Abstract
The small intestine plays an essential role in the health and well-being of animals. Previous studies have shown that Lactobacillus has a protective effect on intestinal morphology, intestinal epithelium integrity and appropriate maturation of gut-associated tissues. Here, gene expression in jejunum tissue of weaned piglets was investigated by RNA-seq analysis after administration of sterile saline, Lactobacillus reuteri, or an antibiotic (chlortetracycline). In total, 401 and 293 genes were significantly regulated by chlortetracycline and L. reuteri, respectively, compared with control treatment. Notably, the HP, NOX1 and GPX2 genes were significantly up-regulated in the L. reuteri group compared with control, which is related to the antioxidant ability of this strain. In addition, the expression of genes related to arachidonic acid metabolism and linoleic acid metabolism enriched after treatment with L. reuteri. The fatty acid composition in the jejunum and colon was examined by GC-MS analysis and suggested that the MUFA C18:1n9c, and PUFAs C18:2n6c and C20:4n6 were increased in the L. reuteri group, verifying the GO enrichment and KEGG pathway analyses of the RNA-seq results. The results contribute to our understanding of the probiotic activity of this strain and its application in pig production.
Similar content being viewed by others
Introduction
The intestines have various functions such as the absorption of nutrients, the absorption and secretion of electrolytes and water, and the secretion of mucin and immunoglobulins, and they provide a selective barrier against harmful antigens and pathogens1. Weaning is one of the most stressful stages in piglet development. During the transition process, piglets must cope with abrupt separation from their mother, a new environment, and a switch from a highly digestible (liquid) milk diet to a less-digestible, more complex, chemically composed solid feed. This process can potentially cause damage to intestinal tissue, resulting in changes in the villus and crypt architecture, and depress the activity of many brush-border digestive enzymes2. Consequently, piglets are more susceptible to intestinal complications such as disturbed gut motility, reduced absorptive function, reduced mucosal immune defence, and reduced intestinal regenerative capability, resulting in poor appetite, lower feed intake, and growth retardation3. Gastrointestinal disturbances occurring immediately post weaning cause large economic losses in the pig farming industry4.
In the recent years, the increased use of antibiotic growth promoters has raised concerns about the development of pathogenic bacterial strains that are resistant to antibiotics and about residual contamination of the food chain with these agents5. Lactobacillus is part of the normal intestinal flora and is thought to alter the intestinal microbiota, thereby affecting physiological intestinal function and the general health of weaned pigs6,7. Studies focusing on intestinal tissues have shown that Lactobacillus has a protective effect on tissue structure, the integrity of the intestinal epithelium, appropriate maturation of gut-associated tissues and the function of the neuro-endocrine system8,9. Recently, the mechanism by which Lactobacillus supports gastrointestinal health in mammals has become of great interest.
With the development of advanced molecular genetics technologies including next-generation sequencing and bioinformatics, transcriptome sequencing (RNA-seq) provides a convenient platform for measuring large-scale gene expression patterns in organisms. RNA-seq also enables the detection of differentially expressed genes (DEGs) with low expression levels10,11,12,13,14. Some studies have analysed the transcriptome profiles of blood15, muscle16,17, gonads18, liver19, backfat20 and uterine endometrium21 in pigs, while Mach et al.22 analysed the gene expression profile along the small intestine and in ileal Peyer’s patches in four young pigs. The above studies have all helped elucidate the mechanisms underlying tissue and organ development and have thus provided valuable information for the pig farming industry. However, studies exploring gene expression in the small intestinal segment in relation to Lactobacillus and antibiotic treatments have not been conducted.
The present study aimed to investigate gene expression in jejunum tissue of weaned piglets. RNA-seq gene expression profiling was conducted on weaned piglets that received one of the following treatments: Lactobacillus reuteri, chlortetracycline (an antibiotic), or sterile saline (control). The fatty acid composition of the jejunal and colon segments of piglets was also determined to verify the RNA-seq results. The current study should help advance our understanding of Lactobacillus and intestinal health and provide basic data for future studies in this area.
Results
Sequencing data summary
In this study, sequencing generated approximately 4.82 Gb of raw 150-bp paired-end reads, comprising 1.77, 1.49 and 1.56 Gb from the control, chlortetracycline, and Lactobacillus groups, respectively. Using the TopHat2 aligner, over 97.82% of the clean reads per sample were mapped back to the Sus scrofa reference genome (Sscrofa v10.2) sequence23, the vast majority of the mapped reads were located in annotated exons (Table 1).
Identification and analysis of DEGs among the three groups
Based on the FPKM values, approximately 93% and a little more than 1.3% of the reference genes were expressed at less than 100 FPKM and more than 500 FPKM. The first and second Principal Component Analysis (PCA) was performed and the result showed the different expression patterns among the three treatments (Supplementary Figure S1). To judge the statistical significance of the DEGs, P-values ≤ 0.05 and absolute values of |logFC| ≥ 1 were used as the thresholds. In total, 11 genes were differentially expressed among the three groups. 401 DEGs were identified in the chlortetracycline vs. control analysis: 240 up-regulated and 161 down-regulated. Furthermore, 293 DEGs were identified in the L. reuteri vs. control analysis: 128 up-regulated and 165 down-regulated. 99 genes were differentially expressed in both the chlortetracycline and L. reuteri groups.
GO and KEGG pathway analysis
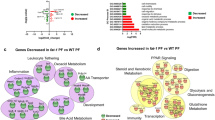
In total, 1374 and 388 DEGs had KEGG pathway annotations in the chlortetracycline vs. control, and L. reuteri vs. control groups, respectively. Gene ontology (GO) analysis was performed to investigate which biological functions are important after treatment. As shown in Fig. 1A,B, the GO analysis was classified into the following three functional categories: biological process, cellular component and molecular function, which comprise 25, 18 and 25 for chlortetracycline vs. control, and 24, 16, and 14 for L. reuteri vs. control. The results also shown that genes related to the GO terms “Antioxidant activity” and “Receptor regulator activity” were significantly expressed in the L. reuteri group compared with the control group. Furthermore, genes related to the GO term “Extracellular matrix part” were significantly expressed by chlortetracycline and L. reuteri compared with control.
Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis was performed to predict the significantly enriched metabolic and immune pathways listed in Fig. 2A,B. In total, 1083 and 991 GO enrichment terms and 78 KEGG enrichment pathways were shared by the chlortetracycline and L. reuteri groups, and 30 and 23 KEGG pathways were significantly enriched by both treatments, respectively.
qRT-PCR confirmation
Using the same RNA samples, the fold change (2−ΔΔCt) in the expression of eight genes was determined using qRT-PCR to validate the RNA-seq results. The expression patterns of the eight genes were generally consistent with the RNA-seq results, suggesting that the RNA-seq results were accurate and reliable (Tables 2 and 3).
GC-MS analysis of the fatty acid composition of the jejunum and colon
The profiles of fatty acids, including saturated fatty acids (SFAs), monounsaturated fatty acids (MUFAs) and polyunsaturated fatty acids (PUFAs), were determined in the jejunum and colon of weaned piglets in the three treatment groups. In the jejunum, the SFAs C12:0, C16:0, C17:0, C18:0, C20:0, C22:0 and C24:0 were decreased by L. reuteri treatment. The MUFA C18:1n9c, and the PUFAs C18:2n6c and C20:4n6 were lower in the L. reuteri group than in the chlortetracycline group (Table 4). However, in the colon, L. reuteri treatment decreased the levels of the SFAs C16:0, C17:0, C18:0, and C20:0, whereas MUFA C18:1n9c, PUFAs C18:2n6c, C18:3n3 and C20:4n6 were significantly increased by supplementation with L. reuteri (Table 5).
Discussion
The mammalian gut is susceptible to stress in early life, in addition to genetic determinants of intestinal health, mammals must adapt to a variety of environments, diets, and stressors to maintain intestinal health, and the health consequences of forced adaptation in the form of early weaning may persist into later life. The expression of intestinal tract genes is regulated by many factors, and the expression patterns change dynamically during periods of growth, with numerous gene families and classes differentially expressed in different cell populations of the gut24. The weaning transition is a complex period, and profound changes occur in the small intestines of piglets undergoing weaning25. Therefore, to better understand the cellular physiology and functionality of the small intestine, the present study used RNA-seq technology to evaluate the transcription profiles of jejunum tissue from weaned piglets treated with L. reuteri, chlortetracycline, or sterile saline (control).
The small intestine is mainly responsible for digestion and absorption, and the jejunal segment participates in the maintenance of biological processes, cell structure, cellular components and molecular functions of the intestine. Our results clearly showed differential gene expression profiles in jejunum tissue in response to the three treatments. In particular, 11 genes, including C-C motif chemokine 22 precursor (CCL22), RNA-binding region containing protein 2-like (S100A9), immune-responsive gene 1 protein homolog (IRG1), cytochrome P450 2C49 precursor (CYP2C49), DNA nucleotidylexotransferase-like (DNTT), putative aldo-keto reductase family 1 member C1 (AKR1C1), inter-alpha-trypsin inhibitor heavy chain H4 precursor (ITIH4), uteroglobin-like (SCGB1A1), serum amyloid A2 precursor (SAA4), serpin A3-8 (ENSSSCG00000030371), and immune-responsive gene 1 protein homolog (ENSSSCG00000030557), were differentially expressed among the three groups. The CYP2C49 gene is associated with lipid metabolism, as coconut oil and beef tallow increased its expression levels, while backfat subcutaneous adipose tissue showed increased SFAs and decreased MUFAs and PUFAs26. Our study showed that CYP2C49 was down-regulated in the L. reuteri group, which may be associated with the fatty acid composition in the intestine. Kim et al.21 reported that CYP genes are also expressed in the uterine endometrial tissues of pigs during pregnancy, with levels increasing throughout gestation and declining at the end of pregnancy (day 114). The other genes, such as CCL22, S100A9, SAA4, and SCGB1A1, have been shown to be associated with immunomodulation27,28,29,30. AKR1C1 has crucial roles in the biosynthesis and inactivation of all classes of steroid hormones31. The above genes have been studied mainly in humans, rats and cell lines in vitro. Our study showed that AKR1C1 was up-regulated, SCGB1A1 was down-regulated, and CCL22 and S100A9 were moderately regulated in the L. reuteri group. Previous studies have suggested that the Lactobacillus genus involved in pro-inflammatory NF-κB signalling, which TLR4 and NOD2 receptors were down-regulated in rat jejunum32. Therefore, the correlation between gene expression and immune signalling pathways affected by L. reuteri in piglet jejunum needs to be studied in the future.
In addition, the DEGs in our study were mainly classified into two important categories according to function: nutrient metabolism and immunoregulation. We detected DEGs associated with nutrient metabolism, including mineral, vitamin, amino acid and lipid metabolism, such as the UDP glucuronosyltransferase 2 family, polypeptide A3 (UGT2A3), lactotransferrin (LTF), and calmegin precursor (CLGN), in the L. reuteri group compared with the control group, solute carrier family 2 (SLC2A8), S100 calcium binding protein A9 (S100A9), fatty acid desaturase 3 (FADS3), alanine-glyoxylate aminotransferase 2 (AGXT2) and vitamin D3 receptor (VDR) were differentially expressed in the chlortetracycline group compared with the control group. The DEGs associated with immunoregulation included immunoglobulin kappa variable region (IGKV-3), which was differentially expressed in the L. reuteri group compared with the control group, and T-cell immunoglobulin and mucin domain containing 4 (TIMD4) and CD79b molecule (CD79B), which were differentially expressed in the chlortetracycline group compared with the control group.
Mach et al.22 confirmed that sucrose-isomaltase (SI), dipeptidyl peptidase (DPP4), and enterocyte sodium/glucose co-transporter (SLC5A1) were significantly expressed in jejunum tissue of Large White male pigs at 70 days of age. Our results indicated that SI, DPP4, and SLC5A1 were expressed in the jejunal segment of weaning piglets in all three treatment groups. However, SI gene expression was significantly higher in the chlortetracycline group than in the control group, while it showed an increasing trend (P = 0.062) in the L. reuteri group compared with the control group. SI is the only enzyme reported to hydrolyse sucrose in the small intestine, and it plays a pivotal role in the digestion and absorption of carbohydrates33. The differences in SI expression in our present study suggested differences in carbohydrate metabolism in response to chlortetracycline treatment. Our previous study indicated that Proteobacteria were significantly more abundant in the chlortetracycline and L. reuteri groups. However, the increase in the relative abundance of Proteobacteria was mostly correlated with an increase in the relative abundance of Actinobacillus in the chlortetracycline group and with increases in the relative abundance of Actinobacillus and Escherichia-Shigella in the L. reuteri group34. Therefore, the results may be explained by changes in the composition of gut microbiota after oral administration of chlortetracycline or Lactobacillus in weaned piglets.
We investigated the biological functions of the DEGs by GO annotation and KEGG pathway analysis. The GO term “Antioxidant activity” was clearly enriched by L. reuteri treatment. Notably, haptoglobin precursor (HP) and NADPH oxidase 1 were significantly expressed in the L. reuteri group. Our previous studies suggested that Lactobacillus strains increase the serum concentrations of superoxide dismutase, glutathione peroxidase and catalase in weaned piglets35. Kumar et al.36 observed that the activity of catalase, superoxide dismutase and glutathione S-transferase (GST) increased in animals treated with probiotics. It is believed that probiotics can increase GST activity through the action of butyric acid. This short chain fatty acid can change the histone acetylation status, thus increasing the expression of GST37. In addition, the GO term “Receptor regulator activity” was also enriched by L. reuteri treatment. Thus, we hypothesised that this Lactobacillus strain can improve oxidative stress and homeostasis in the piglet intestine.
To investigate the biological functions of the DEGs, we performed GO enrichment and KEGG pathway analyses. Our data clearly revealed that the expression of genes related to retinol metabolism, arachidonic acid metabolism, linoleic acid metabolism and the chemokine signalling pathway increased after treatment with L. reuteri treatment. L. reuteri treatment also increased the expression of genes related to mineral absorption, cytokine-cytokine receptor interaction, vitamin digestion and absorption, intestinal immune network for IgA production, glutathione metabolism, glycine, and serine and threonine metabolism compared with control treatment. However, retinol metabolism, linoleic acid metabolism, arachidonic acid metabolism, starch and sucrose metabolism, cytokine-cytokine receptor interaction, ascorbate and aldarate metabolism, pentose and glucuronate interconversion, glycine, serine and threonine metabolism, and mineral absorption were induced by chlortetracycline treatment compared with control treatment. In agreement with our present study on the diversity and composition of the gut microbiota of weaned piglets33, the differences we observed in the biological functions of the DEGs among the three treatment groups may be related to the intestinal microbiota composition.
Our current study also showed that seven categories of SFAs in the jejunum and four in the colon were significantly decreased and that UFAs, such as MUFA C18:1n9c, PUFAs C18:2n6c, C18:3n3 and C20:4n6, were significantly increased in the L. reuteri group compared with the control group, these results were consistent with the DEGs results, in which FABP3, FABP1 and FADS3 were up-regulated in the L. reuteri group, and with the GO enrichment and KEGG pathway analyses, which identified linoleic acid metabolism and arachidonic acid metabolism as significantly increased in the L. reuteri group. Mpofu et al.38 reported higher SFA levels in breast muscle of broilers in the antibiotic group than in the Lippia javanica group. Studies have also suggested that Lactobacillus strains can improve the biosynthesis of linoleic acid or conjugated linoleic acid in vitro 39,40. Swiatkiewicz et al.26 reported that the presence of lipids in pig tissues and their degree of saturation affect the sensory quality of the meat, while adipose tissue metabolism depends on dietary fatty acid composition and is controlled by changes in gene expression. Therefore, we will conduct more experiments to elucidate the effect of Lactobacillus strains on the fatty acid composition of porcine muscle, which is critical for pork quality and human health.
Conclusion
In summary, the gene expression profiles of jejunum tissue from weaned piglets were distinct in response to the three treatments used in this study (control, chlortetracycline, and L. reuteri ZLR003). Our study showed that the L. reuteri ZLR003 strain promoted antioxidant activity, which is related to oxidative stress in pig production. In addition, the fatty acid composition in the jejunum and colon was improved in the L. reuteri ZLR003 group. This study elucidated the probiotic mechanisms of the L. reuteri ZLR003 strain in weaned piglets in vitro and in vivo. Although more research is required, our findings may lead to the future use of Lactobacillus strains to improve the gut health of farmed animals.
Methods
Ethical approval
Protocols and procedures related to animal work throughout the study were approved by the Institutional Animal Care and Use Committee (IACUC) of the Institute of Animal Husbandry and Veterinary Medicine, Beijing Academy of Agriculture and Forestry Sciences, China. The approval number is IACUC-2010. All pigs used in this study were taken care and operated according to the relevant regulations.
Lactobacillus and chlortetracycline preparation
The L. reuteri ZLR003 strain used in the present study was deposited at the China General Microbiological Culture Collection Center (CGMCC) under CGMCC number 11530. The strain was inoculated in Mann, Rogosa and Sharpe (MRS) broth (Merck, Darmstadt, Germany) with an inoculum dose of 1.0% (v/v) for 18 h at 37 °C. Bacterial cells were harvested by centrifugation at 6738 × g for 10 min at 4 °C under aseptic conditions, followed by suspension in a 0.85% sterile saline solution. A liquid preparation containing 2.0 × 109 colony forming units (cfu)/mL of viable cells was used. Chlortetracycline ( > 95% purity, Mellon, Biological Technology Co., Ltd., Dalian, China) was prepared at 100 mg/kg using 0.85% sterile saline solution. A 0.85% sterile saline solution lacking L. reuteri ZLR003 or chlortetracycline was used as the control treatment. The three treatments were prepared concurrently every 2 days and stored at 4 °C.
Animal selection and collection of tissue samples
A total of nine crossbred (Landrace × Large White) piglets weaned at 30 days of age and weighing 8.57 ± 1.28 kg (BW) were used under the same husbandry conditions. Piglets were randomly divided into three groups (four females and five males) with a consistent average weight: 1) control treatment, piglets received 5 mL of 0.85% sterile saline solution orally every morning; 2) L. reuteri treatment, piglets were orally administered 5 mL of L. reuteri ZLR003 (2.0 × 109 cfu/mL); 3) antibiotic treatment, piglets were orally administered 5 mL of 100 mg/kg chlortetracycline. All piglets were given a specifically designed basic diet did not contain any antibiotics41 (Table 6). Piglets were housed in an environmentally controlled stainless steel cage with a room temperature between 25 °C and 28 °C and 60% relative humidity. Feed and water were provided ad libitum through a feeder and nipple drinker throughout the experimental period, which lasted 10 d.
At the end of the experiment, the nine piglets were euthanised under anaesthesia by exsanguination. The jejunum tissues were sampled at 5 cm, 24 cm and 40 cm from the duodenum-jejunum junction, and the three sites were combined into one sample. A total of nine samples were collected in sterile containers, snap-frozen in liquid nitrogen and stored at −80 °C. The contents of the two intestinal segments (jejunum and colon) were collected simultaneously, and all 18 samples were snap-frozen in sterile containers in liquid nitrogen and stored at –80 °C.
RNA extraction
Total RNA was extracted from tissues using TRIzol® Reagent according to the manufacturer’s instructions (Invitrogen, Carlsbad, CA, USA), and genomic DNA was removed using DNase I (TaKara, Japan). RNA quality was determined using a 2100 Bioanalyser (Agilent Technologies, Palo Alto, CA, USA) and quantified using a ND-2000 (Nano Drop Technologies, Wilmington, DE, USA). A high-quality RNA sample (OD260/280 = 1.8–2.2, OD260/230 ≥ 2.0, RIN ≥ 8.0, 28 S:18 S ≥ 1.0, > 10 μg) was used to construct the sequencing library.
Library preparation and Illumina HiSeq. 4000 sequencing
Total RNA (5 μg) was prepared for the RNA-seq transcriptome library using the TruSeq™ RNA sample preparation kit according to the manufacturer’s instructions (Illumina, San Diego, CA, USA). Thereafter, mRNA was isolated using the polyA selection method with oligo (dT) beads, followed by fragmentation (100 to 400 bp) in fragmentation buffer. Next, double-stranded cDNA was synthesised using a Super Script double-stranded cDNA synthesis kit (Invitrogen) with random hexamer primers (Illumina) and then subjected to end-repair, phosphorylation and ‘A’ base addition according to the Illumina library construction protocol. Nine cDNA libraries from the following three groups, and libraries were size-selected for cDNA target fragments of 200–300 bp on 2% Low Range Ultra Agarose followed by 15 cycles of PCR amplification using Phusion DNA polymerase (NEB, USA). After quantitation by TBS-380 (Tumer BioSystems, Sunnyvale, CA, USA), the paired-end RNA-seq library was sequenced on an Illumina HiSeq. 4000 (2 × 150-bp read lengths). The raw reads were deposited in the National Center for Biotechnology Information Sequence Read Archive database (https://www.ncbi.nlm.nih.gov/sra) under accession number: SRR5124832.
Bioinformatics analysis
Read mapping
The raw paired-end reads were trimmed and quality controlled by SeqPrep (https://github.com/jstjohn/SeqPrep) and Sickle (https://github.com/najoshi/sickle) using the default parameters. The clean reads were separately aligned with the reference genome in orientation mode using TopHat2 software (http://tophat.cbcb.umd.edu/)42. The bowtie2 mapping criteria stated that sequencing reads should be uniquely matched to the genome allowing up to two mismatches, without insertions or deletions. Next, the gene region was expanded following the site depths for operon attainment. The whole genome was split into multiple 15-kbp windows that shared 5-kbp of sequence. New transcribed regions were defined as > 2 consecutive windows without an overlapping gene region, where at least two reads mapped per window were in the same orientation.
Differential expression analysis and functional enrichment among the three groups
To identify DEGs between two different samples, the expression levels of each transcript were calculated using the fragments per kb of exon per million (FPKM) mapped reads method. Cuffdiff (http://cufflinks.cbcb.umd.edu/)43 was used for the differential expression analysis. DEGs between two samples were selected using the following criteria: i) the logarithmic fold change was > 2.0, and ii) the false discovery rate was < 0.05. To understand the DEG functions, GO functional enrichment and KEGG pathway analyses were conducted using Goatools (https://github.com/tanghaibao/Goatools) and KOBAS (http://kobas.cbi.pku.edu.cn/home.do)44. DEGs were considered significantly enriched in GO terms and metabolic pathways when the Bonferroni-corrected P-value was less than 0.05.
Quantitative real-time PCR (qRT-PCR) confirmation
To confirm the RNA-seq results, eight candidate genes were selected and examined by quantitative reverse transcription PCR (qRT-PCR) in the same RNA samples as were used for RNA-seq. Reverse transcription was performed with 1 μg of extracted RNA in a total volume of 20 μL using TIANScript M-MLV reverse transcriptase (TianGen, Beijing, China) with specific primers (ACTB-qPCR-R, CYP2C49-qPCR-R, DDX3X-qPCR-R, HP-qPCR-R, LTF-qPCR-R, SAA3-qPCR-R, SIGLEC1-qPCR-R, TBXAS1-qPCR-R and ACACB-qPCR-R), according to the manufacturer’s instructions. The resulting cDNA was stored at −20 °C prior to PCR amplification. For qPCR, SYBR Green Real-time PCR Master Mix (TOYOBO, Osaka, Japan) was used to amplify the cDNA using an iQ5 Multicolor Real-Time PCR Detection System (Bio-Rad Laboratories, Hercules, CA, USA). The PCR conditions comprised initial denaturation at 95 °C for 3 min and 40 cycles of 95 °C for 15 s, 60 °C for 30 s, and 72 °C for 20 s. Melting curve data were collected at 0.5 °C increments in 10-s cycles (55 °C for 80 cycles). The gene expression data were normalised against the ACTB housekeeping gene data, and the relative expression levels were calculated using the 2−ΔΔCt method. All reactions were conducted on one plate in triplicate to ensure reproducibility.
GC-MS analysis of the fatty acid composition in the jejunum and colon
The fatty acid composition was determined by gas chromatography (6890 series, Agilent Technologies, Wilmington, DE, USA) according to the procedures of Sukhija and Palmquist45 with slight modifications. The samples were converted to fatty acid methyl esters using methanolic HCl. Undecanoic acid (C11:0) was used as the internal standard. Aliquots of 1 μL were injected into a capillary column (60 m × 250 μm × 0.25 μm, DB-23, Agilent) with cyanopropyl methyl silicone as the stationary phase. The column temperature was programmed with a 1:20 split. Injector and detector temperatures were maintained at 260 and 270 °C, respectively. Nitrogen was the carrier gas at a flow rate of 2.0 mL/min.
Statistical analyses of unsaturated fatty acid data
The fatty acid data were analysed according to the General Linear Models (GLM) procedure in SAS. Differences among means were tested using Tukey’s test, and differences were considered significant at P < 0.05.
References
Lalles, J. P. et al. Gut function and dysfunction in young pigs: physiology. Anim.Res. 53, 301–316 (2004).
Lalles, J. P., Bosi, P., Smidt, H. & Stokes, C. R. Weaning- a challenge to gut physiologists. Livest Sci. 108, 82–93 (2007).
Isaacson, R. & Kim, H. B. The intestinal microbiome of the pig. Anim Health Res Rev. 13(1), 100–109 (2012).
Lalles, J. P., Bos, P., Smidt, H. & Stokes, C. R. Nutritional management of gut health in pigs around weaning. P. Nutr. Soc. 66, 260–268 (2007).
Vondruskova, H., Slamova, R., Trckova, M., Zraly, Z. & Pavlik, I. Alternatives to antibiotic growth promoters in prevention of diarrhoea in weaned piglets: a review. Vet Med-Czech. 55(5), 199–224 (2010).
Liu, H. et al. Oral administration of Lactobacillus fermentum I5007 favors intestinal development and alters the intestinal microbiota in formula-fed 455 piglets. J. Agric. Food. Chem. 62, 860–866 (2014).
Jiao, L. F. et al. Cello-oligosaccharide influences intestinal microflora, mucosal architecture and nutrient transport in weaned pigs. Anim. Feed. Sci. Tech. 195, 85–91 (2014).
Metzler, B. U., Mosenthin, R., Baumgartel, T. & Rodehutscord, M. Effects of fermentable carbohydrates and low dietary phosphorus supply on the chemical composition of faecal bacteria and microbial metabolites in the gastrointestinal tract of pigs. J. Anim. Physiol. Ann. 93(1), 130–139 (2009).
Ross, G. R., Gusils, C., Oliszewski, R., de Holgado, S. C. & Gonzalez, S. N. Effects of probiotic administration in swine. J. Biosci. Bioeng. 109(6), 545–549 (2009).
Hardcastle, T. J. & Kelly, K. A. Bayseq: empirical bayesian methods for identifying differential expression in sequence count data. BMC Bioinformatics. 11, 422, https://doi.org/10.1186/1471-2105-11-422 (2010).
Robinson, M. D. & Oshlack, A. A scaling normalization method for differential expression analysis of RNA-seq data. Genome Biol. 11, R25 (2011).
Tarazona, S., Alcalde, F. G., Dopazo, J., Ferrer, A. & Conesa, A. Differential expression in RNA-seq: a matter of depth. Genome Res. 21, 2213–2223 (2011).
Trapnell, C. et al. Differential gene and transcript expression analysis of RNA-seq experiments with tophat and cufflinks. Nat Protoc. 7, 562–578 (2012).
Wang, L., Feng, Z., Wang, X., Wang, X. & Zhang, X. Degseq: an R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics. 26, 136–138 (2010).
Jegou, M. et al. Whole blood transcriptomics is relevant to identify molecular changes in response to genetic selection for feed efficiency and nutritional status in the pig. PLoS One. 11(1), e0146550 (2016).
Zhao, X. et al. Comparative analyses by sequencing of transcriptomes during skeletal muscle development between pig breeds differing in muscle growth rate and fatness. PLoS One. 6(5), e19774, https://doi.org/10.1371/journal.pone.0019774 (2011).
Ayuso, M. et al. Comparative Analysis of Muscle Transcriptome between Pig Genotypes Identifies Genes and Regulatory Mechanisms Associated to Growth, Fatness and Metabolism. PLoS One. 10(12), e0145162, https://doi.org/10.1371/journal.pone.0145162 (2015).
Esteve-Codina, A. et al. Exploring the gonad transcriptome of two extreme male pigs with RNA-seq. BMC Genomics. 12, 552–565 (2011).
Ramayo-Caldas, Y. et al. Liver transcriptome profile in pigs with extreme phenotypes of intramuscular fatty acid composition. BMC Genomics. 13, 547–564 (2012).
Xing, K. et al. Integration of Transcriptome and Whole Genomic Resequencing Data to Identify Key Genes Affecting Swine Fat Deposition. PLoS One. 10(4), e0122396, https://doi.org/10.1371/journal.pone.0122396 (2015).
Kim, M. et al. Analysis of Stage-Specific Gene Expression Profiles in the Uterine Endometrium during Pregnancy in Pigs. PLoS One. 10(11), e0143436, https://doi.org/10.1371/journal.pone.0143436 (2015).
Mach, N. et al. Extensive expression differences along porcine small intestine evidenced by transcriptome sequencing. PLoS One., doi, https://doi.org/10.1371/journal.pone.0088515 (2014).
Groenen, M. A. M., Archibald, A. L., Uenishi, H., Tuggle, C. K. & Takeuchi, Y. Analyses of pig genomes provide insight into porcine demography and evolution. Nature 491, 393–398 (2012).
Freeman, T. C. et al. A gene expression atlas of the domestic pig. BMC Biol. 10, 90 (2012).
Boudry, G., Peron, V., Le Huerou-Luron, I., Lalles, J. P. & Seve, B. Weaning induces both transient and long-lasting modifications of absorptive, secretory, and barrier properties of piglet intestine. J. Nutr. 134, 2256–2262 (2004).
Swiatkiewicz, M., Oczkowicz, M., Ropka-Molik, K. & Hanczakowska, E. The effect of dietary fatty acid composition on adipose tissue quality and expression of genes related to lipid metabolism in porcine livers. Anim. Feed. Sci. techno. 216, 204–215 (2016).
Elhussein, M. O. Y., Elazom, A., Mohamed, M. S. & Abdalla, F. B. Local elevation of CCL22: A new trend in immunotherapy (skin model). J. Cell. Immun. 2(2), 79–84 (2016).
Sekiya, S. et al. Enzyme-linked immunosorbent assay for S100A9 in the stool of rats with dextran sulfate sodium-induced colitis. J. Immunol. Methods. 439, 44–49 (2016).
Peri, A., Cordella-Miele, E., Miele, L. & Mukherjee, A. B. Tissue-specific expression of the gene coding for human Clara cell 10-k D protein, a phospholipase A2-inhibitory protein. J. Clin. Invest. 92, 2099–2109 (1993).
Malle, E. & De Beer, F. C. Human serum amyloid A (SAA) protein: a prominent acute-phase reactant for clinical practice. Eur. J. Clin. Invest. 26(6), 427–435 (1996).
Traven, K. et al. Ruthenium complexes as inhibitors of the aldo-keto reductases AKR1C1-1C3. Chem. Biol. Interact. 234, 349–359 (2015).
Kishi, K., Takase, S. & Goda, T. Enhancement of sucrose –isomaltase gene expression induced by luminally administered fructose in rat jejunum. J. Nutr. Biochem 10, 8–12 (1999).
Zhang, D. Y. et al. Changes in the diversity and composition of gut microbiota of weaned piglets after oral administration of Lactobacillus or an antibiotic. Appl. Microbiol. Biot. 100(23), 10081–10093 (2016).
Wang, J. et al. Lactobacillus plantarum ZLP001: In vitro assessment of antioxidant capacity and effect on growth performance and antioxidant status in weaning Piglets. Asian-Aust. J. Anim. Sci. 25(8), 1153–1158 (2012).
Zhang, D. Y. et al. Complete genome sequence of probiotic Lactobacillus reuteri ZLR003 isolated from healthy weaned pig. J. Biotechnol. 228, 69–70 (2016).
Kumar, R. et al. Lactobacillus plantarum AS1 isolated from south indian fermented food Kallappam suppress 1, 2-dimethylhydrazine (DMH)-induced colorectal cancer in male wistar rats. Appl. Biochem. Biotechnol. 166, 620–31 (2012).
Pool-Zobel, B., Veeriah, S. & Bohmer, F. D. Modulation of xenobiotic metabolising enzymes by anticarcinogens - focus on glutathione S-transferases and their role as targets of dietary chemoprevention in colorectal carcinogenesis. Mutat. Res. 591, 74–92 (2005).
Mpofu, D. A., Marume, U., Mlambo, V. & Hugo, A. The effects of Lippia javanica dietary inclusion on growth performance, carcass characteristics and fatty acid profiles of broiler chickens. Anim. Nutr. 2, 160–167 (2016).
Park, J. Y., Lee, S. H., Kim, K. R., Park, J. B. & Oh, D. K. Production of 13S-hydroxy-9(Z)-octadecenoic acid from linoleic acid by whole recombinant cells expressing linoleate 13-hydratase from Lactobacillus acidophilus. J. Biotechnol. 208, 1–10 (2015).
Ortega-Anaya, J. & Hernandez-Santoyo, A. Production of bioactive conjugated linoleic acid by the multifunctional enolase from Lactobacillus plantarum. Int. J. Biol. Macromol. 91, 524–535 (2016).
Feeding Standard of Swine. China Agriculture Press, Beijing, China (2004).
Trapnell, C., Pachter, L. & Salzberg, S. L. Top Hat: discovering splice junctions with RNA-Seq. Bioinformatics. 25, 1105–11 (2009).
Trapnell, C., Hendrickson, D. G., Sauvageau, M., Goff, L. & Rinn, J. L. Differential analysis of gene regulation at transcript resolution with RNA-seq. Nat. Biotechnol. 31, 46–53 (2013).
Xie, C. et al. KOBAS 2.0: a web server for annotation and identification of enriched pathways and diseases. Nucleic Acids Res. 39, 316–322 (2011).
Sukhija, P. S. & Palmquist, D. L. Rapid method for determination of total fatty acid content and composition of feedstuffs and feces. J. Agric. Food Chem. 36, 1202–1206 (1988).
Acknowledgements
This study was funded as a special project by a special project of the Beijing Municipal Science & Technology Commission (grant number Z141100002614002), Modern Agri-industry Technology Research System, Beijing Innovation Consortium of Swine Research System (grant number BAIC02-2016), the Scientific and Technological Innovation Ability Construction project of the Beijing Academy of Agriculture and Forestry Science (grant number KJCX20150404). We thank the Beijing Xiqingminfeng Farm (Beijing, China) for their assistance with these studies.
Author information
Authors and Affiliations
Contributions
Z.D.Y. performed experiments, analysed the data and wrote the manuscript. qPCR was performed by S.T.T., H.Y. analysed the fatty acid data. L.H. and W.S.X. helped with the tables and figures. W.J. and W.Y.M. performed the animal experiments. J.H.F. designed the experiments and analysed the sequence data. Z.R.J. conceived of and designed the experiments and revised the manuscript. All authors contributed to writing the manuscript and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, D., Shang, T., Huang, Y. et al. Gene expression profile changes in the jejunum of weaned piglets after oral administration of Lactobacillus or an antibiotic. Sci Rep 7, 15816 (2017). https://doi.org/10.1038/s41598-017-16158-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-16158-y
This article is cited by
-
Sex-dependent changes in the microbiota profile, serum metabolism, and hormone levels of growing pigs after dietary supplementation with Lactobacillus
Applied Microbiology and Biotechnology (2021)
-
N-acetylcysteine modulates lipopolysaccharide-induced intestinal dysfunction
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.