Abstract
The purpose of this study was to compare the predictive ability of five obesity indices, including body mass index (BMI), waist circumference (WC), waist-to-height ratio (WHtR), waist-to-hip ratio (WHpR) and body adiposity index (BAI), to predict multiple non-adipose metabolic risk factors, including elevated blood pressure (BP), elevated fasting plasma glucose (FPG), elevated triglyceride (TG), reduced high-density lipoprotein cholesterol (HDL-C), elevated serum uric acid (SUA) and non-alcoholic fatty liver disease (NAFLD), in an elderly Chinese population. A total of 5685 elderly Chinese subjects (≥60 years) were recruited into our community-based cross-sectional study. Receiver operating characteristic curve (ROC) analyses were used to compare the predictive ability as well as determine the optimal cut-off values of the obesity indices for multiple metabolic risk factors. According to the areas under the receiver operating characteristic curve (AUC), BMI, WC and WHtR were able to similarly predict high metabolic risk in males (0.698 vs. 0.691 vs. 0.688), while in females, BMI and WC were able to similarly predict high metabolic risk (0.676 vs. 0.669). The optimal cut-off values of BMI, WC and WHtR in males were, respectively, 24.12 kg/m2, 83.5 cm and 0.51, while in females, the values were 23.53 kg/m2 and 77.5 cm.
Similar content being viewed by others
Introduction
Metabolic syndrome (MetS) is a complex disease spectrum with a clustering of metabolic and cardiovascular risk factors1. MetS has a high general prevalence of 33.9% among Chinese adults2. According to the diagnosis criteria proposed by the International Diabetes Federation (IDF) and the American Heart Association (AHA)/National Heart, Lung and Blood Institute (NHLBI) in 20093, obesity, elevated blood pressure (BP), elevated fasting plasma glucose (FPG), elevated triglyceride (TG) and reduced high-density lipoprotein cholesterol (HDL-C) are the main components of MetS. In addition to these factors, numerous studies support the hypothesis that elevated serum uric acid (SUA)4,5,6,7 and non-alcoholic fatty liver disease (NAFLD)8,9,10 are also risk factors for MetS. Uric acid is the end-product of purine metabolism in humans, and an altered SUA level is associated with glucose and lipid metabolism11,12. In numerous studies, hyperuricemia is considered to be a risk factor for MetS or metabolic disorders4,5,6,7. NAFLD is a clinicopathological condition characterized by abnormal lipid deposition in hepatocytes13 and has been suggested to be a hepatic manifestation of MetS14,15. Therefore, obesity, elevated BP, elevated FPG, elevated TG, reduced HDL-C, elevated SUA and NAFLD are considered to be the common and important metabolic risk factors for MetS in our study.
Obesity is already a serious public health burden worldwide16. Up to 50% of elderly Chinese have an abnormally high body mass index, causing a large disease-related healthcare burden17. Various obesity-related anthropometric indices, such as body mass index (BMI), waist circumference (WC), waist-to-height ratio (WHtR), waist-to-hip ratio (WHpR) and body adiposity index (BAI), have been used to predict metabolic risk factors in numerous studies18,19,20,21,22. BMI is a measurement of body fat based on height and weight23, while WC reflects abdominal or central obesity. WHtR and WHpR further reflect the fat distribution based on WC, and all three are considered to be specific alternatives to assess abdominal fat24. In contrast to all of the previous indicators, BAI is a recent indicator and is used to estimate the amount of body fat25,26.
The purpose of this study is to compare the predictive ability of these obesity indices for predicting multiple non-adipose metabolic risk factors in elderly Chinese subjects at or above 60 years of age. To further evaluate the importance of obesity factor in Chinese elderly people, we adopted the five commonly used obesity indices mentioned above as well as more comprehensive metabolic risk factors, including elevated BP, elevated FPG, elevated TG, reduced HDL-C, elevated SUA and NAFLD, in the present study.
Results
Baseline characteristics of study subjects
As shown in Table 1, there were no significant statistical differences by gender for age and BMI. In contrast, WC and WHpR were greater in males compared to females, while female subjects had greater WHtR and BAI compared to males (P value < 0.001). Among the multiple metabolic risk factors, the prevalence of elevated BP, elevated FPG and NAFLD did not differ by gender. The prevalence of elevated TG and reduced HDL-C was greater in females compared to males, while male subjects had an increased prevalence of elevated SUA compared to females (P value < 0.001).
Number of metabolic risk factors and obesity indices
Table 2 shows the association of metabolic risk factors and obesity indices. BMI, WC, WHtR, WHpR and BAI showed an increase in the number of metabolic risk factors in both genders. As shown in Table 2, 2653 elderly subjects (1115 men and 1538 women) had ≥3 metabolic risk factors. We estimated that approximately half of the elderly subjects (46.67% in total, 43.85% in males and 48.95% in females) in our study had high metabolic risk.
AUC of obesity indices for multiple metabolic risk factors
Table 3 shows the areas under the receiver operating characteristic curve (AUC) for BMI, WC, WHtR, WHpR and BAI for each multiple metabolic risk factors in both genders using receiver operating characteristic curve (ROC) analyses. BMI had the strongest predictive ability for elevated BP, elevated FPG and NAFLD in both genders; for the elevated SUA of males; and for the elevated TG of females. WC had the strongest predictive ability for reduced HDL-C of both genders as well as for the elevated TG of males. WHtR had the strongest predictive ability for the elevated SUA of females.
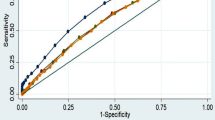
ROC analyses were applied to compare the predictive ability of obesity indices to predict the presence of a high metabolic risk population that had ≥3 non-adipose metabolic risk factors. Figure 1 shows the ROC curves for the five obesity indices to predict high metabolic risk in males and females. The AUC, optimal cut-off values, sensitivity, specificity and Youden index of the five obesity indices for predicting high metabolic risk are shown in Table 4. BMI had the strongest predictive ability for both genders. The statistical significance of the AUC differences of the five obesity indices suggested that the AUC of BMI, WC and WHtR did not differ in males and were all greater than WHpR and BAI; however, in females, the AUC of BMI and WC did not differ and were greater than for WHtR, WHpR and BAI. WHpR and BAI had a low predictive ability in both genders. The optimal cut-off values of BMI, WC and WHtR in males were 24.12 kg/m2, 83.5 cm and 0.51, respectively, while in females, the values were 23.53 kg/m2 and 77.5 cm.
Discussion
In this study, we found BMI, WC and WHtR to be more accurate than WHpR and BAI for predicting multiple metabolic risk factors; BMI, WC and WHtR were able to similarly predict the high metabolic risk population in males, while in females, BMI and WC were able to similarly predict the high metabolic risk population.
Obesity plays a central role in MetS and is closely associated with various metabolic risk factors. In addition to the five main components of MetS, we used elevated SUA and NAFLD as two additional metabolic risk factors in this study as there are numerous studies indicating that elevated SUA and NAFLD are risk factors for MetS and metabolic disorders4,5,6,7,8,9,10. To the best of our knowledge, this is one of the first comprehensive studies to evaluate the predictive ability of obesity indices with multiple metabolic risk factors and to focus on a Chinese elderly population. The strengths of our study include the sample size, the specificity of our population, and the additional use of elevated SUA and NAFLD risk factors.
The results showed that BMI and WC were more useful for predicting multiple metabolic risk factors, in line with Wang et al.’s research19. We also found that WHtR had the same predictive ability as BMI and WC for predicting the high metabolic risk population in males. A European study suggested that BMI, WC and WHtR were associated with clustered cardiometabolic risk factors and that the magnitudes of the associations were similar for BMI, WC and WHtR using mixed-effect regression and ROC analyses27. Liu et al. have found that the BMI, WC and WHtR values were able to equivalently predict the presence of multiple metabolic risk factors using ROC analyses in a general Chinese adult population21. Our results validated these findings in elderly males, while we found that BMI and WC had more significant predictive value than WHtR in elderly females. Moreover, we found that both WHpR and BAI in either gender was a much weaker predictor, supporting Yu et al.’s research28.
In the present study, the optimal cut-off values of the appropriate obesity indices were determined to predict the high metabolic risk population according to gender among the Chinese elderly. The BMI values for both genders were approximately 24 kg/m2, in line with Wang et al.’s research in north China19, and were higher than Liu et al.’s research, which was conducted in a general adult population in northeast China21 (22.85 and 23.30 kg/m2 in males and females). The WC values in both genders were lower than in Wang et al.’s results19 (88.05 and 87.10 cm in males and females aged 65–85 years), Liu et al.’s results21 (91.3 and 87.1 cm in males and females) and Guan et al.’s results conducted in Chinese rural adults18 (85 and 80 cm in males and females). The WHtR value in males was in line with Liu et al.’s results21 and was higher than that of Guan et al.’s results18 (0.50 in males). Wang et al.’s research suggested that the obesity indices for the prediction of MetS changed with age distribution19. Our study focuses on elderly Chinese people aged 60 years and above in Shanghai, and the differences in the results may be due to the age and regional distributional differences of the samples. As these obesity indices change with age and gender29, “one for all” cut-off values of obesity indices for all age groups are not appropriate to assess multiple metabolic risk factors.
There are, however, several limitations of this study. The subjects in this study were all from Shanghai, and the sample may not fully reflect all Chinese elderly. This study included quite a large number of Chinese elderly, but we only enrolled subjects who had completed the comprehensive health check study, which may have biased our primary findings. Further studies with multi centers and a larger sample size are needed to identify the association of obesity indices with more comprehensive metabolic risk factors.
Methods
Study subjects
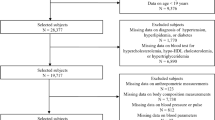
This study is a community-based cross-sectional investigation for the elderly population in Shanghai, China. A total of 5736 residents from Zhangjiang community of Shanghai aged ≥60 years were recruited into our study between April and July 2015. The study was performed according to the guidelines of the Helsinki Declaration. A standard protocol was designed by Shanghai innovation center of TCM health service and was approved by the Ethics Committee of Shanghai University of Traditional Chinese Medicine. Written informed consents were obtained from all subjects.
The inclusion criteria included age ≥ 60 years, local residents in Shanghai, complete data measurements and informed consents. Subjects with mental disorders, malignant tumors or incomplete recorded information were excluded from this project based on their medical records. After investigation, 51 subjects were excluded from the study. A total of 5685 Chinese elderly subjects (2543 males and 3142 females) with complete data were finally included in this study.
Data measurements
The anthropometric indices included WC, hip circumference (HC), height and weight. WC and HC were measured to the nearest 0.1 cm using a flexible metric measuring tape (Pudong CDC; Shanghai, China). Height and weight were measured to the nearest 0.1 cm and 0.1 kg using electronic measurement instrument (Shengyuan; Zhengzhou, China). All subjects were measured wearing light clothing without hats and shoes. BMI was calculated as bodyweight (kg)/height2 (m2). WHtR was calculated as WC (cm)/height (cm). WHpR was calculated as WC (cm)/HC (cm). BAI was calculated as HC (cm)/height1.5 (m) minus 1826.
Blood pressure was measured with electronic sphygmomanometers (Biospace; Cheonan, South Korea) using the standard recommended procedures. Blood samples were obtained from the antecubital vein in the morning after an overnight fasting period. FPG, TG, HDL-C and SUA were measured using an automatic biochemistry analyzer (Hitachi; Tokyo, Japan). The color ultrasound system (TOSHIBA; Tokyo, Japan) was used by two experienced radiologists to screen for fatty liver.
Criteria for multiple metabolic risk factors and the “high metabolic risk population”
According to the diagnosis criteria of MetS in 20093, the multiple non-adipose metabolic risk factors included the following items: (a) elevated BP: systolic blood pressure (SBP) ≥ 130 mmHg or diastolic blood pressure (DBP) ≥ 85 mmHg, or ongoing antihypertensive medications; (b) elevated FPG: FPG ≥ 5.6 mmol/L, or ongoing anti-diabetic treatment; (c) elevated TG: TG ≥ 1.7 mmol/L; and (d) reduced HDL-C: HDL-C < 1.04 mmol/L in males and HDL-C < 1.30 mmol/L in females. In addition to the regular MetS components, we also enrolled: (e) elevated SUA: SUA > 420 μmol/L according to the diagnosis criteria of hyperuricemia in 200030; (f) NAFLD: no history of heavy drinking, liver ultrasound imaging for diffuse fatty liver, and no drug-induced liver disease or viral hepatitis according to the diagnosis criteria of NAFLD in 201231. Subjects with three or more of the six non-adipose metabolic risk factors were defined as the “high metabolic risk population” in present study.
Statistical analyses
All of the descriptive statistics for all of the variables were calculated. Continuous variables were expressed as mean ± standard deviation and compared using two-sided t tests. Categorical variables were expressed as counts or percentages and compared using Pearson’s χ 2 tests. To compare the predictive ability and determine the optimal cut-off values of the obesity indices for predicting multiple metabolic risk factors, ROC analyses were used. AUCs were calculated, and the optimal cut-off values were identified from the maximum Youden index (sensitivity plus specificity-1) to determine the appropriate obesity indices.
P values < 0.05 were set as significant for all of the statistical tests for bilateral contrasts. All statistical analyses were conducted using SPSS version 17.0 (SPSS; Chicago, USA). The statistical significance of the differences in the AUCs was analyzed using MedCalc version 17.1.0 (MedCalc; Ostend, Belgium) with the algorithm developed by DeLong’s research team32.
References
Alberti, K. G., Zimmet, P. & Shaw, J. Metabolic syndrome–a new world-wide definition. A Consensus Statement from the International Diabetes Federation. Diabet Med 23, 469–480 (2006).
Lu, J. et al. Metabolic Syndrome Among Adults in China: The 2010 China Noncommunicable Disease Surveillance. J Clin Endocrinol Metab 102, 507–515 (2017).
Alberti, K. G. et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 120, 1640–1645 (2009).
Kuwabara, M. et al. Asymptomatic Hyperuricemia Without Comorbidities Predicts Cardiometabolic Diseases: Five-Year Japanese Cohort Study. Hypertension 69, 1036–1044 (2017).
Khichar, S. et al. Serum uric acid level as a determinant of the metabolic syndrome: A case control study. Diabetes Metab Syndr 11, 19–23 (2017).
Yang, T. et al. Uric acid level as a risk marker for metabolic syndrome: a Chinese cohort study. Atherosclerosis 220, 525–531 (2012).
Lin, W. Y. et al. In addition to insulin resistance and obesity, hyperuricemia is strongly associated with metabolic syndrome using different definitions in Chinese populations: a population-based study (Taichung Community Health Study). Ann Rheum Dis 67, 432–433 (2008).
Naderian, M. et al. Assessment of Lean Patients with Non-alcoholic Fatty Liver Disease in a Middle Income Country; Prevalence and Its Association with Metabolic Disorders: A Cross-sectional Study. Arch Iran Med 20, 211–217 (2017).
Lee, S. W. et al. The association of non-alcoholic fatty liver disease and metabolic syndrome in a Chinese population. Hepatobiliary Pancreat Dis Int 16, 176–180 (2017).
Caserta, C. A. et al. Association of non-alcoholic fatty liver disease and cardiometabolic risk factors with early atherosclerosis in an adult population in Southern Italy. Ann Ist Super Sanita 53, 77–81 (2017).
Chizynski, K. & Rozycka, M. [Hyperuricemia]. Pol Merkur Lekarski 19, 693–696 (2005).
Kanbay, M. et al. Uric acid in metabolic syndrome: From an innocent bystander to a central player. Eur J Intern Med 29, 3–8 (2016).
Brunt, E. M. Nonalcoholic steatohepatitis: definition and pathology. Semin Liver Dis 21, 3–16 (2001).
Gianotti, G. et al. Diastolic dysfunction and cardiovascular risk in old subjects: possible association with NAFLD? Arch Gerontol Geriatr 58, 188–195 (2014).
Dowman, J. K., Tomlinson, J. W. & Newsome, P. N. Systematic review: the diagnosis and staging of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis. Aliment Pharmacol Ther 33, 525–540 (2011).
Ng, M. et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 384, 766–781 (2014).
Zhang, M., Jiang, Y., Li, Y., Wang, L. & Zhao, W. [Prevalence of overweight and obesity among Chinese elderly aged 60 and above in 2010]. Zhonghua Liu Xing Bing Xue Za Zhi 35, 365–369 (2014).
Guan, X. et al. Associations between metabolic risk factors and body mass index, waist circumference, waist-to-height ratio and waist-to-hip ratio in a Chinese rural population. J Diabetes Investig 7, 601–606 (2016).
Wang, F. et al. Waist circumference, body mass index and waist to hip ratio for prediction of the metabolic syndrome in Chinese. Nutr Metab Cardiovasc Dis 19, 542–547 (2009).
Zhang, Z. Q. et al. Comparison of various anthropometric and body fat indices in identifying cardiometabolic disturbances in Chinese men and women. PLoS One 8, e70893 (2013).
Liu, Y., Tong, G., Tong, W., Lu, L. & Qin, X. Can body mass index, waist circumference, waist-hip ratio and waist-height ratio predict the presence of multiple metabolic risk factors in Chinese subjects? BMC Public Health 11, 35 (2011).
Guasch-Ferre, M. et al. Waist-to-height ratio and cardiovascular risk factors in elderly individuals at high cardiovascular risk. PLoS One 7, e43275 (2012).
Baumgartner, R. N., Heymsfield, S. B. & Roche, A. F. Human body composition and the epidemiology of chronic disease. Obes Res 3, 73–95 (1995).
Budimir, D. et al. Sex-specific association of anthropometric measures of body composition with arterial stiffness in a healthy population. Med Sci Monit 18, CR65–71 (2012).
Bennasar-Veny, M. et al. Body adiposity index and cardiovascular health risk factors in Caucasians: a comparison with the body mass index and others. PLoS One 8, e63999 (2013).
Bergman, R. N. et al. A better index of body adiposity. Obesity (Silver Spring) 19, 1083–1089 (2011).
Sardinha, L. B. et al. A Comparison between BMI, Waist Circumference, and Waist-To-Height Ratio for Identifying Cardio-Metabolic Risk in Children and Adolescents. PLoS One 11, e0149351 (2016).
Yu, J. et al. Optimal cut-off of obesity indices to predict cardiovascular disease risk factors and metabolic syndrome among adults in Northeast China. BMC Public Health 16, 1079 (2016).
Reas, D. L., Nygard, J. F., Svensson, E., Sorensen, T. & Sandanger, I. Changes in body mass index by age, gender, and socio-economic status among a cohort of Norwegian men and women (1990-2001). BMC Public Health 7, 269 (2007).
Fang, J. & Alderman, M. H. Serum uric acid and cardiovascular mortality the NHANES I epidemiologic follow-up study, 1971–1992. National Health and Nutrition Examination Survey. JAMA 283, 2404–2410 (2000).
Chalasani, N. et al. The diagnosis and management of non-alcoholic fatty liver disease: practice guideline by the American Gastroenterological Association, American Association for the Study of Liver Diseases, and American College of Gastroenterology. Gastroenterology 142, 1592–1609 (2012).
DeLong, E. R., DeLong, D. M. & Clarke-Pearson, D. L. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44, 837–845 (1988).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 81620108030, No. 81603411,No. 81573814) and Key project of Shanghai 3-year plan from Longhua Hospital (ZY3-CCCX-2-1002/ZY3-CCCX-3-4001), Shanghai, China.
Author information
Authors and Affiliations
Contributions
G.J., L.Z., B.C.L., D.L., and Z.G. designed research, B.C.L., Y.L.H., P.H.Z., J.Y.W., and X.J.H. performed experiments, Z.G. and H.Y.H. analyzed the data, Z.G. wrote the main manuscript text. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gu, Z., Li, D., He, H. et al. Body mass index, waist circumference, and waist-to-height ratio for prediction of multiple metabolic risk factors in Chinese elderly population. Sci Rep 8, 385 (2018). https://doi.org/10.1038/s41598-017-18854-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-18854-1
This article is cited by
-
Association of changes in waist circumference, waist-to-height ratio and weight-adjusted-waist index with multimorbidity among older Chinese adults: results from the Chinese longitudinal healthy longevity survey (CLHLS)
BMC Public Health (2024)
-
Independent and joint associations of body mass index, waist circumference, waist-height ratio and their changes with risks of hyperuricemia in middle-aged and older Chinese individuals: a population-based nationwide cohort study
Nutrition & Metabolism (2021)
-
Body roundness index is a superior indicator to associate with the cardio‐metabolic risk: evidence from a cross‐sectional study with 17,000 Eastern-China adults
BMC Cardiovascular Disorders (2021)
-
Factors associated with glycemic control in community-dwelling elderly individuals with type 2 diabetes mellitus in Zhejiang, China: a cross-sectional study
BMC Endocrine Disorders (2019)
-
The role of frailty in predicting mortality and readmission in older adults in acute care wards: a prospective study
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.