultured using a protocol developed in our laboratory32,55 (Supplementary materials and methods). To assess the efficiency of electroporation, one negative control (P: PBS only) and one electroporation control (D: only plasmid injection without electroporation) were included.
The cultures were maintained for more than four weeks. Starting from d3, SSCs were examined regularly for EGFP expression under a fluorescence microscope (Leica, Germany).
In vivo gene transfer to the goat testis using electroporation
After the in vitro experiment, the left testis of each experimental animal was injected in vivo with the optimized volume and concentration of the aqueous solution of linearized plasmid DNA. A plasmid concentration of 1 µg/µl in PBS was used, at volumes of 1.0 ml 1.5 ml for pre-pubertal and adult goat testes, respectively. In the prepubertal goat, the right testis was kept completely untouched throughout the experiment as a control. However, in adult goats, the right testes were removed immediately after electroporation to prevent the dilution of transgenic sperms.
Before the electroporation procedure, regional anesthesia of the testicular area was achieved by epidural infiltration of lignocaine 2% (Xylocaine 2%, Astra Zeneca, UK). The animal was placed in lateral recumbency with the left hind limb up and away by exposing the surgical field. The scrotum and surrounding area were clipped and prepared aseptically for surgery. An incision was made on the caudoventral surface of the testis through the skin and tunica dartos. The testis was exposed in the scrotal sack and then fixed with the tip of fingers to avoid retraction during the injection. Injection and electroporation conditions were similar to those of the in vitro experiment. After electroporation, the testis was replaced. The non-injected testis was removed by separating the cremaster from the vascular testicular cord. Each of these structures was ligated by transfixation suturing. Finally, the cord was transected distal to the ligatures and the testis was removed. The muscle and skin layers were sutured leaving a small gap for the exudation (Supplementary Figure S2). An antiseptic dressing containing povidone-iodine was provided for healing. All the animals were housed in individual shelters and administered with broad-spectrum antibiotics.
Detection of EGFP expression in the in vivo transfected testes
On d21 post-electroporation, testes (i.e., the left experimental and right control) from the prepubertal goats were surgically removed and were transported immediately to the laboratory on ice. In the laboratory, the epididymis was separated from the testis and testicular tissues were processed for further analysis.
A piece of each testis was cut into thin sections and the obtained testicular tissues were teased apart in PBS to separate the seminiferous tubules. The isolated seminiferous tubules were examined directly under a fluorescence microscope to assess the EGFP expression. A portion of the isolated seminiferous tubules was stored in RNAlater (Qiagen, Germany) for total RNA isolation, and another portion in T-PER reagent (Thermo Scientific, USA) for protein isolation, at −20 °C until further use. Histomorphology and IHC analysis of the testes were performed with tissue fixed in 4% paraformaldehyde (PFA) and sectioned at 5 µm thickness. IHC staining of fixed tissue samples was performed using anti- GFP antibody (Supplementary materials and methods).
Total RNA was extracted from the seminiferous tubules using TRI-reagent (Sigma, USA) as per the recommended protocol. One microgram of DNase-treated total RNA was reverse-transcribed using a cDNA synthesis kit (RevertAid H Minus First Strand cDNA Synthesis Kit, Fermentas, USA). The expression of EGFP was analyzed by qPCR on an Applied Biosystems 7500 Real-time PCR system (Supplementary materials and methods). β-actin (ACTB) was used as a reference gene for qPCR analysis. Total protein was extracted from the seminiferous tubules using T-PER reagent according to the manufacturer’s instructions. Total protein (30 μg per lane) was analyzed by western blotting according to standard procedures (Supplementary materials and methods).
Evaluation of semen parameters
On d30, d60 and d120 after electroporation, semen samples were collected from the bucks (n = 3) using a sterilized artificial vagina (AV). Immediately after the semen collection, the tube containing semen was placed in a water bath (37 °C). Semen samples were analyzed to evaluate viability, motility, membrane integrity and acrosomal integrity. To assess the progressive motility, semen was diluted (1:10) with sodium citrate-glucose (SCG) buffer. One drop of the diluted semen was placed on a glass slide, and after placing a cover slip, it was immediately examined under the high power (40×) objective of a phase contrast microscope. At least 20 fields were examined and the average number of motile sperm cells (%) was determined. Eosin-Nigrosin based differential staining was used to determine the live/dead percentage of the sperms. A hypo-osmotic swelling test (HOST) was employed to assess the functional integrity of the sperm cell membranes56. The acrosomal integrity was determined by staining with fluorescein isothiocyanate coupled with peanut agglutinin (FITC-PSA)57.
Estimation of transgene integration in the sperm
To assess the integration of the transgene into the sperm, semen samples from the transfected testes of all three bucks were collected at five different time intervals: d 21, 30, 60, 90 and 120 post-electroporation. Each 50 µl semen sample was washed four times with PBS and centrifuged for 5 min at 800 g to pellet the sperms. Total DNA from the pelleted sperm was isolated using a GeneipureIDTM DNA isolation kit (Genei, India) according to the manufacturer’s instructions. The presence of the EGFP gene in the sperm of transfected animals at different intervals was assessed using qPCR. β-actin was used as an internal control (Supplementary materials and methods). At d60, a portion of the semen samples was examined directly under a fluorescence microscope to detect EGFP expression in sperm. Approximately 200 sperms were examined from three representative microscopic fields for each semen sample and EGFP-positive sperms were counted. Further, RT-PCR analysis of sperm RNA was performed at d60 post-electroporation.
Fertilizing ability of the sperms from the bucks with transfected testes
Semen samples from in vivo transfected testes were used for in vitro fertilization (IVF) to evaluate the effects of electroporation on the fertilizing ability of the sperm. Goat ovaries were obtained from the local abattoir and aspirated oocytes were matured, fertilized and cultured in vitro (Supplementary materials and methods). The cleaved embryos were assessed for EGFP expression under a fluorescence microscope. Fluorescent embryos were pooled, and total RNA was extracted using an RNeasy Mini Kit (Qiagen, Germany). Then, RT-PCR analysis was performed to assess the abundance of EGFP transcripts in the embryos.
Transgenic screening of kids born out of natural mating of bucks with transfected testis
After d60 post transfection, bucks were allowed to mate naturally with the adult female goats (does) of similar age. Blood genomic DNA was isolated from all the kids born using phenol-chloroform extraction followed by ethanol precipitation58. The genotypes of all the kids were verified by PCR (Supplementary materials and methods) and Southern blot analysis (Supplementary materials and methods) to determine the presence of the EGFP reporter gene. In vivo EGFP fluorescence in the kids was assessed using a portable light source (excitation maximum = 488 nm; emission maximum = 507 nm). EGFP gene expression was also evaluated by RT-PCR analysis of blood and skin samples.
Statistical analysis
The transfection efficiency of in vitro electroporation was evaluated using two-way ANOVA. One-way ANOVA was used to assess the effects of in vivo electroporation on the different semen parameters, the relative expression of EGFP mRNA in testis and the presence of the EGFP gene in sperm. The percentage data of the semen parameters were subjected to arcsine transformation before statistical analysis. A P-value < 0.05 was considered to be statistically significant.
References
Hadjantonakis, A., Gertsenstein, M., Ikawa, M., Okabe, M. & Nagy, A. Generating green fluorescent mice by germline transmission of green fluorescent ES cells. Mech. Dev. 76, 79–90 (1998).
Wall, R. J. Pronuclear microinjection. Cloning Stem Cells. 3, 209–220 (2001).
Lois, C., Hong, E. J., Pease, S., Brown, E. J. & Baltimore, D. Germline transmission and tissue specific expression of transgenes delivered by lentiviral vectors. Science. 295, 868–872 (2002).
Liu, C., Xie, W., Gui, C. & Du, Y. Pronuclear microinjection and oviduct transfer procedures for transgenic mouse production. Methods Mol. Biol. 1027, 217–232 (2013).
Rogers, C. S. et al. Production of CFTR-null and CFTR-ΔF508 heterozygous pigs by adeno-associated virus–mediated gene targeting and somatic cell nuclear transfer. J. Clin. Invest. 118, 1571–1577 (2008).
Chen, X., Zhang, Z., Chang, X., Niu, Y. & Cui, H. Production of transgenic mice expressing tumor virus A under ovarian specific promoter 1 control using testis mediated gene transfer. Mol. Med. Rep. 9, 955–960 (2014).
Maione, B., Lavitrano, M., Spadafora, C. & Kiessling, A. A. Sperm-mediated gene transfer in mice. Mol. Reprod. Dev. 50, 406–409 (1998).
Dhup, S. & Majumdar, S. S. Transgenesis via permanent integration of genes in repopulating spermatogonial cells in vivo. Nat. Methods. 5, 601–603 (2008).
Yamazaki, Y., Yagi, T., Ozaki, T. & Imoto, K. In vivo gene transfer to mouse spermatogenic cells using green fluorescent protein as a marker. J. Exp. Zool. 286, 212–218 (2000).
Sehgal, L. et al. Lentiviral Mediated Transgenesis by In Vivo Manipulation of Spermatogonial Stem Cells. PLoS ONE. 6, e21975, https://doi.org/10.1371/journal.pone.0021975 (2011).
Blanchard, K. T. & Boekelheide, K. Adenovirus-Mediated Gene Transfer to Rat Testis In Vivo. Biol. Reprod. 56, 495–500 (1997).
Amaral, M. G. et al. Testis-mediated gene transfer in mice: comparison of transfection reagents regarding transgene transmission and testicular damage. Biol. Res. 44, 229–234 (2011).
Kubota, H., Hayashi, Y., Kubota, Y., Coward, K. & Parrington, J. Comparison of two methods of in vivo gene transfer by electroporation. Fertil. Steril. 83, 1310–1318 (2005).
Hibbitt, O. et al. In vivo gene transfer by electroporation allows expression of a fluorescent transgene in hamster testis and epididymal sperm and has no adverse effects upon testicular integrity or sperm quality. Biol. Reprod. 74, 95–101 (2006).
Huang, Z. et al. In vivo transfection of testicular germ cells and transgenesis by using the mitochondrially localized jellyfish fluorescent protein gene. FEBS Lett. 487, 248–251 (2000).
Ebert, K. M. et al. Transgenic production of a variant of human tissue-type plasminogen activator in goat milk: generation of transgenic goats and analysis of expression. Nature Biotechnol. 9, 835–838 (1991).
Meade, H. M. et al. Expression of recombinant proteins in the milk of transgenic animals. Gene Expression Systems: Using Nature for the Art of Expression (eds Fernandez, J. M. & Hoeffler, J. P.), 399–427 (1998).
Jänne, J. et al. Transgenic bioreactors. Biotechnol. Annu. Rev. 4, 55–74 (1998).
Houdebine, L. M. Production of pharmaceutical proteins by transgenic animals. Comp. Immunol. Microbiol. Infect. Dis. 32, 107–121 (2009).
Freitas, V. J. et al. Production of transgenic goat (Capra hircus) with human Granulocyte Colony Stimulating Factor (hG-CSF) gene in Brazil. An. Acad. Bras. Cienc. 79, 585–592 (2007).
Bokhoven, M. et al. Insertional gene activation by lentiviral and gammaretroviral vectors. J. Virol. 83, 283–294 (2009).
Modlich, U. et al. Insertional transformation of hematopoietic cells by self-inactivating lentiviral and gammaretroviral vectors. Mol. Ther. 17, 1919–1928 (2009).
Biffi, A. et al. Lentiviral vector common integration sites in preclinical models and a clinical trial reflect a benign integration bias and not oncogenic selection. Blood 117, 5332–5339 (2011).
Thomas, C. E., Ehrhardt, A. & Kay, M. A. Progress and problems with the use of viral vectors for gene therapy. Nat. Rev. Genet. 4, 346–358 (2003).
Meng, J. et al. Autologous skeletal muscle derived cells expressing a novel functional dystrophin provide a potential therapy for Duchenne Muscular Dystrophy. Sci. Rep. 6, 19750 (2016).
Kumar, R. et al. Testicular biometry and seasonal variations in semen parameters of Black Bengal goats. Indian J. Anim. Sci. 84, 635–639 (2014).
Pathak, A., Katiyar, R. S., Sharma, D. N. & Farooqui, M. M. Postnatal developmental anatomy of testes and epididymis of Gaddi goats. Int. J. Morphol. 32, 1391–1398 (2014).
Kabiraj, S. K., Hoque, S. A. M., Khandoker, M. A. M. Y. & Husain, S. S. Testicular biometry and its relationship with body weight and semen output of black Bengal bucks in Bangladesh. J. Cell Anim. Biol. 5, 27–32 (2011).
Chen, T., Segall, E. M., Langer, R. & Weaver, J. C. Skin electroporation: rapid measurements of the transdermal voltage and flux of four fluorescent molecules show a transition to large fluxes near 50 V. J. Pharm. Sci. 87, 1368–74 (1998).
Oatley, J. M., de Avila, D. M., Reeves, J. J. & McLean, D. J. Spermatogenesis and germ cell transgene expression in xenografted bovine testicular tissue. Biol. Reprod. 71, 494–501 (2004).
Yomogida, K., Yagura, Y. & Nishimune, Y. Electroporated transgene-rescued spermatogenesis in infertile mutant mice with a sertoli cell defect. Biol. Reprod. 67, 712–717 (2002).
Raina, A., Kumar, S., Shrivastava, R. & Mitra, A. Testis Mediated Gene Transfer, in Vitro Transfection in Goat Testis by Electroporation. Gene. 554, 96–100 (2015).
Usmani, A. et al. A non-surgical approach for male germ cell mediated gene transmission through transgenesis. Sci. Rep. 3, 3430, https://doi.org/10.1038/srep03430 (2013).
Chandrashekran, A. et al. Lentiviral vector transduction of spermatozoa as a tool for the study of early development. FEBS Open Bio. 4, 266–275 (2014).
Hofmann, A. et al. Efficient transgenesis in farm animals by lentiviral vectors. EMBO Rep. 4, 1054–1060 (2003).
Garrels, W. et al., Genotype-independent transmission of transgenic fluorophore protein by boar spermatozoa. PLoS ONE 6, e27563, https://doi.org/10.1371/journal.pone.0027563 (2011).
Villuendas, G. CMV-driven expression of green fluorescent protein (GFP) in male germ cells of transgenic mice and its effect on fertility. Int. J. Androl. 24, 300–305 (2001).
Kramer, J. A. & Krawetz, S. A. RNA in spermatozoa: implications for the alternative haploid genome. Mol. Hum. Reprod. 3, 473–478 (1997).
Grunewald, S., Paasch, U., Glander, H. J. & Anderegg, U. Mature human spermatozoa do not transcribe novel RNA. Andrologia 37, 69–71 (2005).
Feugang, J. M. et al. Transcriptome analysis of bull spermatozoa: implications for male fertility. Reprod. Biomed. Online 21, 312–24 (2010).
Sendler, E. et al. Stability, delivery and functions of human sperm RNAs at fertilization. Nucleic Acids Res. 41, 4104–4117 (2013).
França, L. R., Becker-Silva, S. C. & Chiarini-Garcia, H. The length of the cycle of seminiferous epithelium in goats Capra hircus. Tissue Cell. 313, 274–280 (1999).
Pramod, K. R., Kumar, R. & Mitra, A. Transgenic expression of green fluorescent protein in caprine embryo produced through electroporation-aided sperm-mediated gene transfer. Gene. 576, 505–511 (2016).
Gandolfi, F. et al. Failure to produce transgenic offspring by intra-tubal insemination of gilts with DNA-treated sperm. Reprod. Fertil. Dev. 8, 1055–1060 (1996).
García-Vázquez, F. A. et al. Factors affecting porcine sperm mediated gene transfer. Res. Vet. Sci. 913, 446–453 (2011).
Maione, B., Pittoggi, C., Achene, L., Lorenzini, R. & Spadafora, C. Activation of endogenous nucleases in mature sperm cells upon interaction with exogenous DNA. DNA Cell Biol. 16, 1087–1097 (1997).
Chang, K. T. et al. Production of transgenic rats and mice by the testis-mediated gene transfer. J. Reprod. Dev. 45, 29–36 (1999).
Sato, M., Gotoh, K. & Kimura, M. Sperm-mediated gene transfer by direct injection of foreign DNA into mouse testis. Transgenics 2, 357–369 (1999).
Brinster, R. L. et al. Somatic expression of herpes thymidine kinase in mice following injection of a fusion gene into eggs. Cell 27, 223–231 (1981).
Jaenisch, R. Transgenic animals. Science 240, 1468–1474 (1988).
Bishop, J. O. & Smith, P. Mechanism of chromosomal integration of microinjected DNA. Mol. Biol. Med. 6, 283–298 (1989).
Cui, X. Targeted integration in rat and mouse embryos with zinc-finger nucleases. Nat. Biotechnol. 29, 64–67 (2011).
Park, T. S., Lee, H. J., Kim, K. H., Kim, J. S. & Han, J. Y. Targeted gene knockout in chickens mediated by TALENs. Proc. Natl. Acad. Sci. USA 111, 12716–12721 (2014).
Dow, L. E. Inducible in vivo genome editing with CRISPR-Cas9. Nat. Biotechnol. 33, 390–394 (2015).
Pramod, K. R. & Mitra, A. In vitro culture and characterization of spermatogonial stem cells on Sertoli cell feeder layer in goat (Capra hircus). J. Assist. Reprod. Genet. 31, 993–1001 (2014).
Jeyendran, R. S., Van Der Ven, H. H., Perez-Pelaez, M., Crabo, B. G. & Zaneveld, L. J. D. Development of an assay to assess the functional integrity of the human sperm membrane and its relationship to other semen characteristics. J. Reprod. Fertil. 70, 219–228 (1984).
Sukardi, S., Curry, M. R. & Watson, P. F. Simultaneous detection of the acrosomal status and viability of incubated ram spermatozoa using ûuorescentmarkers. Anim. Reprod. Sci. 46, 89–96 (1997).
Haraguchi, S. & Nakagawara, A. A. Simple PCR Method for Rapid Genotype Analysis of the TH-MYCN Transgenic Mouse. PLoS ONE. 4, e6902, https://doi.org/10.1371/journal.pone.0006902 (2009).
Acknowledgements
This work is supported by a National Fellow project, Indian Council of Agricultural Research (ICAR), Government of India. The authors duly acknowledge ICAR-Senior Research Fellowship to R.K.P. during his Ph.D. program, and further acknowledge Dr. G.K. Das, Principal Scientist, Animal Reproduction Division, IVRI, Bareilly, Uttar Pradesh and Dr. Ratan K. Choudhary, Assistant Professor, School of Animal Biotechnology, GADVASU, Ludhiana for critical reading of the manuscript.
Author information
Authors and Affiliations
Contributions
R.K.P. prepared and drafted the manuscript and helped in designing the experiments. Experiments were performed by R.K.P. A.M. conceived the initial idea of the study, designed and coordinated the whole study and helped in preparing the manuscript. Both the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pramod, R.K., Mitra, A. Intratesticular injection followed by electroporation allows gene transfer in caprine spermatogenic cells. Sci Rep 8, 3169 (2018). https://doi.org/10.1038/s41598-018-21558-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-21558-9
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.