Abstract
We, for the first time, provide the experimental demonstration on the band gap engineering of layered hexagonal SnSe2 nanostructured thin films by varying the thickness. For 50 nm thick film, the band gap is ~2.04 eV similar to that of monolayer, whereas the band gap is approximately ~1.2 eV similar to that of bulk for the 1200 nm thick film. The variation of the band gap is consistent with the the theoretically predicted layer-dependent band gap of SnSe2. Interestingly, the 400–1200 nm thick films were sensitiveto 1064 nm laser iradiation and the sensitivity increases almost exponentiallly with thickness, while films with 50–140 nm thick are insensitive which is due to the fact that the band gap of thinner films is greater than the energy corresponding to 1064 nm. Over all, our results establish the possibility of engineering the band gap of SnSe2 layered structures by simply controlling the thickness of the film to absorb a wide range of electromagnetic radiation from infra-red to visible range.
Similar content being viewed by others
Introduction
Single and few layered metal dichalogenide materials such as WS2, MoS2, MoSe2, MoTe2, TaS2 have been widely explored to develop various electronic devices such as transistors1, photo-detectors2, energy storage3, and humidity sensors4,5,6, etc because of their unique layer dependent structural and electronic properties such as tunable band gaps4,5,6,7,8,9. Among the layered materials, SnSe2 is an earth abundant n-type binary semiconductor whose band gap can be tuned for a wide range of electromagnetic spectrum from 1–2 eV making SnSe2 attractive material for various elctronic device applications7. The semiconducting nature of SnSe2 was discoveved in 1955 during an investigation into the fundamental factors that are responsible for intrinsic semiconductivity in certain intermetallic compounds and compounds formed by the metalloids Se and Te8. The carrier concentration varies from 1017–1019 cm−3, electron mobilities µe between 0.6–85 cm2/V s9,10,11,12,13,14,15, a direct and indirect band gap between 0.9–2.04 eV13,16,17,18, high absorption coefficient of >104 cm−1. It crystallizes in the CdI2 hexagonal lattice19 and is a prototype of transitional metal dichalogenides, composed of two-dimensional Se-Sn-Se sheets stacked on the top of one another and characterized by strong covalent bonding between Se-Sn-Se atoms and week interlayer Van der Waal’s bonding.
SnSe2 thin films have been prepared by spin coating20,21, spray pyrolysis14,22,23, chemical vapour deposition(CVD)15,18, molecular beam epitaxy10, thermal evaporation of Sn and Se elements24, and sputtering25. Here, we deposited Sn films of various thicknesses on soda lime glass (SLG) substrate and annealed the films in selenium atmosphere for 1 h at 450 °C to obtain hexagonal SnSe2. Interestingly, the band gap varied from 2.04 eV for 50 nm thick film to 1.20 eV for 1200 nm thick one. Furthermore, 400–1200 nm thick films were shown to be sensitive to 1064 nm laser iradiation, while films with 50–140 nm thick is insensitive which is due to the fact that the band gap of thinner films is greater than the energy corresponding to 1064 nm. Over all, our results establish the possibilty of engineering the band gap of SnSe2 layered structure by simply controlling the thickness of the film to absorb a wide range of electromagnetic radiation from infra-red to visible range.
Results and Discussion
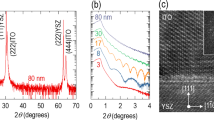
The thickness of the deposited Sn films on SLG varied from 30–600 nm and after selenisation, it increased to 50–1200 as shown in Figure S1 (Supporting Information) and were measured using Dektak profilometer (deviation is ± 10 nm) and Atomic force microscopy (AFM) for 50 nm thick film only. The overall thickness of SnSe2 films is almost double the thickness of the Sn films. X-ray diffraction (XRD) patterns of SnSe2 thin films are shown in Fig. 1a and matches well with the ICSD data as evident from Fig. 1b. It is observed that hexagonal pure phase of SnSe2 with space group P-3 m1 was formed with no evidence of any secondary phases. Rietveld refinement using GSAS26 software program was used to fit the crystal structure for the 1000 thick film as shown in Fig. 1c. The criteria for fitting is based on least squares refinement theory which gives indicators of the quality of refinement (SI). The details of the least squares refinement and associated parameters obtained in this study are shown in SI (Tables S1 and S2). The fitting parameters yield a = b = 0.3818 nm and c = 0.6152 nm with a strain of 0.12% from the Williamson-Hall plot (Fig. 1d).
Figure S2 shows the room temperature Raman spectra of SnSe2 films. From Figure S2b, two Raman active modes at 115 and 183.5 cm−1 are observed which belong to SnSe2 hexagonal phase6,10,19,25,27,28. The peak at 115 cm−1 is for Eg mode and is due to in-plane strecthing, while the peak located at 183.5 cm−1 is for A1g mode due to out of plane stretching of selenium atoms29. The intensity of A1g mode increases with the thickness (Figure S2c) as is the case of layer dependency7.
In order to investigate the chemical electronic states of the prepared SnSe2 thin films, x-ray photoelectron spectroscopy (XPS) study was performed. In the survey spectrum of SnSe2, the peaks corresponding to Sn3d, C1s and Se3d were identified as shown in Fig. 2a. In addition, O peak observed may be from the SLG substrate. The spectra of Sn3d and Se3d were measured to determine the oxidation states of the constituent elements. The spectra of the constituent elements is shown in Fig. 2b and c. Sn3d state splits into two states of Sn3d3/2 and Sn3d5/2 with binding energy of 493.01 and 484.55 eV respectively with a peak to peak separation of 8.46 eV, which is an indication of Sn4+ 30,31,32. The peaks at 54.46 and 53.59 eV are related to Se3d3/2 and Se3d5/2 states, respectively30,31,32,33,34. Figure S3 shows the energy dispersive x-ray (EDX) spectrum of 1200 nm thick SnSe2 films. The presence of Cu and C in the spectrum emerges from carbon coated copper grids. We can conclude that the SnSe2 thin films on SLG, confirms the oxidation states of constituent elements of Sn and Se with no presence of other oxidation states.
Transmission electron microscope (TEM), high resolution TEM (HRTEM) images and selected area electron diffraction (SAED) pattern shown in Fig. 3(a–f) confirm SnSe2 hexagonal crystal structure. The strong reflections in SAED pattern shown in Fig. 3b and HRTEM fringes presented in Fig. 3c and d strongly confirm further the high cystallinity and layered hexagonal crystal structure of SnSe2 with inter layer distances of about 0.643 nm15. The obtained fringes of ~0.6430 and 0.291 nm in HRTEM images shown in Fig. 3(c–e) and f correspond to (001) and (100) crystal plane of the hexagonal crystal system35,36. In the SAED pattern (Fig. 3b), the crystal planes of (101), (003), (202) and (301) correspond to d-spacings of 0.2894, 0.2100, 0.1475 and 0. 1034 nm, respectively of the hexagonal crystal system with space group P-3m118,37,38. These results are consistent with XRD planes for the hexagonal crystal system with space group P-3m1 shown in Fig. 1b. HRTEM images shown in Fig. 3(g–i) for 900, 400 and 140 nm thick films also show similar features as other thicknesses. The diffraction spots in SAED of Fig. 3b appear elongated and may suggest the formation of small crystalline domains inside the SnSe2 layers which are disoriented with respect to each other39,40.
The optical properties of SnSe2 thin films on SLG substrate were evaluated by taking diffuse reflectance spectra (DRS) between 300–2000 nm of wavelength as shown in Fig. 4a. Using the Kubelka-Munk (KM) function41, the DRS was then converted to an equivalent absorption spectra as shown in Fig. 4b. The KM function at any wavelength is given by \(F({R}_{\infty })=\frac{{(1-{R}_{\infty })}^{2}}{2{R}_{\infty }}=\frac{\alpha }{S}\), where \({R}_{\infty }\) is the reflectance of the film relative to the reference material (i.e. \(\frac{{R}_{sample}}{{R}_{reference}}\)), α is the absorption coefficient and S is the scattering coefficient. The scattering coefficient is weakly dependent on energyand therefore, \(F({R}_{\infty })\) is assumed to be proportional to the absorption42. The optical band gap of SnSe2 thin films were then estimated from Tauc plot43 as shown in Fig. 4c and Figure S4 i.e. a plot of (αhυ)2 against hυ for direct band gap material and the variation in band gap with thickness is shown in Fig. 4d. From Fig. 4d, it can be noted that the band gap depends on the thickness of the film which in turn depends on the number of layers7. It is interesting to note that the variation of energy corresponding to maximum absorption is similar to that of band gap (Figure S5) and the band gap varies linearly with the inverse of the thickness (inset of Fig. 4d). For 50 nm thick film, the band approximates 2.04 eV similar to that of monolayer thick film, whereas the band gap is approximately 1.2 eV for the 1200 nm thick film similar to that of bulk7. The variation of band gap with thickness is in excellent agreement with the theoretical prediction of layer dependency and is in accordance with other reports4,7,42,43,44,45. The large band gap observed for 50 nm thick film may be due to quantum confinement of layered d-electron dichalcogenides (Se2−) and has been observed in other transitional metal dichalcogenides46,47,48,49,50.
(a) DRS of SnSe2 thin films on SLG substrate of various thicknesses, (b) absorbance versus wavelength of SnSe2 thin films, (c) Tauc plot for 50, 140 and 1200 nm thin films, and (d) variation of band gap with thickness. Inset shows the change in band gap as a function of the inverse of the thickness. The solid lines are guide to the eye.
Room temperature Hall measurements for all the films revealed that SnSe2 films are n-type materials with a mobity in the range of µe = 2.0–8.0 cm2 V−1 s−1, resistivity of ρ = 5–30 Ω cm and carrier concentration of ne = (0.95–6) × 1017 cm−3. The variation of resisitivity, mobilty and carrier concentration with thin film thickness are shown in Figure S6. It is interesting to note that the resistivity is optimum for a thickness of 700–900 nm, while the mobility peaks around 900 nm. Similarly, the carrier concentartaion is optimum around 700–800 nm. The better connectivity (Figure S7) of grains in the film is believed to be the reason for the better electrical properties. Over all, the mobility and carrier concentration are optimum for 700–900 nm thick films.
It can be noted from Fig. 4d that the band gap of thicker films (>400 nm) is below 1.3 eV, while that of thinner films (<140 nm) is above 1.6 eV. This indicates that thinner films can not be used for IR (1064 nm = 1.165 eV) photodetection. In order to substantiate this speculation, the photoresponse of SnSe2 thin films under 1064 nm laser illumination was evaluated using the device configuration shown in Fig. 5a. The optical photograph of the typical device structure is shown in the backgraound. Figure 5b shows the current (I)-voltage (V) linear behaviour of SnSe2 under dark and 1064 nm laser illumination with different power density (0 to 250 mW/cm2) and we observe that there is an increase in current upon the laser illumination on 1200 nm thick SnSe2 film. The photocurrent is measured under alternating dark and 1064 nm laser light. Figure 5c and S8 show the temporal photoresponse of 1200 nm thick SnSe2 film under 1064 nm laser illumination with different power densities upto 250 mW/cm2 at a bias voltage of 10 and 5 V, respectively.
It can be noted that the photocurrent increases with increasing bias and power density. The dependence of photocurrent on power density shown in Fig. 5(d) is fitted using the power law51: I ph α P m and the exponent m which determines the response characteristic of a photodetector with incident power density, is found to increase from 0.7 to 0.9 when the bias is increased from 5 to 10 V. The value of m indicates that trap states and interactions between the photogenerated carriers (electron-hole pairs) are involved in the recombination kinetics of photo-carriers51 at lower bias and the value of m is close to ideal unity value at higher bias which indicates that the photo-generated current can be attributed to efficient separation of electron-hole pairs with less trap states and interaction between photo-generated carriers.
Photoresponse of different films with a power density of 250 mW/cm2 and a bias of 10 V are shown in Figure S9(a–f) and the photocurrent as a function of thickness is shown in Fig. 6(a). The sensitivity (defined as S = Iλ/Idark, Iλ = Ilight − Idark, I is the current) as a function of thickness is shown in the inset of Fig. 6(a). It is interesting to note that thinner films do not respond to the 1064 nm irradiation, while the sensitivty increases with thickness for thicker films. As indicated before, the band gap of thinner films is higher than the energy corresponding to 1064 nm and hence, not responsive (Figure S10). In order to testify the band gap dependency, we irradiated 140 nm thick film with white light. Interestingly, the film is found to be sensitive to white light (Figure S11) ensuring the thinner film is insensitive to IR because of the higher band gap. The sensitivity is found to be 90.4% for visible light with 100 mW/cm2. Figure 6b shows the variation of responsivity (defined as Rλ = Iλ/(PλA), A is the effective surface area and Pλ is the power density. Figure 6c and d show the variation of external quantum efficiency (defined as EQE = hcRλ/qλ) and detectivity (defined as D* = Rλ/(2qIo)1/2)52 with power density. Similar dependency has also been observed for 5 V (Figure S12). It may be noted that Rλ, EQE and D* increase with thickness as well as with power density (Figure S13). At this point, we would like to note that the photocurrent and sensitivity (Fig. 6(a)) as well as the dark current (Figure S14) increases with the thickness of the films. As the thickness increases the band gap decreases that promotes the enhancement of the darkcurrent. Similarly, the decrease in band gap also promotes more charge carriers to be generated as depicted in Fig. 7 leading to higher photocurrent and hence, the sensitivity.
Figure S15(a–d) shows the ON and OFF IR response characteristic of the device which is well retained even after six cycle repeatations with their respective fitted growth and decay rate constants at 10 and 5 V and are determined by fitting one cycle of the photo-response curve using the second order exponential equation given by I(t)growth = Idark + α exp[t/τ1] + βexp[t/τ2] and I(t)decay = Idark + χexp[−t/τ1] + γ exp[−t/τ2 ] respectively53, where α, β, χ and γ are scaling constants, τ1 and τ2 are time constants, t is the time for ON or OFF cycles and Idark is the dark current. From the fits, we estimate the time constants for growth and decay. When the bias voltage was set to 10 V, the photocurrent rises very fast within 0.38 s upon illumination followed by a slower component of 16 s before saturation. The average response time constant for this process is about 2.5 s. The average time constant was calculated from τavearage = (ατ1 + βτ2)/(α + β). After switching off the exciting laser, the photo-current decay follows a second order exponential relaxation process with an estimated time constant of 0.36 and 8.32 s with an average time constant of 3.68 s before reaching the initial dark current. When the bias voltage was 5 V, the response and decay of photoresponse follow a second order exponential relaxation process as is the case of 10 V. The time constants are 0.28 and 15.33 s with an average time constant of ≈7.76 s for the sensor response, while the time constansts for decay are 0.59 and 14.47 s with an average time constant of ≈7.52 s. It can be noted that the fastest response/decay was obtained at a bias voltage of 10 V and is consistent with the power law and is attributed to efficient seperation of electron-hole pairs. The general response is quiet slow as compared to other previously reported SnSe2 layered device15. This slow response may be due to defects or charge impurity states inside the band gap which act as recombination centres for the photo-generated charge carriers2,54. Previous reports with other layered 2D transitional metal dichalogenides have mainly focused on detecting visible light, a narrow range of the electromagnetic spectrum. The cut-off wavelength for our device is 1064 nm which is superior to those obtained by others15,55,56,57 and suggests that SnSe2 of about 1200 nm thick grown on SLG can also be used as an efficient IR photodetector. The IR-response and sensitivity of other prepared films in this study are compared in Table S3. Overall, our results suggest that the SnSe2 thin film can be explored as an excellent material for photodetection.
Conclusion
We have fabricated and characterized layered SnSe2 thin films of different thickness on SLG substrate by DC sputtering of Sn metal target followed by selenisation. Hall measurements confirm that SnSe2 is n-type material with carrier mobility between 2.0–8.0 cm2 V−1 s−1, resistivity between 5–30 Ω cm and carrier concentration ne between (0.95–6) × 1017 cm−3. We have observed that the band gap depends on thin film thickness. For 50 nm thick film, the band gap is ~2.04 eV similar to that of monolayer, whereas it is approximately 1.2 eV for the 1200 nm thick film similar to that of bulk. The IR photodetection response of SnSe2 demonstrates a sensitivity of ~3% for a 1000 nm thick film and the response time constant is 0.38 s at a bias of 10 V. On the other hand, the sensitivity is 90.4% for 140 nm thick film at 100 mW/cm2 which is insesitive to IR. Overall, our results suggest that the SnSe2 thin film can be explored as an excellent material for photodetection and the sensitivity with response time can be improved further by increasing the applied bias.
Methods
Thin film Deposition
Prior to deposition, soda lime glass substrates (SLG) were cleaned by placing them in dilute HCl for 10 minutes, followed by sonication in de-ionised (DI) water and then boiled in iso-propanol alcohol (IPA) at 82.5 °C for about 15 minutes. The substrates were then purged with nitrogen gas to remove any contaminants such as water vapour. The vaccum chamber was first evacuated to a base pressure of about 5.0 × 10−6 mbar. Sn was then sputtered onto SLG between 15–47 W for 5–20 minutes using argon as plasma source flowing at 1.5 sccm. The chamber working pressure was maintained at 4.0 × 10−3 mbar throughout the deposition to obtain films of various thicknesses ranging from 30 to 600 nm. The substrate was maintained at 20 rotations per minute to obtain films of uniform thickness. The Sn films on SLG was annealed in a calibrated tube furnace as shown in Fig. 8. 1 g of Se powder in Alumina boat was placed in a zone with temperature of about 300 °C (above the melting point of Se), whereas Sn thin films on SLG was placed in a temperature zone of about 450 °C. Prior to selenization process, argon gas was passed through the tube furnace for 5 minutes to drive out the oxygen gas present. The tube furnace ramp rate was maintained at 3 °C per minute to a temperature of 450 °C and held constant for 1 h. The furnace was cooled down to room temperature naturally. Argon gas flow was maintained at 20 sccm throughout the annealing and cooling processes.
Thin film Characterization
The crystal structure of the films have been accessed by X-ray diffraction (XRD) using CuKα (1.5418 Å) (X’pert-PRO PANAlytical instruments). The crystalline properties of the film were further determined using 200 kV FETEM (JEM-2100F) and were prepared by scratching the film surface. The powder collected was dispersed in IPA, sonicated for about 10 minutes and dropped onto carbon coated copper grids. The surface morphology of the films, roughness and thickeness were determined using non-contact mode AFM (A.P.E. Research A100-AFM). Veeco Dektak 6 M surface profilometer was used to measure the thickness of the film. Diffuse Reflection Spectrum (DRS) of the thin film was obtained using UV-Vis-NIR spectrophotometer (Perkin Elmer-lambda 750 Instruments). Raman study was carried out at room temperature in the range of 50–600 cm−1 using Visible LabRAM HR instruments with a 532 nm laser. X-ray photoelectron spectroscopy (XPS) measurements were performed using AXIS Ultra DLD X-ray photoelectron spectrometer with MgKα X-ray source. The system was maintained in ultra-high vacuum at a base pressure of 6.8 × 10−9 Torr. The C1s peak at 284.60 eV was taken as reference to correct the binding energy values of our samples.
Device fabrication and current-voltage measurements
The electrical contacts of Cr/Au (6 nm/80 nm) was thermally evaporated and deposited on top of SnSe2 thin films. The current-voltage characteristics were measured using a Keithley SMU2400 source meter and 1064 nm laser with varrying intensity. The Hall measurements were conducted at room temperature in the presence of 0.55 T magnetic field using Ecopia HMS 5000 Hall effect measurement system and measurements were taken in the Van der Pauw geometry.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Choi, W. et al. High-detectivity multilayer MoS2 phototransistors with spectral response from ultraviolet to infrared. Adv. Mater. 24, 5832–5836 (2012).
Chang, Y. et al. Monolayer MoSe2 Grown by Chemical Vapor Deposition for Fast Photodetection. ACS Nano 8, 8582–8590 (2014).
Chhowalla, M. et al. The chemistry of two-dimensional layered transition metal dichalcogenide nanosheets. Nat. Chem. 5, 263–275 (2013).
Wang, Q. H., Kalantar-Zadeh, K., Kis, A., Coleman, J. N. & Strano, M. S. Electronics and optoelectronics of two-dimensional transition metal dichalcogenides. Nat. Nanotechnol. 7, 699–712 (2012).
Bharatula, L. D., Erande, M. B. Mulla, I. S. Rout, C. S. & Late, D. J. SnS2 nanoflakes for efficient humidity and alcohol sensing at room temperature. RSC Adv. 6, (2016).
Pawbake, A. S., Date, A., Jadkar, S. R. & Late, D. J. Temperature dependent raman spectroscopy and sensing behavior of few layer SnSe2 nanosheets. ChemistrySelect 1, 5380–5387 (2016).
Gonzalez, J. M. & Oleynik, I. I. Layer-dependent properties of SnS2 and SnSe2 two-dimensional materials. Phys. Rev. B - Condens. Matter Mater. Phys. 94, 1–10 (2016).
Mooser, E. & Pearson, W. B. New Semiconducting Compounds. Phys. Rev. 101, 492–493 (1956).
Evans, B. L. & Hazelwood, R. A. Optical and electrical properties of SnSe2. J. Phys. D. Appl. Phys. 2, 1507–1516 (2002).
Lin, Z. et al. Molecular beam epitaxy of large-area SnSe2 with monolayer thickness fl uctuation. 2D Materials, 2016, 4, 014006.
Barrios-Salgado, E., Nair, M. T. S. & Nair, P. K. Thin films of n-type SnSe2 produced from chemically deposited p-type SnSe. Thin Solid Films 598, 149–155 (2016).
Julien, C., Eddrief, M., Samaras, I. & Balkanski, M. Optical and electrical characterizations of SnSe, SnS2 and SnSe2 single crystals. Mater. Sci. Eng. B15, 70–72 (1992).
Schlaf, R., Pettenkofer, C. & Jaegermann, W. Band lineup of a SnS2/SnSe2/SnS2 semiconductor quantum well structure prepared by van der Waals epitaxy. J. Appl. Phys. 85, 6550 (1999).
Amalraj, L., Jayachandran, M. & Sanjeeviraja, C. Preparation and characterization of tin diselenide thin film by spray pyrolysis technique. Mater. Res. Bull. 39, 2193–2201 (2004).
Zhou, X. et al. Ultrathin SnSe2 Flakes Grown by Chemical Vapor Deposition for High-Performance Photodetectors. Adv. Mater. 27, 8035–8041 (2015).
Gonzalez, J. M. & Oleynik, I. I. Layer-dependent properties of SnS2 and SnSe2 novel two-dimensional materials. https://doi.org/10.1103/PhysRevB.94.125443
Guo, P., Luo, Y. W. & Jia, Y. Tuning band gap and optical properties of SnX2 nanosheets: Hybrid functional studies. Mod. Phys. Lett. B30, 1650120 (2016).
Huang, Y. et al. Designing the shape evolution of SnSe2 nanosheets and their optoelectronic properties. Nanoscale 7, 17375–17380 (2015).
Anwar, S., Gowthamaraju, S., Mishra, B. K., Singh, S. K. & Anwar, S. Spray pyrolysis deposited tin selenide thin films for thermoelectric applications. Mater. Chem. Phys. 153, 236–242 (2015).
Mitzi, D. B., Milliron, D. J., Copel, M., Murray, C. & Kosbar, L. High mobility solution-deposited chalcogenide films for flexible applications. 2005 IEEE VLSI-TSA - Int. Symp. VLSI Technol. - VLSI-TSA-TECH. Proc. Tech. Pap 4, 41–44 (2005).
Wang, R. Y. et al. Electronic and optical switching of solution-phase deposited SnSe 2 phase change memory material. J. Appl. Phys. 109, (2011).
Narro-Rios, J. S., Ramachandran, M., Martínez-Escobar, D. & Sánchez-Juárez, A. Ultrasonic spray pyrolysis deposition of SnSe and SnSe2 using a single spray solution. J. Semicond. 34, 13001 (2013).
Anwar, S., Mishra, B. K. & Anwar, S. Optimized substrate temperature range for improved physical properties in spray pyrolysis deposited Tin Selenide thin films. Mater. Chem. Phys. 175, 118–124 (2016).
Hady, D. A., Soliman, H., El-Shazly, A. & Mahmoud, M. S. Electrical properties of SnSe2 thin films. Vacuum 52, 375–381 (1999).
De Groot, C. H. et al. Highly selective chemical vapor deposition of tin diselenide thin films onto patterned substrates via single source diselenoether precursors. Chem. Mater. 24, 4442–4449 (2012).
Larson, A. C. & Von Dreele, R. B. General Structure Analysis System (GSAS). Structure 748, 86–748 (2004).
Smith, A. J., Meek, P. E. & Liang, W. Y. Raman scattering studies of SnS2 and SnSe2. J. Phys. Chem C1321, 1321 (1977).
Guo, C., Tian, Z., Xiao, Y., Mi, Q. & Xue, J. Field-effect transistors of high-mobility few-layer SnSe2. 203104, (2016).
Cheng, C., Li, Z., Dong, N., Wang, J. & Chen, F. Tin diselenide as a new saturable absorber for generation of laser pulses at 1 μm. Optics Express 25(6), 6132–6140 (2017).
Boscher, N. D., Carmalt, C. J., Palgrave, R. G. & Parkin, I. P. Atmospheric pressure chemical vapour deposition of SnSe and SnSe2 thin films on glass. Thin Solid Films 516, 4750–4757 (2008).
Kim, S. I. et al. Metallic conduction induced by direct anion site doping in layered SnSe2. Sci. Rep. 6, 19733 (2016).
Du, C.-Fb, Li, J.-R. & Huang, X.-Y. Microwave-assisted ionothermal synthesis of SnSex nanodots: A facile precursor approach towards SnSe2 nanodots/graphene nanocomposites. RSC Adv. 6, 9835–9842 (2016).
Patil, S. J., Lokhande, V. C., Lee, D. & Lokhande, C. D. Electrochemical impedance analysis of spray deposited CZTS thin fi lm: Effect of Se introduction. Opt. Mater. (Amst). 58, 418–425 (2016).
Ma, R. et al. Fabrication of Cu2ZnSn(S,Se)4 (CZTSSe) absorber films based on solid-phase synthesis and blade coating processes. Appl. Surf. Sci. 368, 8–15 (2016).
Serra, A., Rossi, M., Buccolieri, A. & Manno, D. Solid-to-solid phase transformations of nanostructured selenium-tin thin films induced by thermal annealing in oxygen atmosphere. AIP Conf. Proc 1603, 31–39 (2014).
Saha, S., Banik, A. & Biswas, K. Few Layer Nanosheets of n-type SnSe2. Chem. - A Eur. J. 44, 1–12 (2016).
Fang, Z. et al. The enhanced photoelectrochemical response of SnSe2 nanosheets. Cryst Eng Comm 16, 2404 (2014).
Huang, L., Yu, Y., Li, C. & Cao, L. Substrate Mediation in Vapor Deposition Growth of Layered Chalcogenide Nanoplates: A Case Study of SnSe Substrate Mediation in Vapor Deposition Growth of Layered Chalcogenide Nanoplates: A Case Study of SnSe2. The Journal of Physical Chemistry C 117(no. 12), 6469–6475 (2013).
Song, Y. et al. Pseudocapacitance-tuned high-rate and long-term cyclability of NiCo2S4 hexagonal nanosheets prepared by vapor transformation for lithium storage. J. Mater. Chem. A5, 9022–9031 (2017).
Song, Y. et al. Bottom-up Approach Design, BandStructure, and Lithium Storage Properties of Atomically Thin gamma-FeOOH Nanosheets. ACS Appl. Mater. Interfaces 8, 21334–21342 (2016).
Fiechter, S., Martinez, M., Schmidt, G., Henrion, W. & Tomm, Y. Phase relations and optical properties of semiconducting ternary sulfides in the system Cu-Sn-S. J. Phys. Chem. Solids 64, 1859–1862 (2003).
Džimbeg-malčić, V., Barbarić-mikočević, Ž. & Itrić, K. Kubelka-Munk Theory in Describing Optical Properties of Paper (I). Tehnički vjesnik 1, 117–124 (2011).
Chand, P., Gaur, A. & Kumar, A. Structural, optical and ferroelectric behavior of hydrothermally grown ZnO nanostructures. Superlattices Microstruct. 64, 331–342 (2013).
Im, H. S. et al. Facile phase and composition tuned synthesis of tin chalcogenide nanocrystals. RSC Adv. 3, 10349 (2013).
Bindu, K. & Nair, P. K. Semiconducting tin selenide thin films prepared by heating Se–Sn layers. Semicond. Sci. Technol. 19, 1348–1353 (2004).
Mak, K. F., Lee, C., Hone, J., Shan, J. & Heinz, T. F. Atomically thin MoS2: A new direct-gap semiconductor. Phys. Rev. Lett. 105, 2–5 (2010).
Kuc, A., Zibouche, N. & Heine, T. Influence of Quantum Confinement on the Electronic Structure of the Transition Metal Sulfide TS2. Phys. Rev. B83, 245213 (2011).
Yun, W. S., Han, S. W., Hong, S. C., Kim, I. G. & Lee, J. D. Thickness and strain effects on electronic structures of transition metal dichalcogenides: 2H-MX2semiconductors (M = Mo, W; X = S, Se, Te). Phys. Rev B85, 33305 (2012).
Ding, Y. et al. First principles study of structural, vibrational and electronic properties of graphene-like MX2 (M = Mo, Nb, W, Ta; X = S, Se, Te) monolayers. Phys. B Condens. Matter 406, 2254–2260 (2011).
Liu, L., Kumar, S. B., Ouyang, Y. & Guo, J. Performance limits of monolayer transition metal dichalcogenide transistors. IEEE Trans. Electron Devices 58, 3042–3047 (2011).
Jia, Z. et al. Enhanced Photoresponse of SnSe-Nanocrystals-Decorated WS2 Monolayer Phototransistor. ACS Appl. Mater. Interfaces 8, 4781–4788 (2016).
Murali, B. & Krupanidhi, S. B. Facile synthesis of Cu2CoSnS4 nanoparticles exhibiting red-edge-effect: Application in hybrid photonic devices. J. Appl. Phys. 114, (2013).
Zang, C. et al. Photoresponse properties of ultrathin Bi2Se3 nanosheets synthesized by hydrothermal intercalation and exfoliation route. Appl. Surf. Sci. 316, 341–347 (2014).
Tongay, S. et al. Defects activated photoluminescence in two-dimensional semiconductors: interplay between bound, charged, and free excitons. Sci. Rep. 3, 2657 (2013).
Chang, R.-P. & Perng, D.-C. Near-infrared photodetector with Cu(In,Al)Se2 thin film. Appl. Phys. Lett. 99, 81103 (2011).
Chang, R. P. & Perng, D. C. Nano-structured Cu(In,Al)Se2 near-infrared photodetectors. Thin Solid Films 529, 238–241 (2013).
Akgul, F. A., Akgul, G., Gullu, H. H., Unalan, H. E. & Turan, R. Enhanced diode performance in cadmium telluride-silicon nanowire heterostructures. J. Alloys Compd. 644, 131–139 (2015).
Acknowledgements
Emma P. Mukhokosi is grateful to Indian Institute of Science through the Office of International Relations for the Ph.D scholarship, gratefull to Dr. Mustaque Ali, Dr. Sandra Dias and Mr. Nagabhushan Patel for their support in carrying out the experiments. The authors acknowledge Indian Institute of Science for providing the TEM facility.
Author information
Authors and Affiliations
Contributions
Idea was conceived through discussion of all authors. E.P.M. performed the experiments, S.B.K. and K.K.N. co-supervised the work. All authors contributed to analysis of data, discussion of the results and writing of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mukhokosi, E.P., Krupanidhi, S.B. & Nanda, K.K. Band Gap Engineering of Hexagonal SnSe2 Nanostructured Thin Films for Infra-Red Photodetection. Sci Rep 7, 15215 (2017). https://doi.org/10.1038/s41598-017-15519-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-15519-x
This article is cited by
-
Diversiform gas sensors based on two-dimensional nanomaterials
Nano Research (2023)
-
Influence of device architectures and mobility on response/recovery time of metal halide perovskites: A review
Journal of Materials Science (2022)
-
The effect of molybdenum-doped tin selenide semiconductor material (SnSe) synthesized via electrochemical deposition technique for photovoltaic application
Journal of Materials Science: Materials in Electronics (2022)
-
Applications of 2D-Layered Palladium Diselenide and Its van der Waals Heterostructures in Electronics and Optoelectronics
Nano-Micro Letters (2021)
-
Size- and temperature-control optical direct/indirect band tuning in layered compounds: band gap engineering
Optical and Quantum Electronics (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.