Abstract
We conducted a retrospective study based on the Surveillance, Epidemiology, and End Results Program (SEER) database to establish a novel nomogram prognostic prediction system and to estimate the association between overall survival and prognostic factors, as well as to explore surgical treatment strategies for adenocarcinoma of the jejunum and ileum. A total of 883 patients from the SEER database were included in this study. Eight potential prognostic factors were included in a nomogram model and discriminatory power and accuracy were examined using the Harrell’s C-index and Akaike Information Criterion (AIC) index. In comparison with the AJCC TNM staging system, the nomogram prediction system was more accurate and homogeneous (Harrell’s C-index, 0.731 vs. 0.667; AIC index, 4852.9 vs. 4913.723). For surgical management, resection of more than 12 local lymph nodes could improve the likelihood of survival. This study demonstrates that our nomogram model is more accurate and homogeneous than the traditional AJCC TNM staging system, and proper surgical strategies for mesenteric lymphadenectomy improve overall survival.
Similar content being viewed by others
Introduction
Small bowel neoplasms, whether benign or malignant, are extremely rare1. The American Cancer Society estimates that there will be approximately 10190 new cases and 1390 deaths from small bowel neoplasms in 20172. Although the mucosa surface of the small bowel accounts for more than 90% of the gastrointestinal tract, only 3% of neoplasms of the digestive tract originate in the small bowel1. Due to this relatively rare incidence, little clinical attention is paid to these neoplasms.
There are over 40 different histological types in both benign and malignant small bowel neoplasms3. Adenomas, lipomas and leiomyomas are the major types of benign neoplasms4,5, while adenocarcinomas and carcinoids (neuroendocrine tumours) are the most common types of the malignant tumours6. Several high-risk conditions also warrant additional attention due to their association with an increased risk of adenocarcinoma of the small bowel: familial adenomatous polyposis (FAP), Lynch syndrome, Peutz-Jeghers syndrome and Crohn’s disease7,8,9,10.
In comparison with other gastrointestinal neoplasms, small bowel tumours always present as non-specific complaints such as irregular abdominal pain, diarrhoea, gastrointestinal bleeding or intestinal obstruction, making it difficult to correctly diagnose the neoplasm11. Benign tumours often remain asymptomatic and are discovered either during surgery or occasionally during an autopsy. In recent years, the development of advanced imaging equipment and techniques enables the use of more reliable and accurate methods to detect these lesions in the small bowel12.
Nomogram, a graphical mathematical model that estimates several clinical factors in an integrative way, is considered to be a reliable clinical outcomes prediction tool13,14. After including significant risk factors, a nomogram algorithm can statistically estimate and predict several clinical endpoint events, such as prognosis and overall survival15,16. To date, nomogram models have been applied to prognostic prediction in many types of cancers, such as nasopharyngeal carcinoma (NPC), small cell lung cancer (SCLC), gynaecologic malignancies and gastrointestinal tumours, and are considered to be a more precise model than current staging systems17,18,19,20. Unfortunately, prognostic predicting tools for small bowels malignancies are rare.
Regarding small bowels malignancies, several studies have explored several prognostic factors, including race, gender, age, marital status, TNM staging and cancer-specific survival rate21,22,23,24. A number of studies have reported an association between the number of mesenteric lymph nodes (LNs) resected and patients’ prognosis25,26. Although lymphadenectomy is recommended in the National Comprehensive Care Network (NCCN) guidelines27, the impact on prognosis, particularly for patients with adenocarcinoma of the jejunum and ileum, is still undetermined. In our study, using the Surveillance, Epidemiology, and End Results (SEER) database, an open cancer statistical database established by the National Cancer Institute (NCI)28, we explored several prognostic factors and the surgical management of mesenteric lymph nodes of adenocarcinoma of the jejunum and ileum.
Results
Demographic and clinicopathological characteristics
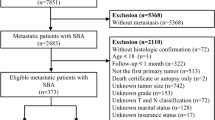
Overall, 883 patients with adenocarcinoma of the jejunum and ileum were identified in the study cohort. Within this cohort, approximately half were male (n = 501, [56.7%]). The median age at diagnosis was 63 years (ranged from 20 to 100 years old), with average age of 62.88 ± 12.47 years. Regarding clinicopathologcal characteristics, half of the patients (n = 490, [55.5%]) were diagnosed with jejunum adenocarcinoma, while the remaining 393 patients were diagnosed with ileum adenocarcinoma. In 492 patients (55.7%), the adenocarcinoma had invaded the serosa, in the remaining 308 patients (34.9%), the tumour had invaded the serosa. Only 23.1% of patients (n = 204) received a lymph nodes examination. Additionally, 186 patients (21.1%) were diagnosed with distant metastasis, and nearly 283 patients (33%) were estimated to have poorly or undifferentiated adenocarcinoma of the jejunum or ileum.
Overall survival and prognosis predictive factors for patients with adenocarcinoma of the jejunum and ileum
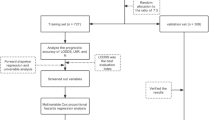
In this study, 10 clinicopathological factors were selected as potential prognosis predictors from the SEER database: age at diagnosis, gender, race, marital status, status of primary tumours (T stage), examined lymph nodes, lymph nodes status (N stage), histological grade, status of distance metastasis (M stage) and tumour locations (Table 1). Using a Cox proportional hazards regression model, seven factors were identified as associated with overall survival: age, marital status, T stage, N stage, number of lymph nodes examined, M stage and histological grade. Except for the histological grade, all factors were significantly associated with overall survival in multivariate analysis (P < 0.05).
Nomogram prediction system for patients with adenocarcinoma of the jejunum and ileum
The nomogram prediction system was a novel model to estimate overall survival based on eight prognostic factors: histological grade, the TNM stage, age at diagnosis, number of lymph nodes examined, gender and primary tumour site. Each factor was ascribed a weighted point and the total points implied the prognosis. For example, 70 years of age was associated with 53 points, female was associated with zero points, depth of invasion (T2) was associated with zero points, no metastasized lymph nodes (N0) was associated with zero points, 30 examined lymph nodes was associated with three points, well-differentiated adenocarcinoma was associated with zero points, and distant metastasis was associated with 100 points, leading to a total score of 156. In addition, for each patient with adenocarcinoma of the jejunum or ileum, more total points in nomogram model indicated a worse prognosis. The factors and final nomogram model that estimate overall survival are shown in Fig. 1. Given that age was a non-linear prognostic factor, the restricted cubic spline (RCS) curve and hazard ratio are shown in Fig. 2.
To evaluate the predictive accuracy in both the nomogram prediction system and traditional AJCC TNM staging system, the Harrell’s C-index and AIC index were calculated. For the nomogram model, the Harrell’s C-index was 0.731 versus 0.667 in AJCC TNM system, indicating that our nomogram model was more accurate or discriminative in comparison with the traditional AJCC TNM staging system. Figure 3 presents the calibration plots of 5-year survival in the nomogram model, which revealed an overall survival predicted by nomogram model that closely approached the actual survival and was within a 10% margin of error. The AIC index was also calculated to avoid overfitting the prognostic models, and was 4852.9 for nomogram model and 4913.723 for AJCC TNM system, respectively. These results demonstrated that the nomogram model did not overestimate actual overall survival in this cohort and could be a better prognosis prediction system than the traditional AJCC TNM staging system.
Surgical management of adenocarcinoma of the jejunum and ileum
Proper and beneficial surgical management strategies for patients with adenocarcinoma of the jejunum and ileum are still unclear. As mesenteric lymph nodes metastasis could be a vital factor in both prognostic prediction and therapeutic strategy, we explored the correlation between overall survival and the number of lymph nodes intraoperatively resected. In our study we analysed this correlation using an X-tile test. This test was used to determine the optimal cutoff point for predicting cancer-specific survival according to the number of lymph nodes examined. We observed that a resection of 5 or 12 lymph nodes could significantly improve overall survival (Fig. 4A and B). In consideration of the biological behaviour of adenocarcinoma of the jejunum and ileum and the guidelines for other gastrointestinal adenocarcinomas29, resection of more than 12 local lymph nodes could benefit patients with adenocarcinoma of the jejunum and ileum. In our study, these patients had improved cancer-specific survival (P < 0.05) (Fig. 4C).
(A) The distribution of number of patients with adenocarcinomas of jejunum or ileum according to lymph nodes examined. (B) X-tile plots for number of lymph nodes constructed by patients with adenocarcinomas of jejunum or ileum. The plots show the χ2 log-rank values produced, dividing them into 2 groups by the cutoff point 12. (C) Kaplan-Meier survival curves of cumulative survival of patients with adenocarcinomas of jejunum or ileum.
Discussion
Malignant neoplasms of the jejunum and ileum, particularly adenocarcinomas, are difficult to diagnosis and estimate before proper therapies because of their rare incidence and asymptomatic characteristics. Based on the selected risk factors, we assessed the prognosis of patients with adenocarcinoma of the jejunum and ileum using our newly established nomogram prediction model, which demonstrated higher accuracy and improved homogeneity as compared to traditional TNM stages. Notably, we determined that patients who underwent lymphadenectomy for more than 12 local lymph nodes could improve 5-year survival (Fig. 4C). This trend was in accordance with previous studies for patients with neuroendocrine tumours of the small bowel26,30.
In this study, we investigated several clinical characteristics that could potentially be prognostic predictors and survival outcomes of patients with adenocarcinoma of the jejunum and ileum. Consistent with previous studies, young age was a positive prognosis prediction factors in malignant tumours of the small bowel22,31. Additionally, marital status could be considered to be a protective factor, as we observed a Hazard Ratio of approximately 0.90 in married patients in comparison with single patients. However, in our study, we did not identify any significant differences in gender, race or locations of tumours, which might be other potential predictors in further investigations.
To analyse the association between survival outcomes and the potential prognostic factors, we utilized a nomogram prediction model as a novel prognosis prediction system and estimated the accuracy and homogeneity of both the nomogram model and the traditional AJCC TNM system. Based on the prognostic data of 883 patients who underwent surgery, we analysed eight factors to estimate the prognostic outcomes of those patients with adenocarcinoma of the jejunum and ileum. In the nomogram model, each factor was ascribed a weighted point to evaluate the effect on overall prognosis and the sum of the weighted points was considered to be an integrated factor to analyse the association between these prediction factors and overall prognosis: a higher scores indicated a worse prognosis. Furthermore, we examined the prediction accuracy and homogeneity of the nomogram model in comparison with the traditional AJCC TNM system. Harrell’s C indices and AIC indices were calculated, the results suggested our nomogram model was relatively close to the actual survival and more homogeneous (AIC index of 4852.9 for the nomogram model versus 4913.723 for the AJCC TNM staging system) than the traditional TNM system.
Surgical treatment is considered to be an important therapeutic strategy in gastrointestinal tumours27. Due to the rare incidence of adenocarcinoma of the jejunum or ileum, treatment and management strategies are still controversial. In previous studies, surgical excision has been demonstrated as a proper first-line therapy for patients with small bowel neoplasms, especially in carcinoid tumours of the small bowel and adenocarcinomas of the duodenum32,33. In surgery, local lymph nodes, including mesentery lymph nodes, are considered to be one of the essential factors tightly correlated with overall rates of survival. In our study drawing from a cohort of SEER database patients, we observed that lymphadenectomy of local lymph nodes could potentially predict prognosis. For patients with no nodal metastasis, intraoperativly increasing lymph nodes examination was significantly associated with an improvement in survival. Previous studies have shown that ≥ 15 lymph nodes resected for adenocarcinoma of the duodenum and ≥ 8 lymph nodes resected for carcinoid of the small bowel were positively correlated with overall survival26,34. In alignment with these studies, we analysed the clinicopathological data of 883 patients with adenocarcinoma of the jejunum and ileum and found that lymphadenectomy for more than 12 lymph nodes was significantly associated with positive patient prognosis (P < 0.022). This result also demonstrated that local lymph node estimation was essential for both stage assessment and the prognosis prediction system. Local lymph nodes identification is vital for both survival prediction and the management of therapies for adenocarcinoma of the small bowel. Because of its potentially metastatic behaviour, in adenocarcinoma of the duodenum, pancreaticoduodenectomy combined with extant lymphadenectomy, such as in patients with > 15 lymph nodes resected as mentioned previously, is essential and necessary for a better prognosis. In contrast, due to the rare incidence of adenocarcinoma of the jejunum and ileum invading contiguous organs, wide excision of tumours is easier to process. However, our study still holds the view that extant lymphadenectomy for more than 12 lymph nodes could improve prognosis for patients with adenocarcinoma of the jejunum and ileum.
Although increasing lymph nodes resection for patients with small bowel neoplasms might improve survival, challenges still exist during lymphadenectomies. Compared with other gastrointestinal tumours, adenocarcinoma of the jejunum or ileum presents more atypically and in emergency scenarios, such as with intestinal obstruction or bleeding, which makes it difficult for surgeons to identify the status of local lymph nodes. Additionally, because of stimulation from growth factors secreted from tumours or mesentery, fibrotic reaction could result leading to encasement of mesenteric vessels26. Excessive resection could damage these vessels and threaten the blood supply to the small bowel, leading to an incomplete local mesenteric lymphadenectomy of more than 12 lymph nodes.
It should be noted that multidisciplinary therapies, such as surgical resection combined with adjuvant chemotherapies, have been widely utilized for suitable patients with adenocarcinoma of the jejunum or ileum. Several clinical trials are exploring proper chemotherapeutic strategies and other factors impacting the efficacy of various chemotherapy reagents35,36. For instance, Gao, et al. elucidated that patients with Stage II colorectal cancer (CRC) could gain survival benefits from fluorouracil-based chemo reagents by utilizing a novel cancer hallmark-based gene signatures sets (CSS sets) model based on analysing 1005 patients with Stage II CRC from 13 cohorts37. Thus, precise and robust prognostic predictors are required and need to be further explored for multidisciplinary therapeutic management, particularly for adjuvant chemotherapy. For patients with Stage IV adenocarcinoma, peritoneal metastases (PM) occurs more easily in the jejunum or ileum than in duodenum because of its unique biological behaviour in its invasion into the peritoneum38. Systematic therapies for Stage IV adenocarcinoma of the jejunum or ileum warrant further investigation, whereas in case of local lesions of adenocarcinoma combined with PM, the proper therapeutic strategy is cytoreductive surgery followed by hyperthermic intraperitoneal chemotherapy (HIPEC), benefitting patients with PM or pseudomyxoma peritonei (PMP)39.
In addition, for prediction algorithms which are applied for patients with malignant tumours, accuracy and concordance or homogeneous are wildly considered. In comparison with the traditional TNM staging system, which only considers tumour size and extension (T stage), lymph nodes involvement (N stage) and distant metastasis (M stage)17, a nomogram prediction model with multiple factors has major benefits. Except for clinicopathological predictors, algorithms focusing on metastasis-driving genes or mutant gene signal pathways could also be precise prognosis prediction models based on the statistical analysis of tumour gene microarrays and Multiple Survival Screening (MSS) algorithm40. In breast cancer, the PIK3CA-mutated signalling pathway and 26 S proteasome genes were proved to be associated with patients’ prognosis by gene microarrays and signalling network analysis41,42. Therefore, a potential prognostic prediction model combining clinicopathological characteristics and bioinformatics approaches, such as genome sequencing data and network analysis, could be a more accurate and robust model, which requires further exploration.
Although our study is based on 10 years SEER database data, there are still several limitations. First, this study utilizes retrospective population-based data across several specific states in the United States but not worldwide. Second, certain clinical details such as specific details of chemotherapy, the approaches of surgery or information on disease-free survival were not complete, which may influence the prognosis of patients in our cohort. Furthermore, the histological grade might cause bias due to relatively subjective diagnostic standards among different pathologists or clinicians.
In summary, our study demonstrated that the nomogram prognosis prediction model could estimate overall survival more accurately and with improved homogeneity as compared to the traditional AJCC TNM staging system. Additionally, our analysis elucidated that mesenteric lymphadenectomy for patients with adenocarcinoma of the jejunum or ileum could improve overall survival.
Patients and Methods
Patients
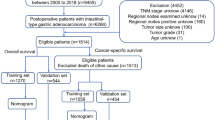
In our study, data collected included the demographic and clinicopathological characteristics and survival of patients with adenocarcinoma of the ileum and jejunum. All patients were identified between 2003 and 2014 in the SEER database. The inclusion criteria were follows: 1. Patients clinicopathologically diagnosed with adenocarcinoma of the ileum or jejunum; 2. Patients who underwent surgery and for whom exact pathological details were available; 3. Patients who survived for more than three months postoperatively. In our study, a signed SEER research data agreement form was provided to the SEER program and we were given approval to access and analyse SEER data.
Statistical analysis
All patients were regrouped according to the 7th AJCC TNM staging system43. Continuous data were presented as means ± standard deviations. Categorical variables were compared using the χ2 test or Fisher’s exact test. Continuous variables were compared using the Student’s t-test. Both univariate and multivariate Cox proportional hazard regression models were used to explore the association between all factors and overall survival. Overall survival was estimated using the Kaplan-Meier estimator, and differences in survival were examined using the log-rank test. Restricted cubic splines were applied to transform continuous non-liner prediction factors to fit the test statistic. X-tile analysis was used to determine the optimal cutoff point for predicting survival according to the number of lymph nodes examined in patients with adenocarcinoma of the ileum and jejunum. Preselected multiple potential factors were tested as nomogram parameters irrespective of significance. To estimate the accuracy and identification abilities of those predictors, Akaike’s Information Criterion (AIC) and Harrell’s C statistic were used in this study. All statistical tests were two-sided, and P values < 0.05 were considered to be statistically significant. Statistical analyses were performed using SPSS 13.0 and R software version 3.3.0 (http://www.r-project.org) with the “SEERaBomb”, “rms” and “AICcmodavg” packages.
References
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2015. CA: a cancer journal for clinicians 65, 5–29, https://doi.org/10.3322/caac.21254 (2015).
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer Statistics, 2017. CA: a cancer journal for clinicians 67, 7–30, https://doi.org/10.3322/caac.21387 (2017).
O’Riordan, B. G., Vilor, M. & Herrera, L. Small bowel tumors: an overview. Digestive diseases (Basel, Switzerland) 14, 245–257 (1996).
Lee, Y. T. Leiomyosarcoma of the gastro-intestinal tract: general pattern of metastasis and recurrence. Cancer treatment reviews 10, 91–101 (1983).
Gill, S. S., Heuman, D. M. & Mihas, A. A. Small intestinal neoplasms. Journal of clinical gastroenterology 33, 267–282 (2001).
Neugut, A. I., Jacobson, J. S., Suh, S., Mukherjee, R. & Arber, N. The epidemiology of cancer of the small bowel. Cancer epidemiology, biomarkers & prevention: a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology 7, 243–251 (1998).
Koornstra, J. J. Small bowel endoscopy in familial adenomatous polyposis and Lynch syndrome. Best practice & research. Clinical gastroenterology 26, 359–368, https://doi.org/10.1016/j.bpg.2012.01.022 (2012).
Westerman, A. M. & Wilson, J. H. Peutz-Jeghers syndrome: risks of a hereditary condition. Scandinavian journal of gastroenterology. Supplement 230, 64–70 (1999).
Koornstra, J. J., Kleibeuker, J. H. & Vasen, H. F. Small-bowel cancer in Lynch syndrome: is it time for surveillance? The Lancet. Oncology 9, 901–905, https://doi.org/10.1016/s1470-2045(08)70232-8 (2008).
Ribeiro, M. B. et al. Adenocarcinoma of the small intestine in Crohn’s disease. Surgery, gynecology & obstetrics 173, 343–349 (1991).
Pourmand, K. & Itzkowitz, S. H. Small Bowel Neoplasms and Polyps. Current gastroenterology reports 18, 23, https://doi.org/10.1007/s11894-016-0497-x (2016).
Horton, K. M., Kamel, I., Hofmann, L. & Fishman, E. K. Carcinoid tumors of the small bowel: a multitechnique imaging approach. AJR. American journal of roentgenology 182, 559–567, https://doi.org/10.2214/ajr.182.3.1820559 (2004).
Iasonos, A., Schrag, D., Raj, G. V. & Panageas, K. S. How To Build and Interpret a Nomogram for Cancer Prognosis. Journal of Clinical Oncology 26, 1364–1370, https://doi.org/10.1200/jco.2007.12.9791 (2008).
Balachandran, V. P., Gonen, M., Smith, J. J. & DeMatteo, R. P. Nomograms in oncology: more than meets the eye. The Lancet Oncology 16, e173–e180, https://doi.org/10.1016/s1470-2045(14)71116-7 (2015).
Wang, Y. et al. Prognostic nomogram for intrahepatic cholangiocarcinoma after partial hepatectomy. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 31, 1188–1195, https://doi.org/10.1200/jco.2012.41.5984 (2013).
Liang, W. et al. Development and validation of a nomogram for predicting survival in patients with resected non-small-cell lung cancer. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 33, 861–869, https://doi.org/10.1200/jco.2014.56.6661 (2015).
Liang, W. et al. Development and validation of a nomogram for predicting the survival of patients with non-metastatic nasopharyngeal carcinoma after curative treatment. Chinese journal of cancer 35, 98, https://doi.org/10.1186/s40880-016-0160-9 (2016).
Pan, H. et al. Nomogram prediction for the survival of the patients with small cell lung cancer. Journal of thoracic disease 9, 507–518, https://doi.org/10.21037/jtd.2017.03.121 (2017).
Polterauer, S. et al. Nomogram prediction for overall survival of patients diagnosed with cervical cancer. British journal of cancer 107, 918–924, https://doi.org/10.1038/bjc.2012.340 (2012).
Han, D. S. et al. Nomogram predicting long-term survival after d2 gastrectomy for gastric cancer. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 30, 3834–3840, https://doi.org/10.1200/jco.2012.41.8343 (2012).
Lepage, C., Rachet, B. & Coleman, M. P. Survival from malignant digestive endocrine tumors in England and Wales: a population-based study. Gastroenterology 132, 899–904, https://doi.org/10.1053/j.gastro.2007.01.006 (2007).
Wu, L. et al. Survival outcomes and surgical intervention of small intestinal neuroendocrine tumors: a population based retrospective study. Oncotarget 8, 4935–4947, https://doi.org/10.18632/oncotarget.13632 (2017).
Aydin, D. et al. Evaluation of prognostic factors and treatment in advanced small bowel adenocarcinoma: report of a multi-institutional experience of Anatolian Society ofMedical Oncology (ASMO). Journal of B.U.ON.: official journal of the Balkan Union of Oncology 21, 1242–1249 (2016).
Dabaja, B. S., Suki, D., Pro, B., Bonnen, M. & Ajani, J. Adenocarcinoma of the small bowel: presentation, prognostic factors, and outcome of 217 patients. Cancer 101, 518–526, https://doi.org/10.1002/cncr.20404 (2004).
Hellman, P. et al. Effect of surgery on the outcome of midgut carcinoid disease with lymph node and liver metastases. World journal of surgery 26, 991–997, https://doi.org/10.1007/s00268-002-6630-z (2002).
Landry, C. S. et al. Resection of at-risk mesenteric lymph nodes is associated with improved survival in patients with small bowel neuroendocrine tumors. World journal of surgery 37, 1695–1700, https://doi.org/10.1007/s00268-013-1918-8 (2013).
Kulke, M. H. et al. Neuroendocrine tumors, version 1.2015. Journal of the National Comprehensive Cancer Network: JNCCN 13, 78–108 (2015).
Altekruse, S. F. et al. SEER cancer registry biospecimen research: yesterday and tomorrow. Cancer epidemiology, biomarkers & prevention: a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology 23, 2681–2687, https://doi.org/10.1158/1055-9965.EPI-14-0490 (2014).
Benson, A. B. III et al. Metastatic colon cancer, version 3.2013: featured updates to the NCCN Guidelines. Journal of the National Comprehensive Cancer Network: JNCCN 11, 141–152, quiz152 (2013).
Landerholm, K., Zar, N., Andersson, R. E., Falkmer, S. E. & Jarhult, J. Survival and prognostic factors in patients with small bowel carcinoid tumour. The British journal of surgery 98, 1617–1624, https://doi.org/10.1002/bjs.7649 (2011).
Bilimoria, K. Y. et al. Small bowel cancer in the United States: changes in epidemiology, treatment, and survival over the last 20 years. Annals of surgery 249, 63–71, https://doi.org/10.1097/SLA.0b013e31818e4641 (2009).
Coco, C., Rizzo, G., Manno, A., Mattana, C. & Verbo, A. Surgical treatment of small bowel neoplasms. European review for medical and pharmacological sciences 14, 327–333 (2010).
Han, S. L. et al. Surgically treated primary malignant tumor of small bowel: a clinical analysis. World journal of gastroenterology 16, 1527–1532 (2010).
Ecker, B. L. et al. Lymph node evaluation and survival after curative-intent resection of duodenal adenocarcinoma: A matched cohort study. European journal of cancer (Oxford, England: 1990) 69, 135–141, https://doi.org/10.1016/j.ejca.2016.09.027 (2016).
Halfdanarson, T. R., McWilliams, R. R., Donohue, J. H. & Quevedo, J. F. A single-institution experience with 491 cases of small bowel adenocarcinoma. American journal of surgery 199, 797–803, https://doi.org/10.1016/j.amjsurg.2009.05.037 (2010).
Ecker, B. L. et al. Efficacy of adjuvant chemotherapy for small bowel adenocarcinoma: A propensity score-matched analysis. Cancer 122, 693–701, https://doi.org/10.1002/cncr.29840 (2016).
Gao, S. et al. Identification and Construction of Combinatory Cancer Hallmark-Based Gene Signature Sets to Predict Recurrence and Chemotherapy Benefit in Stage II Colorectal Cancer. JAMA oncology 2, 37–45, https://doi.org/10.1001/jamaoncol.2015.3413 (2016).
Rovers, K. P., de Bree, E., Yonemura, Y. & de Hingh, I. H. Treatment of Peritoneal Metastases from Small Bowel Adenocarcinoma. International journal of hyperthermia: the official journal of European Society for Hyperthermic Oncology, North American Hyperthermia Group 1–35, https://doi.org/10.1080/02656736.2016.1266700 (2016).
Elias, D. et al. Role of hyperthermic intraoperative peritoneal chemotherapy in the management of peritoneal metastases. European journal of cancer (Oxford, England: 1990) 50, 332–340, https://doi.org/10.1016/j.ejca.2013.09.024 (2014).
Li, J. et al. Identification of high-quality cancer prognostic markers and metastasis network modules. Nat Commun 1, 34, https://doi.org/10.1038/ncomms1033 (2010).
McGee, S. R., Tibiche, C., Trifiro, M. & Wang, E. Network Analysis Reveals A Signaling Regulatory Loop in the PIK3CA-mutated Breast Cancer Predicting Survival Outcome. Genomics, proteomics & bioinformatics 15, 121–129, https://doi.org/10.1016/j.gpb.2017.02.002 (2017).
Fu, C., Li, J. & Wang, E. Signaling network analysis of ubiquitin-mediated proteins suggests correlations between the 26S proteasome and tumor progression. Molecular bioSystems 5, 1809–1816, https://doi.org/10.1039/B905382D (2009).
Egner, J. R. AJCC cancer staging manual. JAMA 304, 1726–1727 (2010).
Acknowledgements
We thank all authors of included studies in this study. We also thank the Surveillance, Epidemiology, and End Results (SEER) database which provided the free data, and free R software which is needed in analyzing program (http://www.r-project.org). This research was supported by the National Natural Science Foundation of China (No. 81502442).
Author information
Authors and Affiliations
Contributions
All authors have participated in the conception and design of this study. X.X., Z.J.Z. and Y.C.S. have been in charge of data obtainment and analysis. X.X. and Z.J.Z. drafted the manuscript. C.X.D. and H.Z. have revised the manuscript before submission. All authors have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xie, X., Zhou, Z., Song, Y. et al. Surgical Management and Prognostic Prediction of Adenocarcinoma of Jejunum and Ileum. Sci Rep 7, 15163 (2017). https://doi.org/10.1038/s41598-017-15633-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-15633-w
This article is cited by
-
Small Bowel Tumors – Case Series Analysis: Prognostic Factors and Survivals
Indian Journal of Surgery (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.