Abstract
In mammals, CD46 is involved in the inactivation of complement by factor I (FI). In teleost, study on the function of CD46 is very limited. In this study, we examined the immunological property of a CD46 molecule (CsCD46) from tongue sole, a teleost species with important economic value. We found that recombinant CsCD46 (rCsCD46) interacted with FI and inhibited complement activation in an FI-dependent manner. rCsCD46 also interacted with bacterial pathogens via a different mechanism to that responsible for the FI interaction, involving different rCsCD46 sites. Cellular study showed that CsCD46 was expressed on peripheral blood leukocytes (PBL) and protected the cells against the killing effect of complement. When the CsCD46 on PBL was blocked by antibody before incubation of the cells with bacterial pathogens, cellular infection was significantly reduced. Consistently, when tongue sole were infected with bacterial pathogens in the presence of rCsCD46, tissue dissemination and survival of the pathogens were significantly inhibited. These results provide the first evidence to indicate that CD46 in teleosts negatively regulates complement activation via FI and protects host cells from complement-induced damage, and that CD46 is required for optimal bacterial infection probably by serving as a receptor for the bacteria.
Similar content being viewed by others
Introduction
The complement system is a crucial constituent of the immune system and provides effective protection via mechanisms involving both innate and adaptive immune defenses1,2. The complement system consists of three pathways of activation: the classical pathway, the alternative pathway, and the lectin pathway3. The three pathways converge at the cleavage of C3 to C3a and C3b, which leads to a series of events involving cleavage of C5 and assembly of C5b, C6, C7, C8, and C9 to generate the membrane attack complex that induces osmotic lysis of the target cells4,5.
Complement activation is regulated by many factors6. Factor I, a serine protease, is one of the complement regulatory proteins7. In humans, factor I regulates the complement activation cascades of both classical and alternative pathways by cleavage and degradation of C4b and C3b in the presence of certain cofactors, thereby preventing the assembly of the C3 and C5 convertases8,9,10. CD46 is a cell surface molecule that functions in monomeric form as a key regulator of the classical and alternative complement activation cascades11. CD46 is a cofactor of factor I and regulates complement activation by facilitating the proteolysis of deposited C3b and C4b by factor I, thus preventing amplification of the complement cascade on the cells on which CD46 is expressed12,13. CD46 consists of four homologous complement control protein (CCP) repeats, a serine-threonine-proline-rich (STP) domain, a transmembrane hydrophobic domain, a cytoplasmic anchor, and a cytoplasmic tail14,15.
In addition to its role in complement activation, CD46 is also employed as a cellular receptor by several viruses and bacteria11. Human-specific pathogens targeting CD46 include measles virus, adenovirus groups B and D, herpes virus, Neisseria gonorrhoeae, Neisseria meningitidis, Streptococcus pyogenes, Escherichia coli, and Fusobacterium nucleatum 16,17,18,19. In bovine, CD46 is a receptor for bovine viral diarrhea virus, an enveloped RNA virus20. A recent study indicated a direct link between CD46 and components of the autophagy machinery17. Recognition of pathogens by CD46 is thought to trigger autophagy, which serves as a critical step in controlling infection11.
Studies on fish complement system have revealed the existence of almost all the orthologues of mammalian complement components in teleost, and multiple isoforms of some components are present in fish21. A few complement molecules, such as C3 and C1q, have been reported to be highly conserved in terms of functional properties between teleosts and mammals, though the immune functions of most fish complement components are unknown21,22. CD46-like sequences have been identified in several teleost species; however, only two reports on CD46 have been documented23,24. In one of these reports, a recombinant Chinese hamster ovary cell line expressing carp CD46 showed significantly increased tolerance against complement lysis and less deposition of C3, and that anti-CD46 antibody enhanced the depositions of C3 on autologous erythrocytes23. Another study examined the expression patterns of membrane-bound regulators of complement activation (gTecrem) in peripheral blood leukocytes (PBL) and erythrocytes of ginbuna crucian carp24. Despite these studies, the biological functions of CD46 in fish, especially in host fish in vivo, remain to be investigated. The present study, with an aim to gain more insights into the function of teleost CD46, we examined the biological role in complement activation and during bacterial infection in tongue sole (Cynoglossus semilaevis).
Results
The sequence feature of CsCD46
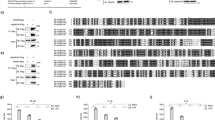
CsCD46 is composed of 340 amino acid residues with a calculated molecular mass of 36.8 kDa and a theoretical pI of 5.74. CsCD46 is a transmembrane protein with a signal peptide sequence (residues 1 to 20), an extracellular region containing four CCP modules (residues 23 to 77, 82 to 135, 140 to 194, and 199 to 254), a transmembrane region (residues 276 to 304), and an intracellular region (residues 310 to 332). CsCD46 was predicted to localize in the cytoplasmic membrane and contained 11 putative O-glycosylation sites. CsCD46 shared 25.5–40.9% overall sequence identities with CD46 from a number of teleost species including Takifugu rubripes, Poecilia formosa, Poecilia latipinna, Nothobranchius furzeri, Lates calcarifer, Neolamprologus brichardi, Oreochromis niloticus, Kryptolebias marmoratus, Austrofundulus limnaeus, Fundulus heteroclitus, Pundamilia nyererei, Cyprinus carpio, and Danio rerio (Fig. 1). CsCD46 also shared 25.7% overall sequence identity with human CD46, mainly in the CCP regions (Fig. 1).
Alignment of the sequences of CsCD46 homologues. Dots denote gaps introduced for maximum matching. Numbers in brackets indicate overall sequence identities between CsCD46 and the compared sequences. The consensus residues are in red, the residues that are ≥75% identical among the aligned sequences are in grey. The signal peptide sequence is indicated by a black line, the CCP domain is indicated by star signal, the transmembrane region is indicated by triangles, and the intracellular region is indicated by circles. The GenBank accession numbers of the aligned sequences are as follows: Takifugu rubripes, XP_011616176.1; Poecilia formosa, XP_016527734.1; Poecilia latipinna, XP_014910555.1; Nothobranchius furzeri, XP_015822576.1; Lates calcarifer, XP_018539190.1; Neolamprologus brichardi, XP_006794358.1; Oreochromis niloticus, XP_013130666.1; Kryptolebias marmoratus, XP_017274702.1; Austrofundulus limnaeus, XP_013866187.1; Fundulus heteroclitus, XP_012731019.1; Pundamilia nyererei, XP_013770344.1; Cyprinus carpio, BAM68364.1; Homo sapiens, ABK81636.1; Danio rerio, BAM68365.1.
Effect of recombinant CsCD46 (rCsCD46) on complement activation
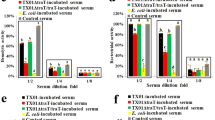
To examined the potential effect of rCsCD46 on complement activity, dilutions of tongue sole serum were incubated with rCsCD46 or the control protein rTrx that was purified under the same condition as rCsCD46 (Fig. S1). Subsequent analysis showed that serum treated with rCsCD46 at 8-, 16-, and 32-fold dilutions exhibited significantly lower hemolytic and bactericidal activities than untreated normal serum or rTrx-treated serum (Fig. 2), suggesting that rCsCD46 inhibited complement activation.
Effect of rCsCD46 on complement activation. Normal and inactivated serum of tongue sole in different dilutions was incubated with or without rCsCD46, rTrx, and the hemolytic (A) and bactericidal (B) activities of the serum were subsequently determined. Data are the means of three independent experiments and presented as means ± SEM. ** P < 0.01.
Involvement of factor I in rCsCD46-mediated effect on complement
With the above observation, we wondered whether factor I was involved in rCsCD46-mediated effect on complement. To investigate this question, tongue sole factor I (CsFI) in the serum was blocked by antibody against rCsFI, and its effect on rCsCD46 activity was subsequently determined. The results showed that for serum treated with rCsCD46 plus anti-rCsFI antibody, the hemolytic activity was significantly lower than that of the control serum, but significantly higher than that of the serum treated with rCsCD46 or rCsCD46 plus preimmune antibody (Fig. 3A), suggesting that the presence of anti-rCsFI antibody significantly reduced the inhibitory effect of rCsCD46 on complement activation. In contrast, treatment with anti-rCsFI antibody and preimmune antibody alone or plus rTrx had no apparent effect on the hemolytic activity. Similar to these results, when rCsCD46 was incubated with serum from which CsFI had been removed by affinity column absorption to anti-rCsFI antibody, the hemolytic activity of the serum was significantly increased compared with rCsCD46-treated normal serum (Fig. 3B). Co-IP and Western blot analyses showed that following incubation with serum, rCsCD46 was co-immunoprecipated with CsFI (Fig. 3C), suggesting that rCsCD46 interacted directly with serum CsFI. However, when rCsCD46 was incubated with rCsFI, no binding between these two proteins was detected (data not shown), suggesting that the interaction between rCsCD46 and rCsFI depended on other factors in the serum.
Involvement of CsFI in rCsCD46-mediated effect on complement activation. (A) Tongue sole serum in various dilutions was incubated with or without (control) rCsCD46 plus anti-rCsFI antibody, rCsCD46 plus preimmune antibody, rCsCD46, anti-rCsFI antibody, preimmune antibody, rTrx plus anti-rCsFI antibody, and rTrx plus preimmune antibody. The hemolytic activity of the serum was subsequently determined. (B) Dilutions of tongue sole serum that had been subjected to anti-rCsFI antibody absorption to deplete CsFI or anti-rTrx antibody absorbance (control serum) were incubated with rCsCD46, and the hemolytic activity of the serum was determined. (C) Interaction between rCsCD46 and CsFI. Tongue sole serum treated with different concentrations of rCsCD46 (80 µg/ml, 40 µg/ml, and 20 µg/ml) or Trx (80 µg/ml) was immunoprecipitated with anti-His antibody and then immunoblotted with anti-rCsFI antibody. For (A) and (B), data are the means of three independent experiments and presented as means ± SEM. Values with different letters indicate significantly different (P < 0.05).
Protective effect of CsCD46 on PBL against complement damage
Immunofluorescence microscopy showed that when PBL was treated with anti-rCsCD46 antibody, the antibody was found to be bound on the cells, whereas no cell-associated antibody was detected in PBL treated with anti-rTrx antibody (Fig. 4A), suggesting that CsCD46 was expressed by and exposed on the surface of PBL. To examine whether the CsCD46 on PBL had any effect against serum-induced cellular damage, PBL were treated with serum in the presence or absence of anti-rCsCD46 antibody, and cell death was determined by flow cytometry. The results showed that in PBL treated with serum plus anti-rCsCD46 antibody, the amount of cell death (30.2% ± 2.8%) was significantly (P < 0.01) higher than that in the PBL treated with serum alone (13.4% ± 1.5%) (Fig. 4B). In contrast, the amounts of cell death in PBL treated with serum plus anti-rTrx antibody (10.1% ± 1.1%), anti-rCsTLR2 antibody (13.5% ± 1.9%), and preimmune antibody (12.2% ± 1.3%) were comparable to that in the PBL treated with serum alone (Fig. 4B). When PBL were treated with the serum that had been depleted of factor I by anti-rCsFI antibody, the amount of cell death (31% ± 3.2%) was significantly higher than that of the control cells treated with normal serum (13.4% ± 1.5%) (Fig. S2).
Expression of CsCD46 on peripheral blood leukocytes (PBL) and its effect on complement damage. (A) PBL were incubated with anti-rCsCD46 antibody (Aa and Ab) or anti-rTrx antibody (Ad and Ae) and then treated with FITC-labeled secondary antibody and stained with DAPI. The cells were subjected to microscopy with green fluorescence light (Aa and Ad) or blue fluorescence light (Ab and Ae). Ac is a merged image of Aa and Ab; Af is a merged image of Ad and Ae. (B) PBL were treated with serum in the presence or absence of anti-rCsCD46 antibody, anti-rTrx antibody, preimmune antibody, or anti-CsTLR2 antibody for 1 h; the control cells were untreated with serum or antibody. Cellular damage was then determined by Annexin V-PI assay.
Binding of rCsCD46 to bacteria via a different mechanism from that for binding to CsFI
To examine the potential of rCsCD46 to interact with bacteria, rCsCD46 at different concentrations was incubated with the Gram-negative bacteria Edwardsiella tarda, Escherichia coli, Pseudomonas fluorescens, Vibrio anguillarum, and Vibrio harveyi, and the Gram-positive bacteria Bacillus subtilis and Streptococcus iniae. Among these bacteria, E. tarda, P. fluorescens, V. anguillarum, V. harveyi, and S. iniae are common fish pathogens. Subsequent ELISA analysis showed that rCsCD46 exhibited apparent binding to E. tarda, V. harveyi, V. anguillarum, and P. fluorescens in a dose-dependent manner (Fig. 5A). Relatively high binding indexes were observed with P. fluorescens and E. tarda. In contrast, heat-inactivated rCsCD46 or rTrx did not bind any bacteria (data not shown). Plate count analysis showed that rCsCD46 had no apparent effect on the survival of E. tarda and P. fluorescens (data not shown). To examine whether rCsCD46 bound bacteria and CsFI via the same site in rCsCD46, P. fluorescens was incubated with rCsCD46 in the presence or absence of rCsFI. Subsequent ELISA analysis showed that the presence of rCsFI had no significant effect on the binding index of rCsCD46 to bacteria (Fig. 5B).
Binding of rCsCD46 to bacteria. (A) Edwardsiella tarda, Escherichia coli, Pseudomonas fluorescens, Vibrio anguillarum, Vibrio harveyi, Bacillus subtilis, and Streptococcus iniae were incubated with or without (control) different concentrations of rCsCD46, and bacteria-protein binding was determined by ELISA. (B) P. fluorescens was incubated with or without (control) rCsCD46 plus rCsFI, rCsCD46 plus rTrx, rCsCD46, rCsFI, or rTrx (all proteins were at a final concentration of 40 µg/ml), and protein-bacteria binding was determined as above. Data are the means of three independent assays and presented as means ± SEM. **P < 0.01.
Effect of CsCD46 on bacterial infection
In vitro effect on cellular infection
With the above results, which showed that rCsCD46 interacted with fish bacterial pathogens and that PBL expressed CsCD46 on the cell surface, we investigated the role of CsCD46 in bacterial infection. To investigate this question, E. tarda and P. fluorescens were incubated with PBL in the presence or absence of anti-rCsCD46 antibody or anti-rTrx antibody, and bacterial infection was assessed by plate counts to determine the number of PBL-associated live bacteria at different times of infection. The results showed that at 1 h, 2 h, and 4 h post-infection, the numbers of E. tarda recovered from Anti-rCsCD46 antibody-treated PBL were significantly lower than that recovered from control cells or cells treated with anti-rTrx antibody or preimmune antibody (Fig. 6A). Similar results were obtained with P. fluorescens (Fig. 6B).
Effect of CsCD46 on bacterial infection in peripheral blood leukocytes (PBL). Edwardsiella tarda (A) and Pseudomonas fluorescens (B) were incubated with PBL in the presence or absence (control) of anti-rCsCD46 antibody, anti-rTrx antibody, or preimmune antibody, and bacterial infection was determined by plate count at various hours post-incubation. Data are the means of three independent assays and presented as means ± SEM. **P < 0.01.
In vivo effect on tissue dissemination and survival
To examine the in vivo effect of rCsCD46 on bacterial infection, tongue sole were infected with E. tarda and P. fluorescens in the presence of rCsCD46 or rTrx, and pathogen loads in the kidney, spleen, and blood of the fish were determined at 12, 24, and 48 h post-infection. The results showed that at all examined time points, the amounts of E. tarda recovered from all tissues in the fish infected with E. tarda plus rCsCD46 were significantly lower than those from the fish infected with E. tarda plus rTrx or E. tarda alone (Fig. 7A). Similarly, at 12, 24, and 48 h post-infection, the amounts of P. fluorescens recovered from all tissues in the fish infected with P. fluorescens plus rCsCD46 were significantly lower than those from the fish infected with P. fluorescens plus rTrx or P. fluorescens alone (Fig. 7B).
Effect of rCsCD46 on bacterial infection in tongue sole. Tongue sole were infected with Edwardsiella tarda (A) and Pseudomonas fluorescens (B) in the presence of rCsCD46, rTrx, or PBS (control), and the amounts of bacteria in kidney, spleen, and blood were determined at different times after infection. Data are the means of three independent assays and presented as means ± SEM. **P < 0.01.
Discussion
In this study, we investigated the biological role of CsCD46 in the regulation of the complement system and pathogen infection. Sequence analysis showed that CsCD46 possessed the four conserved CCP modules typical of that observed in mammalian and fish CD46. The CCP modules are known to be the sites responsible for complement regulation25. Consistent with the functional importance of these modules, the sequence identities/similarities between CsCD46 and human CD46 were clustered within the CCP regions. CsCD46 was similar in primary structure to most known fish CD46, however, like most fish CD46, CsCD46 does not possess the stretch of threonine residues immediately downstream of the CCP modules found in CD46 of N. brichardi and P. nyererei, suggesting that some specific features of CD46 differ among fish.
Studies in mammalian models have indicated that CD46 regulates the complement system via its ability to promote the cleavage of C3b and C4b by the serine protease factor I12,25,26. The CCP modules of CD46 offer binding sites for C3b, C4b, or factor I25. In the complement regulation process, CD46 binds to the substrate (C4b or C3b), and then factor I binds to CD46 to cleave the substrate27,28,29,30. In teleost, the only functional study of CD46 was carried out with the common carp CD46-like molecule Tecrem23. In this study, it was shown that Chinese hamster ovary cell line expressing Tecrem exhibited higher tolerance against complement damage and less deposition of C3, while an anti-Tecrem antibody enhanced the depositions of C3 and C4 on autologous erythrocytes23. However, the role of factor I in the action of CD46 was unclear. In our study, we found that rCsCD46 diminished the hemolytic and bactericidal activities of tongue sole serum resulting from activation of the serum complement, suggesting that rCsCD46 exerted a negative regulatory effect on complement activation. Functional CD46 in mammals is known to be a monomer, and it is possible that the rCsCD46 obtained in our study was in a monomeric form and exhibited a structure similar to that of the natural protein. We further observed that anti-rCsFI antibody significantly decreased the inhibitory effect of rCsCD46 on the complement system, most likely due to antibody blocking of natural CsFI in the serum. This conclusion was supported by the fact that serum CsFI co-immunoprecipated with rCsCD46, and that when serum was depleted of factor I by anti-rCsFI antibody absorption, the effect of rCsCD46 on serum complement was significantly reduced. These results indicated that CsCD46 was able to interact directly with CsFI to form the CsCD46–CsFI complex, which was essential for execution of the complement regulatory activity of CsCD46.
In mammals, host cells are protected from complement activation by regulatory proteins31. CD46 is one of such proteins that is widely expressed on most human cells, except erythrocytes and, combining with factor I, arrests propagation of the complement activation pathway by inactivation of C3b25,32,33. In our study, we found that CsCD46 was localized on the surface of PBL, consistent with its structural prediction, and that anti-rCsCD46 antibody significantly increased the sensitivity of PBL to serum-induced cellular destruction. These results, together with the observed complement-inhibitory effect of rCsCD46, indicate that like mammalian CD46, CsCD46 is a membrane-bound cofactor that plays a role in protecting host cells from complement attack via its capacity to control complement activation.
Previous studies have shown that some pathogens employ complement regulatory proteins to escape complement-mediated lysis34,35,36,37. CD46 acts as a receptor for a wide variety of viral and bacterial pathogens38. Measles virus and adenovirus have been reported to bind to the extracellular CCP1 and CCP2 domains of CD46, and N. gonorrhoeae and N. meningitidis bind CD46 via CCP modules and the STP region18,39,40,41. In our study, rCsCD46 was shown to interact with four Gram-negative bacterial pathogens but not with Gram-positive bacteria, possibly because of differences in the surface components between these bacterial species. Interestingly, the interaction between bacteria and rCsCD46 was unaffected by the presence of rCsFI, suggesting that rCsFI and bacteria bound to different regions of rCsCD46. In vitro bacterial infection analysis showed that the attachment of E. tarda and P. fluorescens to PBL was significantly reduced by the presence of anti-rCsCD46 antibody, supporting the idea that CsCD46 acts as a cellular receptor for these pathogens on PBL. In line with this idea, in vivo infection analysis showed that in the presence of rCsCD46, the tissue dissemination and colonization abilities of E. tarda and P. fluorescens were significantly impaired, suggesting that the interaction between the bacteria and rCsCD46 prevented binding of the bacteria to CsCD46 on host cells, thereby reducing bacterial attachment to host cells and resulting in attenuated bacterial invasion. These observations indicated that, like other pathogens that utilize CD46 for host infection, E. tarda and P. fluorescens have likely evolved the capacity to employ CD46 as a binding receptor for cellular invasion, representing the development of a virulence strategy by these pathogens during their battle with the host system.
In conclusion, we demonstrated for the first time that a teleost CD46 inhibited complement activation through recruitment of factor I, and was able to facilitate bacterial infection by serving as a cellular receptor for bacterial pathogens. These results add new insights into the function of teleost CD46 and the regulation of complement activation in fish.
Materials and Methods
Ethics statement
All experiments involving live animals conducted in this study were approved by the Ethics Committee of Institute of Oceanology, Chinese Academy of Sciences. All methods were carried out in accordance with the relevant guidelines, including any relevant details.
Fish
Clinically healthy tongue sole were purchased from a commercial fish farm in Shandong Province, China. Fish were maintained at 20 °C in aerated seawater and fed daily with commercial dry pellets. Before experiment, the fish were verified to be clinically healthy by plate count as reported previously42. Fish were euthanized with an overdose of tricaine methanesulfonate (Sigma, St. Louis, MO, USA) before tissue collection.
Bacterial strains and culture conditions
E. tarda TX1, P. fluorescens TSS, V. harveyi T4D, V. anguillarum C312, and S. iniae SF1 are bacterial pathogens isolated from diseased fish43,44,45. E. coli BL21 (DE3) and DH5α were purchased from TransGen (Beijing, China). B. subtilis was purchased from China General Microbiological Culture Collection Center (CGMCC). All strains were cultured in Luria–Bertani broth (LB) medium at 37 °C (for E. coli and B. subtilis) or 28 °C (for other bacteria).
Sequence analysis
The amino acid sequence of CsCD46 (GenBank accession no. XP_016891345.1) was analyzed using the BLAST program at the National Center for Biotechnology Information (NCBI). Domain search was performed with the conserved domain search program of NCBI. Multiple sequence alignment was carried out with DNAMAN. O-glycosylation prediction was performed with the glycosylation site prediction tool of http://www.cbs.dtu.dk/services/NetOGlyc/.
Preparation of cDNA
Total RNA was isolated from the tongue sole blood with EZNA Total RNA Kit (Omega Bio-tek, Doraville, GA, USA). The cDNA was synthesized with the Thermo Scientific RevertAid Reverse Transcriptase (Thermo Scientific HyClone, Beijing, China) according to manufacturer’s instructions.
Plasmid construction
The coding sequence of CsCD46 was cloned by PCR with primers F1 (5′-ATGATGTGGAAAATCTCAGTC-3′) and R1 (5′-TCAAGAAGTTCCTGAATCACGGA-3′), which were designed based on the CD46 sequence published previously (GenBank accession no. XP_016891345.1)46. To construct pEtCsCD46, which expresses recombinant CsCD46 (rCsCD46) with His tags, the coding sequence of CsCD46 without the signal peptide sequence (residues 21 to 340) was amplified by PCR with primers CsCD46F (5′-GATATCATGCAAGAGTGTAATGAACCTGTG-3′, underlined sequence, EcoRV site) and CsCD46R (5′-GATATCAGAAGTTCCTGAATCACGGA-3′, underlined sequence, EcoRV site), and PCR program was 94 °C for 4 min; 5 cycles at 94 °C for 30 s, 54 °C for 30 s, 72 °C for 1 min; 25 cycles at 94 °C for 30 s, 60 °C for 30 s, 72 °C for 1 min; followed by 72 °C for 8 min. The PCR product was ligated with the T−A cloning vector T-Simple (TransGen Biotech., Beijing, China), and the recombinant plasmid was digested with EcoRV to retrieve the CsCD46-containing fragment, which was inserted into pET25947 at the SwaI site, resulting in pEtCsCD46. To construct pEtCsFI, which expresses His-tagged recombinant CsFI (rCsFI), CsFI without signal peptide sequence was amplified by PCR with primers CsFIF (5′-GATATCATGACAGATCCTCAAAGTCTATATT-3′, underlined sequence, EcoRV site) and CsFIR (5′-GATATCAGAATTAAACCTAGTGACTGTTG-3′, underlined sequence, EcoRV site). The PCR product was inserted into pET259 as above.
Purification of recombinant proteins and preparation of antibody
E. coli BL21 (DE3) was transformed with pEtCsCD46, pEtCsFI, and pET32a (which expresses the Trx tag). rTrx was purified under the same conditions as rCsCD46 and rCsFI, so that it could be used as a control protein in subsequent experiments that analyzed the activities of rCsCD46 and rCsFI. The transformants were cultured in LB medium at 37 °C to mid-log phase, and the expression of rCsCD46, rCsFI, and rTrx were induced by adding isopropyl-β-D-thiogalactopyranoside to a final concentration of 1 mM. After growth at 16 °C for an additional 16 h, the cells were harvested by centrifugation, and recombinant proteins were purified under denaturing conditions using nickel-nitrilotriacetic acid columns (GE Healthcare, Piscataway, NJ, USA) as recommended by the manufacturer. The purified proteins were reconstituted as described previously48. The reconstituted proteins were treated with Triton X-114 to remove endotoxin as reported previously49. The proteins were dialyzed for 24 h against phosphate-buffered saline (PBS) and concentrated using PEG 20000 (Solarbio, Beijing, China). The concentrations of the purified proteins were determined using the Bradford method with bovine serum albumin as a standard. Mouse antibodies against rCsCD46, rCsFI, and rTrx were prepared as described previously50. The antibodies were purified using rProtein G Beads (Solarbio, Beijing, China). The specificity and titer of the serum antibodies were determined as reported previously51 by enzyme-linked immunosorbent assay (ELISA).
Antibody detection of CsCD46 and CsIF in PBL and serum
Tongue sole PBL were prepared with Percoll as reported previously52. The proteins of ~109 cells were prepared by lysing the cells with 200 µl 1% Triton X-100. The proteins were resolved by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to a nitrocellulose membrane. Western blot was performed as reported previously49 with anti-rCsCD46 antibody (1 µg/ml). To detect CsFI in serum, blood was drawn from the caudal vein of tongue sole and placed on ice immediately. The blood was allowed for clotting for 2 h. Serum was then collected by centrifugation at 4000 rpm for 10 min. For Western blot analysis, the serum was concentrated approximately 20-fold and subjected to SDS-PAGE; the proteins were then transferred to a nitrocellulose membrane, and Western blot was performed as above with anti-rCsFI antibody. Western blot examining binding of anti-rCsFI and anti-rCsCD46 antibodies to rCsFI and rCsCD46, respectively, was performed as above. The results showed that anti-rCsCD46 and anti-rCsFI antibodies detected CsCD46 and CsFI, respectively, in PBL and serum (Fig. S3). The molecular mass of the natural CsCD46 appeared to be larger than the theoretical molecular mass, which is probably due to glycosylation of the protein.
Complement activity assay
Complement activity was determined as reported previously53. To examine the effect of rCsCD46 on complement activation, tongue sole serum was diluted serially in Hank′s Balanced Salt Solution (HBSS) (Solarbio, Beijing, China). Inactivated serum was prepared by heating at 56 °C for 30 min and serum dilutions were then added with or without rCsCD46 or rTrx to a final concentration of 40 µg/ml. The mixtures were then incubated at 22 °C for 1 h. To measure hemolytic activity, rabbit red blood cells (Guangzhou Future Technology Co., Ltd, Guangzhou, China) were washed and resuspended in HBSS, and 10 µl of suspension was then added to 50 µl of each of the serum mixtures or heat-inactivated serum in a 96-well culture plate, followed by incubation at 22 °C for 30 min. After incubation, the supernatant was collected by centrifugation and the absorbance at 450 nm was determined. For easy of comparison, the hemolytic activity of the control sample at 1/8 serum dilution was defined as 1. For bactericidal activity assay, the above serum mixtures were either untreated or heated at 56 °C for 30 min. E. coli DH5α was cultured in LB medium to an optical density (OD)600 of 0.8; the cells were washed and resuspended to 2 × 106 colony-forming units (CFU)/ml in HBSS. The bacterial suspension was combined with an equal volume of heated or unheated serum mixture. After incubation at 22 °C for 1 h, the sample was serially diluted and plated in triplicate in LB agar plates. The plates were incubated at 28 °C for 24 h, and the colonies that appeared on the plates were enumerated. The genetic nature of the colonies was verified by PCR. For easy of comparison, the bactericidal activity of the control sample at 1/8 serum dilution was defined as 1.
Co-immunoprecipitation
Interaction between rCsCD46 and CsFI was determined by co-immunoprecipitation using an Immunoprecipitation Protein A/G Plus Agarose Kit (Sangon Biotech, Shanghai, China) according to the manufacturer’s instructions. Tongue sole serum containing rCsCD46 (20 µg/ml, 40 µg/ml, or 80 µg/ml) or rTrx (80 µg/ml) was mixed with E. coli (106 CFU/ml) and incubated at 22 °C for 1 h. The mixture was passed through a 0.22-µm microfilter to remove bacterial cells. The filtered serum was treated with anti-His antibody (1 µg/ml) at 4 °C for overnight. After treatment, the mixture was transferred to a spin column bound with protein-A/G and incubated at 4 °C for 2 h. After incubation, the spin column was centrifuged and washed three times. Fifty microliters loading buffer was added to the column, and the column was treated at 95 °C for 5 min. The immunoprecipitated complex was collected by centrifugation and concentrated 20-fold with PEG 20000. The immunoprecipitated complex and rCsFI protein (positive control) were resolved by SDS-PAGE and transferred to a nitrocellulose membrane (Amersham, Cambridge, UK). Western blot was performed as above with anti-rCsFI antibody (1 µg/ml). The membrane of Western blot was washed with PBS and detected with the HRP-DAB kit (Tiangen, Beijing, China). The color reaction was stopped after 10 min by several washes with water, and the membrane was air dried.
Interaction between rCsCD46 and rCsFI was determined by co-immunoprecipitation as above. rCsCD46 (80 µg/ml) was mixed with rCsFI (80 µg/ml) and incubated at 22 °C for 1 h. The mixture was treated with anti-rCsCD46 antibody (1 µg/ml) at 4 °C for overnight. Co-immunoprecipitation and Western blot were performed as above.
Involvement of CsFI in rCsCD46-mediated effect on complement activation
To examine the effect of anti-rCsFI antibody on rCsCD46-mediated inhibition of complement activation, tongue sole serum was diluted serially in HBSS. Anti-rCsFI antibody or preimmune antibody (2 µg/ml) was mixed with the serum dilutions in the presence or absence of 40 µg/ml rCsCD46 or rTrx. The control serum was mixed with the same volume of HBSS. The mixtures were incubated at 22 °C for 1 h, and hemolysis activity was measured as above. To examine the effect of CsFI depletion on rCsCD46-mediated inhibition of complement activation, tongue sole serum was treated with anti-rCsFI antibody (1 µg/ml) or anti-rTrx antibody (1 µg/ml) at 4 °C for overnight. After incubation, the mixture was transferred to the spin column bound with protein-A/G and incubated at 4 °C for 2 h. The spin column was then centrifuged, and the serum was collected. The serum was diluted serially in HBSS. rCsCD46 was mixed with anti-rCsFI antibody-treated serum, anti-rTrx antibody-treated serum (control serum), or normal serum, and the mixture was determined for hemolytic activity as above.
Fluorescence microscopy
Tongue sole PBL were prepared with Percoll as above. Detecting of CsCD46 on PBL by fluorescence microscopy was performed as reported previously54. Briefly, PBL were resuspended in PBS to 107 cells/ml. Anti-rCsCD46 antibody or anti-rTrx antibody (2 µg/ml) was added to PBL suspension. The cells were incubated at 22 °C for 2 h and then centrifuged at 300 g for 10 min. The cells were collected, washed three times with PBS, and resuspended in PBS. Fluorescein isothiocyanate (FITC)-labeled goat anti-mouse IgG (Bioss, Beijing, China) (1/1000 dilution) was added to the cells, followed by incubation at 22 °C for 2 h in the dark. The cells were centrifuged and washed as above. The cells were resuspended in PBS and observed with a fluorescence microscope (Nikon E800, Japan).
Flow cytometry analysis of cell damage
PBL prepared above were resuspended in L-15 medium (Thermo Scientific HyClone, Beijing, China) to 1 × 107 cells/ml. Anti-rCsCD46 antibody, anti-rTrx antibody, anti-rCsTLR2 antibody55, preimmune antibody, all at 2 µg/ml, or PBS was added to PBL. After incubation at 22 °C for 1 h, the cells were centrifuged at 300 g for 10 min. The cells were washed three times with PBS and resuspended in serum, followed by incubation at 22 °C for 1 h. The control cells were untreated with serum or antibody. To measure cellular damage, PBL were treated with annexin V and propidium iodide (PI) (Majorbio Biotech, Shanghai, China) for 15 min in the dark according to the manufacturer’s instructions. The cells were then subjected to flow cytometry using a FACSort Flow Cytometer (BD Biosciences, USA). Data analysis was performed using FlowJo software 7.6.1 (Tree Star Inc, Ashland, OR, USA). To examine the effect of factor I on CsCD46-induced protection of PBL against complement damage, anti-rCsFI antibody or anti-rTrx antibody (2 µg/ml) was mixed with serum and incubated at 22 °C for 1 h. Treatment of PBL with the serum and measure of cellular damage were performed as above.
Binding of rCsCD46 to bacteria
Bacteria were cultured in LB medium to OD600 0.8 and resuspended in coating buffer (15 mM Na2CO3, 35 mM NaHCO3, pH 9.6) to 108 CFU/ml. Bacteria–protein interaction was determined by ELISA as reported previously56. Briefly, the bacterial suspension was added to 96-well ELISA plates and incubated at 4 °C for overnight. rCsCD46 was heated at 100 °C for 10 min to prepare inactivated rCsCD46. After blocking with 5% skim milk and washing with PBST (PBS containing 0.1% Tween- 20), different concentrations (2.5 µg/ml, 5 µg/ml, 10 µg/ml, 20 µg/ml, 40 µg/ml, and 80 µg/ml) of rCsCD46, heat inactivated rCsCD46, rTrx, or PBS were added to the plates, and the plates were incubated at 22 °C for 2 h. The plates were then washed with PBST, and mouse anti-His antibody (Tiangen, Beijing, China) and horseradish peroxidase (HRP)-conjugated goat anti-mouse IgG (Bioss, Beijing, China) were added to the plates sequentially as reported previously56. The TMB Kit (Tiangen, Beijing, China) was used to observe color development. The plates were read at 450 nm using a Precision microplate reader (Molecular Devices, Toronto, Canada). Positive readings were defined as values at least twice that of the control. The assay was performed three times and the results were expressed as the binding index, defined as follows: A 450 of protein/A 450 of PBS.
To examine the effect of rCsCD46 on bacterial survival, E. tarda and P. fluorescens (104 CFU) were incubated with 40 µg/ml rCsCD46, rTrx, or PBS for 4 h at 22 °C. The cells were then diluted, and the dilutions were plated in triplicate on LB agar plates. The plates were incubated at 28 °C for 24 h, and the bacterial numbers were counted.
To determine the effect of rCsFI on the interaction between rCsCD46 and bacteria, P. fluorescens suspension was prepared as above and added to a 96-well ELISA plate. The plate was incubated at 4 °C, blocked with skim milk, and washed with PBST as above. PBS (control), or rCsCD46 plus rCsFI, rCsCD46 plus rTrx, rCsCD46, rCsFI, or rTrx (all proteins being at 40 µg/ml) was added into the plate. The plate was incubated at 22 °C for 2 h and washed as above. ELISA was performed as above.
Effect of anti-rCsCD46 antibody on bacterial attachment to PBL
Anti-rCsCD46 antibody (1 µg/ml), anti-rTrx antibody (1 µg/ml), preimmune antibody (1 µg/ml), or PBS was added to PBL. The cells were incubated at 22 °C for 1 h. E. tarda and P. fluorescens were cultured in LB medium to an OD600 of 0.8 and resuspended in PBS to 1 × 106 CFU/ml and 1 × 107 CFU/ml, respectively. One hundred microliters of bacterial suspension was added to each well of PBL (~105 cells/well). The plate was incubated at 22 °C. At 1, 2, and 4 h post-incubation, the cells were washed three times with PBS to remove uninfected bacteria. The cells were lysed by adding 100 µl of 1% Triton X-100; the lysate was diluted in LB medium and plated on LB agar plates. The plates were incubated at 28 °C for 24 h, and the colonies that emerged on the plates were counted. The genetic nature of the colonies was verified by PCR with primers reported previously57.
Effect of rCsCD46 on bacterial infection in fish
E. tarda and P. fluorescens were cultured as above and resuspended in PBS to 2 × 106 CFU/ml and 2 × 107 CFU/ml, respectively. rCsCD46 or rTrx was added to the cells to the final concentration of 40 µg/ml. The control cells were added with PBS. Tongue soles were divided randomly into three groups and injected intramuscularly with 50 μl suspensions of bacteria, bacteria plus rCsCD46, or bacteria plus rTrx. At 12 h, 24 h, and 48 h post-infection, kidney, spleen, and blood were collected from the fish (five at each time point). The tissues were homogenized in PBS. The homogenates were serial diluted and plated in triplicate on LB agar plates. After incubation at 28 °C for 48 h, the colonies that appeared on the plates were enumerated. The genetic identification of these colonies was verified by PCR as above.
Statistical analysis
All experiments were repeated three times. Statistical analyses were carried out with SPSS 17.0 software (SPSS Inc., Chicago, IL, USA). Data were analyzed with analysis of variance (ANOVA), and statistical significance was defined as P < 0.05. In Fig. 3A and B, statistical significance represented by different letters was defined as P < 0.05.
References
Walport, M. J. Complement. First of two parts. N. Engl. J. Med. 344, 1058–1066 (2001).
Merle, N. S., Church, S. E., Fremeaux-Bacchi, V. & Roumenina, L. T. Complement system part I– molecular mechanisms of activation and regulation. Front. Immunol. 6, 262 (2015).
Ricklin, D., Hajishengallis, G., Yang, K. & Lambris, J. D. Complement: a key system for immune surveillance and homeostasis. Nat. Immunol. 11, 785–797 (2010).
Endo, Y., Takahashi, M. & Fujita, T. Lectin complement system and pattern recognition. Immunobiology 211, 283–293 (2006).
Sarma, J. V. & Ward, P. A. The complement system. Cell Tissue Res. 343, 227–235 (2011).
Longhi, M. P., Harris, C. L., Morgan, B. P. & Gallimore, A. Holding T cells in check – a new role for complement regulators? Trends Immunol. 27, 102–108 (2006).
Rose, K. L. et al. Factor I is required for the development of membranoproliferative glomerulonephritis in factor H–deficient mice. J. Clin. Invest. 118, 608–618 (2008).
Terado, T., Nonaka, M. I., Nonaka, M. & Kimura, H. Conservation of the modular structure of complement factor I through vertebrate evolution. Dev. Comp. Immunol. 26, 403–413 (2002).
Gros, P., Milder, F. J. & Janssen, B. J. Complement driven by conformational changes. Nat. Rev. Immunol. 8, 48–58 (2008).
Noris, M. & Remuzzi, G. Overview of complement activation and regulation. Seminars Nephrol. 33, 479–492 (2013).
Persson, B. D. et al. Structure of the extracellular portion of CD46 provides insights into its interactions with complement proteins and pathogens. PLoS Pathog. 6, e1001122 (2010).
Barilla-LaBarca, M. L., Liszewski, M. K., Lambris, J. D., Hourcade, D. & Atkinson, J. P. Role of membrane cofactor protein (CD46) in regulation of C4b and C3b deposited on Cells. J. Immunol. 168, 6298–6304 (2002).
Cardone, J., Le Friec, G. & Kemper, C. CD46 in innate and adaptive immunity: an update. Clin. Exp. Immunol. 164, 301–311 (2011).
Riley-Vargas, R. C., Gill, D. B., Kemper, C., Liszewski, M. K. & Atkinson, J. P. CD46: expanding beyond complement regulation. Trends Immunol. 25, 496–503 (2004).
Liszewski, M. K. & Atkinson, J. P. Complement regulator CD46: genetic variants and disease associations. Hum. Genomics 9, 7 (2015).
Marttila, M. et al. CD46 is a cellular receptor for all species B adenoviruses except types 3 and 7. J. Virol. 79, 14429–14436 (2005).
Joubert, P. E. et al. Autophagy induction by the pathogen receptor CD46. Cell Host Microbe 6, 354–366 (2009).
Weyand, N. J., Calton, C. M., Higashi, D. L., Kanack, K. J. & So, M. Presenilin/g-secretase cleaves CD46 in response to Neisseria infection. J. Immunol. 184, 694–701 (2010).
Greenstone, H. L., Santoro, F., Lusso, P. & Berger, E. A. Human herpesvirus 6 and measles virus employ distinct CD46 domains for receptor function. J. Biol. Chem. 277, 39112–39118 (2002).
Krey, T. et al. Function of bovine CD46 as a cellular receptor for bovine viral diarrhea virus is determined by complement control protein 1. J. Virol. 80, 3912–3922 (2006).
Boshra, H., Li, J. & Sunyer, J. O. Recent advances on the complement system of teleost fish. Fish Shellfish Immunol. 20, 239–262 (2006).
Gao, Z., Li, M., Ma, J. & Zhang, S. An amphioxus gC1q protein binds human IgG and initiates the classical pathway: Implications for a C1q-mediated complement system in the basal chordate. Eur. J. Immunol. 44, 3680–3695 (2014).
Tsujikura, M. et al. A CD46-like molecule functional in teleost fish represents an ancestral form of membrane-bound regulators of complement activation. J. Immunol. 194, 262–272 (2015).
Nur, I., Harada, H., Tsujikura, M., Somamoto, T. & Nakao, M. Molecular characterization and expression analysis of three membrane-bound complement regulatory protein isoforms in the ginbuna crucian carp Carassiusauratus langsdorfii. Fish Shellfish Immunol. 35, 1333–1337 (2013).
Richards, A. et al. Mutations in human complement regulator, membrane cofactor protein (CD46), predispose to development of familial hemolytic uremic syndrome. Proc. Natl. Acad. Sci. USA 100, 12966–12971 (2003).
Liszewski, M. K., Post, T. W. & Atkinson, J. P. Membrane cofactor protein (MCP or CD46): newest member of the regulators of complement activation gene cluster. Annu. Rev. Immunol. 9, 431–455 (1991).
Masaki, T., Matsumoto, M., Nakanishi, I., Yasuda, R. & Seya, T. Factor I-dependent inactivation of human complement C4b of the classical pathway by C3b/C4b receptor (CR1, CD35) and membrane cofactor protein (MCP, CD46). J. Biochem. 111, 573–578 (1992).
Kim, D. D. & Song, W. C. Membrane complement regulatory proteins. Clin. Immunol. 118, 127–136 (2006).
Lublin, D. M. & Atkinson, J. P. Decay-accelerating factor: biochemistry, molecular biology, and function. Annu. Rev. Immunol. 7, 35–58 (1989).
Farkas, I. et al. CD59 blocks not only the insertion of C9 into MAC but inhibits ion channel formation by homologous C5b–8 as well as C5b–9. J. Physiol. 539, 537–545 (2002).
Morgan, B. P. & Harris, C. L. Complement regulatory proteins. 283–284 (London: Academic Press, 1999).
Nakanishi, I. et al. Identification and characterization of membrane cofactor protein (CD46) in the human kidneys. Eur. J. Immunol. 24, 1529–1535 (1994).
Liszewski, M. K., Farries, T. C., Lublin, D. M., Rooney, I. A. & Atkinson, J. P. Control of the complement system. Adv. Immunol. 61, 201–283 (1996).
Rooijakkers, S. H. & van Strijp, J. A. Bacterial complement evasion. Mol. Immunol. 44, 23–32 (2007).
Zipfel, P. F., Würzner, R. & Skerka, C. Complement evasion of pathogens: common strategies are shared by diverse organisms. Mol. Immunol. 44, 3850–3857 (2007).
Blue, C. E., Spiller, O. B. & Blackbourn, D. J. The relevance of complement to virus biology. Virology 319, 176–184 (2004).
Stoermer, K. A. & Morrison, T. E. Complement and viral pathogenesis. Virology 411, 362–373 (2011).
Russel, S. CD46: a complement regulator and pathogen receptor that mediates links between innate and acquired immune fuction. Tissue Antigens 64, 111–118 (2004).
Nemerow, G. R., Pache, L., Reddy, V. & Stewart, P. L. Insights into adenovirus host cell interactions from structural studies. Virology 384, 380–388 (2009).
Yamamoto, H., Fara, A. F., Dasgupta, P. & Kemper, C. CD46: the ‘multitasker’ of complement proteins. Int. J. Biochem. Cell Biol. 45, 2808–2820 (2013).
Gill, D. B. & Atkinson, J. P. CD46 in Neisseria pathogenesis. Trends Mol. Med. 10, 459–465 (2004).
Zhou, Z. J. & Sun, L. CsCTL1, a teleost C-type lectin that promotes antibacterial and antiviral immune defense in a manner that depends on the conserved EPN motif. Dev. Comp. Immunol. 50, 69–77 (2015).
Zhang, M., Sun, K. & Sun, L. Regulation of autoinducer 2 production and luxS expression in a pathogenic Edwardsiella tarda strain. Microbiology 154, 2060–2069 (2008).
Wang, H. R., Hu, Y. H., Zhang, W. W. & Sun, L. Construction of an attenuated Pseudomonas fluorescens strain and evaluation of its potential as a cross protective vaccine. Vaccine 27, 4047–4055 (2009).
Cheng, S., Hu, Y. H., Jiao, X. D. & Sun, L. Identification and immunoprotective analysis of a Streptococcus iniae subunit vaccine candidate. Vaccine 28, 2636–2641 (2010).
Chen, S. et al. Whole-genome sequence of a flatfish provides insights into ZW sex chromosome evolution and adaptation to a benthic lifestyle. Nat. Genet. 46, 253–260 (2014).
Hu, Y. H., Zheng, W. J. & Sun, L. Identification and molecular analysis of a ferritin subunit from red drum (Sciaenops ocellatus). Fish Shellfish Immunol. 28, 678–686 (2010).
Li, M. F., Wang, C. & Sun, L. Edwardsiella tarda MliC, a lysozyme inhibitor that participates in pathogenesis in a manner that parallels ivy. Infect. Immun. 83, 583–90 (2015).
Zhang, B. C. & Sun, L. Tongue sole (Cynoglossus semilaevis) prothymosin alpha: cytokine-like activities associated with the intact protein and the C-terminal region that lead to antiviral immunity via Myd88-dependent and –independent pathways respectively. Dev. Comp. Immunol. 53, 96–104 (2015).
Wang, W., Zhang, M., Xiao, Z. & Sun, L. Cynoglossus semilaevis ISG15: a secreted cytokine-like protein that stimulates antiviral immune response in a LRGG motif-dependent manner. PLoS One 7, e44884 (2012).
Sun, Y., Zheng, W. J., Hu, Y. H., Sun, B. G. & Sun, L. Edwardsiella tarda Etal, an in vivo induced antigen that is involved in host infection. Infect. Immun. 80, 2948–2955 (2012).
Li, Y. X., Hu, Y. H., Sun, J. S. & Sun, L. CsCXCe1: a novel Cynoglossus semilaevis CXC chemokine that functions as a chemoattractant and an immunomodulator for peripheral blood leukocytes. Dev. Comp. Immunol. 37, 54–64 (2012).
Li, M. F., Sun, L. & Li, J. Edwardsiella tarda evades serum killing by preventing complement activation via the alternative pathway. Fish Shellfish Immunol. 43, 325–329 (2015).
Li, M. F., Li, J. & Sun, L. CsMAP34, a teleost MAP with dual role: A promoter of MASP-assisted complement activation and a regulator of immune cell activity. Scientific reports 6, 39287 (2016).
Li, X. P. & Sun, L. Toll-like receptor 2 of tongue sole Cynoglossus semilaevis: signaling pathway and involvement in bacterial infection. Fish Shellfish Immunol. 51, 321–328 (2016).
Li, M. F., Li, Y. X. & Sun, L. CD83 is required for the induction of protective immunity by a DNA vaccine in a teleost model. Dev. Comp. Immunol. 51, 141–147 (2015).
Zheng, W. J. & Sun, L. Evaluation of housekeeping genes as references for quantitative real time RT-PCR analysis of gene expression in Japanese flounder (Paralichthys olivaceus). Fish Shellfish Immunol. 30, 638–645 (2011).
Acknowledgements
This work was supported by the grants of Scientific funds for outstanding young scientists of Shandong Province Award (ZR2016CB01), the National Natural Science Foundation of China (31330081 and 31602197), the AoShan Talents Program Supported by Qingdao National Laboratory for Marine Science and Technology (No. 2015ASTP), and the Taishan Scholar Program of Shandong Province.
Author information
Authors and Affiliations
Contributions
L.S. and M.F.L. conceived and designed the experiments, M.F.L. and Z.H.S. performed the experiments and analyzed the data, and M.F.L. and L.S. wrote the paper.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, Mf., Sui, Zh. & Sun, L. A teleost CD46 is involved in the regulation of complement activation and pathogen infection. Sci Rep 7, 15028 (2017). https://doi.org/10.1038/s41598-017-15124-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-15124-y
This article is cited by
-
Edwardsiella tarda TraT is an anti-complement factor and a cellular infection promoter
Communications Biology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.