Abstract
The deep abiogenic synthesis of hydrocarbons is possible under the conditions of the asthenosphere. We have found that this process can also occur under the mineral and thermobaric conditions of subducting slabs. We have investigated the abiogenic synthesis of hydrocarbon systems at pressures of 2.0–6.6 GPa and temperatures of 250–600 °C. The determined lower thermobaric limit of the reaction at 280–300 °C and 2–3 GPa corresponds to a depth of 70–80 km during cold subduction. The hydrocarbon fluid formed in the slab can migrate upwards through the network of faults and fractures to form petroleum deposits.
Similar content being viewed by others
Introduction
The origin of hydrocarbons addresses such topics as petroleum accumulation1, global carbon and hydrocarbon cycles, and the origin of life2. During the last decades, theoretical and experimental investigations of hydrocarbon systems under high thermobaric conditions were published3,4,5. The experiments and computations focused on the properties and phase transformation of single hydrocarbons4, 6 and complex hydrocarbon systems7,8,9 with chemical compositions similar to those of natural hydrocarbon systems. In the abovementioned studies, the abiogenic formation of hydrocarbons was attributed to the high pressure-temperature chemical reactions between the natural donors of carbon (mineral or fluid) and hydrogen (presumably aqueous fluids) in the mantle. In most of these works, these reactions were attributed to high pressures at temperatures of approximately 1000 °C5. These thermobaric conditions correspond with the Earth’s asthenosphere conditions. Previous experiments demonstrated that hydrocarbons can be formed from deep rock-fluid interactions under a wide range of temperatures and pressures of 500–1500 °C and 3.0–8.0 GPa6, 8,9,10,11. The results of these experiments showed that the water-carbonate interaction under reduced mantle conditions is one of the most preferable paths, leading to the deep formation of hydrocarbons12.
However, the subducting slab environment could also be considered for hydrocarbon formation due to the enrichment of slab sedimentary rocks with carbonates13, 14 and the abundance of particularly calcite-rich rocks in subduction zones15,16,17,18.
In this study, we have attempted to determine the lowest thermobaric conditions (and the corresponding depth) at which hydrocarbons could be generated in the slab from carbonates and water (the source of which could be any hydroxyl group-containing mineral presented in the investigated geological environment, e.g. serpentine) in the Earth’s interior.
The experiments were carried out in Toroid-type high-pressure chamber. The analysis of the gases produced was performed using gas chromatography from the gas extracting cell. Raman spectroscopy was used to analyze the solid phase products (see Method). The high-pressure equipment was pressure and temperature calibrated (see Supplemental Materials). The design of high-pressure equipment allowed us to measure the pressure and temperature with an accuracy of 0.1 GPa and 10 °C throughout the experiment, respectively. The quantity of the gas produced was only calculated relatively due to the present technical difficulties of the gas extracting cell calibration (see Supplemental Materials).
According to Kutcherov et al.19, the general pathway for the formation of hydrocarbons:
To determine the lower thermobaric limit of reaction (1), we conducted 15 experiments in a temperature range of 250 to 750 °C at pressures ranging from 2.0 to 6.6 GPa. Thermobaric conditions were chosen according to the Earth’s interior pressure-temperature profiles20. Experimental conditions and product characterizations are shown in Table 1. The description of high and low yields is presented in the Supplemental Materials (Supplementary Note).
We replaced initial iron with iron(II) oxide in experimental runs #2 and #5, slightly increasing the oxygen fugacity of the system (reaction from Kutcherov et al.19).
It was observed that the water-carbonate interaction in the reduced conditions leads to the formation of hydrocarbons in the majority of our experiments. We divided the experiments into two sets: experiments where the formation of hydrocarbons was detected, and experiments in which no hydrocarbons were observed. The results of the first set of experiments show that the hydrocarbon mixture formed contains a prevalence of methane and remains stable at temperatures higher than 250 °C over the entire pressure range investigated (Fig. 1). From the chromatograms shown in Fig. 1, one can see that although methane is the main component of the hydrocarbon system (up to 95%), the mixture contains other saturated hydrocarbons to heptane, their isomers, and even benzene. Although the distribution of hydrocarbons was observed in previous works8, 19, this is the first time that the aromatic component, benzene, has been observed as the product of the reaction between calcite and water in the reduced environment. No signs of oxygen-containing hydrocarbons were detected, though, their formation in similar systems was reported in recent works11, 21, probably due to either lower temperatures in this research, or the limits of the analysis technique and/or different reaction components, including capsule material.
The results of the second set of experiments (#12, #13, #14 and #15) demonstrate that at a temperature of 250 °C within the pressure range of 2.3–6.6 GPa, hydrocarbons were not formed. These thermobaric conditions lay outside of the reaction zone.
Figure 2 illustrates the performed experiments along with slab geotherms. In some of the first set of experiments conducted at lower temperatures (#7, #8, #9, #10 and #11), the hydrocarbon yield was significantly low. Combining this result with the results of the second set of experiments suggests that the hydrocarbon formation at pressures from 2.0 to 6.6 GPa occurs at temperature higher than 250 °C. No significant pressure influence was observed. According to the pT-profile of the Earth’s interior20, a temperature range of 250–300 °C at 2.0–3.0 GPa corresponds to a depth of 70–80 km of a cold subducting slab. Therefore, the results from our experiments show that the formation of hydrocarbon systems from carbonates and water may occur in the cold slab at a depth of 70–200 km.
Experiments and geotherms20. The blue curve is a cold slab, the red curve is a hot slab, and the black curve is the upper mantle pT profiles. The white diamonds depict experiments where hydrocarbons are not detected. The small gray diamonds correspond to experiments with a low hydrocarbon yield. The big gray diamonds correspond to experiments with a high hydrocarbon yield. The error bars are smaller than the symbol size. The pink sector indicates the thermobaric zone where hydrocarbon formation is favored.
The results of the experiments #1 and #2 at the thermobaric conditions corresponding to the conditions in a hot slab show that the formation of complex hydrocarbon systems may also begin at a depth of 70–80 km (Fig. 2).
The solid product formed in experiment #2 was analyzed using Raman spectroscopy (Supplemental Materials, Supplementary Figure 1). The obtained Raman spectra revealed the presence of heavier hydrocarbons in the solid product. Heavy hydrocarbons were observed previously from the system MgCO3–Ca(OH)2–Fe–SiO2 after exposure at 3 GPa and 1400 °C for 24 hours11. No free carbon (diamond or graphite) was detected. This result differs from the study of carbonated mafic mineral aqueous transformations, where diamond formation was observed at a pressure of 5.0 GPa and a temperature of 900 °C22. This discrepancy is a consequence of the significantly lower temperature used in our experiments compared to that in the experiments conducted by Sverjensky & Huang (2015)22.
In Martirosyan et al.23, the formation of iron carbide was observed as a result of a reaction between iron and magnesite at 6.0 GPa and 1000–1500 °C. In our experiments, iron carbide was not observed. In contrast to the experiments described23, we used water as one of the initial components of the reaction.
The observed differences imply that the behavior of the carbonate-iron-water system strongly depends on the thermobaric conditions and could differ in the subducting zone and asthenosphere.
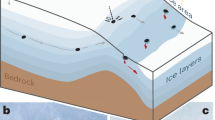
Fluid migration pathways is an important issue to discuss in the formation of petroleum deposits. Several studies discuss different mechanisms of migration such as hydrocarbon-bearing melt migration within a mantle plume24, and volcanic activity25. In our opinion, the most favorable path for the formed hydrocarbons to migrate from the subduction zone upwards is by migration along the weakened surface of the slab (Fig. 3).
To conclude, we have demonstrated that the formation of complex hydrocarbon mixtures could occur in the subducting slabs at a depth of 70–80 to 200 km. The formed hydrocarbons can migrate upwards along the weakened surface of the slab to the Earth’s crust and form hydrocarbon deposits. These results may further enlighten the abiogenic origin of hydrocarbons and mechanism of petroleum deposit formation.
Method
Experiments in a Toroid-type high-pressure chamber
Pure CaCO3 (>99%, Sigma Aldrich), Fe (>99%, Sigma Aldrich), FeO (>99%, Sigma Aldrich), and distillated water were used as starting materials in our investigations. The high pressure-temperature conditions corresponding to those in a subducting slab were modeled using a Toroid-type high-pressure chamber with resistive heating26.
CaCO3, Fe(FeO) and H2O (molar ratio 1:1:1) were mixed and tightly loaded into a steel capsule with a working volume of ~0.5 cm3. Stainless steel (Fe~70%) was used as the capsule material to avoid any influence of any additional components on the reaction course since iron was one of the reaction components. The capsule design allowed contact between the mixture and the atmosphere to be avoided. The mixture contained in the sealed capsule was placed in the toroid chamber. Two heaters (graphite/Al2O3) were placed above and below the capsule, allowing resistive heating to achieve temperatures up to 1800 °C. Reference materials used for pressure (Bi, PbSe, PbTe) and temperature (Ti, Sn, Pb) calibration using the resistance differential (Supplementary Figure 2 in the Supplemental Materials) were placed on the top of the capsule inside the paper disk and covered with aluminum or copper foil for improved conductivity. The toroid chamber with the capsule inside was put between two hard alloy anvils of the high pressure equipment. The assemblage is shown in Supplementary Figure 3 in the Supplemental Materials. The main experimental parameters were indirectly controlled by the “URS-2” press program. The hydraulic pressure and power applied to the anvils was set according to a calibration to create the desired pressure and temperature in the capsule. The program also allowed the rate of heating to the required temperature, the duration of exposure, and the cooling rate to be adjusted. All performed experiments were heated in 60 s, exposed for 2 h, and cooled instantly (quenched).
Analysis by means of gas chromatography
The analysis of the gaseous products was carried out by means of a gas chromatograph Chromatek-5000 using a gas extracting cell. The cell is an improved version of a previously described modification19. The present gas extracting cell (Supplementary Figure 4 in the Supplemental Materials) allows us to send the gas mixture directly from the capsule into the gas chromatograph. After the capsule was taken from the pressure equipment it was placed inside the cell and sealed inside with a rubber ring. After air was taken from the cell by the first blow, a hard alloy needle penetrated the capsule. After penetration, the gas spread out inside the cell and was then sent to the chromatograph for analysis by the second blow.
The specification of the chromatograph Chromatek-5000 was focused on the detection and separation of light hydrocarbons by an Agilent GS-GasPro capillary column in an increasing temperature regime from 60 to 140 °C in 50 min. The column allows to detect even neglectable quantity of the hydrocarbon gas produced. The detection of the separated hydrocarbon components was carried out by the FID detector.
Analysis by means of Raman spectroscopy
After the analysis of the gaseous product, the solid phase of experiment #2 was extracted from the capsule and analyzed by Raman spectroscopy using a He-Ne laser (wavelength 632.8 nm, power 2 mW) at Bayerisches Geoinstitut. The solid powder was evenly distributed on the surface of a glass plate and examined in a few different areas using the laser with a ~30 µm spot size.
Data Availability
All data generated or analyzed during this study are included in this published article (and its Supplementary Information files).
References
Kutcherov, V. G. & Krayushkin, V. A. Deep-seated abiogenic origin of petroleum: From geological assessment to physical theory. Rev. Geophys. 48, 1–30 (2010).
Hazen, R. M., Jones, A. P. & Baross, J. A. Carbon in Earth. 75, (Mineralogical society of america geochemical society, 2013).
Kenney, J. F., Kutcherov, Va, Bendeliani, Na & Alekseev, Va The evolution of multicomponent systems at high pressures: VI. The thermodynamic stability of the hydrogen-carbon system: the genesis of hydrocarbons and the origin of petroleum. Proc. Natl. Acad. Sci. USA 99, 10976–10981 (2002).
Spanu, L., Donadio, D., Hohl, D., Schwegler, E. & Galli, G. Stability of hydrocarbons at deep Earth pressures and temperatures. 108, 6843–6846 (2011).
Kolesnikov, A. Y., Saul, J. M. & Kutcherov, V. G. Chemistry of Hydrocarbons Under Extreme Thermobaric Conditions. Chem. Sel. Rev. 2, 1336–1352 (2017).
Scott, H. P. et al. Generation of methane in the Earth’s mantle: In situ high pressure-temperature measurements of carbonate reductio. n. Proc. Natl. Acad. Sci. USA 101, 14023–14026 (2004).
Kolesnikov, A., Kutcherov, V. G. & Goncharov, A. F. Methane-derived hydrocarbons produced under upper-mantle conditions. Nat. Geosci. 2, 566–570 (2009).
Kutcherov, V. G. et al. Synthesis of complex hydrocarbon systems at temperatures and pressures corresponding to the Earth’s upper mantle condition. s. Dokl. Phys. Chem. 433, 132–135 (2010).
Sharma, A., Cody, G. D. & Hemley, R. J. In situ diamond-anvil cell observations of methanogenesis at high pressures and temperatures. Energy and Fuels 23, 5571–5579 (2009).
Chen, J. Y., Jin, L. J., Dong, J. P., Zheng, H. F. & Liu, G. Y. Methane formation from CaCO3 reduction catalyzed by high pressure. Chinese Chem. Lett. 19, 475–478 (2008).
Sonin, V. M. et al. Synthesis of heavy hydrocarbons under P-T conditions of the Earth’s upper mantle. Dokl. Earth Sci. 454, 32–36 (2014).
Etiope, G. & Sherwood Lollar, B. Abiotic methane on earth. Rev. Geophys. 51, 276–299 (2013).
Dasgupta, R. et al. Carbon-dioxide-rich silicate melt in the Earth’s upper mantle. Nature 493, 211–5 (2013).
Manning, C. E. Geochemistry: A piece of the deep carbon puzzle. Nat. Geosci. 7, 333–334 (2014).
Luth, R. In Mantle Petrology: Field Observations and High Pressure Experimentation 297–316 (Geochem. Soc. Spec., 1999).
Dasgupta, R. & Hirschmann, M. M. The deep carbon cycle and melting in Earth’s interior. Earth Planet. Sci. Lett. 298, 1–13 (2010).
Hammouda, T. & Keshav, S. Melting in the mantle in the presence of carbon: Review of experiments and discussion on the origin of carbonatites. Chem. Geol. 418, 171–188 (2015).
Poli, S. Melting carbonated epidote eclogites: carbonatites from subducting slabs. Prog. Earth Planet. Sci., doi:10.1186/s40645-016-0105-6 (2016).
Kutcherov, V. G., Bendeliani, N. A., Alekseev, V. A. & Kenney, J. F. Synthesis of hydrocarbons from minerals at pressures up to 5 GPa. Dokl. Phys. Chem. 387, 328–330 (2002).
Litasov, K. D., Shatskiy, A. & Ohtani, E. In Physics and Chemistry of the Deep Earth (ed. Karato, S.) 38–65, doi:10.1002/9781118529492.ch2 (John Wiley & Sons, Ltd., 2013).
Tomilenko, A. A. et al. The synthesis of methane and heavier hydrocarbons in the system graphite–iron–serpentine at 2 and 4 GPa and 1200 °C. High Temp. Press. 44, 451–465 (2015).
Sverjensky, D. A. & Huang, F. Diamond formation due to a pH drop during fluid–rock interactions. Nat. Commun. 53, 1689–1699 (2015).
Martirosyan, N. S., Litasov, K. D., Shatskiy, A. F. & Ohtani, E. Reactions of iron with calcium carbonate at 6 GPa and 1273–1873 K: Implications for carbonate reduction in the deep mantle. Russ. Geol. Geophys. 56, 1322–1331 (2015).
Belonoshko, A. B., Lukinov, T., Rosengren, A., Bryk, T. & Litasov, K. D. Synthesis of heavy hydrocarbons at the core-mantle boundary. Nat. Publ. Gr., doi:10.1038/srep18382 1–6 (2015).
Dobretsov, N. L., Koulakov, I. Y. & Litasov, Y. D. Migration paths of magma and fluids and lava compositions in Kamchatka. RGG 53, 1253–1275 (2012).
Khvostantsev, L. G., Slesarev, V. N. & Brazhkin, V. V. Toroid type high-pressure device: history and prospects. High Press. Res. 24, 371–383 (2004).
Acknowledgements
We thank D. Kudryavtsev for performing the Raman analysis and L. Dubrovinsky and Bayerisches Geoinstitut (Bayreuth, Germany) for the provision of the Raman equipment. We thank A. Serovajsky for his help during the high-pressure equipment calibration and interpretation of the chromatographic spectra. We thank the anonymous reviewer for valuable comments. This research was supported by grants from the Sloan Foundation through the Deep Carbon Observatory.
Author information
Authors and Affiliations
Contributions
V.K. designed the study. E.M. conducted and analyzed the experiments. V.K., E.M. and A.K. discussed the results and wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mukhina, E., Kolesnikov, A. & Kutcherov, V. The lower pT limit of deep hydrocarbon synthesis by CaCO3 aqueous reduction. Sci Rep 7, 5749 (2017). https://doi.org/10.1038/s41598-017-06155-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-06155-6
This article is cited by
-
Contribution of Deep Hydrocarbons in Gas Hydrate Formation
Chemistry and Technology of Fuels and Oils (2023)
-
The composition of the fluid phase in inclusions in synthetic HPHT diamonds grown in system Fe–Ni–Ti–C
Scientific Reports (2022)
-
In-situ abiogenic methane synthesis from diamond and graphite under geologically relevant conditions
Nature Communications (2021)
-
Formation of complex hydrocarbon systems from methane at the upper mantle thermobaric conditions
Scientific Reports (2020)
-
Raman Spectroscopy Study on Chemical Transformations of Propane at High Temperatures and High Pressures
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.