Abstract
Monitoring resistance and investigating insecticide resistance mechanisms are necessary for controlling the small brown planthopper, Laodelphax striatellus. The susceptibility to four common insecticides of L. striatellus collected from Jiangsu, Anhui, Zhejiang and Jilin provinces of China in 2015 was monitored. The results showed that all field populations remained susceptible to chlorpyrifos and thiamethoxam with resistance ratios (RRs) of 2.3- to 9.5 and 1.6- to 3.3, respectively, while the insects had developed moderate pymetrozine resistance with RRs of 18.7 to 34.5. Resistance against buprofezin had developed to an alarmingly high level in three southeastern provinces of China with RRs of 108.8 to 156.1, but in Jilin it had an RR of only 26.6. Moreover, in line with both the buprofezin and pymetrozine resistance levels, we found LsCYP6CW1 to be over-expressed in all field L. striatellus populations, which indicated that it might be important for cross-resistance between buprofezin and pymetrozine. RNA interference (RNAi) ingestion resulted in the effective suppression of LsCYP6CW1 expression, and significantly increased susceptibility to both buprofezin and pymetrozine compared with the control, which further confirmed that overexpression of LsCYP6CW1 was involved in the cross-resistance to buprofezin and pymetrozine in field L. Striatellus populations.
Similar content being viewed by others
Introduction
The small brown planthopper (Laodelphax striatellus Fallén) (Homoptera: Delphacidae) is a notorious agricultural pest with a wide distribution range from Southeast Asia to Siberia and Europe that attacks several important agricultural crops including rice, corn, wheat, oat and barley1. Damage is inflicted not only by direct feeding but also by transmitting several plant viruses2. In China, this pest has been found in all rice-growing areas and has been a serious problem since 1999. In recent years, the outbreak frequency of this insect increased greatly3. It is believed that insecticide resistance may be the main contributor to such a population surge, as the control of this species mainly depends on applications of chemical insecticides4. Therefore, to better provide guidelines for the scientific use of pesticides in the resistance management of field L. striatellus, monitoring resistance to common insecticides and clarifying the development of resistance is necessary.
In China, the main insecticides used to control the small brown planthopper include pymetrozine, chlorpyrifos, buprofezin and thiamethoxam, and multiple reports indicate that L. striatellus has developed resistance to chlorpyrifos, buprofezin and pymetrozine5,6. The increasing resistance of rice planthopper to these insecticides was thought to be related to the widespread use of these insecticides to control L. striatellus. However, other possible factors might also be important, including shared detoxification resistance among different insecticides.
Increases in metabolism mediated by detoxification enzymes, such as cytochrome P450s, esterases and glutathione S-transferase, have been considered important for insecticide resistance7,8. Among the three detoxification enzymes, cytochrome P450s appear to be the major type of detoxification enzyme and are extensively involved in different types of insecticide resistance9,10,11. Cytochrome P450s constitute a large enzyme family found in almost all living organisms from bacteria to humans9. Insect P450s can metabolize diverse endogenous and exogenous compounds, such as insecticides and plant allele chemicals, and have been divided into four major clades: CYP2, CYP3, CYP4, and mitochondrial P450s12,13. CYP6s belong to the CYP3 family and are extensively involved in the development of insecticide resistance. In insects, the overexpression of some CYP6 genes has been associated with the increased metabolism of insecticides in resistant strains, including Drosophila melanogaster (CYP6G1), Musca domestica (CYP6D1), Bemisia tabaci (CYP6CM1), Culex pipiens pallens (CYP6F1), Nilaparvata lugens (CYP6ER1, CYP6AY1), Locusta migratoria (CYP6F) and Aphis gossypii (CYP6A2)14,15,16,17,18,19,20,21. Our previous study found that overexpressed CYP6CW1 was closely associated with buprofezin resistance in a lab-selected resistant strain22, but whether or not a similar resistance mechanism occurs in field populations is unclear.
This study aimed to (1) evaluate the susceptibility of the small brown planthopper to four commonly used insecticides in seven different field populations; (2) analyse the synergistic roles of piperonyl butoxide (PBO) and conduct P450 gene expression analyses of L. Striatellus in seven resistant field populations and the susceptible YN population; and (3) investigate the cross-resistance functions of LsCYP6CW1 between buprofezin and pymetrozine via RNA interference (RNAi). The results will help to understand the insecticide detoxification function of LsCYP6CW1 and benefit resistance management.
Results
Susceptibility of L. striatellus to insecticides
The susceptibility of field L. Striatellus populations to various tested insecticides is reported in Table 1. The LC50 of chlorpyrifos ranged from 17.8 mg/L to 74.6 mg/L, with RRs (resistance ratios) ranging from 2.3- to 9.5-fold, which suggests that field populations of L. Striatellus have developed low and moderate resistance to chlorpyrifos; the LC50 of pymetrozine ranged from 173.6 mg/L to 319.7 mg/L, with RRs ranging from 18.7- to 34.5-fold and indicating a moderate level of resistance. The LC50 of buprofezin was from 35.8 mg/L to 210.8 mg/L, with RRs from 26.6- to 156.1-fold, and resistance occurred at moderate or extremely high levels. Finally, the LC50 of thiamethoxam ranged from 7.3 mg/L to 15.3 mg/L, and these results show thiamethoxam susceptibility in L. striatellus field populations.
Synergistic effect evaluated
The results showed that PBO had no significant synergistic effect on chlorpyrifos in most field populations except for LJ, where the synergistic rate was 1.24. However, PBO exerted a negative synergistic effect (synergistic rate 0.37) on chlorpyrifos in the JX population. A moderate synergistic effect of PBO on pymetrozine was found in six field populations but not in the relatively susceptible CC population, with the synergistic rate varying from 1.11 to 1.86. Surprisingly, only three field populations of LY, JY and LJ showed synergistic effects of PBO on buprofezin, with rates of 1.62-, 1.50- and 1.50- fold, respectively. The other four field populations showed no synergistic effect of PBO on buprofezin. In line with the result of buprofezin, thiamethoxam showed a synergistic effect of PBO in only some field populations, with a synergistic rate of 1.87 in JH, 1.27 in JX and 1.26 in LJ, in contrast to the absence of an effect in the other populations (Table 1).
Screening of L. striatellus P450 genes associated with insecticide resistance in the field strain
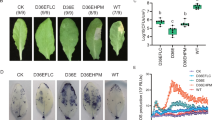
Of the 47 P450 genes of L. striatellus, LsCYP6CW1 showed an 8.87-fold increase in expression for NJ, 14.96-fold for LY, 9.96-fold for JH, 22.24-fold for JY, 2.21-fold for CC, 11.92-fold for JX, and 16.92-fold for LJ, respectively, compared with the susceptible YN population (Fig. 1a). These expression levels of LsCYP6CW1 were to some extent in accord with pymetrozine and buprofezin resistance levels in these field populations. Apart from LsCYP6CW1, other P450 genes also showed increased expression, including LsCYP304H1V3, which exhibited 2.58-fold higher expression for the LY population, 3.23-fold for JH, 2.23-fold for JY and 3.58-fold for LJ, respectively (Fig. 1b). LsCYP305 showed 2.10-fold higher expression for NJ, 4.46-fold for LY, 4.59-fold for JH, 5.59-fold for JY, 3.37-fold for CC and 6.59-fold for the LJ population (Fig. 1c), respectively, and LsCYP4B showed 2.00-fold higher expression for NJ, 4.96-fold for LY, 6.28-fold for JH, 2.10-fold for CC and 2.93-fold for LJ (Fig. 1d). Furthermore, some P450 genes of L. striatellus showed lower expression in the field populations than in the YN population. These genes included LsCYP4, LsCYP4DE1 and LsCYP439A1V3, which had 0.52-, 0.35- and 0.30-fold expression, respectively, for the NJ population. LsCYP439A1V3 showed 0.29-fold expression for LY, LsCYP4G115 had 0.5-fold expression for JH, and both LsCYP4DE1 and LsCYP439A1V3 had 0.23 and 0.39-fold lower expression, respectively, for JX (Fig. 1e).
Relative expression fold changes of P450 genes in field populations (black represents NJ, LY, JH, JY, CC, JX and LJ) and the susceptible YN population (grey represents YN) of L. striatellus. Each bar indicates the mean of three biological samples, each of which involved 3 technical replicates. Error bars represent the standard deviation from the mean. Data were normalized to the expression of ADP ribosylation factor (ARF). The significantly different expression of P450 genes in the field populations compared with the control YN population (figure only shows expression differences of more than 2-fold) are marked by asterisks. *means significantly different at the 0.05 level and **means significantly different at the 0.01 level. (a) represents LsCYP6CW1, (b) represents LsCYP304H1V3, (c) represents LsCYP305, (d) represents LsCYP4B and (e) represents lower-expression genes in the field populations.
Knockdown of LsCYP6CW1 increases sensitivity to both pymetrozine and buprofezin in field populations
LsCYP6CW1 exhibited the highest expression in all field populations compared to the susceptible YN population, and the expression levels of LsCYP6CW1 were consistent with the pymetrozine and buprofezin resistance levels. We therefore further evaluated the role of LsCYP6CW1 in pymetrozine and buprofezin resistance via RNAi. Before conducting RNAi, the siRNA candidate target regions of LsCYP6CW1 were identified through https://www.genscript.com/ssl-bin/app/rnai (Genscript Corporation). Moreover, previous studies found that RNAi ingestion could effectively suppress gene expression in small rice planthopper nymphs23,24. In this study, the 1st instar nymphs of L. striatellus were fed a dose of 0.2 mg/mL of dsLsCYP6CW1 and dsGFP. The qPCR data showed that the LsCYP6CW1 mRNA levels were dramatically decreased at 5 d. As shown in Fig. 2, the ingestion of 0.2 mg/mL of dsLsCYP6CW1 for 5 d resulted in 0.28-fold lower expression than dsGFP ingestion and 0.37-fold lower expression than ingestion of an artificial diet only (control). LC50-discriminating doses of the JY and JX populations were used to investigate differences in susceptibility after RNAi. Knockdown of LsCYP6CW1 in both the JY and JX populations significantly increased susceptibility to pymetrozine and buprofezin compared with the control; 75% and 72% died in the JY population and 79% and 82% died in the JX population following exposure to pymetrozine and buprofezin, respectively. These figures were significantly higher than those observed in the control groups, in which 49% and 46% of JY insects from the dsGFP treatment died following exposure to pymetrozine and buprofezin, respectively; equivalent figures for insects fed the artificial diet were 51% and 48%. For the JX population, 46% and 53% of insects died following exposure to pymetrozine and buprofezin, respectively, in the dsGFP treatment, and the equivalent figures were 52% and 51% when the insects were fed only an artificial diet (Fig. 3).
LsCYP6CW1 mRNA expression levels after nymphs ingested artificial feed; dsGFP, dsRNA of green fluorescent protein (GFP); or dsLsCYP6CW1, dsRNA of LsCYP6CW1. The time from ingestion was 5 d, and the dose was 200 μg/mL. The levels of LsCYP6CW1 transcription in LsCYP6CW1 dsRNA-treated and untreated insects were normalized compared to dsGFP-treated individuals. Data are presented as the average of three biological replicates, each conducted in duplicate and normalized to a control gene with error bars representing SEM. Means depicted with different letters are significantly different by ANOVA(P < 0.05).
Mortality of LsCYP6CW1-silenced L. striatellus nymphs after buprofezin and pymetrozine treatment in the JY and JX populations, respectively. Control (nymphs that ingested artificial feed), dsGFP (nymphs that ingested dsRNA GFP), and dsCYP6CW1 (nymphs that ingested dsRNA LsCYP6CW1). Data are presented as the average of three biological replicates, each conducted in duplicate and normalized to a control gene with error bars representing SEM. Means depicted with different letters are significantly different by ANOVA (P < 0.05).
Discussion
In order to maintain the long-term efficacy of insecticides, it is necessary to periodically assess the susceptibility levels and cross-resistance profiles of agricultural pests to the insecticides. In this study, the monitored field populations of L. Striatellus came mainly from eastern China, such as Jiangsu, Zhejiang and Anhui provinces, and these regions have developed higher levels of resistance than other regions in China5,6,25. One field population came from Jilin, in the northeast rice area of China, where resistance monitoring data are scarce. The results showed no strong L. striatellus resistance to chlorpyrifos, a broad-spectrum organophosphate used to control L. striatellus, with RRs ranging from 2.27 to 9.52 in seven field L. Striatellus populations, while Ban et al. (2012) reported chlorpyrifos RRs of 10–61.8 with 11 (out of 13) L. Striatellus populations having RRs > 226. This apparent increase in chlorpyrifos susceptibility could be a consequence of the reduced use of chlorpyrifos, which is being rotated with other insecticides, such as thiamethoxam and pymetrozine, to slow down resistance development in field populations. Pymetrozine is a new insecticide with a novel mode of action against sucking insects such as the planthopper. Previous results found that L. Striatellus was susceptible or had a low level of resistance to pymetrozine (RR = 0.5–6.7) in 18 field populations tested between 2010 and 2013 in China5,6. Pymetrozine use has been increasing among Chinese farmers with government subsidy in recent years, and we found that field L. striatellus had developed a moderate level of resistance to pymetrozine (RR of 18.7–34.5). This result suggests that pymetrozine has been overused, and action is required to slow down the development of resistance.
Buprofezin is an insect growth regulator and has a long history of use in planthopper control. High levels of buprofezin resistance have developed in several pest species6,26,27. The current study showed that L. striatellus in eastern China has developed an alarmingly high resistance level to buprofezin with high RR (108.8–156.1), but in the northeast Changchun population, only moderate resistance to buprofezin with an RR of 26.6 is present. This finding suggests that buprofezin use should be limited in eastern China by rotation with other insecticides. Thiamethoxam is a neonicotinoid insecticide that has been used for planthopper control in rice since the 1990s, and no apparent small brown planthopper resistance to thiamethoxam was observed in this study, in line with Ban et al. (2012) and Zhang et al. (2014)5,6, which suggests that L. striatellus still maintains its susceptibility to thiamethoxam, which continues to be an effective pesticide for L. striatellus. Overall, the current study revealed that thiamethoxam and chlorpyrifos could control L. striatellus. Buprofezin should be avoided in eastern China, and pymetrozine should be rotated with other insecticides.
P450 monooxygenase-mediated detoxification is among the major insecticide resistance mechanisms, involving the altered expression of multiple genes in many insect species15,16,17,21. Our previous study found the expression levels of LsCYP6CW1 to be associated with buprofezin resistance levels22. In this study, in line with the buprofezin resistance levels, we found LsCYP6CW1 to be uniquely over-expressed in all field populations of L. striatellus, which further implicates overexpressed LsCYP6CW1 in field resistance to buprofezin. LsCYP6CW1 expression could provide an effective molecular marker to detect buprofezin resistance in the field in future tests. Moreover, the overexpression of P450 genes might also be important for cross-resistance among different insecticides28. We found that the overexpression of LsCYP6CW1 was to some extent consistent with field resistance to pymetrozine, so we propose that overexpressed LsCYP6CW1 might also be important for cross-resistance between buprofezin and pymetrozine in field populations. Other relatively less overexpressed P450 genes, such as LsCYP304H1V3, LsCYP305 and LsCYP4B, might play secondary roles in insecticide resistance in field L. striatellus populations.
Previous studies have reported that the ingestion of double-stranded RNA (dsRNA) can effectively suppress target genes in L. striatellus, and this ability has been widely used in gene function research23,24,29. We further evaluated the resistance function of LsCYP6CW1 in field populations via RNAi. The ingestion of 0.2 mg/mL of dsLsCYP6CW1 for 5d resulted in an effective suppression of LsCYP6CW1 expression and significantly increased susceptibility to both buprofezin and pymetrozine compared to the control in two selected field-resistant populations, where the expression of LsCYP6CW1 was significantly higher than for the other P450 genes. This result further implicated overexpressed LsCYP6CW1 was an important factor that conferring cross-resistance to these two insecticides in field L. striatellus populations. Moreover, the resistance mechanism may be complicated in field populations because of the application background of different insecticides. In this study, we found that overexpressed CYP6CW1 was involved in buprofezin and pymetrozine resistance in field L. striatellus, but the regulatory mechanism of overexpressed CYP6CW1 in the field is unclear. One situation that could occur in the field populations of L. striatellus is that the overexpressed CYP6CW1 might be regulated by other insecticide selection pressures but not correlated or weakly correlated with the application of buprofezin and pymetrozine, however, as CYP6CW1 could detoxify both buprofezin and pymetrozine, further studies on metabolism of buprofezin and pymetrozine by P450 enzymes in field populations of L. striatellus could throw light on the association of overexpression of CYP6CW1 vis a vis metabolism of these insecticides.
Although the results of this study suggest that LsCYP6CW1 is involved in cross-resistance to both buprofezin and pymetrozine in field L. striatellus, our work showed that piperonyl butoxide (PBO) had no substantial synergistic effect on buprofezin and pymetrozine resistance in field populations of L. striatellus. There are several possible explanations for the results. 1) A number of P450 enzymes are associated with detoxification of a different groups of insecticides, there may be a possibility the PBO has more affinity for other P450 enzymes than the products of LsCYP6CW1. 2) Rice planthoppers are plant-sucking pests, and whether or not the rice-stem dipping bioassays used in the current study provided an effective systemic delivery of PBO to the rice seedlings deserves further exploration. 3) A previous study also found that the PBO synergist could decrease the activation of a proinsecticide, which indicated that PBO might be a poor inhibitor of the P450(s) responsible for resistance to some insecticides30, and any resistance suppression in this study could be misleading.
Methods
Insects
The susceptible (YN) population of L. striatellus, which contained thousands of individuals, was collected from the Yunnan province of China in July 2001 and has been reared without insecticides since that time. Seven field populations of L. striatellus were collected in 2015: Nanjing (NJ), Liyang (LY), Jianhu (JH) and Jiangyan (JY) of Jiangsu, Changchun (CC) of Jilin, Jiaxing (JX) of Zhejiang and Lujiang (LJ) of Anhui province. The insects were all reared on rice seedlings at 27 ± 1 °Cunder a 14:10 h light:dark cycle. Field collected insects were mass mated. The third-instar nymphs of F1 or F2 progeny were used for bioassays.
Bioassays
The degrees of resistance of the NJ, LY, JH, JY, CC, JX, LJ and YN populations against chlorpyrifos, pymetrozine, buprofezin and thiamethoxam were assayed by the rice seedling dip bioassay method based on previous approaches26. The formulated insecticides were diluted in distilled water to generate six serial dilutions. A group of four rice seedlings was immersed individually into the insecticide dilutions for 20 s. After air-drying, the seedlings were placed in disposable plastic cups with moistened paper to maintain the wetness of the rice seedling roots. Fifteen 3rd-instar nymphs were placed in each treated plastic cup. Rice seedlings dipped in distilled water that contained 1% TritonX-100 were used as controls. For each dilution, three replicates each containing 15 individuals were treated. For analysis of the synergistic effect of the enzyme inhibitors on these four insecticides, 15 mg L−1 PBO was added to each dilution. All tests were maintained at 27(±1) °C, and mortality was recorded after 48 h, 120 h, 120 h and 120 h for chlorpyrifos, pymetrozine, buprofezin and thiamethoxam, respectively. Bioassay data were subjected to probit analysis using the Polo Plus software.
Screening of L. striatellus P450 genes associated with insecticide resistance
The candidate P450 genes used in this study came from previous research7,22. We newly integrated these p450 genes and deleted the short segments. After the candidate P450 genes were obtained, a BlastP search was performed in NCBI for further support of the annotation predictions, and we finally retained 47 P450 gene sequences in L. striatellus. Total RNA was extracted from 3rd-instar nymphs of the field and YN populations using the SV Total RNA Isolation system (Promega). Three independent RNA preparations were made for each population, each containing 15 individuals. The first-strand cDNA was synthesized from 2 µg of total RNA using an oligo(dT)15 primer and Superscript III reverse transcriptase (Promega). The relative expression levels of the 47 P450 genes in the YN and field populations were determined by qPCR with ADP ribosylation factor (ARF) as a reference31. The PCR primer sequences and the expected size of each PCR product are shown in a previous study and in Table 2 31. The qPCR was performed on three biological samples for each L. striatellus population, and each sample was analysed in three technical replicates on an Applied Biosystems 7500 thermocycler. The PCR mixture contained 10 µL of SYBR Premix Ex Taq™ (Takara, Japan), 1 µL of cDNA, 0.4 µL of ROX Reference Dye (50×), and 0.4 µL of 10 µM sense and antisense primers in a total volume of 20 µL. The optimized cycling programme was 1 cycle of 95 °C for 30 s, 40 cycles of 95 °C for 5 s, 60 °C for 31 s, and a final disassociation stage that was automatically added by the 7500 System SDS software. The relative expression was calculated using the 2−ΔΔCTmethod32. Student’s t-test was performed to determine whether the differences in the relative expression of the 47 P450 genes in the field and YN populations were significant.
Resistance functional analysis through RNA interference (RNAi)
A 372 bp segment of the LsCYP6CW1 cDNA and a 426 bp fragment of the green fluorescent protein (GFP) gene as a control were amplified by PCR. Both PCR products were individually sub-cloned into the pGEM-T easy vector (Promega), and the diluted plasmids were used as templates for the amplification of these target sequences with specific primers that were combined with the T7 RNA polymerase promoter (Table 2). The PCR products were purified with Wizard® SV Gel (Promega) and used as templates for dsRNA synthesis with the T7 Ribomax TM Express RNAi System, according to the manufacturer’s instructions (Promega). The dsRNAs of LsCYP6CW1 and GFP were dissolved in ultrapure water, and the quality and concentrations of the dsRNAs were determined by agarose gel electrophoresis and a Nanodrop 2000 spectrophotometer (Thermo Scientific, Wilmington, DE, USA). Forty 1st-instar nymphs were carefully transferred into the chamber of a glass cylinder (12 cm in length and 2.8 cm in internal diameter) with a liquid artificial diet between two layers of stretched Parafilm® M (Pechiney Plastic Pack ageing Company, Chicago, IL, USA); the stretched Parafilm® M was placed at one end of the chamber, and dense breathable gauze was placed at the other end. The diet was changed and dead nymphs removed daily. Six independent preparations were made for the 200 mg/mL ingestion treatment. Forty-five insects were collected 5 d after ingestion to determine the expression levels of LsCYP6CW1 using qPCR. A group of insects not fed dsRNA were also tested. Furthermore, we selected two populations in which the expression fold of LsCYP6CW1 was significantly higher than that of other P450 genes of L. striatellus to evaluate the resistance role of LsCYP6CW1 between pymetrozine and buprofezin in the field populations. Changes in the susceptibility of the JY and JX populations against buprofezin and pymetrozine after RNAi were evaluated using the rice seedling dipping methods as mentioned above.
References
Kisimoto, R. Flexible diapause response to photoperiod of a laboratory selected line in the small brown planthopper, Laodelphax striatellus Fallén. Appl. Entomol. Zool. 24, 157–159 (1989).
Kisimoto, R. Genetic variation in ability of a planthopper vector-Laodelphax striatellus (Fallén) to acquire rice stripe virus. Virology. 32, 144–152 (1967).
Zhu, J. L. et al. Effect of rice sowing date on occurrence of small brown planthopper and epidemics of planthopper-transmitted rice stripe viral disease. Sci. Agric. Sin. 41, 332–341 (2009).
Liu, X. D., Zhai, B. P. & Liu, C. M. Outbreak reasons of Laodelphax striatellus population (in Chinese with English summary). Chin. Bull. Entomol. 43, 141–146 (2006).
Ban, L. F. et al. Resistance Monitoring and Assessment of Resistance Risk to Pymetrozine in Laodelphax striatellus (Hemiptera: Delphacidae). J. Econ. Entomol. 105, 2129–2135 (2012).
Zhang, K. et al. Susceptibility of Sogatella furcifera and Laodelphax striatellus (Hemiptera: Delphacidae) to Six Insecticides in China. J. Econ. Entomol. 107, 1916–1922 (2014).
Xu, L., Wu, M. & Han, Z. J. Overexpression of Multiple Detoxification Genes in Deltamethrin Resistant Laodelphax striatellus (Hemiptera: Delphacidae) in China. PLos one. 8, e79443 (2013).
Li, X. C., Schuler, M. A. & Berenbaum, M. R. Molecular mechanisms of metabolic resistance to synthetic and natural xenobiotics. Annu. Rev. Entomol. 52, 231–253 (2007).
Guengerich, F. P. Common and uncommon cytochrome P450 reactions related to metabolism and chemical toxicity. Chem. Res. Toxicol. 14, 611–650 (2001).
David, J. P., Ismail, H. M., Proust, A. C. & Paine, M. J. I. Role of cytochrome P450s in insecticide resistance: impact on the control of mosquito-borne diseases and use of insecticides on earth. Philos Trans R Soc Lond B Biol Sci. 368, 20120429 (2013).
Liu, N. & Zhu, F. House fly cytochrome P450s: their role in insecticide resistance and strategies in the isolation and characterization, In: Recent Advances in Entomological Research. Springer 14, 246–257 (2012).
Li, X. C., Schuler, M. A. & Berenbaum, M. R. Molecular mechanisms of metabolic resistance to synthetic and natural xenobiotics. Annu. Rev. Entomol. 52, 231–253 (2007).
Feyereisen, R. Evolution of insect P450. Biochem. Soc. Trans. 34, 1252–1255 (2006).
Daborn, P., Boundy, S., Yen, J., Pittendrigh, B. & Ffrench-Constant, R. DDT resistance in Drosophila correlates with Cyp6g1 over-expression and confers cross-resistance to the neonicotinoid imidacloprid. Mol. Genet. Genomics. 266, 556–563 (2001).
Liu, N. & Scott, J. G. Increased transcription of CYP6D1 causes cytochrome P450-mediated insecticide resistance in house fly. Insect Biochem. Mol. Biol. 28, 531–535 (1998).
Karunker, I. et al. Over-expression of cytochrome P450 CYP6CM1 is associated with high resistance to imidacloprid in the B and Q biotypes of Bemisia tabaci (Hemiptera: Aleyrodidae). Insect Biochem. Mol. Biol. 38, 634–644 (2008).
Gong, M. Q. et al. Cloning and overexpression of CYP6F1, a cytochrome P450gene, from deltamethrin-resistant Culex pipiens pallens. Acta Biochim. Biophys. Sin. 37, 317–326 (2005).
Ding, Z. et al. Biochemical mechanisms of imidacloprid resistance in Nilaparvata lugens: over-expression of cytochrome P450 CYP6AY1. Insect Biochem. Mol. Biol. 43, 1021–1027 (2013).
Bass, C. et al. Overexpression of a cytochrome P450 monooxygenase, CYP6ER1, is associated with resistance to imidacloprid in the brown planthopper, Nilaparvata lugens. Insect Mol. Biol. 20, 763–773 (2011).
Guo, Y. Q. et al. RNA interference of cytochrome P450 CYP6F subfamily genes affects susceptibility to different insecticides in Locusta migratoria. Pest Manag. Sci. 72, 2154–2165 (2016).
Peng, T. F. et al. Over-expression of CYP6A2 is associated with spirotetramat resistance and cross-resistance in the resistant strain of Aphis gossypii Glover. Pestic. Biochem. Physiol. 126, 64–69 (2016).
Zhang, Y. L. et al. Overexpression of a P450 gene (CYP6CW1) in buprofezin-resistant Laodelphax striatellus (Fallén). Pestic. Biochem. Physiol. 104, 277–282 (2012).
Jia, S., W, P. J., Zhou, L. T., Mu, L. L. & Li., G. Q. Knockdown of a putative Halloween gene shade reveals its role in ecdysteroidogenesis in the small brown planthopper Laodelphax striatellus. Gene. 531, 168–174 (2013).
Wan, P. J., Shuang., J., Li, N., Fan, J. M. & Li, G. Q. RNA interference depletion of the Halloween gene disembodied implies its potential application for management of planthopper Sogatella furcifera and Laodelphax striatellus. PLoS One. 9, e86675 (2014).
Gao, B. L., Wu, J., Huang, S. J., Mu, L. F. & Han, Z. J. Insecticide resistance in field populations of Laodelphax striatellus Fallén (Homoptera: Delphacidae) in China and its possible mechanisms. Int. J. Pest. Manage. 54, 13–19 (2008).
Wang, L. H., Fang, J. C. & Liu, B. S. Relative toxicity of insecticides to Laodelphax striatellus (Fallén) (Homoptera:Delphacidae) and the resistance of field populations from different areas of East China. Acta Entomol. Sin. 9, 930–937 (2008).
Wang, Y. H. et al. Advances in the research of insecticide resistance of the small brown planthopper, Laodelphax striatellus. Plant Prot. 36, 29–35 (2010).
Mitchella, S. N. et al. Identification and validation of a gene causing cross-resistance between insecticide classes in Anopheles gambiae from Ghana. PNAS. 109, 6147–6152 (2011).
Zhang, Y. L. et al. Knockdown of NADPH-cytochrome P450 reductase results in reduced resistance to buprofezin in the small brown planthopper, Laodelphax striatellus (fallén). Pestic. Biochem. Physiol. 127, 21–27 (2016).
Feyereisen, R. Insect CYP genes and P450 enzymes. In: Gilbert LI (ed) Insect molecular biology and biochemistry. Elsevier Press, Oxford, pp 236–316 (2012).
Zhang, Y. L. et al. Overexpression of a P450 gene (CYP6CW1) in buprofezin-resistant Laodelphax striatellus (Fallén). Pestic. Biochem. Physiol. 104, 277–282 (2012).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using realtime quantitative PCR and the 2−△△CT method. Methods. 25, 402–408 (2001).
Acknowledgements
The authors sincerely thank Prof. Ary Hoffmann for proof reading and editing the manuscript. This work was supported by the National Natural Science Foundation of China (31301697), the Natural Science Foundation of Jiangsu Province (BK20170072), the Jiangsu Fund for Innovation of Agricultural Science and Technology (CX(15)1055), the Natural Science Foundation of Jiangsu Province (BK20130724).
Author information
Authors and Affiliations
Contributions
J.C.F. and L.H.W. designed the research and wrote the main manuscript text. Y.L.Z., Y.C.H., Y.Q. and B.S.L. performed experiments; H.F.G. and Z.W.L. helped interpret the data; Y.L.Z. prepared and wrote an early version of the paper, and all of the authors reviewed the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, Y., Han, Y., Liu, B. et al. Resistance monitoring and cross-resistance role of CYP6CW1 between buprofezin and pymetrozine in field populations of Laodelphax striatellus (Fallén). Sci Rep 7, 14639 (2017). https://doi.org/10.1038/s41598-017-15294-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-15294-9
This article is cited by
-
Emergence of fipronil resistant Rhipicephalus microplus populations in Indian states
Experimental and Applied Acarology (2020)
-
Neurological alterations induced by formulated imidacloprid toxicity in Japanese quails
Metabolic Brain Disease (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.