Abstract
We engaged pet salamander owners in the United States to screen their animals for two amphibian chytrid fungal pathogens Batrachochytrium dendrobatidis (Bd) and B. salamandrivorans (Bsal). We provided pet owners with a sampling kit and instructional video to swab the skin of their animals. We received 639 salamander samples from 65 species by mail, and tested them for Bd and Bsal using qPCR. We detected Bd on 1.3% of salamanders (95% CI 0.0053–0.0267) and did not detect Bsal (95% CI 0.0000–0.0071). If Bsal is present in the U.S. population of pet salamanders, it occurs at a very low prevalence. The United States Fish and Wildlife Service listed 201 species of salamanders as “injurious wildlife” under the Lacey Act (18 U.S.C. § 42) on January 28, 2016, a precautionary action to prevent the introduction of Bsal to the U.S. through the importation of salamanders. This action reduced the number of salamanders imported to the U.S. from 2015 to 2016 by 98.4%. Our results indicate that continued precautions should be taken to prevent the introduction and establishment of Bsal in the U.S., which is a hotspot of salamander biodiversity.
Similar content being viewed by others
Introduction
Emerging fungal pathogens are an increasing threat to global biodiversity1,2,3. The pathogenic amphibian chytrid fungus Batrachochytrium dendrobatidis (Bd), is a major driver of global amphibian declines and extinctions4,5,6. From 2010–2013, catastrophic enigmatic declines of fire salamanders (Salamandra salamandra) in the Netherlands resulted in the description of a second species belonging to the genus Batrachochytrium, the salamander-eating fungus B. salamandrivorans (Bsal)7. Known host suitability and susceptibility to Bsal is restricted to salamanders (Urodela)7,8. Salamanders belonging to the family Salamandridae are highly susceptible to disease7,8,9. Although few frogs (Anura) have been tested in susceptibility trials, most have been found to be resistant to infection7,8. However, the common midwife toad (Alytes obstetricans) has been shown to maintain subclinical infections of Bsal for several weeks and may act as infection reservoirs10. It is hypothesized that Bsal is endemic to salamanders in Asia and was introduced to Europe via the pet trade8. This hypothesis is supported by recent Bsal detection at endemic levels in newt populations of Vietnam9. Bsal has now been detected in the environment in the Netherlands, Belgium, Germany, and Vietnam9,11,12, and in captive individuals in the United Kingdom and Germany11,13.
Salamanders from Asia represent the majority of salamander species traded internationally for the pet trade and pose a significant threat for the spread of disease14,15,16. The U.S. is a global salamander biodiversity hotspot and the threat of Bsal introduction via the pet trade generated significant concern15,16. As a precautionary approach to avoid Bsal introduction, on 28 January 2016, the U.S. Fish and Wildlife Service included 20 genera of salamanders representing 201 species in the “injurious wildlife” list under the Lacey Act (18 U.S.C. § 42). This was intended to restrict interstate transport and importation of injurious species (https://www.fws.gov/injuriouswildlife/salamanders.html), but recent legal determinations on the scope of the Lacey Act (18 U.S.C. § 42) authority for injurious wildlife may reduce these restrictions17. An urgent priority is to determine whether, in fact Bsal already exists in captive or wild salamanders in the U.S. The Bsal Task Force and Partners in Amphibian and Reptile Conservation’s Disease Task Team call for determining the prevalence and biosecurity risk of Bsal in captive collections16. Therefore, we initiated a citizen science project engaging pet salamander owners to screen their salamanders for Bd and Bsal to evaluate the prevalence of these pathogens in U.S. private salamander collections.
Results
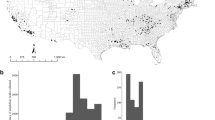
A total of 639 of 4,462 (13.8%) swabs were returned by mail. These 639 swabs were submitted by 56 private salamander owners from 29 states in the U.S. Returned swabs represented 65 species, 22 genera, and three families of salamanders (Ambystomatidae, Plethodontidae, and Salamandridae) (Table 1). Twelve of the genera, containing 40 species tested, are now prohibited from importation to the U.S. under the “injurious wildlife” provision of the Lacey Act (18 U.S.C. § 42). Of the 639 salamanders tested, 486 individuals (76.1%) belong to genera containing at least one species known to be susceptible to Bsal 8,9. Salamander species tested represent 73% of the species of salamanders imported to the U.S. from 2010–2016. Our return rate may have been low due to (i) the limited time window for returning the testing kits given the 12 January 2016 announcement listing 201 species of salamanders under the “injurious wildlife” provision and (ii) the return shipping costs not being covered.
We detected Bd on 1.3% of salamanders (95% CI 0.0053–0.0267) and did not detect Bsal (95% CI 0.0000–0.0071). Seven samples tested positive for Bd, but with low (<100) genomic equivalents that are likely associated with sub-clinical infections (Table 2). We are 95% confident that the maximum prevalence of Bsal in the U.S. captive salamander population is less than 0.71%, if it is in the U.S., and that the prevalence of Bd is between 0.53–2.67%.
Discussion
Bsal has not been detected to date in captive collections or in wild populations in the U.S.8,18,19. Our results suggest that if Bsal is present in private captive U.S. salamander collections, it occurs at extremely low prevalence. The non-detection of Bsal in captive and wild U.S. salamanders suggests that Bsal has not yet been introduced into the U.S., justifying continued monitoring and precautions to prevent its spread. The prevalence of Bd was similar to other surveys of captive collections20.
We screened a diversity of native and non-native salamander species, with higher relative numbers of non-native species. Many of the non-native species we tested have not been imported in recent years indicating that hobbyists may be sustaining captive-bred populations of these animals in the U.S. Many species native to the U.S. were represented by low sample sizes, which may be due to laws protecting the collection of these species, complex captive husbandry and rearing, or low desirability in keeping native species as opposed to exotic species. Cynops ssp. and Paramesotriton ssp. represent 91.6% of individuals imported from 2010–2016 (Table 1), but compose 26.0% of salamanders tested. These species were often sold by wholesalers and chain pet stores, but may not be as popular in the community of hobbyists that responded to our swabbing request. Our sampling may have been biased towards more actively engaged hobbyists. The number of salamanders in private collections in the U.S. remains unknown.
On April 7, 2017, the United States Court of Appeals ruled that the “injurious wildlife” provision of the Lacey Act (18 U.S.C. § 42) does not restrict interstate transport of listed species in the continental U.S.17, which has implications for the potential introduction or spread of Bsal in the U.S. At the time of this publication, neither the United States Fish and Wildlife Service nor Department of Interior have issued official guidance on this court ruling, it is currently unclear if permit requirements or enforcement exists when transporting species listed as “injurious wildlife” across state borders. Interstate trade moratorium of the 201 salamander species listed as “injurious wildlife” on January 28, 2016 may no longer be in effect in the continental U.S. If this interpretation is upheld by the agencies, the ruling will allow for any animals that may be infected with Bsal or Bd to be transported across the U.S. state boundaries. This interpretation may also affect the interstate transport of other invasive taxa that are currently listed as “injurious wildlife”, i.e., Asian carp (Hypophthalmichthys ssp.) and Snakeheads (Channidae). In absence of the Lacey Act (18 U.S.C. § 42) regulating interstate transport of “injurious wildlife” species, enforcement is reliant upon state laws to prohibit the transport or introduction of invasive species. Invasive species are a significant threat to biodiversity, and this ruling sets a precarious precedent for the future of federal management of invasive species in the U.S. The Lacey Act of 1900 (18 U.S.C. § 42) was the first federal law protecting wildlife and was not written to protect wildlife from pathogens. Wildlife agencies with a mandate to manage and mitigate risks to wildlife involving emerging diseases would be better served by comprehensive wildlife health legislation.
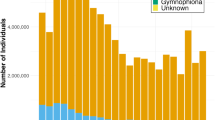
Current import restrictions are limited to 201 species of salamanders known to be susceptible to Bsal. The listing of these species as “injurious wildlife” effectively halted importation of salamanders to the U.S. In 2015, there were 186 importations of salamanders containing 68,102 individuals. The declared value of all salamander imports in 2015 was $43,501 (USD). A total of 7 importations of salamanders containing 1,062 individuals occurred in 2016. Four of these importations contained species to be listed as “injurious wildlife” and were imported before the “injurious wildlife” listing went into effect on January 28, 2016. A total of 22 salamanders were imported after the “injurious wildlife” listing came into effect in 2016, one shipment contained 18 Paramesotriton honkongensis, an “injurious wildlife” species. Although the “injurious wildlife” listing of 201 salamander species reduced the volume of salamanders imported to the U.S., we do not have a comprehensive understanding of the potential for all urodelans, anurans or gymnophionans to act as Bsal hosts. One of the current limitations to verifying that an animal is pathogen free is that the diagnostic assays for Bd and Bsal use real-time qPCR which is (i) time intensive, (ii) requires specialized equipment, and (iii) requires trained personnel. The use of qPCR for diagnosing these pathogens makes it challenging to screen animals at ports of entry or sale. The recent development of lateral flow assay (LFA), a portable, rapid, inexpensive test to detect Bd and Bsal has potential to be an invaluable tool for detecting these pathogens21. This technology could be deployed for rapid testing of imported amphibians at port of entry, in private collections, zoos and in the field.
Communication and engagement with the amphibian hobbyist community is imperative to reduce the risk of pathogen introduction into the environment. Newts belonging to the genus Cynops are potential reservoirs for Bsal 8. Release of unwanted pets into the environment has led to the introduction of C. pyrrhogaster into the wild in Massachusetts22, and C. orientalis in Florida23, and presents a potential pathway for Bsal introduction into native salamander populations in the U.S. There are no proven mitigation methods for preventing the spread of Bsal once introduced into the environment, and Bsal introduction in the U.S. would likely be catastrophic for salamander biodiversity. Release of unwanted pets into the environment should never be considered an option. We encourage hobbyists to test their animals for Bsal and Bd, and disinfect wastewater and materials from enclosures before disposal to prevent pathogen introduction to the environment.
Our citizen science project introduced pet owners to the risks of these emerging pathogens and to biosecurity protocols that can reduce the risk of pathogen introduction to the environment. A Bsal Task Force (salamanderfungus.org) has been established to provide informational resources to respond to possible cases of Bsal in the U.S. Continued public outreach, salamander disease testing, preventing release of unwanted pets, and education about biosecurity should be a priority to prevent Bsal introduction.
Methods
Salamander Testing
Free testing for Batrachochytrium salamandrivorans and Batrachochytrium dendrobatidis was offered to salamander hobbyists through the Amphibian Survival Alliance website (http://www.amphibians.org/salamanderheros/) between November 2014 and January 2016. Experimental methods were approved by the Smithsonian National Zoological Park Institutional Animal Care and Use Committee approval #14–36. All methods were carried out in accordance to these guidelines and regulations. Testing kits were mailed to salamander hobbyists and included one of the following for each salamander: MW113 Swab (Medical Wire, United Kingdom), a pair of nitrile powder-free gloves, 1.5 ml microcentrifuge tube, and a plastic ziplock bag. A swabbing protocol, instructional video (https://www.youtube.com/watch?v=U5h5srxXAaY&), and data sheet were provided. Each salamander was captured in a clean plastic bag and swabbed 10 times on the ventral surface, five times on each foot, and five unilateral strokes on the tail. The swab was air-dried for 5 minutes, then placed in the 1.5 ml centrifuge tube and returned via mail with a corresponding data sheet noting the species and swab number. Participants were instructed to return their swabs before the 28 January 2016 interstate movement moratorium came into effect. After testing, participants were informed whether Bsal and Bd was detected on their salamanders. Hobbyists with salamanders that tested positive for Bd were advised to consult a veterinarian for treatment.
DNA Extraction and qPCR
Dried swabs returned by mail were stored at −20 °C until extracted using a Qiagen DNeasy Blood and Tissue Kit (Germantown, MD, USA) with the manufacturer’s protocol. Real-time PCR procedures followed Blooi et al.24 using the CFX96 real-time system (Bio-Rad Laboratories, Hercules, CA), except that we assayed B. dendrobatidis and B. salamandrivorans independently and in duplicate samples per individual, rather than using the duplex approach, and a FAM-labeled STerC probe was used in place of a Cy5-labeled STerC probe. If only one of the duplicate samples tested positive (n = 639), the real-time PCR was repeated in duplicate to verify the Bsal or Bd status.
We verified that our protocol successfully extracted and amplified DNA, in the potential absence of amplification of Bd or Bsal by attempting to amplify a small section of salamander mitochondrial DNA. We designed the primers Salcytb1 (forward: 5′CAATGGCCCACACTAYACGA3′ and reverse: 5′TGCTGAGTGTGTGTCTGCTG3′) and Salcytb2 (forward: 5′AGGGGCCACAGTCATYACHA3′ and reverse: 5′CCTGTTGGGTTGTTTGABCC3′) to amplify an approximately 200 bp DNA fragment in the mitochondrial cytochrome b (ctyb) gene by using available salamander ctyb sequences on Genbank. We tested 95 individuals that represented multiple DNA extraction plates, 27 salamander species and 17 pet owners. Each 25-ul PCR assay consisted of 1.25U of AmpliTaq Gold DNA Polymerase (ThermoFisher), 2.5 uM MgCl2, 1x PCR Buffer, 200 nM dNTPs, 600 nM reverse primer, 600 nM forward primer, and 5 ul DNA template. PCR conditions were 95 °C for 10 m, followed by 45 cycles of 95 °C for 15 s, 51 °C for 30 s, 72 °C for 30 s, and a final extension (72 °C for 5 m).
Data Analysis
We obtained Law Enforcement Management Information System (LEMIS) records of live salamander trade imports from 2015–2016 using a Freedom of Information Act request to the U.S. Fish and Wildlife Service to determine the effect of the “injurious wildlife” listing of 201 salamander species on the number of salamanders imported to the U.S (see Supplementary Table S2). Previously obtained and published LEMIS records of salamander importations from 2004–2014 were used to compare the relative proportion of species imported vs. maintained in captivity16.
We calculated Clopper-Pearson 95% Confidence Intervals for Bd and Bsal prevalence. We successfully amplified salamander ctyb DNA in 80/95 swabs, indicating that 84% of samples had good DNA quality. Samples that did not amplify had significantly lower DNA concentrations than those that amplified (Wilcoxon rank sum test, W = 54, p = 0.026). We did not observe a pattern in samples that did not amplify; other samples from the same pet owners amplified, and samples from the same salamander species sampled by other pet owners amplified. It is possible that instructions for swabbing salamanders were not properly followed for a small subset of individual salamanders, DNA degraded during shipping, or that substrate on the swab inhibited DNA extraction. We calculated our confidence intervals to incorporate our confidence in DNA quality (639 samples tested × 84% = 537 samples; Table 2). With no positive samples of Bsal, we assumed the prevalence was 1% for calculating the probability of detecting at least one positive individual8.
References
Fisher, M. C. et al. Emerging fungal threats to animal, plant and ecosystem health. Nature 484, 186–194 (2012).
Frick, W. F. et al. An Emerging Disease Causes Regional Population Collapse of a Common North American Bat Species. Science 329, 679–682 (2010).
Lorch, J. M. et al. Snake fungal disease: an emerging threat to wild snakes. Phil Trans R Soc B 371, 20150457 (2016).
Stuart, S. N. et al. Status and Trends of Amphibian Declines and Extinctions Worldwide. Science 306, 1783–1786 (2004).
Lips, K. R. et al. Emerging infectious disease and the loss of biodiversity in a Neotropical amphibian community. Proc. Natl. Acad. Sci. USA 103, 3165–3170 (2006).
Wake, D. B. & Vredenburg, V. T. Are we in the midst of the sixth mass extinction? A view from the world of amphibians. Proc. Natl. Acad. Sci. 105, 11466–11473 (2008).
Martel, A. et al. Batrachochytrium salamandrivorans sp. nov. causes lethal chytridiomycosis in amphibians. Proc. Natl. Acad. Sci. 110, 15325–15329 (2013).
Martel, A. et al. Recent introduction of a chytrid fungus endangers Western Palearctic salamanders. Science 346, 630–631 (2014).
Laking, A. E., Ngo, H. N., Pasmans, F., Martel, A. & Nguyen, T. T. Batrachochytrium salamandrivorans is the predominant chytrid fungus in Vietnamese salamanders. Sci. Rep. 7, 44443 (2017).
Stegen, G. et al. Drivers of salamander extirpation mediated by Batrachochytrium salamandrivorans. Nature 544, 353–356 (2017).
Sabino-Pinto, J. et al. First detection of the emerging fungal pathogen Batrachochytrium salamandrivorans in Germany. https://doi.org/10.1163/15685381-00003008 (2015).
Spitzen-van der Sluijs, A. et al. Expanding Distribution of Lethal Amphibian Fungus Batrachochytrium salamandrivorans in Europe. Emerg. Infect. Dis. 22, 1286–1288 (2016).
Cunningham, A. A. et al. Emerging disease in UK amphibians. Vet. Rec. 176, 468–468 (2015).
Kolby, J. E. et al. First Evidence of Amphibian Chytrid Fungus (Batrachochytrium dendrobatidis) and Ranavirus in Hong Kong Amphibian Trade. PLOS ONE 9, e90750 (2014).
Yap, T. A., Koo, M. S., Ambrose, R. F., Wake, D. B. & Vredenburg, V. T. Averting a North American biodiversity crisis. Science 349, 481–482 (2015).
Gray, M. J. et al. Batrachochytrium salamandrivorans: The North American Response and a Call for Action. PLOS Pathogens. 11, e1005251 (2015).
Srinivasan, S. US Association of Reptile Keepers, Inc. et al. Appelles v. Ryan Zinke, the Honorable, in his Official Capacity as the Secretary of the Interior and United States Fish and Wildlife Service, Appellants, Human Society of the United States and Center for Biological Diversity, Appellees. (2017).
Bales, E. K. et al. Pathogenic Chytrid Fungus Batrachochytrium dendrobatidis, but Not B. salamandrivorans, Detected on Eastern Hellbenders. PLOS ONE 10, e0116405 (2015).
Parrott, J. C. et al. Survey of Pathogenic Chytrid Fungi (Batrachochytrium dendrobatidis and B. salamandrivorans) in Salamanders from Three Mountain Ranges in Europe and the Americas. EcoHealth 1–7, https://doi.org/10.1007/s10393-016-1188-7 (2016).
Sluijs, A. Sder et al. Clinically healthy amphibians in captive collections and at pet fairs: A reservoir of Batrachochytrium dendrobatidis. Amphib.-Reptil. 32, 419–423 (2011).
Dillon, M. J. et al. Tracking the amphibian pathogens Batrachochytrium dendrobatidis and Batrachochytrium salamandrivorans using a highly specific monoclonal antibody and lateral‐flow technology. Microb Biotechnol 10, 381–394 (2016).
Cardoza, J.E., Jones, G.S., French, T.W. & Halllwell, D.B. Exotic and translocated vertebrates of Massachusetts. Fauna of Massachusetts Series No. 6. (Massachusetts Division of Fisheries and Wildlife, Westborough, MA., 1993).
Krysko, K L. et al. Verified non-indigenous amphibians and reptiles in Florida from 1863 through 2010: Outlining the invasion process and identifying invasion pathways and stages. Zootaxa 1–64 (2011).
Blooi, M. et al. Duplex Real-Time PCR for Rapid Simultaneous Detection of Batrachochytrium dendrobatidis and Batrachochytrium salamandrivorans in Amphibian Samples. J. Clin. Microbiol. 51, 4173–4177 (2013).
Acknowledgements
We thank Ana Longo (University of Maryland) for assistance in providing qPCR standards and performing the Bsal qPCR protocol. We thank Candace Hansen from the Amphibian Survival Alliance for assisting with the web interface. We thank Anne Keiser and Doug Lapp for their generous donation in support of this project.
Author information
Authors and Affiliations
Contributions
All authors contributed in design of the study. B.K., M.B., R.C.F., C.R.M.W., and B.G. sent testing material to participants and analyzed the results. C.R.M.W. created the swabbing instructional video. All authors wrote and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Klocke, B., Becker, M., Lewis, J. et al. Batrachochytrium salamandrivorans not detected in U.S. survey of pet salamanders. Sci Rep 7, 13132 (2017). https://doi.org/10.1038/s41598-017-13500-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-13500-2
This article is cited by
-
Human activities favour prolific life histories in both traded and introduced vertebrates
Nature Communications (2023)
-
United States amphibian imports pose a disease risk to salamanders despite Lacey Act regulations
Communications Earth & Environment (2023)
-
Broad host susceptibility of North American amphibian species to Batrachochytrium salamandrivorans suggests high invasion potential and biodiversity risk
Nature Communications (2023)
-
Plethodontid salamanders show variable disease dynamics in response to Batrachochytrium salamandrivorans chytridiomycosis
Biological Invasions (2021)
-
Pooled samples and eDNA-based detection can facilitate the “clean trade” of aquatic animals
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.