Abstract
The acceptance of animal products is increasingly associated with standardized animal welfare, which relates to appropriate animal husbandry from birth to slaughter. In particular, shipment to the slaughterhouse is considered as a critical process exposing the animals to a number of, in part severe, stressors. New biomarkers may be useful for the assessment of animal welfare. The IGF-system has been assessed in a commercial pig transport in conjunction with established markers of stress response. Furthermore, the effect of repeated restraint as an experimental model for repeated acute stress was investigated. During shipment from farm to slaughterhouse, plasma concentrations of IGFBP-3 and IGFBP-2 were significantly reduced (p < 0.01). After shipment, the plasma concentrations of IGFBP-5, glucocorticoids and IL-2 increased but decreased after lairage (p < 0.05) whereas IGF-1 decreased after shipment (p < 0.01). Repeated acute stress increased concentrations of IGFBP-3 and IGF-1 in exsanguination blood (p < 0.05). Differential IGF- signatures can indicate altered endocrine or metabolic control and thus contain complex animal-related information. The somatotropic axis may be of particular interest when established biomarkers such as cortisol, glucose, or lactate cannot be used for the assessment of animal stress or welfare.
Similar content being viewed by others
Introduction
Industrialized production of pork is an important element of the agriculture sector with high economic relevance, which results in pork being the most popular meat product worldwide1. Due to the specialization of pork production chain into different segments, there has been an increased need to transport large numbers of pigs, e.g. from the nursery to the growing and/or grow-to-finish farm and eventually to the slaughterhouse. There is an overall agreement that the transportation event includes multifaceted psychological and physical stress factors, which can have detrimental effects on the health and welfare of farm animals2,3,4,5 resulting in the concurrent activation of endocrine, metabolic or immune pathways6. In response to the increasing public demand for the respect of animal welfare in the livestock production practices, the identification and validation of objective biomarkers for a standardized monitoring of the health and welfare of livestock are needed. However, the value of the assessment of stress-associated blood parameters, such as glucocorticoids, catecholamines or glucose, is often limited due to the interference of different stress factors, genotype-specific stress reactions or poor practical feasibility7. Furthermore, it has been shown that repeated short- or long-term stress (i.e. tail biting) can result in a blunted stress response of the HPA-axis in rats and pigs8,9,10. With respect to the adaptive action of moderate repeated stress on the HPA-axis response, unbiased estimation of increased stress levels in animals during the pre-slaughter period is difficult. Also for metabolic markers of stress such as lactate, short-term versus longer-term stress resulted in differential responses in slaughter pigs11,12. In humans, it has been shown that parameters of the somatotropic axis are appropriate for the characterization of different stages of disorders or impairment of health13,14. Growth hormone (GH) is a primary regulator of vertebrate growth and metabolism. In mammals, it is assumed that acute physical stress, energy restriction, or acute phase of severe illness induce an amplification of GH secretion and increased levels of GH13,15,16. Although GH response to psychological stress is rarely seen17, studies in humans indicate that GH response is positively correlated with higher post-stress levels of anxiety18. In addition to direct effects, GH also affects body growth and metabolism indirectly through the stimulation of insulin-like growth factor (IGF) production in a number of tissues including the liver19. In blood, IGF is bound to IGF-binding proteins (IGFBPs) which control IGF availability, but also have IGF-independent functions20. As IGFBPs are sensitive markers to detect changes of the GH-dependent growth21,22, they are important biomarkers for diagnostics and treatment studies in humans14. In addition to the central role of the IGF-system in linking nutritional intake with somatic growth23,24,25 it is known that glucocorticoids influence levels of IGF-1 and IGFBPs26,27,28 suggesting an interference of acute stress with the IGF system. However, to our knowledge, stress-induced changes of the IGF-system have never been studied comprehensively in pigs yet.
Therefore, the aim of the study was to assess the effects of transport stress factors on the plasma circulating levels of IGF-1, IGF-2, and IGFBPs in pigs during different stages of commercial pig transportation in Germany. In addition, the effect of repeated stress by snaring three times within the pre-slaughter period of 28 h was investigated providing an experimental model for repeated acute stress8 in the pre-slaughter period. IGFBPs were evaluated using quantitative Western ligand blotting (qWLB) which provides complex information on IGFBP-profiles in blood plasma29. Furthermore, the calculation of distinct ratios, as IGFBP-3/-2 ratio or IGF-1/total amount of IGFBPs, provides additional information about regulation of the somatotropic axis and IGF-1 bioavailability, respectively. To validate the quality of results related to the activation of the IGF-system in response to stress, a set of established serum markers and stress vocalization reflecting the acute physiological stress response, inflammatory pathways and energy metabolism were also evaluated.
Results
Effects of sampling time on IGF-system, stress response, and energy metabolism
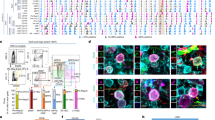
In non-transported pigs on the farm, food removal for a period of 19 h did not affect plasma concentrations of IGF-1, IGFBP-2, and IGFBP-3 (Suppl. Figure 1A). In addition, the time of day between 8 a.m. and 2 p.m. had no effect on IGF-1 or IGFBP-2 in non-transported pigs (Suppl. Figure 1B). However, at 12 p.m. reduced concentrations of IGFBP-3 were present in non-transported pigs if compared to 8 a.m. (p < 0.01). The concentrations of IGFBP-2 and IGFBP-3, which appear to be the dominant IGFBPs in porcine plasma (Fig. 1A), decreased in the course of the pre-slaughter period between 9.45 a.m. and 1.15 pm while the pigs rested in the lairage of slaughterhouse. The lowest levels of both IGFBP-2 and -3 were measured in slaughter blood. More specifically, plasma IGFBP-3 levels were lower at slaughter when compared with basal levels (p = 0.023, Fig. 1B), while IGFBP-2 concentrations were decreased after slaughter compared to basal levels (p = 0.004) and levels after shipment (p = 0.025, Fig. 1C). By contrast, IGFBP-5 was increased after shipment compared to basal levels (p = 0.0004) followed by a decrease after lairage (p = 0.010, Fig. 1D). In plasma, IGF-1 was reduced after shipment (p = 0.001) and after lairage (p = 0.035) compared to basal concentrations measured in the home pen (Fig. 1E), whereas IGF-2 was not affected by the sampling time (Fig. 1F). The total amount of all plasma IGFBPs (IGFBP-2, -3 and -5), simultaneously quantified by qWLB, was reduced in exsanguination blood after CO2 stunning, compared to levels after shipment (p = 0.0006, Fig. 2A). In contrast, the ratio of IGF-1/total IGFBPs just as IGF-1/IGFBP-3 were decreased exclusively after shipment (p = 0.0008, p = 0.008, respectively) compared to basal levels in the home pen on the farm (Fig. 2B). The ratio of IGF-1/IGFBP-5 decreased after shipment (p = 0.002) and after slaughter (p = 0.034) compared to basal concentrations (Suppl. Table 1). The sampling time had no effect on the ratio of IGF-1/IGFBP-2 and IGFBP-3/IGFBP-2. We next asked, if IGFBP-2 may indicate different levels of stress and tested different lengths of transport duration in slaughter pigs (Fig. 3). After 18 h of shipment, the IGFBP-2 concentrations in plasma were significantly reduced if compared to pigs transported for 6 or 12 h, whereas cortisol was similar in all groups irrespective of transportation time. Cortisol was increased after shipment compared to basal levels (p = 7.4E-06), lairage (p = 3.3E-05) and slaughter (p = 0.003, Fig. 4A). Corticosterone (Fig. 4B) was increased after shipment (p = 0.004) followed by a decrease (p = 0.043) at lairage. Distinct to cortisol, corticosterone was not normalized after lairage and was still elevated after slaughter (p = 0.049) compared to basal levels. Adrenaline and noradrenaline (Fig. 4C,D) were massively increased in exsanguination blood (p < 1.2E-12). IL-2 (Fig. 4E) was reduced after lairage if compared to shipment (p = 0.033). The concentrations of SAA were statistically not different between the sampling times (Fig. 4F). Albumin (Fig. 4G) also increased with prolonged sampling time compared to basal blood levels (p = 0.0019, p = 0.006, p = 0.013). Glucose, cholesterol, triglycerides, and lactate (Suppl. Figure 2A–D), were not significantly different in the pre-slaughter period but were increased in exsanguination blood compared to basal levels (p = 0.0008, p = 0.076, p = 0.005, p = 2.35E-05). Stress vocalization was significantly different during loading, transport, unloading, and blood sampling (Fig. 5A,B). The highest and lowest levels of vocalization were recorded during sampling and shipment, respectively. The number of lesions per pig increased after transport compared to home pen (p = 7.56E-11). A percentage of 26% of the gilts had no transport-induced increased lesion score whereas 31% of animals had an increase of score 1 and 43% of score 2 (Fig. 5C,D).
IGF-system at different sampling times of transportation procedure: IGFBP-profile (A), IGFBP-3 ((B); n = 31), IGFBP-2 ((C); n = 31), IGFBP-5 ((D); n = 31), IGF-1 ((E); n = 13) and IGF-2 ((F); n = 13). Quantitative data are presented as LS-means+ SE. rh: recombinant human. *p < 0.05, **p < 0.01, ***p < 0.001, #p < 0.1.
Stress and inflammation associated parameters at different sampling times of commercial transportation procedure: Plasma concentrations of cortisol (A), corticosterone (B), adrenaline (C) and noradrenaline (D), IL-2 (E), SAA (F) and albumin (G). Cortisol, IL-2 and SAA were analyzed with ELISA. Corticosterone was analyzed using LC-MS/MS. Adrenaline and noradrenaline were analyzed using HPLC. Albumin was analyzed using an enzymatic spectrophotometric assay. All Data are presented as LS-means+ SE. Cortisol, adrenaline, noradrenaline, SAA: n = 31 per sampling time. Corticosterone, IL-2, albumin: n = 13 per sampling time. #p < 0.1, *p < 0.05, ** p < 0.01, ***p < 0.001.
Stress vocalization and evaluation of lesions at different events of pig transport. (A) Stress vocalization, presented as LS-means + SE of every event, respectively. (B) Exemplary data of stress vocalization during different events of trial 2. (C) The number of lesions measured in home pen and after transport, presented as LS-means + SE. (D) Percentage of animals per category of transport-induced increase of lesion score. The stress vocalization was analysed using STREMODO and includes all animals in the range of sound recorder. Lesions: n = 65. *p < 0.05, **p < 0.01, ***p < 0.001.
Effects of repeated blood sampling by snaring on plasma parameters, meat and carcass quality in exsanguination blood
Slaughter blood was compared between pigs treated with repeated blood sampling using snaring (treatment group, n = 31) and pigs without blood sampling in the pre-slaughter period (control group, n = 34).The pigs of the treatment group were snared three times before slaughtering, in the home pen on farm (9:00 a.m.), after unloading (9:45 a.m.) and after lairage in the slaughterhouse (11:45 a.m.). Repeated blood sampling affected parameters of IGF-system and stress response. LS-means and p-values of pairwise comparison of plasma parameters, carcass and meat quality between the treatment group (with repeated blood sampling before slaughter) and controls (no repeated blood sampling before slaughter) are presented in Table 1. IGFBP-3, IGF-1 and the IGFBP-3/IGFBP-2 ratio and were higher in animals with repeated blood sampling compared to controls. Corticosterone and lactate were reduced whereas noradrenaline was elevated in the treatment group compared to control. Carcass and meat quality were not significantly affected by the repeated blood sampling.
Correlation of IGF-compounds and established markers of stress and metabolism
With only one exception, exclusively negative correlations were found between compounds from the IGF-system and established biomarkers of stress and metabolism (Table 2). Negative correlations were existent between IGF-1, IGF-2, IGFBP-2 or IGFBP-3 on one hand and cortisol, corticosterone, glucose, albumin, lactate, SAA, cholesterol or triglycerides on the other (p < 0.05). As the only exception, IGFBP-5 was positively regulated with cortisol (p < 0.001). Cortisol correlated positively with albumin and lactate in all samples (P < 0.05).
Discussion
The identification and application of novel biomarkers are needed to ensure reliable monitoring of animal welfare. In the present study, we have assessed the effects of shipment on compounds from the IGF-system. According to the hypothesis of Mesotten and van den Berghe14, the IGF-system and, in particular the signature of different IGFBPs, provide significant information in relation to disturbances of homeostasis in animals.
Effect of shipment
Similarly to previous works6,30, in this study, plasma concentrations of cortisol and corticosterone increased after transportation, including loading and unloading, which may be considered as a response of the HPA-axis to transport associated stress. In addition, increased concentrations of IL-2 have been found in plasma collected after unloading suggesting an effect of transportation on the pigs’ immune function as already reported31,32. With respect to stress vocalization, the data of this study suggest that the loading and unloading procedures are more stressful than road driving, which also is in agreement with previous reports6. Similar to humans, cortisol also in pigs was the predominant circulating glucocorticoid whereas corticosterone was found at more than 20-fold lower concentrations33. In mammals, glucocorticoids are thought to reduce circulating IGF-126. Interestingly, reduced serum concentrations of IGF-1 and IGFBP-5 were observed after 2 h of shipment. In post-weaning piglets at an age of 20 days after birth, 3h-shipment had no effect on serum IGF-1 concentrations34. By contrast in sows, heat stress over 57 days reduced serum concentrations of IGF-1 if compared to shorter exposure times of heat stress, although the IGFBPs have not been assessed in that study35. Also, acute stress reduced serum IGF-I concentrations in adult Yorkshire pigs36. In this study, 5 min of restraint stress depressed serum IGF-1 concentrations for about 20% 150 min after snaring. According to our knowledge, IGFBPs have not been assessed under conditions of stress in pigs. However, in fish, both stress and the administration of dietary cortisol decreased levels of plasma IGF-1 after 2 h but increased plasma levels of up to four different IGFBPs, which were not specified in these studies27,28. These findings support the biomarker potential of IGF-system for evaluation of stress in vertebrates. In any case both, higher levels of plasma IGFBP-5 and lower levels of plasma IGF-1 after shipment, may be related to increased cortisol and corticosterone plasma levels also shortly after transport.
The concentrations of other IGFBPs were not affected by shipment over a period of 2 h. However, in a separate experiment with longer transportation, IGFBP-2 concentrations in plasma from pigs were reduced over time, whereas cortisol was similar after 6, 12, and 18 h of transport. IGFBP-2 is regulated by hormones (GH, steroids, insulin, leptin) but also by proteolysis37. Accordingly, these parameters may be considered as effectors of IGFBP-2 concentrations under conditions of elevated stress. All animals were fasted to a similar extent for 20–24 hours and plasma was derived from slaughter blood at the same time in the morning. Thus, IGFBP-2 concentrations may decrease over time due to the length of shipment, whereas cortisol did not change with prolonged duration of transportation. Thus, we have reason to assume dose-response of the plasma IGFBP-2 concentrations in response to stress. By contrast, cortisol levels possibly due to habituation have no biomarker value under conditions of prolonged stress in transported slaughter pigs.
Effects of lairage and slaughter
During lairage, serum concentrations of corticoids and IL-2 normalized indicating efficient recovery after shipment. In addition, also IGF-I and IGFBP-5 returned to normal if compared to basal levels before transport, indicating responsiveness of IGF-I and IGFBP-5 to intermediate stress. All metabolic parameters were unaffected by treatments other than slaughter. The massive increase of catecholamines in slaughter blood is due to CO2 stunning. The significant reduction of IGFBP-2 in slaughter blood is considered being more the consequence of the prolonged stressful procedure and less due to the acute condition of stress in the CO2 stunner. This assumption is supported by the perpetual decrease of plasma IGFBP-2 concentrations during prolonged transport duration from 6 h up to 18 h of shipment.
IGFBP-2 was reduced also in slaughter blood if compared to basal levels. Because food removal over a period of 19 h did not affect IGFBP concentrations in a control experiment performed with non-transported pigs, these results cannot be explained by the effects of dietary energy levels and fasting status. By contrast, prolonged fasting is well known to regulate IGFBP-237. Accordingly, in newborn pigs prolonged fasting for 48 h decreased IGFBP-2 concentrations measured by Western ligand blotting24. Fasting for 70 h also reduced serum concentrations of IGF-1 and IGFBP-3 but increased serum levels of IGFBP-2 in sheep38. Besides IGFBP-2 also IGFBP-3 was decreased in slaughter blood. However, this reduction also may be due to circadian effects, because at 12:00 o’clock lower IGFBP-3 concentrations have been found if compared to IGFBP-3 levels in the morning at 8:00 a.m.
Skin damage is associated with higher pH values and darker meat colour (potentially dark, firm, dry; DFD) resulting from increased glycogen depletion in the muscle39. In the current study, lesion scoring revealed only a slight increase of skin damage after transportation. Furthermore, pH1, pH2, and drip loss analyses revealed no stress induced effect on meat quality40,41 which is not surprising considering the high standardized pre-slaughter conditions with no mixing in lairage and handling of pigs in small groups without electric prodding and push gates in lairage3,39 applied in this study. In the present study, CO2-stunning induced a significant increase of plasma glucose as a result of the high release of catecholamines such as adrenaline42. The stress response during slaughtering is influenced by breed-specific traits, pre-slaughter conditions such as handling or lairage time, and the type of stunning system43,44, which have to be considered for the evaluation of stress-associated biomarkers.
The effect of restraint stress
Short-term restraint stress by snaring and vena cava blood sampling increased concentrations of catecholamines, cortisol, lactate, and glucose in pigs45,46,47. Therefore, we asked whether repeated restraining for blood sampling of the treatment group influenced blood parameters and meat quality after slaughtering as an experimental model for repeated short-term stress. The results indicate that repeated acute stress by snaring attenuates the corticosterone response to acute slaughter stress. In rats, it was described that repeated short-term stress by restraint potently attenuates acute stress- induced activation of the HPA axis, indicating an adaptive effect of moderate stress on the HPA axis response to acute stress10. This attenuation might be important regarding the validity of glucocorticoids as biomarkers for pre-slaughter stress of pigs in exsanguination blood. In contrast to glucocorticoids, in the present study, noradrenaline was increased in repeatedly restrained pigs suggesting an enhanced stimulation of sympathetic-adrenal medullary system by slaughter-induced stress. In rats, it is described that exposing repeatedly stressed animals to an unrelated stressor results in an enhanced sympathetic-adrenal medullary response indicating a sensitization of this axis48,49. Both habituation and sensitization are described as adaptive processes that allow the organism to physiologically cope with prolonged or intermittent stress exposure preventing deleterious actions but maintaining response flexibility to new threats50. In the current study, IGF-1, IGFBP-3 and the ratio of IGFBP-3/-2 were higher in exsanguination blood of repeatedly restrained pigs indicating a modulation of IGF-system by repeated stress. It is described, that glucocorticoids regulate IGF and IGFBP-expression in a direct or indirect manner51. Glucocorticoids, such as cortisol or dexamethasone, are shown to down regulate IGF-I and IGFBP-3 levels aiming at inhibiting IGF anabolic activity51. The higher levels of corticosterone, found in the exsanguination blood of pigs not subjected to repeated blood sampling may explain their lower levels of IGF-1 and IGFBP-3 at slaughter in this study. As there was no effect of repeated blood sampling on plasma IGFBP-2 concentrations, the increased levels of IGFBP-3 led to an increased IGFBP-3/IGFBP-2 ratio, which is considered as a sensitive marker for GH-induced somatic growth and metabolic homeostasis in humans. During long-term critical illness or low physical performance this ratio decreases14,52 due to a suppression of GH release13,16. Because of the complex regulation of IGF-system, short term effects of acute and repeated stressors on IGFBP-3/IGFBP-2 ratio has still to be investigated. However our study clearly demonstrates acute regulation of the IGF-system during animal transport and the significant correlations with established biomarkers of stress also support novel biomarker potential of the IGF-system.
To summarize and conclude, the signature of the IGF-system contains a complex set of information for specific segments in the transport process from farm to slaughter (Table 3). In both transportation studies, plasma concentrations of IGFBP-2 were reduced over time suggesting dose-dependency of this parameter. Beyond other IGFBPs, IGFBP-5 concentrations were highly dynamic between selected segments of the transportation chain and similar to alterations of IL-2 and glucocorticoids. Finally, plasma IGF-1 concentrations are flexible during animal shipment and may increase or decrease during transportation, while IGF-2 was not regulated acutely during pig transportation. Our results provide substantial evidence that compounds from the IGF-system are specifically regulated by different stressors in the transportation chain. In particular, our results provide evidence that glucocorticoids are less informative under conditions of repeated or prolonged stress. Compounds from the IGF-system may thus represent physiologically relevant biomarkers of repeated or prolonged stress e.g. with extended periods of shipment. Monitoring compounds from the IGF-system in pigs may generate novel biomarker information and improve current standards of animal husbandry. Due to major effects on economical traits, consideration of IGF-compounds in farm animals may guide development of animal husbandry in order to integrate both ethical and economic issues in the future.
Materials and Methods
Animals and pre-slaughter conditions
All experimental procedures adhered to the current guidelines and were approved by the Landesamt für Landwirtschaft, Lebensmittelsicherheit und Fischerei Mecklenburg-Vorpommern as the responsible authorities (reference number LALLF M-V/TSD/7221.3-2-033/14). The pigs were fed a commercial diet (12.8 MJ ME/kg, 15% crude protein, CP) and had free access to water. The animals were grouped at least 4 months before shipment. Social group structure was maintained during the whole experimental procedure on farm, truck and slaughterhouse. A total of 65 market weight (115 ± 7.9 kg) crossbred gilts (Danish Landrace x Yorkshire sows x Piétrain boars, homozygous NN for Hal gene = stress resistant) were randomly selected at a commercial finishing farm and shipped (2 h) to a commercial slaughterhouse (VION, Perleberg, Germany). The ambient temperature during shipment was −5–10 °C. At the farm the pigs were withdrawn of feed for 18 h (since 12.00 pm day 1) and loaded to a commercial truck by a trained crew using boards. The upper decks of the truck were lift up hydraulic. On arrival at the slaughterhouse, pigs were unloaded through a plane ramp using boards. Pigs were kept in lairage for 3 h during which they had free access to water and water sprinkling. For blood sampling pigs were separated in 2 pens (2.5 m2/pig) and control pigs were separated in a third pen (1.25 m2/pig). At the end of lairage, groups of 5 pigs were driven with boards to a mechanical driveway forward to the CO2 stunner (Butina CO2 backloader system, Holbaek, Denmark, 92% CO2) and subsequently exsanguinated in a vertical position. An overview about the experimental design is given in Suppl. Figure 3. In order to test the effect of transport duration on plasma concentrations of IGFBP-2 and cortisol, we analysed samples of an independent shipment study in slaughter pigs53. All pigs (total n = 240) had no access to food for 20–24 h and arrived at the slaughter plant the same time as described before53,54. The effects of food removal and time of day on the IGF-system were assessed in non-transported pigs as described before55. Thereby, blood sampling was performed in catheterized pigs every two hours starting 8 a.m. until 8 a.m. next morning.
Blood collection
Exsanguination blood was gathered from all pigs included in the present study. Basal blood samples from vena cava cranialis were collected in the home pen (21 h before transport). Therefore the pigs were snared and the neck was stretched well upwards for punction of the vena. A total of 31 pigs (treatment group) were used for repeated blood sampling collected in the home pen (basal), directly after unloading (shipment) as well as after lairage (lairage). Repeated bleeding was not performed in a second group of 34 pigs (control group) in order to study the potential effect of the bleeding procedure. Blood samples (approximately 8 ml) were collected in ice-cooled tubes containing 200 µl of 0.1 M EDTA and centrifuged at 2,000 g for 10 min at 4 °C. Then, the plasma was stored in dry ice for transportation to laboratory unit and stored at −20 °C until analysis.
Quantitative Western ligand blotting
IGFBP-2, -3 and 5 were analyzed by quantitative Western ligand blot (qWLB) analysis as described previously29. Due to low abundance, IGFBP-4 was detected but not quantified in the porcine plasma. The analytical range for each IGFBP was 150–15000 ng/ml. Inter-and intra-assay coefficients of variation (CV) were determined by measuring artificial serum samples spiked with low (500 ng/ml) and high (3000 ng/ml) concentrations of IGFBPs. The intra-assay CV (n = 9) for IGFBP-2,-3 and -5 was <15% at high and low concentrations. The inter-assay CV (n = 8) for all IGFBPs was <20% at low and <15% at high concentrations according to recommendations of EMA guideline56. In order to account for the higher variances of Western blotting data compared to ELISA data we decided to assess all samples by quantitative Western ligand blotting.
ELISA assays
All ELISA assays were performed using commercially available ELISA Kits according to the manufacturer’s instructions. Concentrations of IGF-1 and IGF-2 were analyzed in the plasma samples using ELISA Kits E20 and E30 (Mediagnost, Reutlingen, Germany). Cortisol was determined with EIA 1887 (DRG Instruments GmbH, Marburg, Germany) and cross validated with LC-MS/MS revealing a correlation coefficient of r = 0.926 (Spearman correlation). Plasma concentrations of IL-2 were analyzed by Bioglobe GmbH (Hamburg, Germany) using the Bio-Plex Pro™ Human Cytokine Assay (Biorad, Hercules, U.S.A.). Serum amyloid A (SAA) was determined using a Phase SAA ELISA kit (Tridelta Development Ltd., Maynooth, Ireland).
LC-MS/MS and HPLC
Plasma concentration of corticosterone was analyzed using LC-MS/MS technique described previously57. The intra-assay CV at different concentrations (5 ng/ml, 50 ng/ml, 500 ng/ml) for corticosterone were 13.05%, 10.98% and 4.64%, respectively. The inter-assay CV for 100 ng/ml (n = 20) was 5.57%. Plasma concentrations of adrenaline and noradrenaline were analyzed using HPLC with electrochemical detection after extraction from plasma by absorption on aluminum oxide58. The intra- and inter-assay CV were 3.1% and 5.5%, respectively, for adrenaline and 2.0% and 2.5%, respectively, for noradrenaline.
Enzymatic spectro-photometric assays
Plasma lactate was determined by an enzymatic-spectrophotometric assay (Labor + Technik Eberhard Lehmann GmbH, Berlin, Germany). Triglycerides (TG), cholesterol, glucose and albumin were assayed in plasma samples via enzymatic coloured test by using commercial kits (triglycerides: No. LT-TR 0015, total cholesterol: No. LT-CH 0031, glucose: LT-GL 0251; albumin: LT-AB 0103; Labor & Technik Eberhard Lehmann Berlin, Germany, respectively) as described59.
Stress vocalization
Stress vocalization was recorded as described before60. In brief, a commercially available voice recorder was installed at the loading ramp of the truck during loading and unloading of the animals and near the waiting pen during blood sampling. A second voice recorder was fixed within the animal truck which was a self-developed voice recorder for harsh environment based on a digital signal processing (DSP) board. The recordings were divided into the sections loading, unloading, transportation and sampling. The WAV-files of the recordings were retrospectively analysed using the STREMODO software. The software uses a neuronal network to classify time windows of 50 ms into stressful vocalization and non-stressful vocalisation. Stress detections in successive windows were taken as a single event, while allowing interruptions of less than 200 ms. The STREMODO software can give false positive classifications for high-pitched metallic or air flow sounds. Such sounds occur most likely within the truck and during transportation. They were not eliminated manually. However, very short detection events with durations of less than 150 ms were excluded from the statistical evaluation.
Evaluation of carcass and meat quality
Skin lesions on the body of all pigs were registered by the same observer in the home pen before transport and after the second blood sampling essentially as described61. The total number of lesions was evaluated in home pen and after transport. The increase of lesions, that were not present before transport, were counted and lesion scores assigned as follows: 0 – no skin injuries, 1 – one to three wounds; 2 – four or more injuries. The hot carcass weight and lean body content was standardized assessed in the slaughterhouse. The pH value was evaluated in the musculus longissimus dorsi by assessing the pH value of all pigs after 45 min (pH1) and 24 h (pH24) post-mortem. Drip loss was analysed in musculus longissimus dorsi according to standard procedures62.
Statistical analysis
Statistical analyses were performed using SAS software version 9.3 (SAS, Cary, NC, USA). Descriptive statistics and tests for normality were calculated with the UNIVARIATE procedure. The data of all blood parameters were evaluated by ANOVA using MIXED procedure. Repeated measures on the same animal were taken into account using the repeated statement in the MIXED procedure and an unstructured block diagonal residual covariance matrix. The model comprised the fixed effects of sampling time (home pen, shipment, lairage, slaughter), trial (2) and the two-way interaction sampling time x trial. To evaluate the effect of the repeated blood collection procedure on the concentrations of blood parameters at exsanguination the model considered the repeated blood collection act (yes or no), trial (2) and their two-way interactions as fixed effects. For the comparison of stress vocalization and lesions a generalized linear model were employed, applying the GLIMMIX procedure using a Poisson model (distribution = Poisson, link = log). For stress vocalization the model comprised the fixed effect time (loading, unloading, transportation, sampling) and for lesions the fixed effects time (home pen, after transport), trial (2) and group (treatment, control) and their interactions. From the data of IGF-system, for each animal the ratio of IGFBP-3/-2, the total amount of IGFBPs (indicator for IGF-binding capacity), the ratio of IGF-1/total IGFBPs (indicator for IGF-1 bioavailability) and the ratio of IGF-1 to IGFBP-2,-3 and -5 was calculated. Effects and differences were considered significant if p < 0.05. Data, which were below the limit of quantification, were excluded from the statistical analyses.
References
Davis, C. G. & Lin, B.-H. Factors affecting US pork consumption. (US Department of Agriculture, Economic Research Service, 2005).
Grandin, T. & Shivley, C. How Farm Animals React and Perceive Stressful Situations Such As Handling, Restraint, and Transport. Animals (Basel) 5, 1233–1251, doi:10.3390/ani5040409 (2015).
Schwartzkopf-Genswein, K. S. et al. Road transport of cattle, swine and poultry in North America and its impact on animal welfare, carcass and meat quality: a review. Meat Sci 92, 227–243, doi:10.1016/j.meatsci.2012.04.010 (2012).
Von Borell, E. & Schäffer, D. Legal requirements and assessment of stress and welfare during transportation and pre-slaughter handling of pigs. Livestock Production Science 97, 81–87 (2005).
Warriss, P. The welfare of slaughter pigs during transport. Animal welfare 7, 365–381 (1998).
Pérez, M. et al. Effect of transport time on welfare and meat quality in pigs. Meat science 61, 425–433 (2002).
Ciepielewski, Z. M. et al. The effects of ryanodine receptor 1 (RYR1) mutation on plasma cytokines and catecholamines during prolonged restraint in pigs. Veterinary immunology and immunopathology 156, 176–181, doi:10.1016/j.vetimm.2013.10.001 (2013).
Gadek-Michalska, A. & Bugajski, J. Repeated handling, restraint, or chronic crowding impair the hypothalamic-pituitary-adrenocortical response to acute restraint stress. Journal of Physiology and Pharmacology 54, 449–460 (2003).
Valros, A. et al. Physiological indicators of stress and meat and carcass characteristics in tail bitten slaughter pigs. Acta Veterinaria Scandinavica 55, 75 (2013).
Rushen, J., Schwarze, N., Ladewig, J. & Foxcroft, G. Opioid modulation of the effects of repeated stress on ACTH, cortisol, prolactin, and growth hormone in pigs. Physiology & behavior 53, 923–928 (1993).
Edwards, L. N. et al. Use of exsanguination blood lactate to assess the quality of pre-slaughter pig handling. Meat Sci 86, 384–390, doi:10.1016/j.meatsci.2010.05.022 (2010).
Edwards, L. N. et al. The effects of pre-slaughter pig management from the farm to the processing plant on pork quality. Meat Sci 86, 938–944, doi:10.1016/j.meatsci.2010.07.020 (2010).
Møller, N. & Jørgensen, J. O. L. Effects of growth hormone on glucose, lipid, and protein metabolism in human subjects. Endocrine reviews 30, 152–177 (2009).
Mesotten, D. & Van den Berghe, G. Changes within the GH/IGF-I/IGFBP axis in critical illness. Critical care clinics 22, 17–28, v, doi:10.1016/j.ccc.2005.09.002 (2006).
Ranabir, S. & Reetu, K. Stress and hormones. Indian journal of endocrinology and metabolism 15, 18 (2011).
Lawrence, T. L. J., Fowler, V. R. & Novakofski, J. E. Growth of farm animals. (CABI, 2012).
Tamai, H. et al. Effect of psychological stress on human growth hormone response to thyrotropin-releasing hormone in normal controls. Psychotherapy and psychosomatics 46, 122–126 (1986).
Abplanalp, J. M., Livingston, L., Rose, R. M. & Sandwisch, D. Cortisol and growth hormone responses to psychological stress during the menstrual cycle. Psychosomatic Medicine 39, 158–177 (1977).
Jones, J. I. & Clemmons, D. R. Insulin-Like Growth Factors and Their Binding Proteins: Biological Actions*. Endocrine reviews 16, 3–34 (1995).
Hwa, V., Oh, Y. & Rosenfeld, R. G. The Insulin-Like Growth Factor-Binding Protein (IGFBP) Superfamily 1. Endocrine reviews 20, 761–787 (1999).
Bielohuby, M. et al. Validation of serum IGF-I as a biomarker to monitor the bioactivity of exogenous growth hormone agonists and antagonists in rabbits. Disease models & mechanisms 7, 1263–1273, doi:10.1242/dmm.016519 (2014).
Krieger, F. et al. Polyethylene glycol-coupled IGF1 delays motor function defects in a mouse model of spinal muscular atrophy with respiratory distress type 1. Brain 137, 1374–1393 (2014).
McCusker, R., Wangsness, P. J., Griel, L. & Kavanaugh, J. Effects of feeding, fasting and refeeding on growth hormone and insulin in obese pigs. Physiology & behavior 35, 383–388 (1985).
McCusker, R. H., Cohick, W. S., Busby, W. H. & Clemmons, D. R. Evaluation of the developmental and nutritional changes in porcine insulin-like growth factor-binding protein-1 and -2 serum levels by immunoassay. Endocrinology 129, 2631–2638, doi:10.1210/endo-129-5-2631 (1991).
Frystyk, J., Delhanty, P., Skjaerbaek, C. & Baxter, R. Changes in the circulating IGF system during short-term fasting and refeeding in rats. American Journal of Physiology-Endocrinology And Metabolism 277, E245–E252 (1999).
Unterman, T. G., Jentel, J. J., Oehler, D. T., Lacson, R. G. & Hofert, J. F. Effects of glucocorticoids on circulating levels and hepatic expression of insulin-like growth factor (IGF)-binding proteins and IGF-I in the adrenalectomized streptozotocin-diabetic rat. Endocrinology 133, 2531–2539 (1993).
Davis, K. B. & Peterson, B. C. The effect of temperature, stress, and cortisol on plasma IGF-I and IGFBPs in sunshine bass. General and comparative endocrinology 149, 219–225 (2006).
Kajimura, S. et al. Dual mode of cortisol action on GH/IGF-I/IGF binding proteins in the tilapia, Oreochromis mossambicus. Journal of endocrinology 178, 91–99 (2003).
Wirthgen, E. et al. Quantitative Western ligand blotting reveals common patterns and differential features of IGFBP-fingerprints in domestic ruminant breeds and species. Growth hormone & IGF research: official journal of the Growth Hormone Research Society and the International IGF Research Society 26, 42–49, doi:10.1016/j.ghir.2015.11.001 (2016).
Breinekova, K., Svoboda, M., Smutna, M. & Vorlova, L. Markers of acute stress in pigs. Physiological research 56, 323 (2007).
Pineiro, M. et al. Characterisation of the pig acute phase protein response to road transport. The Veterinary Journal 173, 669–674 (2007).
Lv, Q., Zhang, S. & Zhao, R. Transportation stress alters the expression of immunoregulatory cytokines in the porcine thymus. The Veterinary Journal 187, 229–233 (2011).
Nishida, S., Matsumura, S., Horino, M., Oyama, H. & Tenku, A. The variations of plasma corticosterone/cortisol ratios following ACTH stimulation or dexamethasone administration in normal men. The Journal of Clinical Endocrinology & Metabolism 45, 585–588 (1977).
Kojima, C. J., Kattesh, H. G., Roberts, M. P. & Sun, T. Physiological and immunological responses to weaning and transport in the young pig: modulation by administration of porcine somatotropin. Journal of animal science 86, 2913–2919, doi:10.2527/jas.2008-1089 (2008).
Williams, A. M. et al. Effects of a controlled heat stress during late gestation, lactation, and after weaning on thermoregulation, metabolism, and reproduction of primiparous sows. Journal of animal science 91, 2700–2714, doi:10.2527/jas.2012-6055 (2013).
Farmer, C., Dubreuil, P., Couture, Y., Brazeau, P. & Petitclerc, D. Hormonal changes following an acute stress in control and somatostatin-immunized pigs. Domestic animal endocrinology 8, 527–536 (1991).
Hoeflich, A. & Russo, V. C. Physiology and pathophysiology of IGFBP-1 and IGFBP-2 - Consensus and dissent on metabolic control and malignant potential. Best practice & research. Clinical endocrinology & metabolism 29, 685–700, doi:10.1016/j.beem.2015.07.002 (2015).
Ogawa, E. et al. Pretreatment with bovine growth hormone is as effective as treatment during metabolic stress to reduce catabolism in fasted lambs. Endocrinology 137, 1242–1248, doi:10.1210/endo.137.4.8625895 (1996).
Faucitano, L. Causes of skin damage to pig carcasses. Canadian Journal of Animal Science 81, 39–45 (2001).
Adzitey, F. & Nurul, H. Pale soft exudative (PSE) and dark firm dry (DFD) meats: Causes and measures to reduce these incidences-a mini review. International Food Research Journal 18, 11–20 (2011).
Branscheid, W., Honikel, K., Von Lengerken, G. & Troeger, K. Qualität von Fleisch und Fleischwaren. 2., überarbeitete und erweiterte Auflage. Edition Fleisch 1, 247–286 (2005).
Hartung, J., Nowak, B., Waldmann, K. H. & Ellerbrock, S. [CO2-stunning of slaughter pigs: effects on EEG, catecholamines and clinical reflexes]. Dtsch Tierarztl Wochenschr 109, 135–139 (2002).
Peres, L. M. et al. Effect of low or high stress in pre-slaughter handling on pig carcass and meat quality. Revista Brasileira de Zootecnia 43, 363–368 (2014).
Bertoloni, W., Silveira, E. T. F., Ludtke, C. B. & Andrade, J. C. D. Evaluation of pig hybrids stunned with electrical and gaseous (co2) systems. Part 1: blood stress indicators. Food Science and Technology (Campinas) 26, 564–570 (2006).
Neubert, E., Gurtler, H. & Valentin, G. Einfluß der Fixation von Mastschweinen mittels Oberkieferschlinge auf das Verhalten der Plasmakonzentrationen von Catecholaminen, Cortisol, Insulin und Stoffwechselparametern. Berliner und Münchener Tierärztliche Wochenschrift 109, 409–413 (1996).
Roozen, A., Tsuma, V. & Magnusson, U. Effects of short-term restraint stress on plasma concentrations of catecholamines, beta-endorphin, and cortisol in gilts. American journal of veterinary research 56, 1225–1227 (1995).
Merlot, E., Mounier, A.-M. & Prunier, A. Endocrine response of gilts to various common stressors: a comparison of indicators and methods of analysis. Physiology & behavior 102, 259–265 (2011).
Konarska, M., Stewart, R. E. & McCarty, R. Sensitization of sympathetic-adrenal medullary responses to a novel stressor in chronically stressed laboratory rats. Physiol Behav 46, 129–135 (1989).
McCarty, R., Horwatt, K. & Konarska, M. Chronic stress and sympathetic-adrenal medullary responsiveness. Soc Sci Med 26, 333–341 (1988).
Herman, J. P. Neural control of chronic stress adaptation. Front Behav Neurosci 7, 1–12 (2013).
Dell, G., Ward, A., Shokrai, A., Madej, A. & Engström, W. Regulation of the IGF System by Glucocortidoids. Zoological science 16, 377–385 (1999).
van den Beld, A. W. et al. High IGFBP2 levels are not only associated with a better metabolic risk profile but also with increased mortality in elderly men. European journal of endocrinology/European Federation of Endocrine Societies 167, 111–117, doi:10.1530/eje-12-0160 (2012).
Correa, J. A. et al. Welfare of Pigs Being Transported over Long Distances Using a Pot-Belly Trailer during Winter and Summer. Animals: an open access journal from MDPI 4, 200–213, doi:10.3390/ani4020200 (2014).
Goumon, S. et al. Effects of transport duration on maintenance behavior, heart rate and gastrointestinal tract temperature of market-weight pigs in 2 seasons. Journal of animal science 91, 4925–4935, doi:10.2527/jas.2012-6081 (2013).
Metges, C. C. et al. Intrauterine growth retarded progeny of pregnant sows fed high protein:low carbohydrate diet is related to metabolic energy deficit. PloS one 7, e31390, doi:10.1371/journal.pone.0031390 (2012).
Agency, E. M. Guideline on bioanalytical method validation. Committee for Medicinal Products for Human Use (CHMP) (2011).
Kunze, M. et al. Bioanalytical validation for simultaneous quantification of non-aromatic steroids in follicular fluid from cattle via ESI-LC-MS/MS. Journal of chromatography. B, Analytical technologies in the biomedical and life sciences 1007, 132–139, doi:10.1016/j.jchromb.2015.10.010 (2015).
Otten, W. et al. Effects of low and high protein: carbohydrate ratios in the diet of pregnant gilts on maternal cortisol concentrations and the adrenocortical and sympathoadrenal reactivity in their offspring. Journal of animal science 91, 2680–2692 (2013).
Brenmoehl, J. et al. Metabolic adaptations in the liver of born long-distance running mice. Medicine and science in sports and exercise 45, 841–850 (2013).
Schön, P. C., Puppe, B. & Manteuffel, G. Automated recording of stress vocalisations as a tool to document impaired welfare in pigs. Animal Welfare I, 105–110 (2004).
Stukenborg, A., Traulsen, I., Stamer, E., Puppe, B. & Krieter, J. The use of a lesion score as an indicator for agonistic behaviour in pigs. Arch Tierz 55, 163–170 (2012).
Küchenmeister, U., Kuhn, G. & Ender, K. Seasonal effects on Ca 2+ transport of sarcoplasmic reticulum and on meat quality of pigs with different malignant hyperthermia status. Meat science 55, 239–245 (2000).
Acknowledgements
The authors thank Evelin Normann, Regina Wal, Dagmar Mähling, Frank Hintze, Kurt Wendland, Sabine Hinrichs, and Luong Chau from the FBN Dummerstorf, Klaus Voigt and Florian Alex from VION Perleberg GmbH, and Susanne Gellert from ZNVG eG Neumünster for excellent technical and organizational assistance. The authors want to express their gratitude to Prof. Gerhard Manteuffel for substantial encouragement during project initiation. The publication of this article was funded by the Open Access Fund of the Leibniz Association and the Open Access Fund of the Leibniz Institute for Farm Animal Biology (FBN).
Author information
Authors and Affiliations
Contributions
E.W., M.K., W.O., U.G., K.S., P.S., R.P., C.C.M., B.S., K.S. and A.H. designed the study. E.W., M.K., C.W., C.H., M.S., J.B., E.K., M.T., W.O., U.G., P.S., C.M., and R.P. performed experiments. E.W., M.K., C.W., C.H., M.S., J.B., E.K., M.T., W.O., U.G., P.S., C.M., A.T., R.P., S.E., S.G., L.F., and A.H. analysed data. E.W. and A.H. wrote the main manuscript. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
This study was supported by a joint grant from Fleischwerk EDEKA Nord GmbH and VION Perleberg GmbH. AH was supported by grants from the Deutsche Forschungsgemeinschaft (DFG HO2003/6-1) and from the Bundesministerium für Bildung und Forschung (BMBF; Innovationsforum Tierwohl). Sébastien Goumon was supported by grant No MZERO0717 from the Czech Ministry of Agriculture. EW, CH, and AH are related to Ligandis GbR. SE is employed by Fleischwerk EDEKA Nord. All other authors have nothing to declare.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wirthgen, E., Kunze, M., Goumon, S. et al. Interference of stress with the somatotropic axis in pigs – lights on new biomarkers. Sci Rep 7, 12055 (2017). https://doi.org/10.1038/s41598-017-11521-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-11521-5
This article is cited by
-
Finding biomarkers of experience in animals
Journal of Animal Science and Biotechnology (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.