Abstract
Multiple labs have reported that mammalian ovaries contain oogonial stem cells (OSCs), which can differentiate into oocytes that fertilize to produce offspring. However, the physiological relevance of these observations to adult ovarian function is unknown. Here we performed targeted and reversible ablation of premeiotic germ cells undergoing differentiation into oocytes in transgenic mice expressing the suicide gene, herpes simplex virus thymidine kinase (HSVtk), driven by the promoter of stimulated by retinoic acid gene 8 (Stra8), a germ cell-specific gene activated during meiotic commitment. Over a 21-day ablation phase induced by the HSVtk pro-drug, ganciclovir (GCV), oocyte numbers declined due to a disruption of new oocyte input. However, germ cell differentiation resumed after ceasing the ablation protocol, enabling complete regeneration of the oocyte pool. We next employed inducible lineage tracing to fate map, through Cre recombinase-mediated fluorescent reporter gene activation only in Stra8-expressing cells, newly-formed oocytes. Induction of the system during adulthood yielded a mosaic pool of unmarked (pre-existing) and marked (newly-formed) oocytes. Marked oocytes matured and fertilized to produce offspring, which grew normally to adulthood and transmitted the reporter to second-generation offspring. These findings establish that oocytes generated during adulthood contribute directly to ovarian function and natural fertility in mammals.
Similar content being viewed by others
Introduction
Male germline stem cells (GSCs), or spermatogonial stem cells (SSCs), have been identified in the testes of essentially all animal species1, 2. The existence of female GSCs, or oogonial stem cells (OSCs), in adult ovaries has been established and is now widely accepted for flies3 and fish4. Until recently, however, it was thought that female mammals relied on primordial germ cells to generate their entire quota of oocytes during embryogenesis. As such, female GSC function in higher vertebrates was thought to be lost during fetal development, leading to the endowment of a non-renewable pool of ‘resting’ or quiescent primordial oocyte-containing follicles at birth5. Once established, the continuous exit of primordial follicles from this resting pool, due primarily to growth activation to primary follicle stages and beyond – a process in mice that has been estimated to ‘deplete’ the primordial oocyte stockpile by approximately 89 follicles per day during postnatal life6, eventually leads to complete follicular exhaustion as females reach advanced reproductive ages7, 8. This paradigm of a non-renewing pool of oocytes was questioned by a study in 2004 that identified mitotically-active cells expressing DEAD box polypeptide 4 (Ddx4), a conserved germ cell marker9, in ovaries of juvenile and young adult mice10. Additionally, histomorphometry-based counting of viable and atretic oocytes over time, coupled with mathematical modeling, uncovered a pronounced discordance in how quickly primordial oocyte numbers should decline during postnatal life, if this pool is non-renewing, versus what occurs, which is much slower10. This discordance, verified by others later11, raised questions over how the primordial oocyte population, if fixed at birth5, can remain relatively unchanged in numbers during juvenile and young adult life in the face of a constant rate of exit (depletion) through growth activation6.
In 2009, OSCs were isolated from postnatal mouse ovaries using Ddx4 antibody-based sorting12. In addition to reaffirming the mitotic capacity and other characteristic features of these primitive germ cells reported earlier10, this study also showed that GFP-expressing OSCs transplanted into ovaries of chemotherapy-conditioned wild type mice undergo differentiation into oocytes that mature into eggs, which can be fertilized to produce viable offspring12. Many reports followed describing the existence and characteristics of OSCs in ovaries of not just mice, but also rats, cows, non-human primates and humans13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35. Paradigms shifts are not without controversy, however, and the identification of OSCs in mammals is no exception. One area of debate concerns the strategy used to obtain OSCs for characterization studies. The Wu lab first published on the utility of Ddx4 antibody-based magnetic-assisted cell sorting (MACS) to isolate OSCs from mouse ovaries12. These observations were subsequently confirmed and extended by others with the validation of Ddx4 antibody-based fluorescence-activated cell sorting (FACS) for OSC isolation16,17,18,19,20, 22, 26,27,28,29,30,31,32. Nonetheless, conceptual issues have been raised by some scientists disputing the existence of OSCs in mammals based on claims that antigenic sequences in Ddx4 should not be useful for antibody-based sorting of viable OSCs if Ddx4 is a cytoplasmic protein in germ cells36,37,38, as reported previously9, 39. However, these previous reports were published a decade or more prior to the initial purification of OSCs, and thus conclusions drawn regarding Ddx4 localization in germ cells were derived from analysis of embryonic (primordial) germ cells, male germ cells or oocytes, not OSCs.
Following the first report of OSC isolation12, antibody-conjugated microbead technology confirmed that Ddx4 is retained completely inside of oocytes; however, parallel analysis of OSCs identified externalization of the C-terminus of Ddx4 protein16. Extensive FACS-based validation work further showed that extracellular Ddx4 (ecDdx4)-positive cells isolated from adult mouse ovaries through C-terminal antibody binding to viable (non-permeabilized) cell fractions are, in turn, recognized by a different (N-terminal) Ddx4 antibody only after the purified cells are permeabilized16. These types of dual antibody-single protein studies, which are standard practice for identification of cell surface antigens40, not only established the specificity of both antibodies used for analysis of Ddx4 but also the extracellular (C-terminus) versus intracellular (N-terminus) location of different antigenic sequences of Ddx4 in OSCs. Moreover, since both Ddx4 antibodies specifically labeled oocytes in fixed ovarian tissue sections16, the reported inability of C-terminal Ddx4 antibodies to recognize viable oocytes during FACS is due to an absence of this antigenic sequence on the surface of non-permeabilized oocytes – a conclusion supported by antibody-conjugated microbead studies16. It is worth noting that a year before the first report of OSC isolation by Ddx4 antibody-based sorting12, viable germ cells were purified from cultures of human embryonic stem cells using FACS coupled with DDX4 antibodies41.
The existence of OSCs in mammalian ovaries has been documented by other means of purification as well. For example, OG2 transgenic mice [also referred to as Tg(Pou5f1-EGFP)2Mn or ΔPE-Oct4-Gfp transgenic mice], with expression of enhanced green fluorescent protein (EGFP) driven by a modified POU domain class 5 transcription factor 1 (Pou5f1; also referred to as octamer-binding transcription factor-4 or Oct-4) gene promoter fragment to convey germline specificity, have been used to obtain OSCs from postnatal ovarian tissue13. The cells display the hallmark features of OSCs isolated by Ddx4 antibody-based sorting, including a germline gene expression profile, mitotic capacity and growth in vitro, and the ability of OSC-derived oocytes to interact with endogenous ovarian granulosa cells to form follicles13. Another strategy involves immunological sorting with antibodies directed against an undisputed transmembrane-spanning protein in germ cells, interferon-induced transmembrane protein 3 (Ifitm3; also referred to as Fragilis)42, 43. Expression of Ifitm3 is used frequently as an endpoint in studies of primitive germ cells, and Ifitm3 antibody-based sorting of embryonic primordial germ cells is well documented44, 45. Expression of Ifitm3 at both the mRNA and protein levels can be detected in OSCs of multiple species12, 16, 21, 30, and antibodies against an extracellular domain of Ifitm3 have been used to sort OSCs from mouse14, 46 and rat21 ovaries. These cells, like those sorted using Ddx4 antibodies, generate functional eggs and offspring following transplantation21.
Despite these many advances in the study of OSCs and postnatal oocyte formation over the past decade or so, the physiological significance of de-novo oogenesis in the ovaries of adult female mammals remains unknown. One approach for determining the in-vivo function of a specific cell type or process is suicide gene technology. Suicide genes, such as herpes simplex virus thymidine kinase (HSVtk), encode enzymes that are inert in mammalian cells. However, in the presence of target pro-drugs, these enzymes generate cytotoxic metabolites that kill the suicide gene-expressing cells. As examples, suicide gene transgenic mice have been used to define the function of pituitary somatotropes47, neural progenitors48 and bone marrow osteoblasts49 following selective ablation in vivo. A second widely-accepted approach for determination of cell function in vivo is genetic lineage tracing, which uses a cell type-specific promoter to permanently ‘mark’, at both the genomic (recombination) and phenotypic (reporter gene expression) levels, a desired cell in the body and then map its fate. Past studies of hematopoiesis50, neurogenesis51, intestinal crypt cells52, muscle53, hair follicles54, and female GSCs in the teleost medaka4 provide examples of the use of this technology. Herein we sought to combine these two powerful genetic approaches to rigorously explore the contribution, if any, of postnatal oogenesis to adult ovarian function and female fertility in mammals.
Results
Transplanted OSCs generate offspring
Intragonadal transplantation of SSCs expressing a marker gene that can be traced through spermatogenesis to progeny by genotype analysis, a technique first developed over 20 years ago55, 56, remains to this day the undisputed gold standard for establishment of male GSC identity and function57. In 2009, the generation of offspring derived from GFP-expressing OSCs transplanted into the ovaries of wild type female mice was reported12. This outcome, which achieved the exact same bar for functional identity testing of SSCs used without debate for decades55,56,57, has not only been confirmed in mice and extended to rats by this same group15, 21, 34, 35, but has also been verified by others25. As a preface to embarking on studies of the physiological relevance, if any, of OSCs and oogenesis to adult female reproductive function, we independently assessed this experimental paradigm once again. We used young adult OG2 transgenic female mice, which are well characterized and widely utilized in studies of germ cell development due to the restricted expression of EGFP in the germline58,59,60,61, for OSC isolation and intraovarian transplantation into ovaries of young adult wild type recipients16. Past studies have already demonstrated that OG2 transgenic OSCs differentiate into EGFP-positive oocytes that interact with granulosa cells to form follicles both in vitro 13 and in vivo 62. In natural mating trials, 4 transplanted wild type females mated with wild type males delivered a total of 38 offspring over the duration of our study period, 6 of which (15.8%) carried the OG2 transgene and thus were derived from the transplanted OSCs (Supplementary Fig. S1). Of the 4 transplanted females, 3 delivered at least one transgenic pup over the course of the mating trial. Although repeated confirmation of the reproducibility of this outcome is important, intragondal GSC transplantation-based approaches – whether conducted in males55,56,57 or females15, 21, 25, 34, 35 (Supplementary Fig. S1), all suffer from the same major interpretational limitation: the data obtained do not provide insight into the potential contribution of GSCs to adult gonadal function and fertility under normal physiological conditions.
Targeted ablation of differentiating germ cells: validation and controls
Since meiosis is a cellular differentiation process unique to the germline, we next designed a suicide gene-based targeting strategy in mice using a well-characterized 1.4-kb fragment of the promoter of stimulated by retinoic acid gene 8 (Stra8), a germ cell-specific gene activated during meiotic entry in both male and female mice63,64,65,66,67. Our selection of this specific region of the Stra8 promoter offers not only germ cell expression specificity in transgenic animals18, 68, but also the advantage of a brief and defined window of activation during the early meiotic commitment phase of GSC differentiation18, 63,64,65,66,67,68. We considered targeting OSCs directly; however, the lack of a candidate gene with restricted expression in OSCs and not other stem cells or more differentiated germ cells precluded this. This strategy would also not permit phenotype-reversibility studies following suicide gene pro-drug exposure and removal since the originating stem cells would be ablated, and thus unavailable to potentially restore the oocyte-generating (oogenic) pipeline once pro-drug treatment was ceased. Although there are several genes that show restricted expression in oocytes69, targeted ablation of these terminal cells in the female germ cell differentiation program would obscure data interpretation when changes in oocyte numbers represent the readout for de-novo oogenesis. Use of this well-characterized Stra8 promoter fragment to restrict, in pStra8-HSVtk transgenic mice, the cytotoxic actions of HSVtk pro-drug exposure to only early differentiating germ cells formed from OSCs prior to oocyte generation, without targeting OSCs or oocytes directly, would circumvent these technical and interpretational limitations. This would therefore enable us to clearly assess the in-vivo significance, if any, of active oogenesis to adult ovarian function.
Two Stra8 promoter-driven transgene constructs were prepared: one to drive expression of HSVtk (pStra8-HSVtk) (Supplementary Fig. S2), and another to drive expression of GFP (pStra8-Gfp) for use as a control18. To initially test if reversible disruption of differentiation impairs the ability of OSCs to generate oocytes (Fig. 1a), OSCs were purified from ovaries of 2-month-old female mice and established in culture16, 17. Once OSC lines with stable expression of pStra8-Gfp or pStra8-HSVtk were obtained following transfection and G418 selection, the cells were treated with vehicle or the HSVtk pro-drug, ganciclovir (GCV), for 4 days and then split at low density to assess spontaneous formation of in vitro-derived (IVD)-oocytes16, 17, 19. Because IVD-oocytes are poduced in pure germ cell cultures lacking any type of naturally occurring somatic cells (viz., granulosa cells) that are crucial for orchestrating key stages of meiotic arrest required for endogenous oocytes to successfully complete normal maturation, IVD-oocytes are not functional in the sense of fertilization or developmental competency. Nonetheless, this in-vitro culture approach provides a quick, inexpensive and reliable bioassay to study OSC differentiation under experimentally defined conditions16, 17, 19, 33, 35. In vehicle-treated pStra8-HSVtk OSC cultures, the rate of IVD-oocyte formation was comparable to that observed in pStra8-Gfp OSCs cultured with vehicle (Fig. 1b). In cultures of pStra8-Gfp OSCs, used as a negative control to rule out non-specific actions of GCV, IVD-oocyte formation was unaffected by GCV treatment; however, parallel treatment of pStra8-HSVtk OSCs with GCV caused a significant attenuation of IVD-oocyte formation (Fig. 1b). This effect was reversible in that the number of IVD-oocytes produced in pStra8-HSVtk OSC cultures returned to control levels after GCV was removed (Fig. 1b). Endogenous Stra8 expression paralleled the pattern of IVD-oocyte formation in response to GCV exposure and removal (Fig. 1c). These results provided a strong impetus for us to then generate the pStra8-HSVtk transgenic mouse line for evaluation of adult oogenesis in vivo.
Temporal and targeted disruption of OSC differentiation leads to reversible oogenic failure in vitro. (a) Schematic depiction of the inducible HSVtk suicide gene approach for targeted ablation of female germ cells committing to meiosis, and the expected outcome of GCV exposure and removal on oogenesis. (b) Number of IVD-oocytes formed by OSCs expressing pStra8-Gfp or pStra8-HSVtk 48 h after passage and seeding 2.5 × 104 cells per well in 24-well culture plates in the absence (PBS, vehicle) or presence of GCV (2 μM); the post-GCV group depicts pStra8-HSVtk–expressing OSCs cultured with GCV, washed and re-seeded as described above in PBS for assessment of oogenesis 48 h later (mean ± s.e.m., n = 3 independent cultures; *P < 0.05). (c) Changes in endogenous Stra8 expression in OSC cultures described in panel b (mean ± s.e.m., n = 3 independent cultures; *P < 0.05). (d) Representative analysis of OSCs, GFP-expressing ovarian cells, oocytes, ovaries, testes, or adult tail-snip fibroblasts isolated from adult pStra8-Gfp transgenic mice for expression of Stra8-promoter driven expression of Gfp, germ cell markers (Pou5f1, Ddx4, Dppa3), endogenous Stra8, oocyte markers (Nobox, Sohlh1, Zp3; Sohlh1 is also known to be expressed in male germ cells), or β-actin. Complete (uncropped) PCR gels for each target sequence amplified are shown in Supplementary Fig. S3. (e) Representative immunofluorescence analysis of individual GFP-positive cells purified by FACS from ovaries of pStra8-Gfp mice for expression of GFP, Stra8, Nobox or Ddx4 proteins (DAPI nuclear stain, white; scale bars, 10-μm).
To eliminate potential confounding effects of random transgene integration and variability in copy number associated with pronuclear injection, we introduced our transgenes into the neutral Hprt genomic locus for generation of the mouse lines18. In mice, transgene expression controlled by this 1.4-kb fragment of the Stra8 promoter is restricted to the gonads, and more specifically to germ cells undergoing meiotic differentiation18, 68 (Supplementary Fig. S2). Although the Stra8 gene is not actively expressed in mouse oocytes64,65,66,67, 70, we felt it was still important for clear data interpretation in subsequent experiments to characterize the Stra8 promoter-positive cell fraction in ovaries of adult pStra8-Gfp mice to verify this. Gene expression analysis showed that both ecDdx4-positive cells (i.e., OSCs) and GFP-positive cells purified independently by FACS from dispersed ovaries of adult pStra8-Gfp mice expressed the pluripotent stem cell marker Pou5f1, as well as the germ cell markers Ddx4 and developmental pluripotency-associated 3 (Dppa3) (Fig. 1d and Supplementary Fig. S3). The Stra8 promoter-driven GFP-positive ovarian cells also contained endogenous Stra8 mRNA as well as Gfp mRNA – the latter indicative of Stra8 promoter activation, whereas no expression of either endogenous Stra8 or the Stra8 promoter-driven Gfp transgene was detected in freshly purified OSCs or, importantly, in isolated oocytes (Fig. 1d and Supplementary Fig. S3). These findings confirmed that the 1.4-kb Stra8 promoter fragment used does not direct transgene expression in oocytes, consistent with prior reports that Stra8 is shut off once oocytes are formed64,65,66,67, 70.
In further support of this, pStra8-Gfp–positive ovarian cells did not express any markers specific for primordial or early growing immature oocytes (newborn ovary homeobox or Nobox; spermatogenesis and oogenesis helix-loop-helix factor 1 or Sohlh1; zona pellucida glycoprotein 3 or Zp3)69, 71,72,73, whereas expression of all three of these genes was readily detected in isolated oocytes and in whole ovaries containing oocytes (Fig. 1d and Supplementary Fig. S3). As an additional confirmation that the 1.4-kb Stra8 promoter fragment used for transgenic mouse generation is neither active in oocytes nor drives transgene expression in oocytes in vivo, single-cell immunofluorescence analysis of GFP-expressing cells freshly sorted from ovaries of adult pStra8-Gfp mice demonstrated the presence of Ddx4 and endogenous Stra8 proteins but an absence of the well-characterized primordial oocyte protein, Nobox (Fig. 1e). These experiments, taken collectively, documented the fidelity of targeting female germ cells (Pou5f1-, Dppa3- and Ddx4-positive) that are committing to meiosis (Stra8-positive; promoter activity and endogenous gene), but have not yet completed differentiation into newly formed oocytes (Nobox-, Sohlh1- and Zp3-negative), by using this specific Stra8 promoter fragment as a driver for in-vivo transgene expression in mice.
As a final control for the specificity of suicide gene targeting prior to embarking on studies of adult ovaries, we tested our system in adult pStra8-HSVtk male mice since it is well documented that SSCs support spermatogenesis through Stra8-mediated meiotic activation66, 67. Using age-matched pStra8-Gfp male mice treated in parallel to monitor potential toxicity of high doses of GCV to spermatogenesis in the absence of HSVtk expression74, we determined that administration of GCV at 1 mg kg−1 each day for 28 days decreased Stra8 expression in testes of pStra8-HSVtk male mice without affecting Stra8 expression in pStra8-Gfp control males (Fig. 2a). Immunohistochemical and histological evaluations revealed that testes from GCV-treated pStra8-HSVtk male mice showed a significant reduction in the percentage of Stra8-positive seminiferous tubules (Fig. 2b,c) along with decreased cellularity and disrupted spermatogenesis (Fig. 2c). However, these effects were not observed in testes of pStra8-Gfp male mice exposed to GCV in parallel (Fig. 2b,c). Over a 21-day recovery period following cessation of GCV treatment, pStra8-HSVtk male mice regenerated Stra8-expressing germ cells (Fig. 2b,c) to support a resumption of spermatogenesis and a return to normal testicular morphology (Fig. 2c). These findings showed that GSC progeny could be effectively targeted for ablation in a temporally controlled and reversible manner in vivo using this pStra8-HSVtk suicide gene-based approach, and that the GSCs themselves remain competent to continue support of new gamete formation after the cessation of pro-drug treatment.
Reversible spermatogenic failure in adult male pStra8-HSVtk mice following GCV exposure and removal in vivo. (a,b) Stra8 mRNA levels (a) and percentage of Stra8-immunopositive seminiferous tubules (b) in testes of adult pStra8-Gfp or pStra8-HSVtk mice following 28 days of vehicle (PBS) or GCV (1 mg kg−1) exposure, or 21 days after cessation of GCV treatment (Post-GCV). Data represent the mean ± s.e.m. (n = 3 mice per group; *P < 0.05). (c,d) Representative Stra8 protein expression in (c; brown, against a blue hematoxylin counterstain; left panels, ×10 with asterisks marking Stra8-immunopositive tubules; right panels, ×40), and histological appearance of (d; left panels, ×4; right panels, ×40), testes of adult pStra8-Gfp mice following 28 days of GCV exposure (+GCV, 1 mg kg−1), or in testes of adult pStra8-HSVtk mice following 28 days GCV exposure (+GCV) or 21 days after cessation of GCV treatment (Post-GCV). Scale bars, 50-μm.
Oocyte regeneration in ovaries of adult suicide gene transgenic mice
With these controls in place, we next tested if the reversible gametogenic failure observed in pStra8-HSVtk male mice following GCV exposure and removal (Fig. 2) also occurs in adult pStra8-HSVtk female mice treated in a similar manner. As a baseline for study and subsequent data interpretation, oocyte-containing follicle numbers were determined to be comparable in 1.5-month-old (day 48 postpartum) wild type and pStra8-Gfp female mice, and treatment of females of either control genotype with GCV for 21 days had no effect on ovarian Stra8 expression or numbers of existing oocytes (Supplementary Fig. S4). Follicle numbers in 1.5-month-old pStra8-HSVtk female mice exposed to vehicle (Fig. 3a) were likewise comparable to those of wild type and pStra8-Gfp control females (Supplementary Fig. S4). These data collectively confirmed that the existing oocyte reserve is unaffected by either pro-drug administration in the absence of HSVtk expression or, conversely, HSVtk expression in the absence of pro-drug administration. Following daily administration of GCV for 21 days to 1.5-month-old pStra8-HSVtk mice, the ovaries appeared histologically normal (Supplementary Fig. S5), and we observed no significant changes in the number of degenerative (atretic) oocytes during the entire GCV treatment course (Fig. 3b). This was important since it documented a complete absence of any ‘off-target’ oocyte death in response to GCV exposure in this pStra8-HSVtk transgenic model.
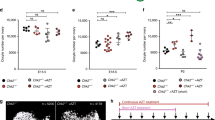
Dynamics of oocyte reserve depletion and regeneration in adult female pStra8-HSVtk mice following GCV exposure and removal in vivo. (a) Number of primordial oocyte-containing follicles in ovaries of young adult pStra8-HSVtk mice following 21 days of treatment with vehicle (PBS) or GCV (10 mg kg−1; completed on postpartum day 69), or 21 days after cessation of GCV treatment (Post-GCV, completed on postpartum day 90). Data represent the mean ± s.e.m. (n = 4–6 mice per group; *P < 0.05). (b) Number of atretic (dying, dead) oocytes in ovaries of young adult pStra8-HSVtk mice treated with vehicle (PBS) or GCV (10 mg kg−1) for up to 21 days (mean ± s.e.m., n = 4–6 mice per group). (c) Stra8 mRNA levels (normalized against β-actin mRNA levels) in ovaries contralateral to those used to derive the oocyte counts shown in panel a (mean ± s.e.m., n = 4–6 mice per group; *P < 0.05). (d) Yield of FACS-purified OSCs (percent of total viable cells sorted) from ovaries of young adult pStra8-HSVtk mice following 21 days of treatment with vehicle (PBS) or GCV (10 mg kg−1). Data represent the mean ± s.e.m., n = 3 mice per group. (e) Sycp3 expression in ovaries of young adult pStra8-HSVtk mice 21 days after ceasing treatment with vehicle (PBS) or GCV (10 mg kg−1). Data represent the mean ± s.e.m., n = 3–4 mice per group; *P < 0.05. (f) Number of Ddx4-positive germ cells with punctate nuclear localization of Sycp3 protein in ovaries contralateral to those used for panel e (mean ± s.e.m., n = 3 mice per group; *P < 0.05). (g) Immunofluorescence images of Ddx4-positive germ cells (blue) co-expressing Sycp3 (red) and Ser139-phospho-H2afx (green) in ovaries of young adult pStra8-HSVtk mice 21 days after ceasing a 3-week course of GCV treatment (10 mg kg−1). DAPI nuclear stain, white; scale bars, 10-μm.
However, ovarian Stra8 expression (Fig. 3c) and primordial oocyte-containing follicle numbers (Fig. 3a) were significantly lower in 1.5-month-old pStra8-HSVtk mice exposed to GCV for 21 days compared to vehicle injected pStra8-HSVtk controls. Quantitative analysis indicated that ovaries of GCV-treated pStra8-HSVtk mice contained almost 1,600 fewer primordial oocytes compared to age-matched pStra8-HSVtk females treated with vehicle in parallel (Fig. 3a), and this was not a result of existing oocyte depletion to due increased oocyte death (Fig. 3b). To test if this oogenic failure phenotype was reversible, 1.5-month-old pStra8-HSVtk female mice were treated with GCV for 21 days (starting on postpartum day 48) and then maintained for 21 additional days after ceasing GCV exposure (post-GCV recovery phase ending on postpartum day 90). A spontaneous return of ovarian Stra8 expression (Fig. 3c) and complete regeneration of the primordial oocyte population (Fig. 3a) occurred over this 21-day recovery period. Since OSCs freshly isolated from adult ovaries do not express Stra8 (Fig. 1d and Supplementary Fig. S3; see also ref. 18), these cells should be unaffected by GCV exposure. This was verified by our findings of a comparable yield of OSCs from ovaries of 1.5-month-old pStra8-HSVtk female mice treated for 21 days with vehicle or GCV (Fig. 3d). Once re-established, the regenerated oocyte pool in pStra8-HSVtk females remained stable such that no differences were observed in numbers of primordial or growing follicles between vehicle- and GCV-treated mice 3 months after cessation of GCV exposure (Supplementary Fig. S6a). In addition, long-term fertility and fecundity parameters were also comparable in pStra8-HSVtk female mice treated with vehicle versus GCV (Supplementary Fig. S6b).
If OSC differentiation is involved in the burst of de-novo oogenesis detected in adult ovaries during the post-GCV oogenic recovery period (Fig. 3a), evidence of active germ cell meiosis should be apparent. Therefore, we next evaluated ovaries of pStra8-HSVtk mice for expression of synaptonemal complex protein 3 (Sycp3), which is widely known to mark germ cells entering prophase-I of meiosis75. During the post-GCV recovery phase, we observed a significant increase in both ovarian Sycp3 expression (Fig. 3e) and numbers of Ddx4-positive germ cells exhibiting a pattern of punctate nuclear Sycp3 protein expression (Fig. 3f,g). These Ddx4-Sycp3 dual-positive ovarian cells also expressed Ser139-phosphorylated H2A histone family member X (P-H2afx) (Fig. 3g), which is known to localize to DNA double-strand breaks in germ cells during the early stages of meiotic prophase76. This evidence of active meiotic differentiation of germ cells, coupled with the reversible oogenic failure phenotype observed in response to specific targeting of germ cells activating Stra8 expression in pStra8-HSVtk transgenic females following GCV exposure and removal (Fig. 3a), collectively support that an important role exists for de-novo oogenesis in maintenance of the adult oocyte reserve.
Oocytes formed during adult life generate offspring
To next assess if oocytes generated during adulthood contribute directly to fertility, we replaced the Gfp coding sequence in our pStra8-Gfp construct with reverse tetracycline-controlled transactivator (rtTA) and generated a knock-in transgenic pStra8-rtTA mouse line. We then introduced two additional alleles into pStra8-rtTA mice: 1) a tetracycline responsive element (TRE)-driven Cre recombinase (TRE-Cre) construct, and 2) a Rosa26-Stop-Yfp reporter construct containing a floxed phosphoglycerate kinase 1 (Pgk) promoter-driven neomycin phosphotransferase (Npt) cassette that prevents Rosa26-driven transcription of the downstream Yfp coding sequence in the absence of Cre recombinase. This approach generated a fluorescent reporter mouse line (pStra8-R26R), in which activation of the Stra8 promoter drives expression of rtTA, leading to Cre recombinase activation and, subsequently, Rosa26-driven Yfp expression through excision of the floxed Pgk-Npt (Stop) sequence only in the presence of doxycycline (Dox). As a result, any germ cells that activate meiosis through Stra8 during a window of Dox exposure will become permanently ‘marked’, thus enabling us to trace the fate of these cells in vivo under normal physiological conditions (Fig. 4a).
Genetic fate mapping strategy and its application to tracing SSC progeny after meiotic differentiation. (a) Schematic depiction of the genetic strategy used to permanently ‘mark’ GSCs committing to meiosis in adult gonads in an inducible manner. (b) Absence of YFP expression in testes of control (R26R or ‘promoterless’ TRE-Cre;Rosa26-Stop-Yfp) transgenic males induced with Dox (1 mg kg−1) for 28 days (DAPI nuclear stain, white). (c) Detection of YFP expression (green) in spermatogonia lining the inner surface of the basement membrane of the seminiferous tubules in pStra8-R26R male mice after 28 days of induction with Dox (1 mg kg−1), and progression of ‘marked’ germ cells to spermatids 3 weeks later (DAPI nuclear stain, white). (d) Genotype analysis of first-generation offspring sired by pStra8-R26R males induced with Dox (1 mg kg−1) for 28 days before housing with wild type females, showing the presence of babies derived from fertilization of eggs by both non-recombined (Stop cassette intact; offspring 1–3) and recombined (Stop cassette excised; offspring 4–6) spermatozoa. Complete (uncropped) PCR gels for each target sequence amplified are shown in Supplementary Fig. S7.
As a positive control, we first performed lineage marking of Stra8-expressing cells in pStra8-R26R male mice following Dox treatment. We observed YFP expression in germ cells along the basement membrane of the seminiferous tubules in the testes (Fig. 4b,c), where endogenous Stra8-expressing cells are known to be located (Fig. 2c). Three weeks after a single Dox induction, differentiated spermatids within the seminiferous tubules were found to be YFP-positive (Fig. 4c). In mating trials with wild type female mice, males induced with Dox sired pups carrying the recombined Rosa26-Yfp allele (Fig. 4d and Supplementary Fig. S7), confirming utility of this system to fate-map germ cells undergoing meiotic differentiation in vivo. In turn, as negative controls we did not observe YFP expression in ovaries of either pStra8-R26R female mice treated with vehicle (n = 5 mice) or R26R (‘promoterless’ TRE-Cre;Rosa26-Yfp) female mice induced with Dox for 21 days (n = 5 mice) (Supplementary Fig. S8).
However, we detected YFP-positive oocytes, enclosed within follicles and co-expressing the immature oocyte transcription factor, Nobox, in ovaries of adult pStra8-R26R female mice treated with Dox for 21 days (Fig. 5a). Marked oocytes formed during Dox induction were often located adjacent to unlabeled (pre-existing) oocytes, which were also positive for Nobox and contained within follicles (Fig. 5a). To test if marked oocytes formed during adulthood are fully functional, female pStra8-R26R mice were induced for 21 days with Dox and then housed with wild type males. Consistent with formation of a mosaic oocyte pool following induction of the reporter (Fig. 5a), Dox-induced females gave birth to mosaic litters composed of offspring lacking (YFP-negative), and offspring exhibiting (YFP-positive), recombination at the Rosa26-Stop-Yfp locus (Fig. 5b and Supplementary Fig. 9a). These data essentially mirrored those obtained from parallel studies of pStra8-R26R male mice used as a positive control (Fig. 4d and Supplementary Fig. S7). Live-imaging revealed widespread YFP expression in recombined offspring, which grew to adulthood without issue (Fig. 5c). Mating of first-generation (F1) female offspring carrying the recombined Rosa26-Yfp allele with wild type males confirmed germline transmission of the recombined reporter gene to F2 offspring (Fig. 5d and Supplementary Fig. S9b), which by live imaging also exhibited widespread YFP expression (Fig. 5e). These results demonstrated that oocytes newly formed in adult ovaries in vivo contribute directly to natural female fertility under physiological conditions.
Genetic fate mapping of oocytes generated during adulthood to the generation of viable offspring. (a) Representative expression analysis of YFP (green; recombined and activated reporter), Ddx4 (red; germ cell marker) and Nobox (purple; oocyte marker) proteins in ovaries of young adult pStra8-R26R mice after 21 days of induction with Dox (2 mg ml−1); DAPI nuclear stain in the merge panel, white. Note the proximity of oocytes newly formed during the Dox induction phase (YFP+/Ddx4+/Nobox+, arrows) adjacent to oocytes pre-existing before the start of Dox induction (YFP–/Ddx4+/Nobox+, arrowheads). (b) Genotype analysis of first-generation (F1) offspring delivered by pStra8-R26R females induced with Dox (10 mg kg−1) for 21 days before housing with wild type males, confirming the presence of babies derived from natural fertilization of both pre-existing oocytes (non-recombined, Stop cassette intact; offspring 1–3) and oocytes formed during the induction period (recombined, Stop cassette excised; offspring 4–6) in adult ovaries. Complete (uncropped) PCR gels for each target sequence amplified are shown in Supplementary Fig. S9a. (c) Live fluorescence image of a representative F1-generation mouse identified by genotyping (b) as being derived from fertilization of an oocyte formed during adulthood in a Dox-induced pStra8-R26R female. (d,e) Natural mating of recombined F1 generation female offspring with wild type males reveals germline transmission of the recombined Rosa26-Yfp allele to F2 generation offspring (d; complete (uncropped) PCR gels for each target sequence amplified are shown in Supplementary Fig. S9b), which was confirmed by live fluorescence imaging of ubiquitous YFP expression (e; a representative F2-generation pup of each genotype is shown for comparative purposes).
OSC function in adult ovaries declines with age
Past studies with mice have shown that the primordial oocyte pool, once established during the early juvenile period, remains remarkably stable during the first 3 months of life despite a high daily rate of exit (growth activation) and atresia, but then shows significant depletion as the females reach 5–6 months of age6, 10, 11. To determine if progressive loss of the oocyte reserve after 3 months of age is due, at least in part, to a declining capacity for oocyte renewal, we compared the magnitude of the post-GCV oogenic recovery response in pStra8-HSVtk female mice during early, mid- and late reproductive ages (Fig. 6a,b). After 3 weeks of GCV exposure from postpartum days 134 to 155 (mid-reproductive life), primordial oocyte numbers in pStra8-HSVtk females was significantly lower compared to those of vehicle-injected controls (Fig. 6b), in a manner that paralleled the response of pStra8-HSVtk females when GCV was administered during early reproductive life from postpartum days 48 to 69 (Fig. 6a). However, compared to the robust post-GCV oogenic response (1,582 new oocytes) observed in females from postpartum days 69 to 90 (early reproductive life; Fig. 6a), the post-GCV oogenic response spanning postpartum days 155 to 176 (mid-reproductive life) was severely blunted (only 607 new oocytes). In fact, in pStra8-HSVtk females during mid-reproductive life, the size of the primordial oocyte pool after 21 days of post-GCV recovery was not significantly different from the size of the pool when GCV treatment was terminated (Fig. 6b). By 10 months of age (late reproductive life), the primordial oocyte pool in vehicle-treated pStra8-HSVtk females was severely diminished due to advanced maternal age, and 21 days of GCV exposure no longer elicited any change in the size of the oocyte reserve (Fig. 6c). This outcome provided not only evidence of an absence of active oogenesis by this time in life but also additional evidence of a complete lack of off-target killing of oocytes by GCV (see also Fig. 3b). Interestingly, however, GCV treatment still elicited a decrease in ovarian Stra8 expression in females at 10 months of age (Fig. 6c). We interpreted this to indicate that OSCs were still present and attempting Stra8-mediated differentiation even at this advanced maternal age, but the selective ablation of Stra8-expressing cells by GCV treatment effectively ‘removed’ the levels of Stra8 mRNA transcript normally contributed by these cells to the overall pool of ovarian mRNA analyzed.
Aging female mice show a progressively diminished potential for de-novo oogenesis. (a,b) Comparison of changes in the primordial follicle reserve in ovaries of pStra8-HSVtk mice in which GCV treatment (10 mg kg−1) was initiated on postpartum day 48 with the post-GCV regenerative phase occurring between 2–3 months of age (a; early reproductive life, Early), or on postpartum day 134 with the post-GCV regenerative phase occurring between 5–6 months of age (b; mid-reproductive life, Mid). Data represent the mean ± s.e.m., n = 3–6 mice per group; *P < 0.05; n.s.d., not significantly different. (c) Lack of effect of 21 days of GCV treatment (10 mg kg−1) on primordial follicle numbers in pStra8-HSVtk mice treated between 10–11 months of age (late reproductive life, Late); note that while oocyte numbers in pStra8-HSVtk females at advanced (late) reproductive age were unaffected by 21 days of GCV treatment, endogenous ovarian Stra8 expression remained sensitive to GCV exposure in these mice (mean ± s.e.m., n = 4–5 mice per group; *P < 0.05).
Consistent with this, and with recent observations from studies of mouse and human ovarian tissues77, 78, we found that the yield of OSCs from adult ovaries increased slightly with advancing maternal age (Fig. 7a). This observation, coupled with the increased levels of ovarian expression of the primitive germ cell marker, PR domain containing 1 with ZNF domain (Prdm1), in 10–11-month-old females (Fig. 7b), collectively indicated that the loss of oogenic potential with age (Fig. 6a–c) is probably due more to impaired OSC function rather than an aging-dependent disappearance of OSCs from the gonads. In ovaries of pStra8-Gfp mice at 10–11 months of age, we also detected an age-related increase in Stra8 promoter-driven Gfp expression (Fig. 7c), increased numbers of GFP-expressing cells (Fig. 7d), and increased expression levels of the endogenous Stra8 gene (Fig. 7e). However, expression of Sycp3 dropped precipitously in ovaries at late reproductive ages (Fig. 7f), along with an expected decline in expression of the immature oocyte marker, Nobox (Fig. 7g). Thus, OSCs are still present, and likely attempting meiotic differentiation, in ovaries at advanced reproductive ages; however, meiotic progression after Stra8 activation apparently then fails, perhaps due to waning Sycp3 availability.
Evidence that the diminished oogenic potential in reproductively aged females is not due to an absence of OSCs in the ovaries. (a) Yield of FACS-purified OSCs from ovaries of female mice at early, mid- and late reproductive ages (mean ± s.e.m., n = 3–4 mice per group). (b,c) Quantitative analysis of Prdm1 and Stra8 promoter-driven Gfp mRNA levels in ovaries of pStra8-Gfp mice at early and late reproductive ages (mean ± s.e.m., n = 6 mice per group; *P < 0.05). (d) Yield of Stra8 promoter-driven GFP-positive cells from ovaries of pStra8-Gfp mice at early and late reproductive ages (mean ± s.e.m., n = 6–8 mice per group with 2 mice for each sample and 3–4 samples at each age). (e–g) Quantitative analysis of endogenous Stra8, Sycp3 and Nobox mRNA levels in ovaries of pStra8-Gfp mice at early and late reproductive ages (mean ± s.e.m., n = 6 mice per group; *P < 0.05).
Discussion
Through use of two widely accepted genetic approaches for the in-vivo study of adult stem cell function, here we show that adult mouse ovaries actively generate new oocytes that are subsequently used for ovulation, fertilization and the generation of viable offspring. As important as these observations are, the current lack of a gene promoter that is exclusively expressed in OSCs, but not in differentiating premeiotic germ cells or oocytes, does not enable unequivocal identification of OSCs as the cells responsible for supporting postnatal oogenesis. However, the independent verification of the presence of OSCs in the ovaries of a growing number of mammalian species10, 12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35, 46, 78, the ability of OSCs to generate new oocytes and functionally competent eggs in adult females12, 15, 16, 20, 21, 25, 32, 34, 35 (Supplementary Fig. S1), and experimental evidence that resident multi-potent stem cells in adult ovaries are unable to generate oocytes79, collectively support that OSCs are the most logical and likely source of new oocytes formed during adulthood. With that said, a few recent studies have concluded based on negative data that OSCs do not exist in postnatal mouse or human ovaries36, 37, 80. However, a detailed re-assessment of the first of these studies using the experimental mouse models and protocols reported in the original paper36 uncovered several design weaknesses and a lack of important controls in the approaches employed to reach the conclusion that OSCs do not exist; when these were accounted for and corrected, OSCs were purified without issue26. These latter observations were verified and extended by recent work from a different laboratory, which also showed that OSCs could be purified by FACS from ovaries of germline-specific fluorescent gene reporter mice; further, the purified OSCs gave rise to offspring following intraovarian transplantation into wild type female recipients34. Design concerns with other studies questioning the existence of OSCs and postnatal oogenesis37, 80 have also been identified81 and addressed experimentally30. Notably, two very recent studies have provided the first insights into the post-transcriptional and epigenetic determinants of unipotency and other germline properties in OSCs, as well as the identity of factors that maintain OSCs in an undifferentiated state82, 83. Additionally, by tracing individual transplanted OCSs expressing EGFP, Wu and colleagues also recently mapped the in-vivo differentiation of these cells into new oocytes following intraovarian delivery, as well as the high similarity in gene expression networks in follicles containing oocytes formed from transplanted OSCs versus endogenous (pre-existing) wild type follicles35.
In considering our first approach, clear interpretation of data derived from use of suicide gene technology is dependent on several variables. First, specificity of the gene promoter used to target the suicide gene to a desired cell type is critical. In full agreement with a large volume of existing work reporting on the specificity of Stra8 expression in pre-meiotic germ cells in mice18, 63,64,65,66,67,68, 70, our in-depth assessment of the GFP-positive cell fraction isolated from ovaries of pStra8-Gfp transgenic mice (Fig. 1d,e) demonstrated that the 1.4-kb Stra8 promoter fragment used for suicide gene targeting is indeed restricted to germ cells (positive for Pou5f1, Dppa3 and Ddx4) that are committing to meiosis (positive for Stra8, at the level of both promoter activity and endogenous gene expression) but have not yet differentiated into new oocytes (negative for Nobox, Sohlh1 and Zp3). In addition, the reversible spermatogenic failure observed following GCV treatment and removal in pStra8-HSVtk male mice (Fig. 2) – a model in which GSC support of adult gametogenesis is universally accepted1, 2, further indicates that use of this specific Stra8 promoter fragment to drive GCV-sensitive HSVtk expression meets the requirements for targeting early germ cells initiating meiotic commitment. Second, the endpoint examined must be unaffected by the suicide gene in the absence of its pro-drug and by pro-drug exposure in the absence of its suicide gene. In-depth assessment of oocyte dynamics and endogenous Stra8 expression in wild type, pStra8-Gfp and pStra8-HSVtk mice exposed to vehicle or GCV (Fig. 5a and Supplementary Fig. S8) satisfied this requirement. Finally, the readout must reflect the endpoint activity of the cells targeted for ablation. In our case, alterations in OSC daughter cell differentiation into oocytes is the most biologically plausible explanation for the loss and, without question, the subsequent spontaneous regeneration of the primordial oocyte-containing follicle pool in pStra8-HSVtk females after GCV exposure and removal, respectively (Fig. 3a). Further to this point, very recent studies of adult female rats exposed to agents known to be highly toxic to oocytes showed that the initial reduction in primordial follicle numbers observed shortly after drug treatment was followed by a robust period of oogenic recovery, leading to regeneration of the primordial follicle pool84.
It is also worth noting that a ‘bystander effect’ caused by release of cytotoxic metabolites from suicide gene-expressing cells, which may then kill surrounding wild type (off-target) cells, has been raised as a confounding issue in the use of suicide gene systems85, 86. However, bystander killing effects have not been observed in mouse models designed to characterize the role of a specific cell type in vivo 47, 48, 87, 88. Further, a clear absence of any change in the incidence of oocyte death in pStra8-HSVtk females during the entire 21-day course of GCV exposure (Fig. 3b) establishes two important principles: 1) the smaller oocyte pool detected in pStra8-HSVtk females after 21 days of GCV treatment does not result from non-specific or off-target killing of existing oocytes during GCV exposure, a point further reinforced by the complete lack of effect of GCV treatment on the oocyte pool in females at 10 months of age (Fig. 6c); and, 2) the Stra8 promoter fragment used to produce our transgenic lines is not active in existing oocytes. The latter point is also fully consistent with an absence of either Stra8 promoter driven Gfp expression or endogenous Stra8 mRNA in oocytes of pStra8-Gfp females (Fig. 1d), as well as an absence of any well-accepted oocyte markers in the GFP-expressing cell fraction purified from ovaries of pStra8-Gfp females by FACS (Fig. 1d). Even in the unlikely case that undetected bystander killing is occurring in this model and the existing oocyte reserve is somehow negatively affected by GCV in a manner we cannot discern, the spontaneous and complete regeneration of primordial oocyte numbers back to pre-treatment levels after ceasing GCV exposure (Fig. 3a) provides unequivocal evidence for the occurrence of active oogenesis and folliculogenesis in adult ovaries. This conclusion agrees with recent studies of Pou5f1-MerCreMer transgenic mice crossed with R26R-enhanced yellow fluorescent protein transgenic mice to establish a tamoxifen-inducible system for labeling Pou5f1-expressing cells in postnatal ovaries89. Although use of the Pou5f1 gene promoter to drive reporter expression for fate mapping in the ovary is complicated by expression of Pou5f1 in cell types aside from OSCs, including oocytes58 and resident multi-potent stem cells79, evidence for the occurrence of germ cell proliferation, meiotic entry and de-novo replenishment of the primordial follicle pool in adult mouse ovaries was provided89. Notably, our assessment of meiotic activation and progression during the post-GCV recovery phase (Fig. 3e–g) in turn independently verifies the recent report from Gou and colleagues regarding their detection of germ cell meiotic entry in adult mouse ovaries using Pou5f1-MerCreMer transgenic mice89.
As compelling as we believe these observations are using suicide gene technology, the limitation with this approach is uncertainty over what, if anything, oocytes formed during adulthood do. By extending this work through parallel use of genetic lineage tracing from the premeiotic germ cell stage, we have shown that active Stra8-mediated germline differentiation is not only required for maintenance of oocyte numbers in female mice during early to mid-adult life (Figs. 3 and 6), but also that oocytes formed during adulthood contribute directly to the pool of eggs used for the generation of offspring in natural mating trials (Fig. 5). Such documentation of a physiological role for postnatal oogenesis in mammals provides an impetus to reconsider current thinking on many aspects of female reproductive biology, including a reassessment of underlying events responsible for ovarian failure with age. For example, in mammalian testes, SSCs are known to persist into advanced age, even after spermatogenic failure has occurred90, 91. When SSCs from aged atrophic testes are transplanted into young adult testes, the cells resume spermatogenesis90. Likewise, past studies have shown that primordial oocyte formation in aged mouse ovaries also resumes when the tissue is grafted into a young adult ovarian environment77. In considering this prior report with our findings presented herein, depletion of the oocyte pool in female mammals with age, which appears to result from a combination of oocyte loss through growth activation followed by ovulation or atresia coupled with a progressive decline in new oocyte input, might be amenable to prevention or even reversal. Another shift in thinking revolves around the fact that the postnatal oocyte reserve is not a finite entity at birth as previously believed5. This change in thinking, coupled with recent reports that a comparable population of mitotically-active germ cells not only exists in adult human ovaries16, 17, 22, 27,28,29, 31, 33 but can also differentiate into IVD-oocytes in culture and into immature oocytes in human ovarian tissue xenografts in vivo 16, 17, 22, 27, 33, provide a solid foundation on which to further explore the potential of OSC-based technologies for management of ovarian function and female infertility78, 81, 92.
Methods
Animals and treatments
Wild type C57BL/6 mice were from Charles River Laboratories, TRE-Cre (strain: Tg(tetO-cre)1Jaw/J; stock number: 006224), Rosa26-Stop-Yfp (strain: B6.129 × 1-Gt(ROSA)26Sor tm1(EYFP)Cos/J; stock number: 006148) and OG2 (strain: B6; CBA-Tg(Pou5f1-EGFP)2Mnn/J; stock number: 004654) mice were from the Jackson Laboratory, and pStra8-Gfp mice were generated as described18. Transgenic mice with HSVtk or rtTA driven by the 1.4-kb Stra8 promoter fragment were generated by replacing the GFP-coding sequence in the pStra8-Gfp construct18 with cDNA encoding GFP-fused HSVtk (provided by J. Galipeau) or encoding rtTA, and the constructs were then sent to Genoway for generation of the indicated knock-in transgenic lines18. For comparative studies, wild type and transgenic siblings from breeding colonies were used in parallel to rule out any potential effect of background strain on the outcomes. For treatments, GCV (Roche) was dissolved in sterile water at 10 mg ml−1, and then diluted in sterile 1X-concentrated PBS for daily dosing (males: 1 mg kg−1 for 28 days; females: 10 mg kg−1 for 21 days). Treatment protocols were based on prior studies of suicide gene-based ablation of somatic cells47,48,49, 87, 88, and on empirical testing using pStra8-Gfp male and female mice as negative controls for GCV dosing. Doxycycline was administered for 21 days via the drinking water at a concentration of 2 mg ml−1. Where indicated, whole body fluorescence imaging of live mice was performed using a Nikon OVA110 imaging system. All animal studies were approved by the appropriate institutional animal care and use committees at Northeastern University and Massachusetts General Hospital, and all methods used in this study were performed in accordance with all relevant institutional guidelines and regulations.
Germ cell isolation, culture and IVD-oocyte formation
For most experiments (except as indicated below), OSCs were isolated from ovaries of young adult mice (2–3 months of age) by FACS using a C-terminal DDX4-specific antibody (ab13840, Abcam). The cells were analyzed immediately or established in culture without somatic feeder cells, as described16, 17, 46, 93. Purified mouse OSCs propagated under these conditions spontaneously differentiate into IVD-oocytes for up to 72 h after passage until confluence is regained, and the number of IVD-oocytes generated by a fixed number of OSCs seeded per well remains relatively constant over successive passages16, 17, 19. Between passages 32–40, OSCs were transfected with the desired plasmids (pStra8-HSVtk or pStra8-Gfp, each containing a neomycin resistance gene) using Lipofectamine 2000 (Invitrogen) and then selected by G418 (Geneticin, Cellgro) over 2 weeks. Cells were then maintained in G418 for all experiments, and the number of IVD-oocytes generated and released into the medium after treatment with vehicle or GCV (2 μM) was then determined by direct visual counts under a microscope16, 17, 19. In other experiments, GFP-positive cells in ovaries of pStra8-Gfp transgenic female mice were quantitated and then isolated by FACS for gene expression profiling.
Intraovarian OSC transplantation
Mouse OSCs, isolated from ovaries of young adult OG2 transgenic female mice as described above, were injected directly into each ovary (~1 × 104 viable cells per injection) of four recipient wild type C57BL/6 female mice at 2 months of age, as detailed previously16. After a one-week recovery period, adult wild type males were introduced into the cages with the transplanted females for mating trials over a subsequent 4-month period. All offspring were genotyped for the absence or presence of the OG2 transgene.
Oocyte counts
Young adult mouse ovaries were fixed, serially sectioned and processed for histomorphometry-based quantification of the number of healthy or degenerative (atretic) oocyte-containing follicles at the indicated stages of development, as detailed10, 94. All samples were assessed in a completely blinded fashion, and reproducibility was independently confirmed in a blinded fashion by two other observers. In all cases, variation in counts between observers was less than 7% (Supplementary Fig. S10). As an additional verification of the counting method employed, serially sectioned mouse ovaries were processed for immunohistochemical detection of the oocyte-specific marker, Nobox (ref. 71), after which the number of Nobox-positive oocytes contained within immature follicles was quantified for each ovary. These counts, and those obtained by direct visual assessment (oocyte morphology through histology), were then compared and found to produce similar data sets (Supplementary Fig. S10).
Gene expression analysis
Total RNA was extracted using Tri-Reagent (Sigma-Aldrich) and reverse transcribed (Superscript III; Invitrogen) using oligo-dT primers. For some experiments, amplification of target gene sequences was performed by conventional PCR to assess for the absence or presence of Gfp, Pou5f1, Ddx4, Prdm1, Dppa3, Ifitm3, Stra8, Nobox, Sohlh1 or Zp3 expression, as well as of β-actin expression as a sample loading control (see Supplementary Table S1 for details). All products were sequenced to confirm identity. For quantitative analysis of mRNA levels, real-time PCR was performed using a Cepheid Smart Cycler II. For Stra8 expression (normalized against β-actin mRNA levels), primers for Stra8 (FAM-labeled D-LUXTM Pre-designed Gene Expression Assays, MLUX3312362) and β-actin (FAM-labeled certified LUXTM Primer Set for Mouse/Rat β-actin, 101M-01) were obtained from Invitrogen. For assessment of Sycp3, Prdm1, Gfp and Nobox expression (normalized against β-actin mRNA levels), SYBR-based quantitative PCR was conducted by using BioRad SsoAdvanced™ Universal SYBR® Green Supermix along with gene-specific primers (Supplementary Table S1).
Genotyping of pStra8-R26R mice
Recombination at the Rosa26-Stop-Yfp locus was confirmed by genotyping offspring for the presence of the Yfp coding sequence along with excision of the floxed Pgk-Npt (Stop) cassette using primers specific for Yfp and Npt, respectively (Supplementary Table S1). As an internal control for PCR quality, primer sets against intestinal fatty acid binding protein 2 (Fabpi) were included during PCR for detection of Npt (Fabpi short, 194 bp) and of Yfp (Fabpi long, 466 bp) (Supplementary Table S1).
Immunofluorescence (IF) and immunohistochemistry (IHC)
Freshly collected tissues were fixed in 4% paraformaldehyde, embedded in paraffin, and sectioned for analysis using primary antibodies against Stra8 (rabbit polyclonal, ab49602; Abcam), Ddx4 (rabbit polyclonal ab13840, Abcam; goat polyclonal AF2030, R&D Systems), Sycp3 (rabbit polyclonal NB300-230, Novus Biologicals), Ser139-phospho-H2afx (mouse monoclonal 05–636, Millipore) or GFP (chicken polyclonal ab13970, Abcam; rabbit polyclonal ab290, Abcam). For IF, detection was performed using donkey anti-chicken Alexa Fluor 488 (Jackson Immuno), donkey anti-goat Alexa Fluor 647 or donkey anti-rabbit Alexa Fluor 546 (Molecular Probes) as secondary antibody16. For IHC, detection was performed using biotin-conjugated anti-rabbit IgG (Santa Cruz Biotechnology) as secondary antibody for horseradish peroxidase-based DAB detection (Sigma-Aldrich). Images were captured using a Nikon E800/BioRad Radiance 2000 confocal microscope or a Nikon ECLIPSE TE2000-S microscope.
Data analysis
All experiments were independently replicated at least three times, using different mice, tissues collected from different mice, or different populations of cells for each biological replicate. These sample sizes allow for adequate power to detect potential treatment effects while also ensuring that the total number of animals used was minimized. Where possible, assignment of mice to experimental groups was made randomly. Quantitative data from replicate experiments (mean ± s.e.m.) were analyzed by one-way ANOVA followed by Student’s t-test (P < 0.05), whereas the qualitative images provided are representative of outcomes obtained across the replicate experiments.
References
Lin, H. The stem-cell niche theory: lessons from flies. Nat. Rev. Genet. 3, 931–940 (2002).
Brinster, R. L. Male germline stem cells: from mice to men. Science 316, 404–405 (2007).
Kirilly, D. & Xie, T. The Drosophila ovary: an active stem cell community. Cell Res. 17, 15–25 (2007).
Nakamura, S., Kobayashi, K., Nishimura, T., Higashijima, S. & Tanaka, M. Identification of germline stem cells in the ovary of the teleost medaka. Science 328, 1561–1563 (2010).
Zuckerman, S. The number of oocytes in the mature ovary. Rec. Prog. Horm. Res. 6, 63–108 (1951).
Faddy, M. J., Telfer, E. & Gosden, R. G. The kinetics of pre-antral follicle development in ovaries of CBA/Ca mice during the first 14 weeks of life. Cell Tiss. Kinet. 20, 551–560 (1987).
Gosden, R. G., Laing, S. C., Felicio, L. S., Nelson, J. F. & Finch, C. E. Imminent oocyte exhaustion and reduced follicular recruitment mark the transition to acyclicity in aging C57BL/6J mice. Biol. Reprod. 28, 255–260 (1983).
Richardson, S. J., Senikas, V. & Nelson, J. F. Follicular depletion during the menopausal transition: evidence for accelerated loss and ultimate exhaustion. J. Clin. Endocrinol. Metab. 65, 1231–1237 (1987).
Fujiwara, Y. et al. Isolation of a DEAD-family protein gene that encodes a murine homolog of Drosophila vasa and its specific expression in germ cell lineage. Proc. Natl. Acad. Sci. USA 91, 12258–12262 (1994).
Johnson, J., Canning, J., Kaneko, T., Pru, J. K. & Tilly, J. L. Germline stem cells and follicular renewal in the postnatal mammalian ovary. Nature 428, 145–150 (2004).
Kerr, J. B. et al. Quantification of healthy follicles in the neonatal and adult mouse ovary: evidence for maintenance of primordial follicle supply. Reproduction 132, 95–109 (2006).
Zou, K. et al. Production of offspring from a germline stem cell line derived from neonatal ovaries. Nat. Cell Biol. 11, 631–636 (2009).
Pacchiarotti, J. et al. Differentiation potential of germ line stem cells derived from the postnatal mouse ovary. Differentiation 79, 159–170 (2010).
Zou, K., Hou, L., Sun, K., Xie, W. & Wu, J. Improved efficiency of female germline stem cell purification using Fragilis-based magnetic bead sorting. Stem Cells Dev. 20, 2197–2204 (2011).
Zhang, Y. et al. Production of transgenic mice by random recombination of targeted genes in female germline stem cells. J. Mol. Cell Biol. 3, 132–141 (2011).
White, Y. A. R. et al. Oocyte formation by mitotically active germ cells purified from ovaries of reproductive-age women. Nat. Med. 18, 413–421 (2012).
Woods, D. C. & Tilly, J. L. Isolation, characterization and propagation of mitotically active germ cells from adult mouse and human ovaries. Nat. Protoc. 8, 966–988 (2013).
Imudia, A. N. et al. Comparative gene expression profiling of adult mouse ovary-derived oogonial stem cells supports a distinct cellular identity. Fertil. Steril. 100, 1451–1458 (2013).
Park, E. S., Woods, D. C. & Tilly, J. L. Bone morphogenetic protein 4 (BMP4) promotes mammalian oogonial stem cell differentiation via SMAD1/5/8 signaling. Fertil. Steril. 100, 1468–1475 (2013).
Wolff, E. F. et al. Oogonial stem cells generate mature oocytes in an autologous Rhesus macaque transplantation model. Reprod. Sci. 21(Supplement), 119A (2014).
Zhou, L. et al. Production of fat-1 transgenic rats using a post-natal female germline stem cell line. Mol. Hum. Reprod. 20, 271–281 (2014).
Dunlop, C. E., Bayne, R. A., McLaughlin, M., Telfer, E. E. & Anderson, R. A. Isolation, purification, and culture of oogonial stem cells from adult human and bovine ovarian cortex. Lancet 383, S45 (2014).
Xie, W., Wang, H. & Wu, J. Similar morphological and molecular signatures shared by female and male germline stem cells. Sci. Reports 4, 5580 (2014).
Khosravi-Farsani, S., Amidi, F., Roudkenar, M. H. & Sobhani, A. Isolation and enrichment of mouse female germline stem cells. Cell J. 16, 406–415 (2015).
Xiong, J. et al. Intraovarian transplantation of female germline stem cells rescues ovarian function in chemotherapy injured ovaries. PLoS One 10, e0139824 (2015).
Park, E. S. & Tilly, J. L. Use of DEAD-box polypeptide 4 (Ddx4) gene promoter-driven fluorescent reporter mice to identify mitotically active germ cells in postnatal mouse ovaries. Mol. Hum. Reprod. 21, 58–65 (2015).
Grieve, K. M., McLaughlin, M., Dunlop, C. E., Telfer, E. E. & Anderson, R. A. The controversial existence and functional potential of oogonial stem cells. Maturitas 83, 278–281 (2015).
Silvestris, E., D’Oronzo, S., Cafforio, P., D’Amato, G. & Loverno, G. Perspective in infertility: the ovarian stem cells. J. Ovarian Res. 8, 55 (2015).
Fakih, M. H. et al. The AUGMENTSM treatment: physician reported outcomes of the initial global patient experience. JFIV Reprod. Med. Genet. 3, 154 (2015).
Woods, D. C. & Tilly, J. L. Reply to adult human and mouse ovaries lack DDX4-expressing functional oogonial stem cells. Nat. Med. 21, 1118–1121 (2015).
Oktay, K. et al. Oogonial precursor cell derived autologous mitochondria injection (AMI) to improve outcomes in women with multiple IVF failures due to low oocyte quality: a clinical translation. Reprod. Sci. 22, 1612–1617 (2015).
Park, S., Yu, J., Marquis, K., DeCherney, A. H. & Wolff, E. F. Egg from ovarian-derived stem cells develops into embryo after intracytoplasmic sperm injection. Reprod. Sci. 23(Supplement), 264A (2016).
Ding, X. et al. Human GV oocytes generated by mitotically active germ cells obtained from follicular aspirates. Sci. Rep. 6, 28218 (2016).
Zhang, C. & Wu, J. Production of offspring from a germline stem cell line derived from prepubertal ovaries of germline reporter mice. Mol. Hum. Reprod. 22, 457–464 (2016).
Wu, C. et al. Tracing and characterizing the development of transplanted female germline stem cells in vivo. Mol. Ther. 25, 1408–1419 (2017).
Zhang, H. et al. Experimental evidence showing that no mitotically active female germline progenitors exist in postnatal mouse ovaries. Proc. Natl. Acad. Sci. USA 109, 12580–12585 (2012).
Zhang, H. et al. Adult human and mouse ovaries lack DDX4-expressing functional oogonial stem cells. Nat. Med. 21, 1116–1118 (2015).
Hernandez, S. F. et al. Characterization of extracellular DDX4- or Ddx4-positive ovarian cells. Nat. Med. 21, 1114–1116 (2015).
Toyooka, Y. et al. Expression and intracellular localization of mouse Vasa-homologue protein during germ cell development. Mech. Dev. 93, 139–149 (2000).
Carter, P., Smith, L. & Ryan, M. Identification and validation of cell surface antigens for antibody targeting in oncology. Endocr. Relat. Cancer 11, 659–687 (2004).
Richards, M., Fong, C.-Y. & Bongso, A. Comparative evaluation of different in vitro systems that stimulate germ cell differentiation in human embryonic stem cells. Fertil. Steril. 93, 986–994 (2008).
Saitou, M., Barton, S. C. & Surani, M. A. A molecular programme for the specification of germ cell fate in mice. Nature 418, 293–300 (2002).
Lange, U. C., Saitou, M., Western, P. S., Barton, S. C. & Surani, M. A. The fragilis interferon-inducible gene family of transmembrane proteins is associated with germ cell fate specification in mice. BMC Dev. Biol. 3, 1 (2003).
Saitou, M. & Surani, A. Cell surface expressed marker of pluripotency. United States Patent 7,226,994 (issued 5 June 2007).
Saitou, M. & Surani, A. Antibodies for identification of murine Fragilis extracellular domain and methods for identifying pluripotent cells. United States Patent 7,884,193 (issued 8 February (2011).
Navaroli, D., Tilly, J. L. & Woods, D. C. Isolation of mammalian oogonial stem cells by antibody-based fluorescence-activated cell sorting. Methods Mol. Biol. 1457, 253–268 (2016).
Borrelli, E., Heyman, R. A., Arias, C., Sawchenko, P. E. & Evans, R. M. Transgenic mice with inducible dwarfism. Nature 339, 538–541 (1989).
Garcia, A. D., Doan, N. B., Imura, T., Bush, T. G. & Sofroniew, M. V. GFAP-expressing progenitors are the principal source of constitutive neurogenesis in adult mouse forebrain. Nat. Neurosci. 7, 1233–1241 (2004).
Visnjic, D. et al. Hematopoiesis is severely altered in mice with an induced osteoblast deficiency. Blood 103, 3258–3264 (2004).
Rubtsov, Y. P. et al. Stability of the regulatory T cell lineage in vivo. Science 329, 1667–1671 (2010).
Ahn, S. & Joyner, A. L. In vivo analysis of quiescent adult neural stem cells responding to Sonic hedgehog. Nature 437, 894–897 (2005).
Barker, N. et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449, 1003–1007 (2007).
Lepper, C., Conway, S. J. & Fan, C. M. Adult satellite cells and embryonic muscle progenitors have distinct genetic requirements. Nature 460, 627–631 (2009).
Morris, R. J. et al. Capturing and profiling adult hair follicle stem cells. Nat. Biotechnol. 22, 411–417 (2004).
Brinster, R. L. & Zimmermann, J. W. Spermatogenesis following male germ-cell transplantation. Proc. Natl. Acad. Sci. USA 91, 11298–11302 (1994).
Brinster, R. L. & Avarbock, M. R. Germline transmission of donor haplotype following spermatogonial transplantation. Proc. Natl. Acad. Sci. USA 91, 11303–11307 (1994).
Kanatsu-Shinohara, M., Morimoto, H. & Shinohara, T. Fertility of male germline stem cells following spermatogonial transplantation in infertile mouse models. Biol. Reprod. 94, 112 (2016).
Yoshimizu, T. et al. Germline-specific expression of the OCT-4/green fluorescent protein (GFP) transgene in mice. Dev. Growth Differ. 41, 675–684 (1999).
Szabo, P. E., Hübner, K., Schöler, H. & Mann, J. R. Allele-specific expression of imprinted genes in mouse migratory primordial germ cells. Mech. Dev. 115, 157–160 (2002).
Jameson, S. A. et al. Temporal transcriptional profiling of somatic and germ cells reveals biased lineage priming of sexual fate in the fetal mouse gonad. PLoS Genet. 8, e1002575 (2012).
Sabour, D. et al. Germ cell nuclear factor regulates gametogenesis in developing gonads. PLoS One 9, e103985 (2014).
Woods, D. C., White, Y. A. R. & Tilly, J. L. Purification of oogonial stem cells from adult mouse and human ovaries: an assessment of the literature and a view towards the future. Reprod. Sci. 20, 7–15 (2013).
Oulad-Abdelghani, M. et al. Characterization of a premeiotic germ cell-specific cytoplasmic protein encoded by Stra8, a novel retinoic acid-responsive gene. J. Cell. Biol. 135, 469–477 (1996).
Menke, D. B., Koubova, J. & Page, D. C. Sexual differentiation of germ cells in XX mouse gonads occurs in an anterior-to-posterior wave. Dev. Biol. 262, 303–312 (2003).
Baltus, A. E. et al. In germ cells of mouse embryonic ovaries, the decision to enter meiosis precedes premeiotic DNA replication. Nat. Genet. 38, 1430–1434 (2006).
Bowles, J. & Koopman, P. Retinoic acid, meiosis and germ cell fate in mammals. Development 134, 3401–3411 (2007).
Anderson, E. L. et al. Stra8 and its inducer, retinoic acid, regulate meiotic initiation in both spermatogenesis and oogenesis in mice. Proc. Natl. Acad. Sci. USA 105, 14976–14980 (2008).
Guan, K. et al. Pluripotency of spermatogonial stem cells from adult mouse testis. Nature 440, 1199–1203 (2006).
Zheng, P. & Dean, J. Oocyte-specific genes affect folliculogenesis, fertilization, and early development. Semin. Reprod. Med. 25, 243–251 (2007).
Wang, N. & Tilly, J. L. Epigenetic status determines germ cell meiotic commitment in embryonic and postnatal mammalian gonads. Cell Cycle 9, 339–349 (2010).
Suzumori, N., Yan, C., Matzuk, M. M. & Rajkovic, A. Nobox is a homeobox-encoding gene preferentially expressed in primordial and growing oocytes. Mech Dev 111, 137–141 (2002).
Pangas, S. A. et al. Oogenesis requires germ cell-specific transcriptional regulators Sohlh1 and Lhx8. Proc. Natl. Acad. Sci. USA 103, 8090–8095 (2006).
East, I. J., Mattison, D. R. & Dean, J. Monoclonal antibodies to the major protein of the murine zona pellucida: effects on fertilization and early development. Dev. Biol. 104, 49–56 (1984).
Faqi, A. S., Klug, A., Merker, H. J. & Chahoud, I. Ganciclovir induces reproductive hazards in male rats after short-term exposure. Hum. Exp. Toxicol. 16, 505–511 (1997).
Page, S. L. & Hawley, R. S. The genetics and molecular biology of the synaptonemal complex. Annu. Rev. Cell Dev. Biol. 20, 525–558 (2004).
Rogakou, E. P., Pilch, D. R., Orr, A. H., Ivanova, V. S. & Bonner, W. M. DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139. J. Biol. Chem. 273, 5858–5868 (1998).
Niikura, Y., Niikura, T. & Tilly, J. L. Aged mouse ovaries possess rare premeiotic germ cells that can generate oocytes following transplantation into a young host environment. Aging 1, 971–978 (2009).
Woods, D. C. & Tilly, J. L. Autologous germline mitochondrial energy transfer (AUGMENT) in assisted human reproduction. Semin. Reprod. Med. 33, 410–421 (2015).
Gong, S. P. et al. Embryonic stem cell-like cells established by culture of adult ovarian cells in mice. Fertil. Steril. 93, 2594–2601 (2010).
Lei, L. & Spradling, A. C. Female mice lack adult germ-line stem cells but sustain oogenesis using stable primordial follicles. Proc. Natl. Acad. Sci. USA 110, 8585–8590 (2013).
Woods, D. C. & Tilly, J. L. Germline stem cells in adult mammalian ovaries. In Ten Critical Topics in Reproductive Medicine, S. Sanders, Ed., Science/AAAS, Washington DC, pp. 10–12 (2013).
Shen, T. T. et al. Genome wide profiling of alternative polyadenylation in mouse female germline stem cells. Nan Fang Yi Da Xue Xue Bao 36, 157–162 (2016).
Zhang, X. L. et al. Integrative epigenomic analysis reveals unique epigenetic signatures involved in unipotency of mouse female germline stem cells. Genome Biol. 17, 162 (2016).
Borovskaya, T. G., Dygai, A. M., Fomina, T. I. & Vychuzhanina, A. V. Quantitative evaluation of primordial follicles in rat ovaries during early and delayed terms after different cytostatic exposures. Bull. Exp. Biol. Med. 161, 61–63 (2016).
Ishii-Morita, H. et al. Mechanism of ‘bystander effect’ killing in the herpes simplex thymidine kinase gene therapy model of cancer treatment. Gene Ther. 4, 244–251 (1997).
Touraine, R. L., Ishii-Morita, H., Ramsey, W. J. & Blaese, R. M. The bystander effect in the HSVtk/ganciclovir system and its relationship to gap junctional communication. Gene Ther. 5, 1705–1711 (1998).
Bush, T. G. et al. Fulminant jejuno-ileitis following ablation of enteric glia in adult transgenic mice. Cell 93, 189–201 (1998).
Chen, J. et al. A restricted cell population propagates glioblastoma growth after chemotherapy. Nature 488, 522–526 (2012).
Guo, K., Li, C.-h, Wang, X.-y, He, D.-j & Zheng, P. Germ stem cells are active in postnatal mouse ovary under physiological conditions. Mol. Hum. Reprod. 22, 316–328 (2016).
Ryu, B. Y., Orwig, K. E., Oatley, J. M., Avarbock, M. R. & Brinster, R. L. Effects of aging and niche microenvironment on spermatogonial stem cell self-renewal. Stem Cells 24, 1505–1511 (2006).
Zhang, X., Ebata, K. T., Robaire, B. & Nagano, M. C. Aging of male germ line stem cells in mice. Biol. Reprod. 74, 119–124 (2006).
Woods, D. C. & Tilly, J. L. The next (re)generation of human ovarian biology and female fertility: is current science tomorrow’s practice? Fertil. Steril. 98, 3–10 (2012).
Navaroli, D., Tilly, J. L. & Woods, D. C. Isolation of oogonial stem cells by antibody-based fluorescence-activated sorting. Meth. Mol. Biol. 1457, 253–268 (2016).
Jones, E. C. & Krohn, P. L. The relationships between age, numbers of oocytes and fertility in virgin and multiparous mice. J. Endocrinol. 21, 469–495 (1961).
Acknowledgements
The authors thank V. Dubus, F. Bertaux and the scientific team at Genoway for generation of the transgenic mouse lines, and J. Galipeau for provision of Gfp-fused HSVtk cDNA. The authors also thank L. Prickett-Rice and K. Folz-Donahue of the Harvard Stem Cell Institute Flow Cytometry Core Facility, the MGH Center for Systems Biology Imaging Core Facility, and M. Cooper (Cooper Graphics) for expert graphic arts assistance with commissioned preparation of the schematic models depicted in Figures 1a and 4a. This work was supported by a Method to Extend Research in Time (MERIT) Award from the National Institute on Aging (NIH R37-AG012279 to J.L.T.), an Exploratory/Developmental Research Grant Award from the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NIH R21-HD072280 to J.L.T.), and a Glenn Foundation for Medical Research Award (to J.L.T.). This work was conducted while N.W. was supported by a Pathway to Independence Award from the National Institute on Aging (NIH K99/R00-AG039512), while C.S. was supported by Mahidol University of Thailand, and while D.C.W. was supported by a Ruth L. Kirschstein National Research Service Award from the National Institute on Aging (NIH F32-AG034809). This work was initiated while E.S.P., D.C.W. and J.L.T. were at Massachusetts General Hospital and Harvard Medical School, and then completed at Northeastern University.
Author information
Authors and Affiliations
Contributions
N.W. and J.L.T. designed the experiments. N.W., C.S., Y.O., E.S.P. and D.C.W. conducted the experiments. All authors analyzed results. N.W. and J.L.T. wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
N.W., C.S., E.S.P. and Y.O. declare no competing financial interests. D.C.W. discloses interest in intellectual property described in U.S. Patent 8,642,329, U.S. Patent 8,647,869 and U.S. Patent 9,150,830, and is a recipient of a corporate sponsored research award from OvaScience, Inc. (Waltham, MA). J.L.T. discloses interest in intellectual property described in U.S. Patent 7,195,775, U.S. Patent 7,850,984, U.S. Patent 7,955,846, U.S. Patent 8,642,329, U.S. Patent 8,647,869, U.S. Patent 8,652,840, U.S. Patent 9,150,830 and U.S. Patent 9,267,111, is a scientific co-founder of OvaScience, Inc., and is a member of the Scientific Advisory Board of OvaScience, Inc.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, N., Satirapod, C., Ohguchi, Y. et al. Genetic studies in mice directly link oocytes produced during adulthood to ovarian function and natural fertility. Sci Rep 7, 10011 (2017). https://doi.org/10.1038/s41598-017-10033-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-10033-6
This article is cited by
-
Stem Cells in Adult Mice Ovaries Form Germ Cell Nests, Undergo Meiosis, Neo-oogenesis and Follicle Assembly on Regular Basis During Estrus Cycle
Stem Cell Reviews and Reports (2021)
-
Testicular Stem Cell Dysfunction Due to Environmental Insults Could Be Responsible for Deteriorating Reproductive Health of Men
Reproductive Sciences (2021)
-
Ovary does harbor stem cells - size of the cells matter!
Journal of Ovarian Research (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.