Abstract
Carbon accumulation in tropical secondary forests may be limited in part by nitrogen (N) availability, but changes in N during tropical forest succession have rarely been quantified. We explored N cycle dynamics across a chronosequence of secondary tropical forests in the Mata Atlântica of Bahia, Brazil in order to understand how quickly the N cycle recuperates. We hypothesized that N fixation would decline over the course of succession as N availability and N gaseous losses increased. We measured N fixation, KCl-extractable N, net mineralization and nitrification, resin-strip sorbed N, gaseous N emissions and the soil δ15N in stands that were 20, 35, 50, and > 50 years old. Contrary to our initial hypothesis, we found no significant differences between stand ages in any measured variable. Our findings suggest that secondary forests in this region of the Atlantic forest reached pre-disturbance N cycling dynamics after just 20 years of succession. This result contrasts with previous study in the Amazon, where the N cycle recovered slowly after abandonment from pasture reaching pre-disturbance N cycling levels after ~50 years of succession. Our results suggest the pace of the N cycle, and perhaps tropical secondary forest, recovery, may vary regionally.
Similar content being viewed by others
Introduction
More than half of extant tropical forests are regenerating from disturbance1 and in the coming decades these re-growing forests will be a substantial carbon sink (~1 Pg yr−1)2, provide habitat for myriad species, and food, fuel and fiber for millions of people2. In the Neotropics, secondary forests recover biomass relatively quickly, reaching 90% of pre-disturbance biomass levels within ~66 years3. In part, the extent and pace at which secondary forests recover depends on the availability of essential plant nutrients, particularly nitrogen and phosphorus4. Land use activities driving deforestation, such as slash and burn agriculture, timber harvesting, and cattle grazing, remove essential plant nutrients such as nitrogen (N) and phosphorus (P) from the system5,6. While both N and P are lost during fires as particulates, N can also volatilize as nitrogen oxides (NOx) and nitrous oxide (N2O). This sets the stage for greater potential N than P limitation during early stages of secondary succession6,7. However, the possibility for high rates of biological N fixation suggests N may accumulate relatively quickly during secondary forest regrowth8. Both the absolute and relative availability of N and P in these systems is likely to influence the rate at which they recover3,4,5,8.
Biogeochemical theory along with empirical evidence suggests that mature lowland tropical forests on highly weathered soils tend to cycle N in relative excess compared with P9,10 with high N leaching11 and gaseous losses12,13. This is in part because phosphorus is ultimately sourced from parent material and gradually depleted over time14 in the absence of high dust inputs15 or rejuvenation by erosion16. In addition, the lowland neotropics have a relatively high (~10% basal area) abundance of trees and lianas in the Fabaceae family17 (hereafter “legumes”), many of which can form symbiotic relationships with N fixing bacteria. High legume abundance and relatively high levels of asymbiotic N fixation in the warm, wet litter and soil18,19,20,21, suggests inputs of N are 5–10 times higher than in temperate forests21. Thus relatively high N and low P availability may be common in the lowland neotropics, but this pattern can be altered when forests are cleared, since burning (a common land clearing strategy) preferentially removes N compared with P6.
To our knowledge, there has been one study that documented N recovery in secondary neotropical evergreen forests7,22 finding evidence for recuperation of the N cycle during ~70 years of secondary succession in the Brazilian Amazon. In this study, the response of several soil and plant metrics were examined (foliar δ15N, foliar N, litterfall N:P, soil nitrate, soil N2O emissions) across a chronosequence, finding that after ~50 years the N cycle transitioned from a relatively closed N cycle (low soil nitrate, low soil nitrate: ammonium ratio, low N2O losses) to a relatively open one (high soil nitrate, high soil nitrate: ammonium ratio, high N2O losses)23. Despite the apparent importance of N in limiting tree regrowth early in succession22, it is not known whether the pace of N recovery found in the Amazon is similar to that found elsewhere. There are several reasons to suspect that the N cycle trajectory during forest succession might differ even within neotropical forests. First, there is substantial variation in biomass recovery even in similar aged forest stands, both among and within secondary forest landscapes3. This suggests that different processes may regulate the pace of forest regeneration, and by extension regeneration of nutrient cycles. Part of this variation is attributable to climate2, but it is also likely that soils24, plant community assembly25,26, and past land use type and intensity5 are also important factors in N cycle recovery rates.
In this context, we studied N cycling across a chronosequence of secondary forests in the Brazilian Atlantic Forest (or the Mata Atlântica) in the south of Bahia, Brazil. Our study site is located in perhaps the least studied, and most threatened, tropical forest biome on earth1. This ancient forest, as diverse as the Amazon27, once stretched along the entire coastline of Brazil, but is now 85% deforested1. Some of the largest remaining fragments north of São Paulo state are found in the southern Bahia, and our study site is located in the Park Serra Do Conduru, which harbors the largest fragment of contiguous forest (10,000 ha of different aged forests) in the region. The Atlantic forest is a focus of extensive national and international restoration efforts28 and thus nutrient constraints to forest recovery have implications for both biogeochemists and restoration efforts. To our knowledge, our study represents the first comprehensive analysis of the N cycle in the Brazilian Atlantic Forest, and the second in the neotropics7.Our default hypothesis, in the absence of more data, is that the pattern observed in the Amazon would be the same in the Atlantic Forest. Thus we expect the N cycle would recover after ~ 50 years of forest succession. In order to test this hypothesis, we measured major inputs of N (via symbiotic and asymbiotic N fixation pathways), gaseous N losses (e.g. N2O & NOx), recycling (e.g. net mineralization and nitrification) and quantified plant available forms of soil N using two metrics.
Results
Nitrogen and phosphorus availability
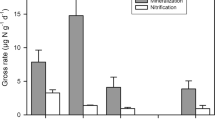
Inorganic N pools of ammonium and nitrate did not significantly vary with forest age in either April (p = 0.48, 0.61, respectively) or August (p = 0.67, 0.91, respectively; K-W test; Fig. 1C). The dominant form of inorganic N was nitrate with levels of ammonium frequently below detection (0.02 mg N- NH4+ kg−1). There was no difference between the concentration of nitrate (p = 0.88) and ammonium (p = 0.36) in August vs. April sampling (Fig. 1C,D). One site, from the 35-year-old forest, consistently had a higher concentration of nitrate near four times greater than all other sites. This site also had higher than average biomass for a 35-year-old forests according to biomass studies in these same study sites29, and ~3 times higher nitrous oxide flux than other 35-year-old forest sites (Fig. 2A). Analyzing data with this plot removed, however, did not change the overall lack of trend between inorganic N concentrations and forest age.
Indicators of nitrogen availability along secondary successional forest chronosequence. (A) Nitrogen mineralization rate, (B) in-situ resin extractable nitrate concentration, (C) instantaneous KCl-extractable soil nitrate concentration. and (D) soil nitrate: ammonium, (E) soil δ15N (0/00 vs. N2). Boxplot with each replicate plot indicated as a point (n = 5, except for mineralization rates in 35-year old forests, n = 3). Data is shown as boxplot with the median value indicated by the band near the middle of the box, the first and third quartiles are the top and bottom of the box, respectively, the maximum and minimum values excluding outliers are the whiskers. Data in panel C and D show data from two sampling times; April = closed circles and August = closed triangles. The effect of forest age is not significant for all N indicators examined (p > 0.05; K-W test).
Nitrogen oxide soil emissions along secondary successional forest chronosequence. (A) nitrous oxide (N2O) emissions. Circles = August sampling and Triangles = November sampling (n = 5), and (B) nitrogen oxide emissions. Circles = NO and Triangles = NO2. (n = 3, except mature forest n = 2). Data is shown as boxplot with the median value indicated by the band near the middle of the box, the first and third quartiles are the top and bottom of the box, respectively, the maximum and minimum values excluding outliers are the whiskers. The effect of forest age is not significant for nitrogen oxide gases examined (p > 0.05; K-W test).
In situ measures of available nitrate, collected using the anion-exchange membranes, did not significantly vary with forest age (p = 0.44; K-W test; Fig. 1B). Resin strip phosphate levels were consistently close to, or below detection limit (0.05 mg P kg−1), with no significant difference among different-aged forests (p = 0.29; K-W test; Table 1).
Net N mineralization was dominated by net nitrification (>98%) and did not significantly vary with forest age (p = 0.20; K-W test; Fig. 1A). Total soil pools of N and carbon (Table 1) did not significantly change with stand age (p = 0.25 & 0.43, respectively; K-W test), nor were there significant differences in the N isotopic signature of soils with stand age (p = 0.58; K-W test; Fig. 1E). δ15N (0/00 vs. N2) of soil had a mean of 4.648 +/− 0.42 and ranged from 3.330 to 6.807.
Nitrogen oxide gas fluxes
Nitrous oxide (N2O) fluxes were highly variable. In August, fluxes ranged from −1.95 µg N-N2O m−2 hr−1 to 29.36 µg N-N2O m−2 hr−1 (Fig. 2A). Mean flux in August was 4.09+/− 1.6 kg µg N-N2O m−2 hr−1. In November, fluxes ranged from 0 to 21.72 µg N-N2O m−2 hr−1. Mean flux in November was 7.95 +/− 1.3 µg N-N2O m−2 hr−1. Soil moisture, a key variable known to influence N2O fluxes30, was greater during the November sampling (p = 0.001). However, soil moisture did not explain significant variation in N2O fluxes in August (R = 0.1; p = 0.27) or November (R = −0.22; p = 0.89).
N2O fluxes did not significantly differ with forest age in either August or November (Fig. 2A). In August, the 35-year-old forests had significantly higher N2O fluxes than the mature forests (p = 0.01), but in November, there were no significant differences in N2O fluxes among forest age classes (p = 0.19). When mature forests were excluded, there were no significant differences observed in N2O fluxes among different aged secondary forests in either August (p = 0.14) or November (p = 0.09). There was no seasonal variation in N2O fluxes in 20-year-old (p = 0.3), 35-year-old (p = 0.80), and 50-year-old forests (p = 0.2; Wilcoxon test). However, N2O fluxes in mature forests differed between August and November (p = 0.007; Wilcoxon test) with a higher N2O flux in November.
Soil emissions of nitrogen oxide gases did not significantly vary between forests of different ages for either NO (p = 0.397) or NO2 (p = 0.498; Fig. 2B). N-NO fluxes ranged from 1.80 +/− 0.85 ppbv in mature forests to 3.60 +/− 0.89 ppbv in 35-year-old forests. N-NO2 fluxes were lower than NO fluxes and ranged from 0.748 +/− 0.24 ng ppbv in 50-year-old forests to 1.421 +/−0.343 ppbv in mature forests.
Biological nitrogen fixation
Despite differences in the abundance of legumes, which were significantly more abundant in the youngest forests (p < 0.05; Table 1), we found no significant differences in symbiotic N fixation across forest succession (F(3, 16) = 0.51; p = 0.68; Fig. 3A). However, rates of SNF were most variable early in forest succession, ranging from 1.06 to 22.75 kg N ha−1 yr−1 in 20-year-old forests (mean 10.29 +/− 4.20 kg N ha−1 yr−1) compared to 0.3 to 8.16 kg N ha−1 yr−1 in mature forests (mean 4.61 +/− 1.7 kg N ha−1 yr−1). Additionally, there was no correlation between percentage of putative legume basal area and rates of SNF (R = 0.20; p = 0.16). Rates of asymbiotic N fixation did not significantly vary with forest age in either the soil layer (p = 0.53) or the leaf litter layer (p = 0.70; Fig. 3B,C). The fraction of total BNF accounted for by FNF varied across sites from 0–100% with no significant trends with stand age.
Biological nitrogen fixation along secondary successional forest chronosequence. (A) symbiotic nitrogen fixation (SNF), (B) leaf litter asymbiotic N fixation (FNF), and (C) soil asymbiotic N fixation. Data is shown as boxplot with the median value indicated by the band near the middle of the box, the first and third quartiles are the top and bottom of the box, respectively, the maximum and minimum values excluding outliers are the whiskers. The effect of forest age is not significant for symbiotic nitrogen fixation (p > 0.05; ANOVA) or asymbiotic nitrogen fixation p > 0.05; K-W test).
Discussion
Contrary to our hypothesis we did not find evidence for changes in the N cycle during the 20 to 50 years of forest succession examined in this study. Instead our data suggests that secondary forests in this region of the Atlantic forest reach pre-disturbance N cycling dynamics by 20 years of succession. We measured several indicators of the N cycle including several different metrics of N availability (Fig. 1), N outputs via nitrogen oxide emissions (N2O, NO, and NO2; Fig. 2), and N inputs via symbiotic and asymbiotic N fixation (Fig. 3) - none of which varied significantly with forest age in our chronosequence, nor between all secondary forests versus mature forests. These findings are in contrast to a similar study7 in the Brazilian Amazon where N cycle indicators increased over the course of 3 to 70 years of succession, a time frame that includes the period examined in this study (20 to 50 years). Given the abundance of legumes and the high soil nitrate: ammonium, we hypothesize that the N cycle recovered rapidly in the first 20 years of succession. Future studies in this region, however, are needed in young forests (<20 years) to test this hypothesis.
In the absence of other major external N sources (such as anthropogenic nitrogen deposition31), biological N fixation especially by legume-rhizobia symbiosis, is thought to be the main pathway for secondary forests to recuperate N losses from prior land use activities and meet the N demands of rapidly re-growing forests8. Rates of SNF in excess of ~10 kg N ha−1 yr−1 could account for the accumulation of N in biomass observed during succession8,21. Available methods for determining rates of biological N fixation are, however, poorly constrained and have their limitations32. Nevertheless, the few studies to quantify N fixation by legume species during secondary succession suggest SNF rates are highest in the first twenty years of forest succession8,21, resulting in increased pools of inorganic N, N mineralization rates, and N2O fluxes33,34,35. While we did not observe a significant difference in SNF with stand age we did observe the highest rates in some of the youngest forests (upwards of 20 kg N ha−1 yr−1), and it is possible that with greater sampling effort we would have more support for the trend towards higher SNF in younger sites. Alternatively, if even ~10% of the recalcitrant forms of soil N were mineralized gradually over time (in top 10 cm of mineral soil there is ~1872 kg N ha−1), then ample N would be available to fuel the biomass recovery observed in study plots29.
Three lines of evidence suggests the N cycle might recover quickly in the first 20 years of succession (Fig. 1). First the ratio of soil nitrate to ammonium is high suggesting soil N has accumulated in excess of plant demand36. This is because ammonium is often the dominant form of inorganic soil N in N-limited systems7. Ammonium is the precursor to the production of nitrate during the mineralization of organic matter. Therefore, increases in the soil nitrate pool tend to only occur once ammonium has accumulated in the soil allowing nitrifying bacteria to become competitive and convert ammonium into nitrate36. Indeed, nitrification was found to be the dominant (>98%) process responsible for mineralization across forest stand ages. Together these two pieces of evidence suggest that N supplies are sufficient for biological demand, even in 20-year old forest stands. Lastly, the lack of a significant change in nitrogen oxide emissions (N2O, NO, NO2) with forest age is also consistent with this hypothesis. The aerobic process of nitrification and anaerobic process of denitrification are the main contributors to nitrogen gas losses, both of which require inorganic N pools in the soil to accumulate past biotic demand (or for microbes to outcompete plants for inorganic N)37. Increases in nitrogen oxide gases thus indicate the recuperation of several N cycle processes. However, nitrous oxide fluxes are known to be highly variable and therefore the lack of a trend with forest age could also be due to insufficient temporal and spatial sampling38. Nevertheless, together these data suggest N has re-accumulated in this system after just 20 years of forest succession. Future studies, however, are needed in the region on forests <20 years to test this hypothesis. Unfortunately, in our study area, owing to the age of the park, forests <20 years old were not available.
Given differences in methods for quantifying N cycle metrics (e.g. different extraction times and degree of shacking in KCl), we were only able to compare our study system to forests on the Osa Peninsula of Costa Rica39. We found our study system had levels of KCl-extractable N, nitrification rates and mineralization rates on the low end of those relatively N poor mature forests in the Osa Pennisula39,40,41. However, the ratio of nitrate:ammonium in our dataset suggests N is accumulating and meeting plant demands. The naturally low N levels observed in this region of the Brazilian Atlantic forests may explain a potential for rapid recovery of the N cycle to pre-disturbance levels. While mature lowland tropical forests are thought to cycle N in excess relative to rock-derived nutrients such as P, recent studies have demonstrated that tropical lowland forests have a wide range of N availability39 at multiple spatial scales42. Overall, our study provides support to a growing body of evidence demonstrating the heterogeneity in tropical forest N cycling at multiple spatial scales42,43.
Differences in prior land use type and intensity, as well as climatic differences in water availability, fire regimes and soil types, may explain the N cycle trajectories observed in this study compared to prior work in the Brazilian Amazon. In our study, we focused on secondary forests growing on areas that experienced slash and burn agriculture with short cultivation phase (2–3 years) while previous work in the Amazon focused on land re-growing on both abandoned pastures and cropland. These two types of land use practices are common in tropical regions and are known to change the initial starting conditions of soil nutrients5; however, pasture management leads to increased soil compaction than non-animal mechanized agriculture44. Intensive prior land use by either practice can inhibit the establishment and growth of woody vegetation during early stages of succession, such as legume tree species, thereby delaying the recovery of the N cycle5. Indeed, legumes are not observed as the dominant species in some young successional forests of the eastern Amazon22. A recent meta-analysis of tropical secondary forest biomass recovery, however, found that there were no significant differences in the biomass recovery between slash and burn agriculture versus pasture3. The authors suggest this may be a result of the high within-category variation observed in land use intensity. Instead differences in biomass recovery were more strongly influenced by variation in water availability, both higher annual rainfall and lower climatic water deficit. In Bahia, rainfall is aseasonal and natural fires are uncommon45. In contrast, the study in the Amazon was in a region that experienced similar rainfall (~1800 mm/year) but with a strong dry season and history of natural forest fires7,22.
Conclusion
Our study examined N cycle dynamics across a chronosequence of secondary tropical forests in the Mata Atlântica of Bahia, Brazil in order to understand how quickly the N cycle recuperates. The pace at which the N cycle recovers has important implications for restoration efforts and conservation priorities. We found that the Atlantic forest reached pre-disturbance N cycling dynamics after just 20 years of succession. These findings suggest that the N cycle recuperates quickly in the first 20 years of succession, however, additional studies in younger forests within the region (<20-year-old stands) are needed. To our knowledge, our study represents the first comprehensive analysis of the N cycle in the Brazilian Atlantic Forest, and the second in the neotropics7. Our study supports emerging evidence that symbiotic nitrogen fixation helps the N cycle recover during secondary succession8,21. However, the pace at which the N cycle recovers appears to vary across tropical secondary forests highlighting the need for future studies to test the controls and factors that influence the pace of N cycle recovery during tropical secondary forests succession.
Methods
Study Site
Our study sites are secondary and mature forests in and around Serra do Condoru State park located in southern Bahia, Brazil (14°25′ S and 39°05′ W) within established forest plots45,46. The park was established in 1997 and encompasses an area of roughly 10,000 ha, the largest contiguous area of Atlantic Forest north of São Paulo state. Forests in the park are a mosaic of different stages of development including secondary forests and remnants of mature forests that have varying degrees of selective logging in the past. During our study period (April to November 2016) mean annual temperature at the site was 24 °C and mean annual precipitation was ~200 mm of rainfall per month. The area does not have significant seasonality in temperature or rainfall except during strong El Niño years when monthly rainfall was typically <25 mm. Our sampling begun at the end of an El Niño year.
Chronosequence – We sampled in 20, 10 × 50-meter study plots consisting of four age classes 20, 35, 50, and mature, with 5 replicate plots per age class. Sites were selected to control for previous land use (slash and burn agriculture followed by 1 to 2 years of manioc cultivation) and differences in soil types (Typic Haplorthox), while including regional variation in topography (120–300 m above sea level and 10–30% slope angle). The estimation of site ages and past land uses were based on a sequence of available aerial photographs and remote sensing data which provided precise and verifiable estimates of site age. Changes in forest cover and land use in a 5000-ha area inside Serra do Conduru State Park were estimated from 1965 to 2007. The remote sensing windows were based on aerial photos taken in 1965, 1975, 1986, 1997, 2002, and 2007. Land use maps were generated for every set of aerial photos and maps of forest age classes were derived using GIS. Information on the type and intensity of past land use was gathered by interviewing local farmers. A total of 95 secondary forest stands (ages ranging from 20 to 50 years old) larger than 3-ha and adjacent to a mature forest were located in the study area. Study plots were randomly selected to represent 5 replicates of four age classes: 20, 35, 50 and >50 years old (or mature). Because the Serra do Conduru state park was legally created in 1997, there were no portions of the park on recently abandoned agricultural fields, which constrained the inclusion of stands younger than 20 years in the chronosequence. Mature forests in the region varied in the degree of human disturbance (e.g. selective logging and hunting) and we selected mature forests sites that captured this variation, as indicated by the range in aboveground biomass for mature forests (300–500 Mg ha−1). At each site, secondary forest plots were located 20 to 50 m from a mature forest fragment and mature forests were at least 10 m from a secondary forest edge. A census for tree size and species identification was done for all plots in 2009. For more information on establishment of study plots see Piotto et al.45,46.
Soil nitrogen and phosphorus availability
We quantified N availability using three techniques: 1) instantaneous 2 N potassium chloride (KCl) extractable inorganic N (NH4+, NO3−), 2) net N mineralization and nitrification, and 3) cumulative in situ extractable NO3− measured using anion exchange resins over a period of two weeks. We measured instantaneous extractable inorganic N in April and August of 2016, while all other measurements were collected only in August 2016. For the analysis of inorganic N pools, net mineralization and net nitrification rates, we collected soils (3 per plot) from the top 10 cm of mineral soil using square 100 cm2 core. Within 4 hours of collection we sieved (2 mm) each soil core, extracted 8-g of soil in 30 mL of 2 N KCl and filtered through Whatman No. 1 filter paper. Net N mineralization and net nitrification were measured from changes in NH4+ and NO3− concentrations during a 4-day aerobic incubation of ~8-g subsamples in the dark at field temperatures. Each extraction was shaken manually for 60 seconds every hour for 4 hours39,47. All extracts were stored frozen until analyzed. ~10 g subsample was dried at 65 °C for 3 days to determine soil moisture. All measurements are reported on a dry-mass basis.
We used anion exchange resins membranes to determine mobile nitrate and phosphate in situ over the course of a two-week period. We prepared anion-exchange membrane strips using sheets of cross-linked copolymers of vinyl monomers from (General Electric #AR204SZRA). We prepared strips (~2 × 10 cm) by shaking them for 24 hours in 0.5 M NaHCO3 for phosphate, and 1 M NaCl for nitrate48. We inserted 12 anion-exchange membranes (6 nitrate, 6 phosphate) into the top 10 cm of each plot. The strips were inserted at a ~45° angle relative to the landscape slope and left in the field for 14 days. After 14 days in the field, we collected the membranes and rinsed them gently with distilled water to remove residual soil. The strips were kept at 4 °C and transported to the laboratory at Brown University. We shook the resin strips for 4 hours in either 30 mL of 2 M KCl solution (for nitrate) or 30 mL of 0.5 M HCl (for phosphate). We then filtered the samples through Swinnex filters and frozen until analysis. The area of the resin strip was determined using ImageJ software, and we report results as nutrient concentration per centimeter squared of resin strips. Concentrations of ammonium, nitrate, and phosphate from extracted samples were analyzed colorimetrically on a Westco SmartChem 200 Discrete Element Analyzer (Westco Instruments, Brookfield, Connecticut).
Soil Properties
In April 2016, three replicate cores per plot (1000 cm3) were collected, passed through a 2-mm sieve, and dried at 65 °C. Soils were sent to the Stable Isotope Facility at the University of California, Davis for analysis of total carbon and nitrogen, as well as the nitrogen isotopic signature. Soil pH and bulk density were quantified when the plots were established in 200945,46.
Soil nitrogen oxide gas emissions
We measured soil emissions of N2O fluxes in August and November of 2016 using syringe sampling of vented static chambers49 made of septic PVC (20 cm diameter) and placed in the soil to a depth of 2 cm after removing leaf litter from the surface. Three replicate chambers were placed in each of the 20 study plots. We waited 25 minutes after installing the collars before the first headspace sample was collected thus allowing for soil emissions to equilibrate after the disturbance created by inserting the collar. We then collected headspace samples every 15 to 45 minutes (a total of 4 time points). To reduce the effect of diel variability, we collected gas samples between the hours of 9:00 and 14:00 hour local time and varied the time at which different aged plots were sampled (for example, within an age class at least one replicate plot was sampled in the morning, mid-day, and afternoon).
All N2O gas samples (20 ml) were stored over pressurized in 12 mL Labco exetainer vials and returned to the lab at Brown University for analysis on Shimadzu GC-2014 equipped with electron capture detector (ECD) for quantifying N2O and flame ionization detector (FID) for quantifying CO2. We used the accumulation of CO2 during the 45 minutes the collar was capped to check for leaks in the collars. In addition, we used a set of standards to test for sample leakage, which traveled with the samples.
For both N2O and CO2 we calculated fluxes from linear regressions of the concentration increase, chamber volume, and air temperature and pressure (collected from the nearby Ilhéus Jorge Amado Airport weather station50) at time of sampling. The low N2O fluxes required longer deployment time (45 minutes) to reach linear increases in [N2O] over time. We used the degree to which [CO2] over time was linear as an indicator of potential chambers leaking. We therefore, did not include samples in our analysis if the [CO2] did not increase over time with a linear R2 > 0.98. However, if [CO2] increased linearly over 30 minutes these samples were retained in the analysis, with emissions rates based on 30 minutes. In total 30 out of 120 replicates were removed, and 10 additional 45-minute time points. N2O fluxes are presented as µg N-N2O m-2 hr−1 following equations in ref.49.
Soil moisture (an important covariate to consider with N2O fluxes30) was determined at the time of sampling using gravimetric methods in August 2016, and a TDR soil moisture probe (FieldScout TDR 300 #6430FS, Spectrum Technologies Inc.) in November 2016. N2O fluxes were coupled with measures of inorganic N for August 2016 sampling dates but not for November 2016. Inorganic N pools were not found to vary seasonally in our plots between April and August sampling dates, and logistical challenges prevented inorganic pools from being sampled in November 2016.
We quantified soil emissions of NOx using ambient air passive samplers consisting of pads that chemically captured NO2 and NOx (NO and NO2) (Ogawa & Co.). Samplers were deployed using a PVC rain shelter and support clips provided by the manufacturer and placed ~ 0.3 meters above the soil surface attached to a small diameter (<5 cm) tree. A set of blanks (pads stored in gas tight brown Nalgene bottles) were carried along with samples to determine potential NOx collection during transport and storage. Samplers were collected after 10 weeks of exposure, and pads were extracted in 8 ml of double distilled water which converted captured nitrogen oxide gases into nitrite. The resulting solution was kept refrigerated prior to analysis for the concentration of nitrogen as nitrite on a Westco SmartChem 200 Discrete Element Analyzer (Westco Instruments, Brookfield, Connecticut). Prior to chemical analysis, we treated the extraction of NOx pads with 1:1 ratio of sample to ether in order to remove a blue dye contained in the pads that interferes with colorimetric analysis of ion concentrations51. The ether precipitates the dye into a supernatant layer that was carefully removed with a pipette and the remaining ether was allowed to evaporate51. Following the protocol by Ogawa & Co., ambient gas phase N-NO2 in ppbv were calculated as C = (QNO2 * \(\propto \) NO2)/t, where C = concentration in ppbv, QNO2 = N-NO2 mass in ng, \(\propto \)NO2 = sampling rate, and t = sampling time in minutes. Sampling rate (\(\propto )\) is dependent on ambient pressure, temperature and humidity, and are determined based upon lookup tables provided by Ogawa. We used the average relative humidity (80%) and average temperature (25 °C) during the 10 weeks of exposure to determine the sampling rate. Similarly, the ambient N-NO concentration was calculated as C = ((QNO2 − QNO2)) * \(\propto \) NO)/t, where QNOx is the mass of N-NOx in ng. Data is presented as N-NO or N-NO2 in units of ppbv. NO estimates are not sensitive to changes in temperature, however, a 10% change in relative humidity results in 4% change in NO concentrations. NO2 estimates are sensitive to both temperature and relative humidity changes; a 5 °C difference in temperature results in 5.5% change in concentrations, while a 10% change in relative humidity results in 3% change in concentrations.
Symbiotic and asymbiotic N fixation
We measured symbiotic rates of N fixation randomly across 20 study plots over the course of 4 weeks in April and early May of 2016. Quantifying SNF requires destructive harvesting and is time consuming thus preventing seasonal measurements. We measured asymbiotic fixation in all 20 plots over the course of one week in April of 2016. Rates of N fixation were coupled with measures of inorganic N pools but were not also coupled with nitrous oxide gas emission sampling efforts due to logistical constraints. We quantified N fixation rates using traditional acetylene reduction assay (ARA) which were calibrated with 15N2 gas incubations for symbiotic nitrogen fixation (details below). We measured rates of symbiotic N fixation using a modified stratified adaptive cluster sampling approach (SACS) described by (Sullivan et al.21) to estimate both nodule biomass and rates of N fixation. Briefly, in each plot we sampled every 2 meters along 5, 10 m transects for a total of 25 cores per plot. When nodules were present in a core, all adjacent cores were examined for nodules until no more nodules were found. We used a 100 cm2 soil core to a depth of 10 cm. All excised nodules found using the SACS method were immediately placed in a 60-ml gas tight jar fitted with septa, exposed to 10% atmosphere of acetylene, and incubated for 1 hour. Acetylene was made in the field using calcium carbide. With each set of ARA, a set of blanks (acetylene with no sample) and controls (sample, no acetylene) were established to determine background ethylene levels present in the acetylene or produced by the sample. Within hours of field collection, nodules were desiccated using silica gel before weighing dry mass.
We measured asymbiotic fixation on decomposing leaf litter and the top 2 cm of the mineral soil horizon. We collected decomposing leaf litter from across the plot area (10 m × 50 m) and homogenized by hand before weighing roughly 2.5 grams of fresh litter into 60 ml acrylic tubes fitted with septa (6 replicates per plot). Soil was collected using a soil core that was 2 cm in diameter. As with excised nodules, soils and litter were exposed to 10% atmosphere of acetylene. Asymbiotic N fixation samples were incubated at field conditions for 18 hours because of the low rates of asymbiotic N fixation. The dry mass and moisture content of soil and litter samples were determined gravimetrically after oven drying for 3 days at 65 °C.
All gas samples from ARA method were stored in pre-evacuated gas tight jars (Labco, Exetainers), and returned to Brown University for analysis of ethylene concentrations using a Shimadzu GC-2014 gas chromatograph equipped with a flame ionization detector (330 °C) and a Poropak-T column at 80 °C. Rates of ARA were calculated as nanomoles ethylene produced dry g−1 hour−1. Rates of acetylene reduced by symbiotic N fixers were converted to N2 fixed using the site specific conversion ratio (2.98 mol ethylene reduced:1 mol N2 fixed) determined by 15N2 incubations following methods described in ref.52. Rates of SNF are reported as kg N fixed ha−1 yr−1 assuming 24 hours of BNF activity year round, thus allowing for comparisons in the literature. Asymbiotic rates were converted to N2 fixed using the theoretical conversion ratio of 3:1. Measures of asymbiotic N fixation were scaled to an area-based flux (e.g. µg N m−1 hr−1) by measuring the mass of litter on the forest floor in a defined area, at the time of collection.
Statistical analyses
All statistical tests were performed on the average of technical replicates from each plot. Data were tested for normality using Shapiro-Wilk test and homoscedasticity using Levene’s test. A 1-way ANOVA on ln-transformed data was used to determine the effect of forest age on rates of symbiotic N fixation, soil C%, soil N%, pH, and legume abundance. All other metrics of the N cycle violated the assumptions of parametric tests even after being ln-transformed, and thus non-parametric Kruskal-Wallis test on the untransformed data was used to determine significant effect of forest age on all N cycle metrics (e.g. inorganic N pools, mineralization rates, resin available P, resin available N, nitrogen oxide emissions, asymbiotic N fixation). A post-hoc Tukey test or Nemenyi test was used to determine significant groupings for data analyzed using parametric versus non-parametric statistics, respectively. Seasonal variation in inorganic N pools and nitrous oxide fluxes were determined using non-parametric t-test, Wilcoxon rank-sum. Statistical analyses were performed using R software v.3.0.2 (R Development Core Team).
References
Ribeiro, M. C., Metzger, J. P., Martensen, A. C., Ponzoni, F. J. & Hirota, M. M. The Brazilian Atlantic Forest: How much is left, and how is the remaining forest distributed? Implications for conservation. Biological Conservation 1–13, https://doi.org/10.1016/j.biocon.2009.02.021 (2009).
Pan, Y. et al. A Large and Persistent Carbon Sink in the World’s Forests. Science 333, 988–993 (2011).
Poorter, L. et al. Biomass resilience of Neotropical secondary forests. Nature 530, 211–214 (2016).
Wang, Y. P. & Houlton, B. Z. Nitrogen constraints on terrestrial carbon uptake: Implications for the global carbon-climate feedback. Geophys. Res. Lett. 36, L24403 (2009).
Nagy, R. C., Rastetter, E. B., Neill, C. & Porder, S. Nutrient limitation in tropical secondary forests following different management practices. Ecological Applications 27, 734–755 (2017).
Kauffman, J. B., Cummings, D. L., Ward, D. E. & Babbitt, R. Fire in the Brazilian Amazon: 1. Biomass, nutrient pools, and losses in slashed primary forests. Oecologia 104, 397–408 (1995).
Davidson, E. A. et al. Recuperation of nitrogen cycling in Amazonian forests following agricultural abandonment. Nature 447, 995–998 (2007).
Batterman, S. A. et al. Key role of symbiotic dinitrogen fixation in tropical forest secondary succession. Nature 502, 224–227, https://doi.org/10.1038/nature12525 (2013).
Vitousek, P. M. Litterfall, nutrient cycling, and nutrient limitation in tropical forests. Ecology 65, 285–298 (1984).
Sanchez, P. A. Properties and management of soils in the tropics. (Wiley, 1977).
Brookshire, E. N. J., Gerber, S., Menge, D. N. L. & Hedin, L. O. Large losses of inorganic nitrogen from tropical rainforests suggest a lack of nitrogen limitation. Ecology Letters 15, 9–16 (2011).
Hall, S. J. & Matson, P. A. Nitrogen oxide emissions after nitrogen additions in tropical forests. Nature 400, 152–155 (1999).
Holtgrieve, G., G., W., Jewett, P. K. & Matson, P. A. Variations in soil N cycling and trace gas emissions in wet tropical forests. Oecologia 146, 584–594 (2005).
Walker, T. W. & Syers, J. K. The fate of phosphorus during pedogenesis. Geoderma 15, 1–19 (1976).
Okin, G. S., Mahowald, N., Chadwick, O. A. & Artaxo, P. Impact of desert dust on the biogeochemistry of phosphorus in terrestrial ecosystems. Global Biogeochemical Cycles 18, https://doi.org/10.1029/2003GB002145 (2004).
Porder, S. & Hilley, G. E. Linking chronosequences with the rest of the world: predicting soil phosphorus content in denuding landscapes. Biogeochemistry 102, 153–166 (2010).
ter Steege, H. et al. Continental-scale patterns of canopy tree composition and function across Amazonia. Nature 443, 444–447 (2006).
Winbourne, J. B. et al. Iron controls over di-nitrogen fixation in karst tropical forest. Ecology 93, 773–781 (2017).
Reed, S. C., Cleveland, C. C. & Townsend, A. R. Functional Ecology of Free-Living Nitrogen Fixation: A Contemporary Perspective. Annu. Rev. Ecol. Evol. Syst. 42, 489–512 (2011).
Reed, S. C., Cleveland, C. C. & Townsend, A. R. Controls over leaf litter and soil nitrogen fixation in two lowland tropical rain forests. Biotropica 39, 585–592 (2007).
Sullivan, B. W., Smith, W. K. & Townsend, A. R. Spatially robust estimates of biological nitrogen (N) fixation imply substantial human alteration of the tropical N cycle. Proceedings of the National Academy of Sciences, https://doi.org/10.1073/pnas.1511978112 (2014).
Davidson, E. A. et al. Nitrogen and phosphorus limitation of biomass growth in a tropical secondary forest. Ecological Applications 14, 150–163 (2004).
Hedin, L. O., Brookshire, E. N. J., Menge, D. N. L. & Barron, A. R. The Nitrogen Paradox in Tropical Forest Ecosystems. Annu. Rev. Ecol. Evol. Syst. 40, 613–635 (2009).
Becknell, J. M. & Powers, J. S. Stand age and soils as drivers of plant functional traits and aboveground biomass in secondary tropical dry forest. Can. J. For. Res. 44, 604–613 (2014).
Asner, G. P. et al. Amazonian functional diversity from forest canopy chemical assembly. Proceedings of the National Academy of Sciences 111, 5604–5609 (2014).
Crouzeilles, R. et al. A global meta-analysis on the ecological drivers of forest restoration success. Nature Communications 7, 11666–8 (2016).
Thomas, W., Carvalho, A., Amorim, A., Garrison, J. & Arbelaez, A. Plant endemism in two forests in southern Bahia, Brazil. Biodiversity and Conservation 7, 311–322 (1998).
Melo, F. P. L. et al. Priority setting for scaling-up tropical forest restoration projects: Early lessons from the Atlantic Forest Restoration Pact. Environmental Science & Policy 33, 395–404 (2013).
Becknell, J. M. et al. Landscape scale LiDAR analysis of aboveground biomass distribution in secondary Brazilian Atlantic Forest. Biotropica. (Accepted).
Butterbach-Bahl, K., Baggs, E. M., Dannenmann, M., Kiese, R. & Zechmeister-Boltenstern, S. Nitrous oxide emissions from soils: how well do we understand the processes and their controls? Philosophical Transactions of the Royal Society B: Biological Sciences 368, 1–13 (2013).
Galloway, J. N. et al. Transformation of the Nitrogen Cycle: Recent Trends, Questions, and Potential Solutions. Science 320, 889–892 (2008).
Warembourg, F. R. Nitrogen fixation in soil and plant systems. In: R. Knowles, T. H. Blackburn (eds). Nitrogen isotope techniques. (Academic Press, San Diego, 1992).
Pearson, H. L. & Vitousek, P. M. Stand dynamics, nitrogen accumulation, and symbiotic nitrogen fixation in regenerating stands of Acacia koa. Ecological Applications 17, 1381–1394 (2001).
Siddique, I. et al. Dominance of legume trees alters nutrient relations in mixed species forest restoration plantings within seven years. Biogeochemistry 88, 89–101 (2008).
Rosenstock, T. S. et al. Agroforestry with N2-fixing trees: sustainable development’s friend or foe? Current Opinion in Environmental Sustainability 6, 15–21 (2014).
Schimel, J. P. & Bennett, J. Nitrogen mineralization: challenges of a changing paradigm. Ecology 85, 591–602 (2004).
Firestone, M. & Davidson, E. Microbiological basis of NO and N2O Production and Consumption in Soil. In: Andreac and Schimel (eds)Exchange of Trace Gases between Terrestrial Ecosystems and the Atmosphere p. 7–21 Wiley & Sons (1989).
Barton, L. et al. Sampling frequency affects estimates of annual nitrous oxide fluxes. Nature Scientific Reports. 5, 15912 (2015).
Osborne, B. et al. Climate, topography, and canopy chemistry exert hierarchical control over soil N cycling in a Neotropical lowland forest. Ecosystems 20, 1–15 (2016).
Taylor, P. et al. Landscape-Scale Controls on Aboveground Forest Carbon Stocks on the Osa Peninsula, Costa Rica. PLoS ONE 10, e0126748 (2015).
Soper, F. M. et al. Modest gaseous N losses support conservative N cycling in a lowland tropical rainforest watershed. Ecosystems. https://doi.org/10.1007/S10021-017-0193-1 (2017).
Balzotti, C. S. et al. Environmental controls on canopy foliar nitrogen distributions in a Neotropical lowland forest. Ecological Applications 26, 2451–2464 (2016).
Townsend, A. R., Asner, G. P. & Cleveland, C. C. The biogeochemical heterogeneity of tropical forests. Trends in Ecology & Evolution 23, 424–431 (2008).
Marin-Spiotta, E., Cusack, D., Ostertag, R. & Silver, W. L. In Chapter 2: Trends in Above and Belowground Carbon with Forest Regrowth After Agricultural Abandonment in the Neotropics 22–72 (Springer, 2007).
Piotto, D. Spatial dynamics of forest recovery after swidden cultivation in the Atlantic forest of southern Bahia, Brazil. PhD dissertation. (2011).
Piotto, D., Montagnini, F., Thomas, W., Ashton, M. & Oliver, C. Forest recovery after swidden cultivation across a 40-year chronosequence in the Atlantic forest of southern Bahia, Brazil. Plant Ecol. https://doi.org/10.1007/s1l258-009-9615-2 (2009).
Weintraub, S. et al. Topographic controls on soil nitrogen availability in a lowland tropical forest. Ecology 96, 1561–1574 (2015).
Qian, P. & Schoenau, J. Practical applications of ion exchange resins in agricultural and environmental soil research. Can. J. Soil Sci. 82, 9–21 (2002).
Holland et al. Soil CO2, N20, and CH4 exchange. In: G. P. Robertson et al. (eds). Standard Soil Methods for Long Term Ecological Research. (Oxford University Press, New York, 1999).
The National Institute of Space Research in Brazil (INPE) (http://clima1.cptec.inpe.br/) (2016).
Dahal, B. & Hastings, M. G. Technical considerations for the use of passive samplers to quantify the isotopic composition of NOx and NO2 using the denitrifier method. Atmospheric Environment 143, 60–66 (2016).
Barron, A. R., Purves, D. W. & Hedin, L. O. Facultative nitrogen fixation by canopy legumes in a lowland tropical forest. Oecologia 165, 511–520 (2010).
Acknowledgements
We would like to thank the staff at Instituto Floresta Viva for logistical assistance, in particular Jucelino Santos for field assistance. Benjamin Sullivan provided advice on symbiotic nitrogen fixation methods. B. Osborne, and J. Becknell provided valuable input on manuscript drafts. This work was funded by SEED grants from Brown University (both the Institute at Brown for Environment and Society, and the Office of the Vice President for Research), the Voss Postdoctoral and Undergraduate Fellows Program at Brown University, the Mellon Mays Undergraduate Fellows program, and an anonymous donor.
Author information
Authors and Affiliations
Contributions
J.B.W., S.P., D.P., and M.H. designed the study. J.B.W., A.F., and L.R. collected the data and performed the analyses. J.B.W. and S.P. led the writing, with contributions from all authors.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Winbourne, J.B., Feng, A., Reynolds, L. et al. Nitrogen cycling during secondary succession in Atlantic Forest of Bahia, Brazil. Sci Rep 8, 1377 (2018). https://doi.org/10.1038/s41598-018-19403-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-19403-0
This article is cited by
-
Soil physicochemical properties and microorganisms jointly regulate the variations of soil carbon and nitrogen cycles along vegetation restoration on the Loess Plateau, China
Plant and Soil (2024)
-
Biological nitrogen fixation in young and old tropical forests under five contrasting edaphoclimatic conditions
Nutrient Cycling in Agroecosystems (2024)
-
Conservative N cycling despite high atmospheric deposition in early successional African tropical lowland forests
Plant and Soil (2022)
-
Site and plant community parameters drive the effect of vegetation on litterfall and nutrient inputs in restored tropical forests
Plant and Soil (2021)
-
Nearby mature forest distance and regenerating forest age influence tree species composition in the Atlantic forest of Southern Bahia, Brazil
Biodiversity and Conservation (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.