Abstract
The human pathogen L. monocytogenes and the animal pathogen L. ivanovii, together with four other species isolated from symptom-free animals, form the “Listeria sensu stricto” clade. The members of the second clade, “Listeria sensu lato”, are believed to be solely environmental bacteria without the ability to colonize mammalian hosts. To identify novel determinants that contribute to infection by L. monocytogenes, the causative agent of the foodborne disease listeriosis, we performed a genome comparison of the two clades and found 151 candidate genes that are conserved in the Listeria sensu stricto species. Two factors were investigated further in vitro and in vivo. A mutant lacking an ATP-binding cassette transporter exhibited defective adhesion and invasion of human Caco-2 cells. Using a mouse model of foodborne L. monocytogenes infection, a reduced number of the mutant strain compared to the parental strain was observed in the small intestine and the liver. Another mutant with a defective 1,2-propanediol degradation pathway showed reduced persistence in the stool of infected mice, suggesting a role of 1,2-propanediol as a carbon and energy source of listeriae during infection. These findings reveal the relevance of novel factors for the colonization process of L. monocytogenes.
Similar content being viewed by others
Introduction
Listeria monocytogenes is a Gram-positive, facultative anaerobic, non-sporulating, rod-shaped bacterium1. The genus Listeria belongs to the phylum of Firmicutes, which is composed of Gram-positive bacteria with low GC-content (36–42%) and also includes the genera Bacillus, Clostridium, Enterococcus, Streptococcus, and Staphylococcus 2,3. L. monocytogenes is ubiquitous in nature and has been isolated from a variety of ecological niches, such as soil, vegetation, water, and feces4. It is a saprophyte that can live on decaying plant material. This prevalence in the environment is promoted by its ability to adapt to salt, acid, and temperature stresses. It can tolerate wide ranges of pH (pH 4.5-9.0) and temperature (0 °C-45 °C) and high concentrations of salt (up to 10% NaCl)5. In addition, it can form biofilms6. The persistence of certain L. monocytogenes strains in processing equipment poses a major challenge for the food industry7.
L. monocytogenes is the causative agent of the foodborne disease human listeriosis. Consumption of contaminated raw and industrially processed foods such as milk and other dairy products, meat products, vegetables, seafood, and ready-to-eat food is the main cause of infection1. Clinical symptoms include gastroenteritis, meningitis, meningoencephalitis, septicemia and prenatal infection8. Listeriosis has a high mortality rate of up to 20–30% regardless of early antibiotic treatment, and infants, elderly and immunocompromised individuals are the main risk groups9. Upon ingestion, the pathogen encounters a variety of stressful conditions during the gastrointestinal passage, including acidic and osmotic stresses. It is believed that these physiological stresses serve as a signal for priming of the cell for invasion and an intracellular lifestyle10,11. For example, acidic and osmotic stresses during passage through the stomach and small intestine trigger the expression of sigma factor B (σB), which induces several stress response- and virulence-associated genes12,13. σB works synergistically with the positive regulatory factor A (PrfA), a thermo-regulated transcription factor active at 37 °C (the body temperature of the host) as well as at ambient temperatures upon induction by low pH14; this transcription factor controls expression of the main virulence genes15.
The genus Listeria currently consists of 17 species. The only other pathogenic member besides L. monocytogenes is L. ivanovii 16, an organism that rarely infects humans17,18, but frequently causes listeriosis in ruminants19,20. Together with L. marthii 21, L. innocua, L. welshimeri, and L. seeligeri, these two species form the “Listeria sensu stricto” group22, one of two distinct clades in the genus Listeria. All members of this clade (clade I) have been found in feces or the gastrointestinal tract of symptom-free animals, as well as in food of animal origin23,24,25,26,27, suggesting a specific interaction of these species with mammalian hosts. Clade II, the “Listeria sensu lato” group, contains the species L. fleischmanni 28, L. weihenstephanensis 29, L. rocourtiae 30, L. aquatica, L. cornellensis, L. riparia, L. floridensis, L. grandensis 31, L. grayi, L. newyorkensis, and L. booriae 32, which have been isolated from food-associated surfaces or the environment.
While the systemic phase of L. monocytogenes infection and the factors involved are well characterized, colonization of the gastrointestinal tract is still underinvestigated. Given that all sensu stricto strains have been found in the gut, but only two strains have been associated with human disease, we postulated that there may be bacterial genes encoded within clade I that promote growth or survival in the mammalian gastrointestinal tract. To identify novel listerial genes that could be involved in adhesion to the mucosal epithelium, metabolism, or chemotaxis and motility, we compared the genomes of the Listeria sensu stricto-group with the genomes of the environmental strains belonging to the Listeria sensu lato group. A total of 151 gene products were identified as being encoded by all sensu stricto strains, but absent from the strains of the sensu lato group. These factors that possibly contribute to the interaction of listeriae with mammals include the flagellum, the metabolic capabilities to utilize ethanolamine and 1,2-propanediol (1,2-PD), and a set of mainly functionally unknown proteins involved in regulation and transport processes. A putative transporter and the 1,2-PD degradation pathway were tested here for their function and their role during infection by L. monocytogenes.
Results
Listeria phylogenetic groups exhibit differences in colonization ability after oral infection in female BALB/c mice
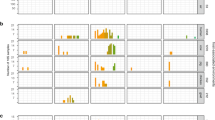
To analyze the proliferation of different species from the Listeria sensu stricto and Listeria sensu lato groups in the gastrointestinal tract and in other organs, female BALB/c mice were orally infected with 4–9 × 108 cell forming units (cfu) of the Listeria sensu stricto species L. monocytogenes and L. welshimeri, as well as the Listeria sensu lato species L. aquatica and L. booriae. The number of luminal Listeria in the ileum and colon was determined 2 days post infection (p.i.) (Fig. 1a and b). There were significantly lower cfu numbers of the three non-pathogenic species in both compartments in comparison with the cfu numbers of L. monocytogenes strain EGDe. The cfu values of L. aquatica and L. welshimeri were similar in the ileum. L. booriae was barely detectable in both compartments and showed significantly lower mean values in comparison to L. welshimeri in the colon, but not in the ileum.
Members of the two listerial phylogenetic groups exhibit different cell numbers in the ileum and colon lumen. Female BALB/c mice were orally infected with 4–9 × 108 cfu of the Listeria sensu stricto species L. monocytogenes (⚫) and L. welshimeri (■), as well as of the Listeria sensu lato species L. aquatica (▲) and L. booriae (▼). After 2 days, cfu numbers for each species were determined in the ileum (a) and colon lumen (b), as well as in the ileum (c) and colon tissue (d). Stool samples (e) were collected at 3 h p.i., 1 day p.i. and 2 days p.i., and the number of cfu per mg feces was calculated. Symbols represent values for individual mice, while horizontal lines indicate the mean value that was pooled from two separate experiments (n = 3 mice per group). Dashed lines represent the detection limit for each sample. Statistical significance was assessed using two-tailed student’s t-test with Welch’s correction.
The number of tissue-associated bacteria, that is, in the mucus, the epithelial layer, or the lamina propria (LP) of the gastrointestinal tract, was also analyzed (Fig. 1c and d). While L. monocytogenes was found in high numbers of 2.5-6.9 × 103 cfu, the three other species were not detectable in these tissues, confirming their non-pathogenic behavior as reported previously for L. welshimeri in mice after intravenous injection33. As expected, L. monocytogenes could also reach the mesenteric lymph nodes and spread to the liver and spleen of infected mice (Supplementary Fig. 1).
The cfu/mg feces from stool samples taken 3 h p.i., 1 day p.i., and 2 days p.i. of the same mice were additionally analyzed. No significant difference in the cfu/mg feces was observed either immediately after infection or within 24 h of infection. In all cases, the number of cfu decreased 1000-fold, suggesting a species-independent initial clearance of listeriae shed in the feces. After an additional 24 h, L. monocytogenes persisted in stool pellets, but the number of cfu decreased significantly for the three non-pathogenic species (Fig. 1e). This effect was more pronounced in the Listeria sensu lato species. For example, for half of the mice fed L. aquatica, no cfu were detected, and only 1 of 6 mice fed L. booriae were still shedding Listeria.
Genome comparison between Listeria sensu stricto and Listeria sensu lato species reveals candidate genes for infection
The findings described above prompted us to identify as yet unknown genes associated with gut colonization by a genome sequence comparison of 16 out of the 17 known Listeria species type strains (all except L. grayi). For this purpose, we calculated the percentage of predicted amino acid sequence identity for each L. monocytogenes protein against the proteins of the other 15 species within both clades. Gene products with at least 70% sequence homology amongst the Listeria sensu stricto species were chosen for further analysis, and filtered against gene products with less than 30% sequence homology amongst the Listeria sensu lato species. Although homology of >40% is suggestive of functional homologs34, we chose the more stringent threshold to focus on the most robust candidate genes. The resulting Table 1 comprises 151 gene products that are present in all clade I species and absent (or significantly divergent) in clade II species. A majority of the gene products are involved in flagellar biosynthesis and the cobalamin (vitamin B12)-dependent utilization of ethanolamine and 1,2-PD. The gain of these clusters has already been described as an evolutionary event that contributed to the taxonomic definition of clade I35. Motility is well known to contribute to successful infection by L. monocytogenes 36,37,38,39,40. Many candidate genes are involved in cell wall and membrane biogenesis, transport processes, transcription, and signaling (Table 1). There is experimental evidence that many of the genes specific for listeriae of clade I contribute to virulence properties, thus validating our approach: Two of the factors involved in peptidoglycan biosynthesis, Lmo0703 and Lmo0717, have been demonstrated to be controlled by the listerial virulence regulators DegU41 and MogR37. Among the transporters, we listed SvpA (Lmo2185) and Lmo2186 with putative heme transport capacity. SpvA is a listerial virulence factor that promotes escape from phagosomes of macrophages42 and/or enables crossing of the digestive barrier43. The ATPase synthase encoded by lmo0090-lmo0093 is known to play a role in intracellular survival44, and deletion of the gene pdeD contributes to a decrease in listerial invasiveness in enterocytes45. Because of their predicted, but not experimentally evaluated function in vitro or in vivo, we chose the putative ATP-binding cassette (ABC) transporter Lmo1131-1132 and the 1,2-PD utilization genes from Table 1 for further functional and infection experiments.
The operon lmo1131-1132 encoding a putative ABC transporter facilitates adhesion and invasion of L. monocytogenes in Caco-2 cells
According to the Basic local alignment search tool on the NCBI homepage, the operon lmo1131-1132 encodes a putative ABC transporter. Both proteins Lmo1131 and Lmo1132 contain an ABC transporter-like transmembrane domain as well as a nucleotide binding domain. For further analysis of the functional role of the operon lmo1131-1132, we constructed the in-frame deletion mutant L. monocytogenes EGDe Δlmo1131-1132 and tested adhesive and invasive capabilities in cell culture assays using Caco-2 (human colon carcinoma) cells and HEp-2 (human larynx squamous cell carcinoma) cells. Adhesion to Caco-2 cells was reduced in the deletion mutant compared with that of the parental strain EGDe by a factor of 2 (Fig. 2a). In contrast, cell culture assays with HEp-2 cells revealed no significant adhesion differences between EGDe and EGDe Δlmo1131-1132 (Fig. 2b), suggesting a specific role of the ABC transporter in the interaction with Caco-2 cells. We also performed a gentamycin protection assay to test the invasion and the intracellular replication properties of the deletion mutant in Caco-2 and HEp-2 cells. Given that the number of intracellular cells detected 1 h after cell culture infection is a measure of the capacity of a strain to enter cells, we observed a more than 20-fold reduction of invasion of Caco-2 cells, but not of HEp-2 cells, by EGDe Δlmo1131-1132 compared with that of the parental strain (Fig. 2c and d). The intracellular replication rates of EGDe [doubling time Td (EGDe) = 83 min] and EGDe Δlmo1131-1132 [Td (EGDe Δlmo1131-1132) = 83 min] however, were identical in the Caco-2 cells, indicating a specific role of the putative transporter in adhesion and/or invasion of this cell type.
Adhesion to and invasion of Caco-2 and HEp-2 cells by a transporter mutant. The percentage of adherent cells of L. monocytogenes EGDe and EGDe Δlmo1131-1132 to approximately 2.5 × 105 eukaryotic Caco-2 cells (a) or HEp-2 cells (b) was analyzed for a MOI of 10. The number of adherent EGDe cells was set to 100%. (c,d) The percentage of invasive and replicating cells of L. monocytogenes EGDe and EGDe Δlmo1113-1132 was determined. Approximately 2.5 × 105 eukaryotic Caco-2 or HEp-2 cells were infected with a MOI of 10, and the numbers of intracellular bacterial cells were determined 1, 3.5 and 7 h p.i. Error bars indicate the standard deviation of three biologically independent experiments including technical replicates. Statistical significance was assessed using two-tailed student’s t-test with Welch’s correction; NS, not significant.
Transcription of lmo1131-1132 shows oxygen- and temperature-dependent regulation
We then examined the transcription of lmo1131 and lmo1132 in L. monocytogenes EGDe at 24 °C and 37 °C under both aerobic and anaerobic growth conditions via quantitative real time (qRT)-polymerase chain reaction (PCR) (Fig. 3). The two transporter genes exhibited similar transcription levels under all four conditions, suggesting transcription of the operon from a common promoter located upstream of lmo1131. Setting the transcriptional level of both genes at 37 °C with oxygen as 100%, we observed a decrease of the lmo1131 and lmo1132 mRNA levels at 37 °C to 4.77% and 6.15%, respectively, upon oxygen depletion. At 24 °C, the transcription of the transporter genes was reduced to 55.80% and 64.02%, respectively, under aerobic conditions and to 3.18% and 4.53% in the absence of oxygen.
Temperature- and oxygen-dependent transcription of lmo1131 and lmo1132. Relative transcription of genes lmo1131 (black bars) and lmo1132 (gray bars) for the conditions 37 °C anaerobic, 24 °C aerobic, and 24 °C anaerobic was compared to that at 37 °C aerobic (preassigned as 100% gene expression for lmo1131). The results were calculated using the 2−ΔΔCT method95 and lmo1759 (pcrA) was used as a reference gene for normalization. Error bars indicate the standard error of three biologically independent experiments including technical duplicates for each condition.
Lack of lmo1131-1132 leads to attenuated colonization of BALB/c mice
To verify the relevance of the candidate genes for the in vivo infection process, a food-borne transmission model in mice was used to enable study of the gastrointestinal phase of listeriosis46. We conducted co-infection experiments with female BALB/c mice aged 6-8 weeks using a 1:1 ratio of EGDe and its mutant EGDe Δlmo1131-1132 lacking the putative transporter genes. The strains were chromosomally tagged with pIMC3kan (EGDe) and pIMC3ery (mutant). Mice were fed a total of 1 × 109 cfu/mouse and the number of Listeria in the lumen of either the ileum or the colon was analyzed 2 days p.i. (Fig. 4a and b). The colon contained 25-fold more L. monocytogenes than the ileum, as was previously reported by others46,47. However, there was no significant difference in the ratio of the two strains recovered from the intestinal contents of either tissue.
Deletion of lmo1131-1132 leads to attenuated colonization of BALB/c mice. Female BALB/c mice were orally infected with a 1:1 ratio of L. monocytogenes EGDe and EGDe Δlmo1131-1132 for a total inoculum of 1 × 109 cfu. After 2 days, the cfu of luminal (a) or cell-associated (c) strains in the ileum and colon were determined using kanamycin- (EGDe) or erythromycin (mutant)-containing plates, and the CI was calculated (b,d). (e) The cfu numbers of both strains in the spleen, liver and MLNs are shown. The numbers of EGDe (circles) or EGDe Δlmo1131-1132 (squares) recovered in each mouse after co-infection are depicted. Solid horizontal lines indicate mean values, which were pooled from at least two separate experiments, while dashed lines represent the detection limit for each sample. (f) The CIs of the strains recovered from the organs are depicted. The geometric mean for each group was compared to the theoretical value of 1.0, and the fold change difference is indicated in parentheses. Statistical significance was assessed using two-tailed student’s t-test with Welch’s correction.
We then assessed the numbers of cfu associated with the flushed tissue, which includes L. monocytogenes trapped in the mucus layer, bacteria adhered to or inside intestinal epithelial cells (ECs) and bacteria that have reached the underlying LP. In both the ileum and the colon, a bimodal distribution was observed, with little or no L. monocytogenes recovered in approximately half of the mice, and up to 104 cfu in the other half (Fig. 4c). In the ileum, the average number of cfu for the latter group was six-fold higher for wildtype EGDe compared with the mutant. Overall, the competitive index (CI) revealed that the wild type strain outcompeted the mutant strain six-fold in the ileum (Fig. 4d). Interestingly, there was no significant difference between EGDe and EGDe Δlmo1131-1132 in the colon.
To further investigate this difference in intestinal colonization, the gut tissues of infected mice were fractionated, and the number of cfu in either the mucus layer, ECs or the LP were determined (Supplementary Fig. 2a). The number of wild type and EGDe Δlmo1131-1132 in the mucus and the epithelial layer of the ileum did not differ significantly. However, wild type L. monocytogenes outnumbered the deletion mutant by five-fold in underlying LP of the ileum, similar to what was observed for whole tissue. As expected, there were no differences in the competitive indexes for any of the colon fractions. To assess the systemic spread of both strains, we also determined the bacterial load in the mesenteric lymph nodes (MLN), spleen and liver 2 days p.i. As shown in Fig. 4e and f, five-fold fewer EGDe Δlmo1131-1132 were recovered from the liver compared to the wild type strain. There was no significant difference between the two strains in the MLN or the spleen. To test whether dissemination of the transporter mutant to peripheral organs was impaired or if the mutant strain had a specific growth defect in the liver, mice were intravenously injected with a 1:1 mixture of EGDe and EGDe Δlmo1131-1132, and the total cfu in liver and spleen were determined 2 days p.i. Approximately equal numbers of both strains were recovered from the liver when the transmission route bypassed the gut (Supplementary Fig. 2b). Together, these data suggest that the transporter Lmo1131-1132 is involved in efficient translocation across the intestinal mucosa in the small intestine, but not the large intestine.
The pduD gene of L. monocytogenes is required for growth with 1,2-PD
Three large gene clusters responsible for the cobalamin-dependent utilization of ethanolamine and 1,2-PD were identified to be specific to the clade sensu stricto species of the genus Listeria 22 (Table 1). These pathways have been demonstrated to play a role in the proliferation of S. Typhimurium in vitro and in vivo 48,49,50, whereas no such experimental data have been available for L. monocytogenes. To investigate the role of 1,2-PD as a potential carbon and energy source for L. monocytogenes, strain EGDe and its mutant EGDe ΔpduD, which lacks a gene essential for 1,2-PD utilization, were grown in minimal medium51 (MM) without glucose, but with 0.5% (w/v) yeast extract under anaerobic conditions at 37 °C. No significant differences in the growth behavior of the two strains were observed. However, when we added 1,2-PD and cobalamin, which is an essential cofactor of PduCDE and whose biosynthesis is encoded by the cob/cbi gene cluster, EGDe grew to an optical density (OD)600 max = 0.35, whereas the cell density of EGDe ΔpduD did not exceed that in the medium without 1,2-PD (Fig. 5a). Therefore, it might be speculated that all nutrients preferred over 1,2-PD are used up during the first 4 h, and that L. monocytogenes only then degrades 1,2-PD to reach a higher cell density.
Improved growth of L. monocytogenes by addition of 1,2-PD. Growth curves of L. monocytogenes EGDe (□) and L. monocytogenes EGDe ΔpduD (○) in MM and 0.5% (w/v) yeast extract without glucose (a) or with 50 mM glucose (b) cultivated at 37 °C under anaerobic conditions. MM was supplemented with 10 mM 1,2-PD and 25 nM cobalamin (filled symbols) or not supplemented (open symbols). OD600 was measured at the indicated intervals using Bioscreen C. Growth curves depict the calculated mean value of three independent biological experiments with technical duplicates, while error bars indicate the standard deviation.
As a control, we compared the growth of both strains in MM containing 50 mM glucose in the absence and presence of 1,2-PD and cobalamin. Under these growth conditions with glucose as an energy source, the addition of 1,2-PD had no significant effect on the cell density of either EGDe or EGDe ΔpduD (Fig. 5b), suggesting that 1,2-PD is utilized by L. monocytogenes only in the absence of glucose.
Supplementation with 1,2-PD and/or cobalamin leads to induction of pdu and cob/cbi genes during the stationary phase
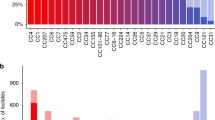
To investigate the transcriptional response of the pdu and the cob/cbi gene clusters to the presence of 1,2-PD and/or cobalamin, a global transcriptome analysis of L. monocytogenes EGDe was conducted via next-generation sequencing (NGS). During growth in brain heart infusion (BHI) medium, in BHI medium with 10 mM 1,2-PD, or in BHI medium with 10 mM 1,2-PD and 25 nM cobalamin, cells were harvested in the mid-logarithmic (exponential) and stationary phases. During the exponential phase, the pdu and the cob/cbi gene clusters showed only weak or no transcription under all three growth conditions. During the stationary phase, transcription of these genes only slightly increased in BHI medium, although levels were still very low (Supplementary Table S1). However, when 1,2-PD was added to the cultures, a strong increase of expression of most pdu and cob/cbi genes in the range from two-fold to 1,605-fold induction was observed (Fig. 6). The transcription of pdu genes was even more elevated (up to more than 10,942-fold) when cobalamin was added, and the presence of this cofactor in the medium resulted in repression of the genes responsible for its biosynthesis (Supplementary Table S1), a finding that confirms a regulatory model in which the presence of cobalamin represses its own synthesis post-transcriptionally via stabilization of an mRNA hairpin52. The transcription levels of pocR, the regulator of both clusters, were only slightly elevated under medium supplemented with 1,2-PD and/or cobalamin. The eut genes of the ethanolamine degradation cluster, which are positioned between the pdu and cbi/cob clusters, were not significantly induced under the growth conditions applied (Supplementary Table S1).
Transcriptome in response to growth with 1,2-PD. L. monocytogenes EGDe grown under different conditions was harvested during the exponential and stationary phases and the mRNA expression profile was analyzed via NGS using the Illumina MiSeq seqencing platform. Fold changes of normalized RPKM (reads per kilobase per million mapped reads) values under the conditions BHI with 10 mM 1,2-PD (BHI + 1,2-PD) and BHI with 10 mM 1,2-PD and 25 nM cobalamin (BHI + 1,2-PD + B12) in comparison to BHI were calculated for both growth phases. The results were visualized using a three-color scheme with red colors indicating negative and blue colors indicating positive fold changes; white means no change. The color intensity corresponds to the magnitude of fold change. Unknown genes refer to genes located in the region of propanediol, ethanolamine, and cobalamin clusters without any known function.
The transcriptome data were succesfully validated via qRT-PCR for the selected genes lmo1146, pocR (lmo1150), pduC (lmo1153), lmo1190 and cbiA (lmo1199) in stationary phase (Table 2). In exponential phase, there is a discrepancy between qRT-PCR and NGS results due to the higher sensitivity of qRT-PCR and the overall low number of RPKM values from the NGS (Suppl. Table S1). The results of both the transcriptome analysis and the qRT-PCR are compatible with the observation that the addition of 1,2-PD and cobalamin to L. monocytogenes EGDe in MM led to a growth advantage after exponential phase when other nutrients had been used up (Fig. 5a).
Deletion of pduD leads to faster clearance in BALB/c mice
We hypothesized that the ability to metabolize 1,2-PD would enhance the ability of L. monocytogenes to compete with gut microbiota and to persist in the intestinal lumen following oral transmission. To test this, female BALB/c mice were co-infected with a 1:1 ratio of wild type EGDe and an isogenic ΔpduD mutant (total of 1 × 109 cfu/mouse) that were differentially tagged with antibiotic resistances. Stools were collected daily for up to 14 days p.i., and the total number of strain per mg of feces was determined. As expected, a significant portion of the inoculum was shed in feces within 3 hours after feeding the mice, and the number of cfu detected 21 hours later had decreased considerably (Fig. 7a). At each time point after that, the average number of ΔpduD mutant bacteria recovered was less than the wildtype strain. By 10 days post-infection, the ΔpduD mutant had been cleared, but wildtype bacteria continued to be shed in the feces in at least some of the mice for up to 4 more days. Accordingly, the competitive index decreased steadily over the course of the experiment with a maximum difference on day 8 when the wild type bacteria outcompeted the mutant bacteria by nearly 40-fold (Fig. 7b). Thus, the presence of the genes encoding the 1,2-PD utilization pathway prolonged the survival of L. monocytogenes EGDe in the mouse gastrointestinal tract.
Deletion of pduD leads to faster clearance in BALB/c mice. Female BALB/c mice were orally infected with a 1:1 ratio of L. monocytogenes EGDe and EGDe ΔpduD for a total inoculum of 1 × 109 cfu. Stool samples were collected 3 h p.i. as well as every 24 h up to 16 days and the mean value of cfu per mg feces was calculated (a). Symbols represent mean values from two separate experiments (n = 4 mice per group) for L. monocytogenes EGDe (circles) and EGDe ΔpduD (squares) and dashed line represents the limit of detection. (b) CIs depict the ratio of EGDe ΔpduD/EGDe. The geometric mean for each group was compared to the theoretical value of 1.0 and the fold change difference is indicated in parentheses.
Discussion
It is believed that a common ancestor of the Listeria sensu stricto group took up a set of virulence genes through horizontal gene transfer, and that these determinants were subsequently lost in the non-pathogenic species of this listerial clade I53,54. Other putative evolutionary processes differentiating Listeria sensu stricto from Listeria sensu lato are the expansion of internalin genes and the acquisition of the metabolically relevant eut/pdu/cbi/cob gene cluster as well as of flagellar genes, which were probably taken up via horizontal gene transfer from an ancestor of the Bacillus cereus complex22. The association of Listeria sensu stricto with fecal samples or the gastrointestinal tract of mostly symptom-free animals or food from animal origins23,24,25,26,27, a reduced colonization rate of the environmental species L. fleischmannii and L. floridensis compared to Listeria sensu stricto species in fecal samples from wild rodents55, as well as the alleviated colonization abilities of the Listeria sensu lato species in our BALB/c infection experiments, prompted us to speculate that a genome comparison of clade I versus clade II strains might identify as yet unknown listerial factors that play a role particularly during the gastrointestinal phase of infection.
Mice and other animal model organisms are considerably more resistant to oral infections than humans, and larger inocula need to be administered (109–1011 cfu/animal) to cause an intestinal infection56. A high species specificity of the surface proteins InlA, which interacts well with ECs of humans or guinea pigs, but not with ECs of mice57,58, contributes to inefficient oral transmission. Recent studies also show that competition with gut microbiota may limit opportunities for L. monocytogenes to invade the gut mucosa47,59. To investigate the relevance of as yet unknown listerial determinants for the gastrointestinal phase of listeriosis in more detail, we used a recently established and highly reproducible feeding model to mimic the natural course of infection46 that overcomes the lack of reproducibility encountered by the conventional administration of listeriae by oral gavage.
Gastrointestinal infection is a multifactorial process and, besides the already mentioned stress conditions, metabolism is one of the key factors of pathogenic bacteria that can only survive in the intestine by searching for a specific niche providing sufficient amounts of nutrients60. A hallmark of pathogens is therefore their metabolic flexibility during infection. Examples of this for L. monocytogenes include the utilization of sugar phosphates and glycerol as alternative carbon sources during intracellular replication61,62,63, the requirement of both thiamine uptake and biosynthesis of thiamine precursors64, or the possible exploitation of uncommon nitrogen sources such as ethanolamine or glucosamine65. L. monocytogenes possesses a 53-kb island harboring genes for the metabolism of 1,2-PD, ethanolamine, and cobalamin, which can be found in all Listeria sensu stricto species22. These compounds can be found in the gastrointestinal tracts of animals and have been shown to play an important role in Salmonella pathogenesis49,66,67. It has been discussed whether these gene clusters are also relevant for the virulence of listeriae, particularly for colonization54. So far, it has been shown that ethanolamine utilization contributes to intracellular replication in ECs62, and that 1,2-PD is metabolized by L. innocua 68. Transcriptional studies in gnotobiotic mice infected with L. monocytogenes showed upregulation of most pdu genes during infection compared to growth in BHI69. Our study not only demonstrates that L. monocytogenes can use 1,2-PD as a nutrient but also provides strong experimental evidence that the ability of L. monocytogenes to catabolize 1,2-PD contributes to its persistence and proliferation during gastrointestinal infection. This is in line with the finding that S. Typhimurium expansion is driven by 1,2-PD and that its utilization requires not only intestinal inflammation, but also the presence of commensal bacteria50. These are assumed to provide 1,2-PD, for example, by the fermentation of fucose that has been cleaved from the mucosal glycans by commensal bacteria70,71.
Toledo-Arana and colleagues showed that lmo1131 but not lmo1132 expression is decreased in intestinal L. monocytogenes of gnotobiotic mice compared with that under BHI growth conditions69. Their reduced transcription at 24 °C and under anaerobic conditions suggests a role in an early stage of infection. A typical prokaryotic ABC transporter is composed of two hydrophobic transmembrane domains (TMDs) and two water soluble nucleotide binding domains (NBDs) at the cytosolic side of the cell membrane. An ABC transporter can be composed of four separate polypeptides, or two identical NBDs and/or TMDs can be present. NBDs and TMDs can also be fused together thus making up either the complete transporter from a single polypeptide or from two homo- or heterodimeric polypeptides72. Lmo1131 and Lmo1132 seem to belong to the group of “half transporters”, with two heterodimeric halves, each containing one NBD and one TMD. In addition to import functions, ABC transporters are involved in sorting molecules to the outer membrane. These molecules include lipoproteins, polysaccharides or fimbriae that might contribute to the interaction with mammalian cells73,74. Examples from human pathogens are the export of a coat protein from EAEC contributing to bacterial dispersion, the polysaccharide-dependent adhesion of Kingella kingae to human ECs, and the fimbriae-mediated adhesion of Streptococcus parasanguis 75,76,77. Another example is protein F as part of an ABC transporter from Haemophilus influenzae whose N-terminus promotes binding to ECs78.
To conclude, non-pathogenic Listeria species have been investigated here for the first time in a mouse model for oral infection, and the results suggest differences in the colonization ability of Listeria sensu stricto and Listeria sensu lato members. Novel Listeria sensu stricto-specific factors involved in the colonization of the gastrointestinal tract were identified, whereas as the Listeria sensu lato species seem to be less adapted to the conditions of the gastrointestinal tract. Because the non-pathogenic Listeria species of the sensu stricto group including L. welshimeri are equipped with these determinants, their potential to occupy niches of the pathogenic species has been underestimated.
Methods
Bacterial strains, plasmids, cell lines and growth conditions
The bacterial strains, cell lines, and plasmids used in this study are listed in Supplementary Table S2. E. coli was grown in Luria-Bertani broth, while L. monocytogenes EGDe was cultivated in BHI or MM51 at 37 °C or 24 °C. If appropriate, the media were supplemented with the following antibiotics: erythromycin (300 μg/ml for E. coli or 10 μg/ml for L. monocytogenes), kanamycin (50 µg/ml), and chloramphenicol (10 µg/ml). For solid media, 1.5% agar (w/v) was added. Human colon ECs (Caco-2 cells, ATCC HTB-37) and human larynx squamous cell carcinoma cells (HEp-2 cells, ATCC CCL-23) were received from the American Type Culture Collection and were cultured at 37 °C and 5% CO2 in Dulbecco’s Modified Eagle Medium (Biochrom KG, Berlin, Germany) supplemented with 10% fetal calf serum (Pan Biotech, Aidenbach, Germany). For RNA isolation, 50.5 ml of BHI was inoculated with 0.5 ml of a L. monocytogenes overnight culture and incubated at 37 °C or 24 °C with shaking (150 rpm). Aerobic growth in broth was conducted in Erlenmeyer flasks with constant shaking; anaerobic growth was performed in sealed Falcon tubes. Growth was monitored by measuring OD600 with a Lambda Bio + spectrophotometer (Perkin Elmer, Waltham, MA, USA). If appropriate, 3.5 ml of BHI supplemented with 10 mM 1,2-PD (Sigma-Aldrich, Taufkirchen, Germany) and/or 25 nm cobalamin (Applichem, Darmstadt, Germany) in a 15-ml glass tube was inoculated with 0.25 ml of an L. monocytogenes overnight culture and incubated at 37 °C with shaking.
Standard procedures
DNA manipulations and isolation of chromosomal DNA were performed in accordance with standard protocols79 and following the manufacturer’s instructions. GeneRulerTM 1 kb DNA Ladder from Thermo Scientific (Waltham, MA, USA) was used as a marker for DNA analysis. A Bio-Rad Gene pulser II was used for electroporation. PCR was carried out with Taq polymerase and with 100-400 ng of chromosomal DNA or an aliquot of a single colony resuspended in 50 µl of H2O as template. Quantitative real-time (qRT)-PCR and whole bacterial RNA isolation were perfomed as described recently80. Transcription of the housekeeping gene 16 S rRNA and lmo1759 (pcrA) was used for normalization. The oligonucleotides used in this study are listed in Supplementary Table S3. For listerial gene annotation, the Listeria homepage of the Institut Pasteur (http://genolist.pasteur.fr/ListiList/) was used.
For genome comparison, type strain genomic sequences of the 16 Listeria species were downloaded from NCBI GenBank. Sequences were uploaded to the RAST server81 and functionally annotated. L. grayi was excluded from this comparison since it is most closely related to Listeria sensu stricto, although it belongs to the Listeria sensu lato group.
In-frame gene deletions were performed as described recently62. Briefly, two flanking fragments of ~1000 bp were amplified from chromosomal DNA derived from the strain EGDe using the oligonucleotides indicated in Supplementary Table S3 and ligated via the introduced BglII sites. Following nested PCR and using the ligation mixture as a template, the resulting fragment was cloned into pLSV101 via SalI and XmaI. Following transformation of the resulting plasmids, erythromycin-resistant EGDe harboring the chromosomally integrated plasmid were selected upon incubation at 42 °C. Cointegrates were resolved, erythromycin-sensitive clones were screened by PCR, and the deletion sites were sequenced to identify the respective mutant strains.
Transcriptome analysis
Whole-transcriptome RNA library preparation was performed as described recently82. Briefly, RNA was extracted, ribosomal RNAs were depleted, and RNA was fragmented via sonication. After dephosphorylation and rephosphorylation, TruSeq Small RNA Sample Kit (Illumina, Munich, Germany) was used, and the resulting cDNAs were size-selected. Libraries were then diluted and sequenced on a MiSeq sequencer (Illumina, Munich, Germany) using a MiSeq Reagent Kit v2 (50 cycles), resulting in 50 bp single-end reads. Illumina FASTQ files were mapped to the reference genome of L. monocytogenes EGDe (GenBank: NC_003210) using Bowtie for Illumina implemented in Galaxy83,84. SAM files were converted to BAM files and indexed. Artemis85,86 was used to visualize and calculate the number of reads mapping on each gene. Gene counts of each library were normalized to the smallest library in the comparison and RPKM (reads per kilobase per million mapped reads) values were calculated. Fold changes between the different conditions were calculated and visualized using a three-color scheme.
EC adhesion and invasion assays
A total of 2.5 × 105 Caco-2 or HEp-2 cells per well were seeded in a 24-well culture plate and cultivated for 48 h until infection. Cells were washed twice with PBS/Mg2+Ca2+ and covered for 35 min (adhesion assay) or 1 h (invasion assay) with 500 µl of DMEM containing approximately 2.5 × 106 bacteria (multiplicity of infection, MOI, =10) from a glycerol stock washed with PBS. For glycerol stocks, strains were grown in 20 ml of BHI medium to mid-log phase (OD600 ~0.85–0.95) and supplemented with glycerol (15% final concentration). Aliquots of 1 ml were frozen at −80 °C. Prior to infection, samples were thawed, and the number of viable bacteria was determined as cfu per ml.
The average MOI was calculated immediately after infection and ranged from 8 to 11. For assessment of adhesion, the Caco-2 or HEp-2 cells were washed thrice with PBS/Mg2+Ca2+ after a 35-min incubation period. Cell layers were lysed in 1 ml of cold Triton X-100 (0.1%) and vortexed for 1 min to disrupt the cells. For invasion assays, Caco-2 or HEp-2 cells were washed twice with PBS/Mg2+Ca2+ after 1 h of incubation. Extracellular bacteria were removed by adding 0.5 ml of DMEM containing 100 μg/ml gentamycin for 1 h, and the medium was then replaced by DMEM with 10 µg/ml gentamycin. At appropriate time points of incubation in the presence of 10 μg/ml gentamycin, the infected eukaryotic cells were washed again with PBS/Mg2+Ca2+ and then lysed in 1 ml of cold Triton X-100 (0.1%) and vortexed. Adhesion and invasion characteristics as well as intracellular replication behavior of the mutants and the wild type were quantified by plating dilutions of the lysed cells on BHI agar plates that had ben incubated at 37 °C for one day. In all experiments, intact eukaryotic cell monolayers were observed prior to cell lysis.
In vitro growth analyses
For growth analysis of L. monocytogenes EGDe and EGDe ΔpduD, we used the Bioscreen C Automated Microbiology Growth Curve Analysis System (Oy Growth Curves Ab Ltd., Helsinki, Finland), allowing automated OD measurement in a microvolume of 200 µl. Overnight cultures of L. monocytogenes grown in BHI at 37 °C were washed with PBS and then diluted in PBS to obtain an OD600 of 1. This cell suspension was further diluted 1:20 in MM51 containing 0.5% (w/v) yeast extract (Oxoid, Wesel Germany). MM was supplemented with 0 or 50 mM glucose (Fluka, Neu-Ulm, Germany). Cultures were incubated at 37 °C with continuous medium shaking (shaking steps: 60), and were overlaid with 200 µl of paraffin oil (Roth, Karlsruhe, Germany) to establish anaerobic conditions. The OD600 was automatically measured every 30 min over a period of 10 h.
Mouse infections
Four-week-old female BALBc/By/J mice from The Jackson Laboratory (Bar Harbor, ME, USA) or BALB/cAnNCrl mice from Charles River Laboratories (Sulzfeld, Germany) were purchased and used for experiments at the age of 6-8 weeks. Mice were maintained in a specific-pathogen-free facility with a 14-h light and 10-h dark cycle. Mice were infected using a model for foodborne infection as described previously46. Mice were placed in a cage with raised wire flooring to prevent coprophagy and denied food for 18-22 h before the infection. Aliquots of frozen L. monocytogenes or other Listeria species were recovered in BHI medium for 1.5 h at 30 °C without shaking. The desired inoculum was resuspended in 2 µl of PBS mixed with 3 µl of salted butter (Kroger, Cincinatti, OH, USA or REWE, Cologne, Germany). Cell suspension was used to saturate a 2- to 3-mm piece of bread (Kroger or REWE). After the onset of the dark cycle, mice were transferred to an empty cage and fed the Listeria-contaminated pieces of bread with sterile forceps. Afterwards, the mice were returned to their raised wire flooring cages and mouse chow was replenished. Sample collection and handling was performed as described recently46. We confirm that all methods were carried out in accordance with relevant guidelines and regulations, and that all experimental protocols were approved by the Regierung von Oberbayern, München, Germany.
Statistics
Statistical analyses for all experiments were performed using the Student’s t-Test with Welch’s correction in Prism6 (GraphPad, La Jolla, CA, USA). P values less than 0.05 were considered significant and are indicated as follows: *(P < 0.05); **(P < 0.01); ***(P < 0.001); NS (not significant, P ≥ 0.05).
References
Farber, J. M. & Peterkin, P. I. Listeria monocytogenes, a food-borne pathogen. Microbiol Rev 55, 476–511 (1991).
Collins, M. D. et al. Phylogenetic analysis of the genus Listeria based on reverse transcriptase sequencing of 16S rRNA. Int J Syst Bacteriol 41, 240–246 (1991).
Sallen, B., Rajoharison, A., Desvarenne, S., Quinn, F. & Mabilat, C. Comparative analysis of 16S and 23S rRNA sequences of Listeria species. Int J Syst Bacteriol 46, 669–674 (1996).
Freitag, N. E., Port, G. C. & Miner, M. D. Listeria monocytogenes - from saprophyte to intracellular pathogen. Nat Rev Microbiol 7, 623–628 (2009).
Thevenot, D., Dernburg, A. & Vernozy-Rozand, C. An updated review of Listeria monocytogenes in the pork meat industry and its products. J Appl Microbiol 101, 7–17 (2006).
Gandhi, M. & Chikindas, M. L. Listeria: A foodborne pathogen that knows how to survive. Int J Food Microbiol 113, 1–15 (2007).
Carpentier, B. & Cerf, O. Review–Persistence of Listeria monocytogenes in food industry equipment and premises. Int J Food Microbiol 145, 1–8 (2011).
Cossart, P. & Toledo-Arana, A. Listeria monocytogenes, a unique model in infection biology: an overview. Microbes Infect 10, 1041–1050 (2008).
Swaminathan, B. & Gerner-Smidt, P. The epidemiology of human listeriosis. Microbes Infect 9, 1236–1243 (2007).
Chaturongakul, S., Raengpradub, S., Wiedmann, M. & Boor, K. J. Modulation of stress and virulence in Listeria monocytogenes. Trends Microbiol 16, 388–396 (2008).
Gray, M. J., Freitag, N. E. & Boor, K. J. How the bacterial pathogen Listeria monocytogenes mediates the switch from environmental Dr. Jekyll to pathogenic Mr. Hyde. Infect Immun 74, 2505–2512 (2006).
Sue, D., Fink, D., Wiedmann, M. & Boor, K. J. sigmaB-dependent gene induction and expression in Listeria monocytogenes during osmotic and acid stress conditions simulating the intestinal environment. Microbiology 150, 3843–3855 (2004).
Kazmierczak, M. J., Mithoe, S. C., Boor, K. J. & Wiedmann, M. Listeria monocytogenes sigma B regulates stress response and virulence functions. J Bacteriol 185, 5722–5734 (2003).
Neuhaus, K., Satorhelyi, P., Schauer, K., Scherer, S. & Fuchs, T. M. Acid shock of Listeria monocytogenes at low environmental temperatures induces prfA, epithelial cell invasion, and lethality towards Caenorhabditis elegans. BMC Genomics 14, 285 (2013).
Scortti, M., Monzo, H. J., Lacharme-Lora, L., Lewis, D. A. & Vazquez-Boland, J. A. The PrfA virulence regulon. Microbes Infect 9, 1196–1207 (2007).
Seeliger, H. P. R., Rocourt, J., Schrettenbrunner, A., Grimont, P. A. D. & Jones, D. Notes: Listeria ivanovii sp. nov. Int J Syst Evol Microbiol 34, 336–337 (1984).
Cummins, J., Casey, P. G., Joyce, S. A. & Gahan, C. G. A mariner transposon-based signature-tagged mutagenesis system for the analysis of oral infection by Listeria monocytogenes. PLoS One 8, e75437 (2013).
Guillet, C. et al. Human listeriosis caused by Listeria ivanovii. Emerg Infect Dis 16, 136–138 (2010).
Alexander, A. V., Walker, R. L., Johnson, B. J., Charlton, B. R. & Woods, L. W. Bovine abortions attributable to Listeria ivanovii: four cases (1988–1990). J Am Vet Med Assoc 200, 711–714 (1992).
Chand, P. & Sadana, J. R. Outbreak of Listeria ivanovii abortion in sheep in India. Vet Rec 145, 83–84 (1999).
Graves, L. M. et al. Listeria marthii sp. nov., isolated from the natural environment, Finger Lakes National Forest. Int J Syst Evol Microbiol 60, 1280–1288 (2010).
Chiara, M. et al. Comparative Genomics of Listeria Sensu Lato: Genus-Wide Differences in Evolutionary Dynamics and the Progressive Gain of Complex, Potentially Pathogenicity-Related Traits through Lateral Gene Transfer. Genome Biol Evol 7, 2154–2172 (2015).
Rocourt, J. & Seeliger, H. P. Distribution of species of the genus Listeria. Zentralbl Bakteriol Mikrobiol Hyg A 259, 317–330 (1985).
Nayak, D. N., Savalia, C. V., Kalyani, I. H., Kumar, R. & Kshirsagar, D. P. Isolation, identification, and characterization of Listeria spp. from various animal origin foods. Vet World 8, 695–701 (2015).
Dahshan, H., Merwad, A. M. & Mohamed, T. S. Listeria Species in Broiler Poultry Farms: Potential Public Health Hazards. J Microbiol Biotechnol 26, 1551–1556 (2016).
Huang, B. et al. Comparison of multiplex PCR with conventional biochemical methods for the identification of Listeria spp. isolates from food and clinical samples in Queensland, Australia. J Food Prot 70, 1874–1880 (2007).
Husu, J. R. Epidemiological studies on the occurrence of Listeria monocytogenes in the feces of dairy cattle. Zentralbl Veterinarmed B 37, 276–282 (1990).
Bertsch, D. et al. Listeria fleischmannii sp. nov., isolated from cheese. Int J Syst Evol Microbiol 63, 526–532 (2013).
Lang Halter, E., Neuhaus, K. & Scherer, S. Listeria weihenstephanensis sp. nov., isolated from the water plant Lemna trisulca taken from a freshwater pond. Int J Syst Evol Microbiol 63, 641–647 (2013).
Leclercq, A. et al. Listeria rocourtiae sp. nov. Int J Syst Evol Microbiol 60, 2210–2214 (2010).
den Bakker, H. C. et al. Listeria floridensis sp. nov., Listeria aquatica sp. nov., Listeria cornellensis sp. nov., Listeria riparia sp. nov. and Listeria grandensis sp. nov., from agricultural and natural environments. Int J Syst Evol Microbiol 64, 1882–1889 (2014).
Weller, D., Andrus, A., Wiedmann, M. & den Bakker, H. C. Listeria booriae sp. nov. and Listeria newyorkensis sp. nov., from food processing environments in the USA. Int J Syst Evol Microbiol 65, 286–292 (2015).
Hof, H. & Hefner, P. Pathogenicity of Listeria monocytogenes in comparison to other Listeria species. Infection 16(Suppl 2), S141–144 (1988).
Rost, B. Twilight zone of protein sequence alignments. Protein Eng 12, 85–94 (1999).
Orsi, R. H. & Wiedmann, M. Characteristics and distribution of Listeria spp., including Listeria species newly described since 2009. Appl Microbiol Biotechnol 100, 5273–5287 (2016).
Bigot, A. et al. Role of FliF and FliI of Listeria monocytogenes in flagellar assembly and pathogenicity. Infect Immun 73, 5530–5539 (2005).
Gründling, A., Burrack, L. S., Bouwer, H. G. & Higgins, D. E. Listeria monocytogenes regulates flagellar motility gene expression through MogR, a transcriptional repressor required for virulence. Proc Natl Acad Sci USA 101, 12318–12323 (2004).
Dons, L. et al. Role of flagellin and the two-component CheA/CheY system of Listeria monocytogenes in host cell invasion and virulence. Infect Immun 72, 3237–3244 (2004).
Bergmann, S., Rohde, M., Schughart, K. & Lengeling, A. The bioluminescent Listeria monocytogenes strain Xen32 is defective in flagella expression and highly attenuated in orally infected BALB/cJ mice. Gut Pathog 5, 19 (2013).
O’Neil, H. S. & Marquis, H. Listeria monocytogenes flagella are used for motility, not as adhesins, to increase host cell invasion. Infect Immun 74, 6675–6681 (2006).
Knudsen, G. M., Olsen, J. E. & Dons, L. Characterization of DegU, a response regulator in Listeria monocytogenes, involved in regulation of motility and contributes to virulence. FEMS Microbiol Lett 240, 171–179 (2004).
Borezee, E., Pellegrini, E., Beretti, J. L. & Berche, P. SvpA, a novel surface virulence-associated protein required for intracellular survival of Listeria monocytogenes. Microbiology 147, 2913–2923 (2001).
Newton, S. M. et al. The svpA-srtB locus of Listeria monocytogenes: fur-mediated iron regulation and effect on virulence. Mol Microbiol 55, 927–940 (2005).
Schauer, K. et al. Deciphering the intracellular metabolism of Listeria monocytogenes by mutant screening and modelling. BMC Genomics 11, 573 (2010).
Chen, L. H. et al. Cyclic di-GMP-dependent signaling pathways in the pathogenic Firmicute Listeria monocytogenes. PLoS Pathog 10, e1004301 (2014).
Bou Ghanem, E. N., Myers-Morales, T. & D’Orazio, S. E. A mouse model of foodborne Listeria monocytogenes infection. Curr Protoc Microbiol 31, 9B 3 1–9B 3 16 (2013).
Becattini, S. et al. Commensal microbes provide first line defense against Listeria monocytogenes infection. J Exp Med 214, 1973–1989 (2017).
Winter, S. E. et al. Gut inflammation provides a respiratory electron acceptor for Salmonella. Nature 467, 426–429 (2010).
Srikumar, S. & Fuchs, T. M. Ethanolamine utilization contributes to proliferation of Salmonella enterica serovar Typhimurium in food and in nematodes. Appl Environ Microbiol 77, 281–290 (2011).
Faber, F. et al. Respiration of Microbiota-Derived 1,2-propanediol Drives Salmonella Expansion during Colitis. PLoS Pathog 13, e1006129 (2017).
Premaratne, R. J., Lin, W. J. & Johnson, E. A. Development of an improved chemically defined minimal medium for Listeria monocytogenes. Appl Environ Microbiol 57, 3046–3048 (1991).
Roth, J. R., Lawrence, J. G. & Bobik, T. A. Cobalamin (coenzyme B12): synthesis and biological significance. Annu Rev Microbiol 50, 137–181 (1996).
Schmid, M. W. et al. Evolutionary history of the genus Listeria and its virulence genes. Syst Appl Microbiol 28, 1–18 (2005).
Buchrieser, C. Biodiversity of the species Listeria monocytogenes and the genus Listeria. Microbes Infect 9, 1147–1155 (2007).
Wang, Y. et al. Isolation and characterization of Listeria species from rodents in natural environments in China. Emerg Microbes Infect 6, e44 (2017).
Jones, G. S. et al. Intracellular Listeria monocytogenes comprises a minimal but vital fraction of the intestinal burden following foodborne infection. Infect Immun 83, 3146–3156 (2015).
Gaillard, J. L., Berche, P., Frehel, C., Gouin, E. & Cossart, P. Entry of L. monocytogenes into cells is mediated by internalin, a repeat protein reminiscent of surface antigens from Gram-positive cocci. Cell 65, 1127–1141 (1991).
Lecuit, M. et al. A single amino acid in E-cadherin responsible for host specificity towards the human pathogen Listeria monocytogenes. EMBO J 18, 3956–3963 (1999).
Quereda, J. J., Meza-Torres, J., Cossart, P. & Pizarro-Cerda, J. Listeriolysin S: A bacteriocin from epidemic Listeria monocytogenes strains that targets the gut microbiota. Gut Microbes 1–8 (2017).
Fuchs, T. M., Eisenreich, W., Kern, T. & Dandekar, T. Towards a systemic understanding of Listeria monocytogenes metabolism during infection. Front Microbiol 3, 137–143 (2012).
Chico-Calero, I. et al. Hpt, a bacterial homolog of the microsomal glucose- 6-phosphate translocase, mediates rapid intracellular proliferation in Listeria. Proc Natl Acad Sci USA 99, 431–436 (2002).
Joseph, B. et al. Identification of Listeria monocytogenes genes contributing to intracellular replication by expression profiling and mutant screening. J Bacteriol 188, 556–568 (2006).
Grubmüller, S., Schauer, K., Goebel, W., Fuchs, T. M. & Eisenreich, W. Analysis of carbon substrates used by Listeria monocytogenes during growth in J774A.1 macrophages suggests a bipartite intracellular metabolism. Front Cell Infect Microbiol 4, 1–14 (2014).
Schauer, K., Stolz, J., Scherer, S. & Fuchs, T. M. Both thiamine uptake and biosynthesis of thiamine precursors are required for intracellular replication of Listeria monocytogenes. J Bacteriol 191, 2218–2227 (2009).
Kutzner, E., Kern, T., Felsl, A., Eisenreich, W. & Fuchs, T. M. Isotopologue profiling of the listerial N-metabolism. Mol Microbiol 100, 315–327 (2016).
Conner, C. P., Heithoff, D. M., Julio, S. M., Sinsheimer, R. L. & Mahan, M. J. Differential patterns of acquired virulence genes distinguish Salmonella strains. Proc Natl Acad Sci USA 95, 4641–4645 (1998).
Klumpp, J. & Fuchs, T. M. Identification of novel genes in genomic islands that contribute to Salmonella typhimurium replication in macrophages. Microbiology 153, 1207–1220 (2007).
Xue, J., Murrieta, C. M., Rule, D. C. & Miller, K. W. Exogenous or L-rhamnose-derived 1,2-propanediol is metabolized via a pduD-dependent pathway in Listeria innocua. Appl Environ Microbiol 74, 7073–7079 (2008).
Toledo-Arana, A. et al. The Listeria transcriptional landscape from saprophytism to virulence. Nature 459, 950–956 (2009).
Staib, L. & Fuchs, T. M. From food to cell: nutrient exploitation strategies of enteropathogens. Microbiology 160, 1020–1039 (2014).
Gill, S. R. et al. Metagenomic analysis of the human distal gut microbiome. Science 312, 1355–1359 (2006).
Wilkens, S. Structure and mechanism of ABC transporters. F1000Prime Rep 7, 14 (2015).
Ito, Y., Kanamaru, K., Taniguchi, N., Miyamoto, S. & Tokuda, H. A novel ligand bound ABC transporter, LolCDE, provides insights into the molecular mechanisms underlying membrane detachment of bacterial lipoproteins. Mol Microbiol 62, 1064–1075 (2006).
Fisher, M. L., Allen, R., Luo, Y. & Curtiss, R. 3rd Export of extracellular polysaccharides modulates adherence of the Cyanobacterium synechocystis. PLoS One 8, e74514 (2013).
Nishi, J. et al. The export of coat protein from enteroaggregative Escherichia coli by a specific ATP-binding cassette transporter system. J Biol Chem 278, 45680–45689 (2003).
Porsch, E. A., Kehl-Fie, T. E. & St Geme, J. W. 3rd. Modulation of Kingella kingae adherence to human epithelial cells by type IV Pili, capsule, and a novel trimeric autotransporter. MBio 3 (2012).
Fenno, J. C., Shaikh, A., Spatafora, G. & Fives-Taylor, P. The fimA locus of Streptococcus parasanguis encodes an ATP-binding membrane transport system. Mol Microbiol 15, 849–863 (1995).
Jalalvand, F. et al. Haemophilus influenzae protein F mediates binding to laminin and human pulmonary epithelial cells. J Infect Dis 207, 803–813 (2013).
Sambrook, J. & Russell, D. W. Molecular cloning: a laboratory manual, 3nd ed. Cold Spring Harbor Laboratory, Cold Spring Harbor, N. Y. (2001).
Muller-Herbst, S. et al. Identification of genes essential for anaerobic growth of Listeria monocytogenes. Microbiology 160, 752–765 (2014).
Overbeek, R. et al. The SEED and the Rapid Annotation of microbial genomes using Subsystems Technology (RAST). Nucleic Acid Res 42(Database issue), D206-14 (2014).
Landstorfer, R. et al. Comparison of strand-specific transcriptomes of enterohemorrhagic Escherichia coli O157:H7 EDL933 (EHEC) under eleven different environmental conditions including radish sprouts and cattle feces. BMC Genomics 15, 353 (2014).
Goecks, J., Nekrutenko, A., Taylor, J. & Galaxy, T. Galaxy: a comprehensive approach for supporting accessible, reproducible, and transparent computational research in the life sciences. Genome Biol 11, R86 (2010).
Blankenberg, D. et al. Galaxy: a web-based genome analysis tool for experimentalists. Curr Protoc Mol Biol Chapter 19, Unit19 10 11–21 (2010).
Rutherford, K. et al. Artemis: sequence visualization and annotation. Bioinformatics 16, 944–945 (2000).
Carver, T., Bohme, U., Otto, T. D., Parkhill, J. & Berriman, M. BamView: viewing mapped read alignment data in the context of the reference sequence. Bioinformatics 26, 676–677 (2010).
Camejo, A. et al. In vivo transcriptional profiling of Listeria monocytogenes and mutagenesis identify new virulence factors involved in infection. PLoS Pathog 5, e1000449 (2009).
Williams, T., Bauer, S., Beier, D. & Kuhn, M. Construction and characterization of Listeria monocytogenes mutants with in-frame deletions in the response regulator genes identified in the genome sequence. Infect Immun 73, 3152–3159 (2005).
Shen, A. & Higgins, D. E. The MogR transcriptional repressor regulates nonhierarchal expression of flagellar motility genes and virulence in Listeria monocytogenes. PLoS Pathog 2, e30 (2006).
Begley, M. et al. The interplay between classical and alternative isoprenoid biosynthesis controls gammadelta T cell bioactivity of Listeria monocytogenes. FEBS Lett 561, 99–104 (2004).
Iurov, D. S. et al. Contribution of L,D-carboxypeptidases in virulence of facultative intracellular pathogenic bacteria Listeria monocytogenes. Zh Mikrobiol Epidemiol Immunobiol, 15–20 (2012).
Bennett, H. J. et al. Characterization of relA and codY mutants of Listeria monocytogenes: identification of the CodY regulon and its role in virulence. Mol Microbiol 63, 1453–1467 (2007).
Mariscotti, J. F., Garcia-del Portillo, F. & Pucciarelli, M. G. The Listeria monocytogenes sortase-B recognizes varied amino acids at position 2 of the sorting motif. J Biol Chem 284, 6140–6146 (2009).
Xiao, Q. et al. Sortase independent and dependent systems for acquisition of haem and haemoglobin in Listeria monocytogenes. Mol Microbiol 80, 1581–1597 (2011).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) Method. Methods 25, 402–408 (2001).
Acknowledgements
This work was supported by a grant to JS within the Research Training Group (Graduiertenkolleg) 1482 of the Deutsche Forschungsgemeinschaft. We thank Lena Riedel, Michael Schneider, Melanie Schoof, Michelle Pitts and Travis Combs for the experimental support, and Ian Monk for kindly providing the plasmids pIMC3ery and pIMC3kan.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: J.S., S.M.H., S.E.F.D. and T.M.F.; performed the experiments: J.S., G.J. and K.S.; analysed the data: J.S., S.M.H., S.E.F.D. and T.M.F.; wrote the manuscript: J.S., S.E.F.D. and T.M.F.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schardt, J., Jones, G., Müller-Herbst, S. et al. Comparison between Listeria sensu stricto and Listeria sensu lato strains identifies novel determinants involved in infection. Sci Rep 7, 17821 (2017). https://doi.org/10.1038/s41598-017-17570-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-17570-0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.