Abstract
Rhabdodontidae is a successful clade of ornithopod dinosaurs, characteristic of Late Cretaceous continental faunas in Europe. A new rhabdodontid from the late Campanian, of southern France, Matheronodon provincialis gen. et sp. nov., is characterized by the extreme enlargement of both its maxillary and dentary teeth, correlated to a drastic reduction in the number of maxillary teeth (4 per generation in MMS/VBN-02-102). The interalveolar septa on the maxilla are alternately present or resorbed ventrally so as to be able to lodge such enlarged teeth. The rhabdodontid dentition and masticatory apparatus were adapted for producing a strict and powerful shearing action, resembling a pair of scissors. With their relatively simple dentition, contrasting with the sophisticated dental batteries in contemporary hadrosaurids, Matheronodon and other rhabdodontids are tentatively interpreted as specialized consumers of tough plant parts rich in sclerenchyma fibers, such as Sabalites and Pandanites.
Similar content being viewed by others
Introduction
Rhabdodontids are basal iguanodontian dinosaurs and characteristic elements of Late Cretaceous dinosaur faunas in Europe1,2,3,4. They have also been described in Early Cretaceous deposits of Spain5. Rhabdodontids are commonly represented, in late Campanian-early Maastrichtian dinosaur faunas of southern France, by two species of the genus Rhabdodon: R. priscus and R. septimanicus 1,2. Rhabdodontid disarticulated elements have recently been discovered at Velaux-La Bastide Neuve, Bouches-du-Rhône Department, southern France. This locality has yielded an abundant and diversified vertebrate fauna, including the titanosaurid sauropod Atsinganosaurus velauciensis 6, ankylosaurian remains, theropod teeth, an ontogenetic series of cranial and postcranial elements of the basal eusuchian crocodile Allodaposuchus precedens 7, pleurodiran and cryptodiran turtles, pterosaurs, hybodont shark teeth, and mawsoniid bones. Here we describe a new rhabdodontid dinosaur, Matheronodon provincialis, from Velaux-La Bastide Neuve, with a quite unusual dentition.
Institutional Abbreviation. MC, Musée de Cruzy, France; MMS/VBN, Musée du Moulin seigneurial/Velaux-La Bastide Neuve. The fossil material is labeled and housed in the municipality palaeontological and archeological structures of Velaux, is under the care of the research association Palaios, and is the property of the department DG 13.
Ornithischia Seeley, 1887
Ornithopoda Marsh, 1881
Iguanodontia Sereno, 1986
Rhabdodontidae Weishampel, Jianu, Csiki, and Norman, 2003
Matheronodon provincialis gen. et sp. nov.
Etymology. Matheron: in honor of Philippe Matheron, who was the first to describe dinosaur remains in Provence; odous (Greek): tooth; provincialis (Latin): from Provence (southern France).
Holotype. MMS/VBN-02-102, a right maxilla; housed in the collections of the Musée du Moulin seigneurial (MMS, Velaux, France).
Referred material. MMS/VBN-93-34, MMS/VBN-09-149a, and MMS/VBN-09-150: maxillary teeth; MMS/VBN-12-22: maxillary tooth crown; MMS/VBN-02-11, MMS/VBN-09-43c, and MMS/VBN-12-A002: dentary teeth.
Horizon and locality. ‘Begudian’ (local stage) sandstones, late Campanian, Late Cretaceous6,8. Velaux-La Bastide Neuve, Aix-en-Provence Basin, Bouches-du-Rhône, southern France.
Diagnosis. Rhabdodontid ornithopod characterized by the following autapomorphies: enlargement of both maxillary and dentary teeth (up to 5 cm in mesio-distal length); reduction of maxillary tooth families (4 per generation in MMS/VBN-02-102); interalveolar septa on the maxilla alternately present or resorbed ventrally, so that one functional tooth is lodged in two paired alveoli; shortened rostral process on the maxilla; broad dorsal shelf along the rostral third of the maxilla; more than 25 vertical and parallel ridges on the labial side of the maxillary teeth.
Description
MMS/VBN-02-102 is a particularly robust right maxilla (rostrocaudal length of 22 cm and maximal height of 10 cm; Fig. 1). The rostral process is particularly short and oriented craniodorsally. In Rhabdodon, the rostral process is much more elongated, forming a horizontal styloid premaxillary process that is nearly as long as the body of the maxilla9; this styloid process is also well developed in Zalmoxes 3. The rostral border of MMS/VBN-02-102 is not eroded, so preservation cannot explain the shortening of its rostral process. In dorsal view, the rostral third of the maxilla is medially broadened to form a triangular and concave rostrodorsal shelf that likely supported the caudolateral (maxillary) process of the premaxilla; this shelf is absent in Rhabdodon 9 and Zalmoxes 3. The medial surface of the rostrodorsal shelf forms an elongated and flattened surface, likely marking the contact with the vomer and/or with the paired maxilla3 (Fig. 1c). Caudally to this surface, the dorsal half of the medial side of the maxilla is dorsoventrally convex, forming dorsal bar as also observed in Rhabdodon 9. At mid-length, the dorsal surface of the bar forms a prominent and laterally-oriented palatine process, with a deep groove along its mediocaudal side forming the articulation with the palatine (Fig. 1c). Caudally to the palatine process, the dorsomedial side of the dorsal bar is pitted and grooved by the articular surface for the pterygoid. The dorsal bar is separated from the flattened dentigerous ventral half by a continuous transverse sulcus (Fig. 1c), corresponding to the neurovascular groove that usually connects the alveolar foramina in ornithopods10. Caudally to the rostral process, the dorsolateral margin of the maxilla forms a prominent, dorsocaudally-inclined, dorsal process. The dorsal portion of this process is expanded along both its rostral and caudal edges; the caudal expansion is more important, forming a triangular caudodorsal wing that participates in the rostrodorsal margin of the elliptical antorbital fossa (Fig. 1b). The dorsal process is much more developed in Matheronodon than in Zalmoxes 3; in Rhabdodon, the dorsal process looks rostrocaudally narrower and is more vertically oriented, as observed in MC-QR99. A large wing-like jugal process extends caudolaterally from the base of the dorsal process up to the caudal quarter of the bone (Fig. 1a,b). It is mediolaterally thin, separated from the main body of the maxilla by a wide and deep sulcus. Along its dorsal edge, a small triangular process marks the caudal end of the antorbital fenestra. A similar process is also present, though it is proportionally higher, in Rhabdodon 9. A short, but strong ectopterygoid ridge connects the caudal end of the jugal process to the main body of the maxilla (Fig. 1a,b). As in Zalmoxes 3, the lateral surface of the maxilla overhangs the tooth row and forms a large labial recess. A dozen foramina are more or less aligned along the dorsal part of the dentigerous portion of the maxilla (Fig. 1b) and likely transmitted neurovascular bundles from the maxillary division of the trigeminal nerve and the maxillary artery3. The rostralmost foramen, just in front of the rostral process, is the largest. Ventrally, the tooth row is relatively straight and composed of only eight alveoli, despite of the large size of this specimen. Up to 10 alveoli are present in much smaller Zalmoxes specimens3 and 11 in MC-QR9, of similar size as MMS/VBN-02-102, from the Campanian of Quarante (Hérault, southern France) and referred to as Rhabododon 9. The interalveolar septa are alternately present or nearly completely resorbed, so the base of the maxillary dental battery in fact contains four tooth positions, each of which is composed of two partially fused alveoli (Fig. 1d). Alveolar resorption has not been observed in Rhabdodon 9 and Zalmoxes 3. The functional maxillary teeth of MMS/VBN-02-102 are not preserved, but the crowns of the replacement teeth emerge from the alveoli. CT-scans of MMS/VBN-02-102 reveal the presence of two generations of replacement teeth (Fig. 2). The first generation emerges from the rostral half of each paired alveolus. The development of the maxillary teeth remained typically reptilian in pattern, progressing in wave-like fashion from the caudal part to the rostral part of the battery11. A second generation of replacement teeth is present in the caudal half of each paired alveolus. It can therefore be concluded that successive generations of teeth alternatively emerged from the rostral, and then from the caudal half of the paired fused alveoli. However, the tooth arrangement and replacement pattern in Matheronodon is not radically different from those in more advanced iguanodontians11. Because of the enlargement of the maxillary crowns (see below), teeth within each fused alveolus were largely imbricated, the rostral one covering the distal one in labial view (Fig. 2). Although they are only partially erupted and a large portion is consequently still embedded within the maxilla, more than 18 vertical ridges are visible along the labial surface of the second and third maxillary crowns (Fig. 1e), and the total number of labial ridges is consequently higher than 25 (see also Fig. 2).
Isolated maxillary teeth (Fig. 3a–d) were found associated to MMS/VBN-02-102, but it cannot be ascertained that they belong to the same individual, as their size is rather heterogeneous and the fossils were obviously transported over a short distance8. However, all have the same morphology and also closely resemble those inside MMS/VBN-02-102, so they evidently belong to the same taxon. All are particularly high (around 6 cm in MMS/VBN-09-149) and mesiodistally broad (around 5 cm in MMS/VBN-09-150). Their crown is typically cleaver-shaped, as also observed in other rhabdodontids including Zalmoxes 3, Mochlodon 4, and Rhabdodon 9. In labial view, the crown is more elevated distally than mesially and the cutting apical edge is oblique and straight. Enamel covers the crown on all sides, but it is much thicker labially than lingually. As also observed in the holotype MMS/VBN-02-102, the labial surface of all recovered maxillary teeth is ornamented by more than 25 vertical and parallel ridges, all subequal in size. All the ridges reach the apical edge of the crown, forming tiny denticles, and then extend farther along the apical portion of the lingual side of the unworn crowns. Some of the ridges are bifurcated at the base of the labial side. The number of labial ridges is much higher in Matheronodon than in other rhabdodontids: a maximum of 19, 13, and 12 labial ridges can be observed in Zalmoxes 3, Mochlodon 4, and Rhabdodon 9, respectively. As already observed3,9, there is apparently no significant correlation between tooth width and number of ridges on the maxillary teeth in rhabdodontids. Basally, the labial enamel surface of the maxillary teeth of Matheronodon is bordered by a thin crenulated cingulum that curves apically along its distal margin. A single wear facet can be observed in MMS/VBN-09-149a, forming an angle of approximately 60° with the horizontal plane, as also observed in the most heavily worn teeth of Zalmoxes 3 and Rhabdodon 9. Scratches on the worn dentine surface are parallel and vertically oriented, as also observed in Zalmoxes 3 and Mochlodon 9. The root of the maxillary tooth is particularly robust, higher than the crown, and lingually curved. Well-developed replacement tooth grooves along both the mesial and distal sides of the roots indicate that functional and replacement teeth were closely imbricated.
Isolated teeth of Matheronodon provincialis gen. et sp. nov. Left maxillary tooth (MMS/VBN- 09–149a) in labial (a) and lingual (b) views. Right maxillary crown (MMS/VBN-12-22) in labial (c) and lingual (d) views? Right dentary tooth (MMS/VBN-02-11) in lingual (e) and labial (f) views? Left dentary tooth (MMS/VBN-12-A002) in lingual view (g). Scale bars = 2 cm.
Three large dentary teeth (Fig. 3e–g) were also found in Velaux-La Bastide Neuve, close to the holotype maxilla. As in other rhabdodontids, unworn dentary crowns have a leaf-shaped, lingual aspect, dominated by a prominent primary ridge, slightly distal to the midline of the crown. As in Zalmoxes 3,12, the thickly-enameled-lingual side of the crown is covered, on either side of the median ridge, by a dozen vertical subsidiary ridges. Subsidiary ridges are less numerous in Rhabdodon (up to 8 on either side of the primary ridge9) and Mochlodon (4–7 on either side4). The subsidiary ridges are slightly divergent and, as it is also the case for the maxillary teeth, they extend onto the apical part of the labial side of the crown, forming small denticles as they cross the apical edge. Unlike in Mochlodon 4, the subsidiary ridges reach the basal margin of the enameled surface of the lingual side of the crown. The enamel is much thinner on the labial side than on the lingual side of the crown. Because these dentary teeth were found in the same fossiliferous pocket as the holotype maxilla and are also characterized by more numerous subsidiary ridges than in Rhabdodon as it is also the case for the maxillary teeth, it is reasonable to refer them to Mochlodon.
Tooth microstructure
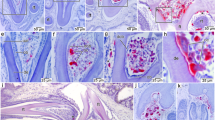
A coronal section in a maxillary tooth (Fig. 4) shows a jagged pattern of ridges and grooves on the labial surface (Fig. 4b,h), corresponding to the numerous vertical ridges, and a rather smooth surface on the lingual side (although small projections are also present). Measurements of enamel thickness (Supplementary Table S1) demonstrate that the ridges on the labial side of the crown have thicker enamel than the grooves in between. The enamel itself has a wavy enamel Schmelzmuster (Fig. 4f,g), similar to hadrosaurid dinosaurs13,14. Unlike in hadrosaurids14, the enamel is present around the entire crown of unworn teeth, but thinner on the lingual side of the sectioned specimen. Measurements on a well-preserved side of the tooth (Fig. 4c) show that Matheronodon has significantly thicker (Supplementary Table S1; independent t-test, N = 11, p < 0.001) enamel (avg. 179 µm) on its dental ridges, compared to the enamel on the central keel of Edmontosaurus teeth (avg. 155 µm, cf. also ‘Anatosaurus’13).
Matheronodon tooth histology. (a) Section plane in labiolingually compressed maxillary tooth MMS/VBN-93-34. (b) Incident light overview of a coronal section as indicated in (a,c) Transmitted light (plane polarized) microscopic view of boxed area in (b) showing enamel thickness variation on the ridges and grooves on the labial surface. (d,e) Transmitted light (plane polarized) microscopic views of boxed areas in (b) with details of the incremental lines in the dentine; Von Ebner lines are indicated by white arrows. (f,g) Transmitted light (plane polarized and cross polarized with lambda filter) microscopic view of boxed area in c, showing the enamel-dentine junction and wavy enamel Schmelzmuster. (h) Detail of the worn crown of MMS/VBN-09-149a (cf. Fig. 3a) showing an occlusal surface with a serrated (black arrows) slicing edge.
The dentine of the sectioned tooth (Fig. 4d,e) does not show any clear variation in the orientation of dentine tubules as in hadrosaurids14. We could also observe a high number of incremental Von Ebner lines (50 to 100 IVELs, Fig. 4e), indicating a long development time for a single tooth15. Unfortunately, the preservation state of the tooth did not allow a precise count of Von Ebner lines in the dentine.
Discussion
Among basal iguanodontians, Rhabdodontidae (all iguanodontians more closely related to Rhabdodon priscus than to Parasaurolophus walkeri 16) form a clade mostly endemic to Europe during the Cretaceous. The following dental characters, regarded as unambiguous synapomorphies for Rhabdodontidae3,4,5 can be observed in Matheronodon: dentary teeth with multiple (more than 10) evenly-spaced accessory ridges around the primary ridge, and maxillary teeth with multiple ridges of similar size and devoid of a primary ridge. Besides Matherodon provincialis, three rhabdodontid genera and six species have currently been named: Rhabdodon priscus from the Campanian-Maastrichtian of France and Spain1, R. septimanicus from the Campanian-Maastrichtian of France2, Zalmoxes robustus and Z. shqiperorum from the Maastrichtian of Romania3,17, Mochlodon suessi from the Campanian of Austria18, and Mochlodon vorosi, from the Santonian of Hungary4.
The maxilla MMS/VBN-02-102 and the teeth that were found in the same fossiliferous pocket at Velaux-La Bastide Neuve are quite different from those described in Rhabdodon, also from late Campanian-early Maastrichtian deposits from Southern France, and in other rhabdodontids, so they clearly belong to a new taxon: the rostral process is short and rostrodorsally oriented, although it is much more elongated, horizontal and styloid in Rhabdodon; the dorsal shelf is much broader on the rostral half of the maxilla; the dorsal process is wider and caudally inclined in MMS/VBN-02-102; alveolar resorption has not been observed in Rhabdodon so far; the number of labial ridges is much higher on the maxillary teeth in Matheronodon (>25) than in Rhabdodon (<12); and the number of subsidiary ridges is also higher on the dentary teeth in Matheronodon (a dozen or more on either side of the primary ridge) than in Rhabdodon (up to 8 on either side of the primary ridge).
The sophistication of their feeding apparatus has long been identified as a key element in the evolutionary success and diversification of ornithopods19,20. Basal, non-iguanodontian ornithopods are characterized by relatively low tooth count (14–17 positions), low, labiolingually-compressed tooth crowns that are bulbous to sub-triangular in shape, and ornamented by numerous apicobasal ridges21,22,23. The marginal denticles are relatively large and triangular20. More advanced iguanodontians, such as Camptosaurus dispar 24, Iguanodon bernissartensis 25, Mantellisaurus atherfieldensis 26, and Ouranosaurus nigeriensis 27, are characterized by a higher tooth count (around 20 positions), proportionally higher, shield-like tooth crowns, a reduced number of apicobasal ridges, and proportionally reduced marginal denticles. The evolutionary explosion of hadrosauroid iguanodontians during the Late Cretaceous was accompanied by the development of true dental batteries, consisting of up to 60 closely packed tooth families. In advanced hadrosaurids, each tooth family is made of three up to seven successional teeth. Tooth miniaturization is accompanied in hadrosaurids by proportionally higher tooth crowns and by a further reduction of ridges to a single median carina (one or two faint subsidiary ridges are occasionally present) and of the marginal denticles11. Enamel restriction to the sides of crowns promoted self-sharpening14. In most hadrosaurids, up to three successional teeth from each tooth row participate in the occlusal surface, forming labiolingually wide pavements across the entire chewing area. This morphological complexity is coupled, in hadrosaurids, with a histological complexity and the development of six dental tissues instead of two in basal genasaurans, for example14. Hadrosaurids therefore developed a complex, dual-function slicing-grinding system presumably for the consumption of moderately tough, cellulose-rich plant diet14.
The rhabdodontid dentition evolved in an opposite way: unlike hadrosaurids, they developed a reduced number of mesiodistally wide, but labiolingually thin teeth4. Extreme tooth enlargement can be observed in Matheronodon, in which the number of maxillary tooth rows is reduced to eight. The high-angled wear surface has a particularly sharp, chisel-like cutting edge. Scratches on the worn dentine surface are usually well marked, parallel and vertically oriented3,4. Moreover, the rhabdodontid quadrate was proportionally low and massive3,9, so that the jaw joint was likely not offset too far from the level of the tooth row. The rhabdodontid skull was likely triangular in dorsal view: the premaxillae and predentary of Zalmoxes are narrow, whereas the maxillae diverge strongly posteriorly, and the quadrates are laterally splayed3. Intracranial mobility was obviously much more limited than in hadrosaurids. In rhabdodontids, the robust jaw, strong and elevated coronoid process and large jaw adductor muscle chamber suggest that jaw closure was powerful and that the motion of lower jaw was limited by the complex jaw joint and tight predentary-dentary suture3. All these characters suggest that the rhabdodontid dentition and masticatory apparatus were adapted for producing a strict and powerful slicing action, resembling a pair of scissors.
The peculiar Matheronodon tooth microstructure also reflects specialization for slicing. As soon as a new tooth erupted, the ridges along the wear-resistant thicker enameled side of the crown formed a self-sharpening serrated and jagged slicing edge (Fig. 4h). Precise measurement of durability would require detailed tribological measurements14,28, but this is beyond the scope of the current paper. Due to the high tooth development time and large size of the teeth, the tooth replacement rate of Matheronodon was probably not as high as in hadrosaurids or ceratopsians14,28.
From a biomechanical point of view, enlarged blade-like teeth, as exemplified by Matheronodon, and also by the mammalian carnassial teeth, are best adapted for fracturing tough (= resistant to crack propagation) foodstuffs28,29,30,31. Hadrosaurids, on the contrary, developed a much more complex dentition forming a dual-function shearing-crushing system, suggestive of a broader dietary range, including leaves, fruits, seeds, and twigs32,33,34; subtle variations in their tooth morphology would have allowed for diversification into more specialized ecological niches14,31. In any case, the important morphological differences in their dentitions likely lead to niche portioning between rhabdodontids and hadrosaurids in Europe by the Late Cretaceous time.
Besides Matheronodon, the Velaux-La Bastide Neuve locality has yielded other herbivorous dinosaur remains, including ankylosaurid elements, and abundant material of the titanosaurid sauropod Atsinganosaurus velauciensis 6. We have not found any trace of hadrosauroids yet. Previous works based on sites in southern France have concluded that an important faunal replacement related to an important late Maastrichtian marine regression occurred in southern Europe during the Late Cretaceous: a Campanian - early Maastrichtian fauna dominated by titanosaurids and rhabdodontids was replaced by a late Maastrichtian assemblage dominated by hadrosaurids35. However, the presence of hadrosauroids, titanosaurids and rhabdodontids in the late Maastrichtian assemblage of Vitrolles-La-Plaine suggests that these animals were still living together in southern France during the latest Cretaceous, but maybe in different environments. Their co-occurrence in the allochthonous assemblage of Vitrolles-La-Plaine can easily be explained by taphonomic processes (hydraulic transport) that concentrated, in the same level, bones of animals living in different places36.
Ceratopsians are particularly rare in Late Cretaceous deposits of Europe. Scarce remains have been described from the Coniacian-Santonian of Belgium37 and from the Campanian of Sweden38. The coronosaurian neoceratopsian Ajkaceratops kozmai was discovered in the Santonian Csehbánya Fm of Iharkút in Hungary39, which has also yielded the rhabdodontid Mochlodon vorosi 4. In contrast, ceratopsians are abundant in Late Cretaceous deposits of Asia and North America, whereas rhabdodontids have not been reported from these areas. Ceratopsian dentitions produced a strict shearing action31, as also hypothesized for rhabdodontids in the present paper. So, both the medium-sized ceratopsians and the rhabdodontids potentially fed on similar tough, woody or fibrous vegetation.
What Matheronodon and Rhabdodon fed on will probably remain unknown, but this issue can be discussed based on some plant fossil assemblages known during the Campanian-Maastrichtian of Europe (see Supplementary Information for more detailed information on the composition of those assemblages): the Grünbach Formation in Austria40,41,42; the Haţeg and Rusca Montană basins in western Romania43, which have also yielded abundant remains of the rhabdodontid Zalmoxes; the Áger, Vallcebre Coll de Nargό and Tremp basins in the southern Spanish Pyrenees44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60; Lo Hueco in the south-western Iberian Ranges61,62, which has also yielded fossils of rhabdodontid and titanosaurid dinosaurs; and Fuveau Basin, Etang de Berre, and Sainte-Baume Massif in southeastern France63,64,65. In Europe, plant assemblages strongly vary in richness and abundance depending on the localities, but overall plant megafossils are marked by the replacement of the conifer Geinitzia by Cunninghamites and the spread of the monocots Sabalites and Pandanites, while plant microfossils are characterized by the abundance of fern spores and Normapolles-group eudicot pollen grains.
Tough woody tissue exists in diverse vascular plant groups, such as gymnosperms and most angiosperms except monocots, and is found in leaves, stems, and roots. It is made of secondary xylem, commonly called wood, which consists of water and mineral conductive cells, called tracheids and vessels, parenchyma cells and facultative sclerenchyma fibers. Sclerenchyma also occur out of secondary xylem, and are particularly abundant in some large leaves such as in some palms; in leaves and stems of many herbaceous and tree plants, sclerenchyma serves the function of support when wood is absent. The relative abundance of secondary xylem vs sclerenchyma fibers in eaten plant parts such as leaves and stems may cause large herbivores to adapt their diet and tooth morphology. Soft eudicot leaves with few or no sclerenchyma fibers can be easily crushed with a pestle in a mortar. Campanian-Maastrichtian conifer leafy stems, such as Geinitzia and Cunninghamites, and eudicot leaves and small stems may be easily and quickly beat into pulp. So they do not only require powerful blade-like teeth. In contrast, many monocot leaves in particular in palms such as the Campanian-Maastrichtian Sabalites longirhachis and Pandanites spp. contain much sclerenchyma fibers along the parallel veins to stiffen the large leaf laminas and petioles. Such fibers must be cut into small fragments with blade-like teeth prior to be swallowed.
It might therefore be cautiously hypothesized that rhabdodontids were adapted to preferentially feed upon tough plant parts rich in sclerenchyma fibers such as Sabalites and Pandanites. In contrast, conifers potentially constituted an important part in the diet of North American hadrosaurids. The supposed gut content of an Edmontosaurus “mummy” mainly consists of conifer needles and branches, together with deciduous angiosperm foliages and numerous small seeds or fruits32,66. Coprolites tentatively referred to Maiasaura primarily contain conifer stem fragments66. Nevertheless, there is no indication of drastic changes in the character or composition of terrestrial vegetation between the early and late Maastrichtian that could easily explain the sudden abundance of hadrosaurids and the purported contemporary decline of rhabdodontids in western Europe. Correlating faunistic and floristic remains highly speculative in the current state of our knowledge.
Methods
Computed tomography (CT) of the maxilla was done at the veterinary faculty of the University of Liege. A 16 multi-slice CT scan was used (Siemens, Somatom 16, Erlangen, Germany) with the following parameters; 120 kV and 46 mA under a Dental 0.75 H60s protocol. The slices of 0.5 mm in depth were digitally reconstructed using Amira 5.3.3 and Drishti v2.6.3.
For investigating tooth microstructure, we sectioned a slightly crushed and isolated maxillary tooth of Matheronodon. The specimen was embedded in epoxy resine (Araldite 2020) and sectioned and ground to a thickness of ~50 µm using standard lapidary methods.
Nomenclatural Act
This published work and the nomenclatural acts it contains have been registered in ZooBank, the proposed online registration system for the International Code of Zoological Nomenclature. The ZooBank life science identifiers can be resolved and the associated information viewed by appending the life science identifiers to the prefix http://zoobank.org/. The life science identifiers for this publication is 1AD18D4B-7063–43F7-A55E-19463D066D78; for Matheronodon: FF4C534D-206A-4659–8592–9154ABF4A8B5, and M. provincialis: A6D62068–15A5–4ADE-83DD-74F0ED529BAB.
References
Matheron, P. Notice sur les reptiles fossiles des dépôts fluvio-lacustres crétacés du bassin à lignite de Fuveau. Mém. Acad. Imp. Sc., Belles-Let., Arts Marseille, 345–379 (1869).
Buffetaut, E. & Le Loeuff, J. Une nouvelle espèce de Rhabdodon (Dinosauria, Ornithischia) du Crétacé supérieur de l’Hérault (Sud de la France). C. R. Acad. Sc. Sér. 2. 318, 943–948 (1991).
Weishampel D. B., Jianu, C.-M., Csiki, Z. & Norman, D. B. Osteology and phylogeny of Zalmoxes (n.g.), an unusual euornithopod dinosaur from the latest Cretaceous of Romania. J. Syst. Palaeont. 1, 65–123 (2003).
Ősi, A., Prondvai, E., Butler, R. & Weishampel, D. B. Phylogeny, histology and inferred body size evolution in a new rhabdodontid dinosaur from the Late Cretaceous of Hungary. PLoS ONE 7(9), 1–44 (2012).
Dieudonné, P.-E., Tortosa, T., Torcida Fernández-Baldo, F., Canudo, J. I. & Díaz-Martínez, I. An unexpected early rhabdodontid from Europe (Lower Cretaceous of Salas de los Infantes, Burgos Province, Spain) and a re-examination of basal iguanodontian relationships. PLoS ONE 11(6), 1–40 (2016).
Garcia, G., Fournier, F., Amico, S., Thouand, E. & Valentin, X. A new titanosaur genus (Dinosauria, Sauropoda) from the Late Cretaceous of southern France and its paleobiogeographic implications. Bull. Soc. Géol. France 181, 269–277 (2010).
Martin, J. E. et al. New specimens of Allodaposuchus precedens from France: intraspecific variability and the diversity of European Late Cretaceous eusuchians. Zool. J. Linn. Soc. 176, 607–631 (2015).
Cincotta, A. et al. Integrated paleoenvironmental reconstruction and taphonomy of a unique Upper Cretaceous vertebrate-bearing locality (Velaux, Southeastern France). PLoS ONE 1(18), 1–20 (2015).
Chanthasit, P. The ornithopod dinosaur Rhabdodon from the Late Cretaceous of France: anatomy, systematics and palaeobiology. Unpublished PhD thesis, Université Claude Bernard Lyon 1 (2010).
Edmund, A. G. On the special foramina in the jaws of many ornithischian dinosaurs. Contrib. Roy. Ontario Mus., Div. Zool. Paleont. 48, 3–14 (1957).
Ostrom, J. H. Cranial morphology of the hadrosaurian dinosaurs of North America. Bull. Am. Mus. Nat. Hist. 122, 37–186 (1961).
Godefroit, P., Codrea, V. & Weishampel, D. B. Osteology of Zalmoxes shqiperorum (Dinosauria, Ornithopoda), based on new specimens from the Upper Cretaceous of Nălaţ-Vad (Romania). Geodiversitas 31, 525–53 (2009).
Sander, P. M. The microstructure of reptilian tooth enamel: terminology, function, and phylogeny. Münch. Geowiss. Abh. 38, 1–102 (1999).
Erickson, G. M. et al. Complex dental structure and wear biomechanics in hadrosaurid dinosaurs. Science 338, 98–101 (2012).
Erickson, G. M. Incremental lines of von Ebner in dinosaurs and the assessment of tooth replacement rates using growth line counts. Proc. Nat. Acad. Sc. USA 93, 14623–14627 (1996).
Sereno, P. C. 1998. A rationale for phylogenetic definitions, with application to the higher-level taxonomy of Dinosauria. N. Jb. Geol. Paläontol. Abhand. 210, 41–83 (1997).
Nopcsa, F. Dinosaurierreste aus Siebenburgen II. (Schädelreste von Mochlodon). Mit einem Anhange: zur Phylogenie der Ornithopodiden. Denk. Akad. Wiss. Wien 74, 149–175 (1902).
Bunzel, E. Die Reptilfauna der Gosau-Formation in der Neuen Welt bei Wiener-Neustadt. Abh. Geol. Reichsanst. Wien 5, 1–18 (1871).
Norman, D. B. & Weishampel, D. B. Ornithopod feeding mechanisms: their bearing on the evolution of herbivory. Am. Nat. 126, 151–164 (1985).
Strickson, E., Prieto-Márquez, A., Benton, M. J. & Stubbs, T. L. Dynamics of dental evolution in ornithopod dinosaurs. Sci. Rep. 6, 28904 (2016).
Galton, P. M. The ornithischian dinosaur Hypsilophodon from the Wealden of the Isle of Wight. Bull. Br. Mus. Nat. Hist., Geol. Series 25, 3–152 (1974).
Scheetz, R. D. Osteology of Orodromeus makelai and the phylogeny of basal ornithopod dinosaurs. Unpublished PhD Thesis (1999).
Jin, L., Chen, J., Zan, S., Butler, R. J. & Godefroit, P. Cranial anatomy of the small ornithischian dinosaur Changchunsaurus parvus from the Quantou Formation (Cretaceous: Aptian–Cenomanian) of Jilin Province, northeastern China. J. Vert. Paleont. 30, 196–214 (2010).
Gilmore, C. W. Osteology of the Jurassic reptile Camptosaurus: with a revision of the species of the genus, and description of two new species. Proc. U.S. Nat. Mus. 332, 197–332 (1909).
Norman, D. B. On the ornithischian dinosaur Iguanodon bernissartensis of Bernissart. Mém. Inst. R. Sci. Nat. Belg. 178, 1–103 (1980).
Norman, D. B. On the anatomy of Iguanodon atherfieldensis (Ornithischia: Ornithopoda). Bull. Inst. R. Sci. Nat. Belg. Sci. Terre 56, 281–372 (1986).
Taquet, P. Géologie et paléontologie du gisement de Gadoufaoua (Aptien du Niger). Cahiers Paléont. CNRS, 1–191 (1976).
Erickson, G. M. et al. Wear biomechanics in the slicing dentition of the giant horned dinosaur Triceratops. Sci. Adv. 1(5), e1500055–e1500055 (2015).
Strait, S. G. Tooth use and the physical properties of food. Evol. Anthr. 5, 199–211 (1997).
Lucas, P. W. Dental functional morphology: How teeth work (Cambridge University Press, (2004).
Mallon, J. C. & Anderson, J. S. The Functional and palaeoecological implications of tooth morphology and wear for the megaherbivorous dinosaurs from the Dinosaur Park Formation (Upper Campanian) of Alberta, Canada. PLoS ONE 9(6), 1–34 (2014).
Kräusel, R. D. Nahrung von. Trachodon. Paläontol. Z. 4, 80 (1922).
Currie, P. J., Koppelhus, E. B. & Muhammad, A. F. “Stomach” contents of a hadrosaurid from the Dinosaur Park Formation (Campanian, Upper Cretaceous) of Alberta, Canada. In Sixth Symposium on Mesozoic Terrestrial Ecosystems and Biota, Short Papers (eds Sun, A. & Wang, Y.) 111–114 (China Ocean Press, 1995).
Tweet, J. S., Chin, K., Braman, D. R. & Murphy, N. L. Probable gut contents within a specimen of Brachylophosaurus canadensis from the Upper Cretaceous Judith River Formation of Montana. Palaios 23, 624–635 (2007).
Le Loeuff, J., Buffetaut, E. & Martin, M. The last stages of dinosaur faunal history in Europe: a succession of Maastrichtian dinosaur assemblages from the Corbières (southern France). Geol. Mag. 131, 625–630 (1994).
Valentin, X. et al. First Late Maastrichtian (Latest Cretaceous) vertebrate assemblage from Provence (Vitrolles-la-Plaine, Southern France. In Bernissart Dinosaurs and Early Cretaceous Terrestrial Ecosystems (ed Godefroit, P.) 582–597 (Indiana Univ. Press, 2012).
Godefroit, P. & Lambert, O. A re-appraisal of Craspedodon lonzeensis Dollo, 1883 from the Upper Cretaceous of Belgium: the first record of a neoceratopsian dinosaur in Europe? Bull. Inst. R. Sci. Nat. Belgique, Sci. Terre 77, 83–93 (2007).
Lindgren, J. et al. The first neoceratopsian dinosaur remains from Europe. Palaeontology 50, 929–937 (2007).
Ösi, A., Butler, R. J. & Weishampel, D. B. A Late Cretaceous ceratopsian dinosaur from Europe with Asian affinities. Nature 465, 466–468 (2010).
Herman, A.B. & Kvaček, J. Late Cretaceous Grünbach flora of Austria. Naturhist. Mus. Wien, 1–216 (2010).
Summesberger, H. The Cretaceous of the Grünbach-Neue Welt Basin. In Climates: past, present and future (eds Kollmann, H. A. & Hubmann, B.) 77–89 (2nd European Palaeontological Congress, Vienna, 1997).
Hradecká, L., Lobitzer, H., Svobodová, M. & Švábenická, L. 2000. Biostratigraphy of selected exposures in the Grünbach-Neue Welt Gosau Group (Late Cretaceous). In 6th International Cretaceous Symposium Abst. Vol., 51 (Vienna, August 27 - September 4, 2000) p. 51, Vienna.
Popa, M. E., Kvaček, J., Vasile, S. & Csiki-Sava, Z. Maastrichtian monocotyledons of the Rusca Montană and Haţeg basins, South Carpathians, Romania. Rev. Palaeobot. Palynol. 210, 89–101 (2014).
Marmi, J., Gomez, B. & Martín-Closas, C. Presencia de macrorrestos parautóctonos de Sabalites cf. longirhachis (Unger, 1850). Kvaček & Herman, 2004 en facies parálicas del Cretácico Superior del Pirineo oriental. Rev. Esp. Paleontol. 23, 7–14 (2008).
Marmi, J., Gomez, B., Villalba-Breva, S. & Martín-Closas, C. A reconstruction of the fossil palm Sabalites longirhachis (Unger) J. Kvaček et Herman from the Maastrichtian of Pyrenees. Rev. Palaeobot. Palynol. 163, 73–83 (2010).
Marmi, J., Gomez, B., Villalba-Breva, S. & Martín-Closas, C. B ergacarpon viladricii gen. et sp. nov., angiosperm seeds and associated fruits from the early Maastrichtian of the eastern Pyrenees (Catalonia, Spain). Rev. Palaeobot. Palynol. 171, 83–94 (2012).
Vicente i Castells, J. Estudi morfològic de la flora cretàcica d’Isona (Pallars Jussà). Institut d’Estudis de la Natura del Barcelonès Nord, Santa Coloma de Gramenet, 223 pp. (2002).
Villalba-Breva, S. et al. Peat-forming plants in the Maastrichtian coals of the Eastern Pyrenees. Geol. Acta 10, 189–207 (2012).
Riera, V., Marmi, J., Oms, O. & Gomez, B. Orientated plant fragments revealing tidal palaeocurrents in the Fumanya mudflat (Maastrichtian, southern Pyrenees): insights in palaeogeographic reconstructions. Palaeogeogr. Palaeoclimatol. Palaeoecol. 288, 82–92 (2010).
Marmi, J., Gomez, B., Martín-Closas, C., Villalba-Breva, S. & Daviero-Gomez, V. Diversified fossil plant assemblages from the Maastrichtian in Isona (southeastern Pyrenees). Rev. Palaeobot. Palynol. 206, 45–59 (2014).
Villalba-Breva, S. et al. Plant taphonomy and palaeoenvironment from the Upper Cretaceous of Isona, Tremp Basin, southern Pyrenees, Catalonia, Spain. Cret. Res. 54, 34–49 (2015).
Médus, J. Palynological zonation of the Upper Cretaceous in Southern France and Northeastern Spain. Rev. Palaeobot. Palynol. 14, 287–295 (1972).
Porta, J., Kedves, M., Solé de Porta, N. & Civis, J. Palinologia del Maastrichtiense del Barranco de la Posa (Lérida, España). Problemática regional. Rev. Invest. Geol. 40, 5–28 (1985).
Ashraf, A. R. & Erben, H. K. Palynologische Untersuchugen an der Kreide/Tertiär-Grenze west-Mediterraner regionen. Palaeontographica B 200, 111–163 (1986).
Médus, J. et al. Prospects for recognition of the palynological Cretaceous/Tertiary Boundary and an iridium anomaly in nonmarine facies of the eastern Spanish Pyrenees: a preliminary report. Newslett. Stratigr. 18, 123–138 (1988).
Lόpez-Martínez, N., Fernández-Marrόn, M. T. & Valle, M. F. The succession of vertebrates and plants across the Cretaceous-Tertiary boundary in the Tremp Formation, Ager valley (South-Central Pyrenees, Spain). Geobios 32, 617–627 (1999).
Fernández-Marrόn, M. T., Lόpez-Martínez, N., Fonollá-Ocete, J. F. & Valle-Hernández, M. F. The palynological record across the Cretaceous-Tertiary boundary in differing palaeogeographical settings from the southern Pyrenees, Spain. In The Palynology and Micropaleontology of Boundaries (eds Beaudoin, A. B. & Head, M. J.). Geol. Soc. London Sp. Public. 230, 243–255 (2004).
Torices, A., Fernández-Marrόn, M. T., Fonollá, F. & Lόpez-Martínez, N. Palynological characterization of a transgressive episode in transitional deposits in the Cretaceous Aren and Tremp formations (south-central Pyrenees, Spain). N. Jb. Geol. Paläont. Abh. 266, 159–172 (2012).
Villalba-Breva, S. et al. Peat-forming plants in the Maastrichtian coals of the Eastern Pyrenees. Geol. Acta 10, 189–207 (2012).
Ortega, F. et al. The biota of the Upper Cretaceous site of Lo Hueco (Cuenca, Spain). J. Iber. Geol. 41, 83–99 (2015).
Peyrot, D., Barroso-Barcenilla, F. & Cambra-Moo Paleoecology of the late Campanian/early Maastrichtian Fossil-Lagerstätte of “Lo Hueco” (Cuenca, Spain): Palynological insights. Palaeogeogr., Palaeoclim., Palaeoecol. 387, 27–39 (2013).
Fritel, P. F. La flore aturienne de Fuveau d’après les matériaux de la collection de Saporta. Bull. Mus. Nat. Hist. Nat. Paris 33, 404–410 (1927).
Carpentier, A. Remarques sur des empreintes de Frenelopsis trouvées dans le Campanien inférieur de la Sainte-Baume. Ann. Mus. Hist. Nat. Marseille 28, 1–14 (1937).
Méon, H., Odin, G. S., Antonescu, E. & Siegl-Farkas, A. Synthetic data on spores and pollen across the Campanian-Maastrichtian boundary at Tercis-les-Bains (SW France). In The Campanian-Maastrichtian stage boundary Characterisation at Tercis les Bains (France) and correlation with Europe and other Continents (Odin, G. S. ed). Dev. Palaeont. Strat. 19, 210–216 (2001).
Horner, J. R., Weishampel, D. B. & Forster, C. A. In The Dinosauria 2 (eds Weishampel, D. B., Dodson, P. & Osmólska, H.) 438–463 (Univ. California Press, 2004).
Chin, K. & Gill, B. D. Dinosaurs, dung beetles, and conifers: participants in a Cretaceous food web. Palaios 11, 280–285 (1996).
Acknowledgements
We gratefully acknowledge the management, logistical and communication assistance from the Velaux Municipality (J.-P. Maggi and L. Melhi) with its heritage, culture and technical services (M. Calvier, and S. Chauvet), the environment department from CD 13 (M. Bourrelly, T. Tortosa, G. Michel, N. Mouly, and S. Amico), the ‘Service Départemental d’Incendie et de Secours’ (SDIS 13), and numerous volunteers during the field campaigns in 2009 and 2012. This work was supported by the Ministry of Education and Communication (research grant VR1013 to Palaios association), the Bouches du Rhône department CD 13 proposals MAPADGAC23112010-1 and MAPADGAC16012014-1-AAPC), by the Belspo BRAIN. be proposal ‘Coldcase’, and by FRIA grants to UL and AC. We are also most grateful to T. Hubin, J. dos Remedios-Esteves, and S. Berton for technical support. And finally, we should like to thank Hai Xing and an anonymous reviewer for their helpful comments on an earlier version of the paper.
Author information
Authors and Affiliations
Contributions
X.V., G.G. and P.G. designed the project. P.G., G.G. and A.C. described the fossils, K.S. described the tooth microstructure, U.L. performed CT scan analyses of the maxilla, B.G. compiled and included the palaeobotanical data. All authors discussed the results and provided input on the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Godefroit, P., Garcia, G., Gomez, B. et al. Extreme tooth enlargement in a new Late Cretaceous rhabdodontid dinosaur from Southern France. Sci Rep 7, 13098 (2017). https://doi.org/10.1038/s41598-017-13160-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-13160-2
This article is cited by
-
Reappraisal of the braincase anatomy of the ornithopod dinosaurs Telmatosaurus and Zalmoxes from the Upper Cretaceous of the Haţeg Basin (Romania) and the taxonomic reassessment of some previously referred specimens
PalZ (2023)
-
Multi-proxy dentition analyses reveal niche partitioning between sympatric herbivorous dinosaurs
Scientific Reports (2022)
-
Unrelated ornithopods with similar tooth morphology in the vicinity of Salas de los Infantes (Burgos Province, Spain): an intriguing case-study
Journal of Iberian Geology (2020)
-
The oldest freshwater crabs: claws on dinosaur bones
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.