Abstract
An important safety concern in the use of human pluripotent stem cells (hPSCs) is tumorigenic risk, because these cells can form teratomas after an in vivo injection at ectopic sites. Several thousands of undifferentiated hPSCs are sufficient to induce teratomas in a mouse model. Thus, it is critical to remove all residue-undifferentiated hPSCs that have teratoma potential before the clinical application of hPSC-derived cells. In this study, our data demonstrated the cytotoxic effects of cardiac glycosides, such as digoxin, lanatoside C, bufalin, and proscillaridin A, in human embryonic stem cells (hESCs). This phenomenon was not observed in human bone marrow mesenchymal stem cells (hBMMSCs). Most importantly, digoxin and lanatoside C did not affect the stem cells’ differentiation ability. Consistently, the viability of the hESC-derived MSCs, neurons, and endothelium cells was not affected by the digoxin and lanatoside C treatment. Furthermore, the in vivo experiments demonstrated that digoxin and lanatoside C prevented teratoma formation. To the best of our knowledge, this study is the first to describe the cytotoxicity and tumor prevention effects of cardiac glycosides in hESCs. Digoxin and lanatoside C are also the first FDA-approved drugs that demonstrated cytotoxicity in undifferentiated hESCs.
Similar content being viewed by others
Introduction
Human embryonic stem cells (hESCs) and induced pluripotent stem cells (iPSCs) are human pluripotent stem cells (hPSCs) that have unique self-renewal (ability to replicate almost indefinitely) and pluripotency (ability to differentiate into all cell types of the human body except for placental cells) properties. These abilities make hPSCs promising resources for regeneration therapy1. However, substantial challenges remain to be overcome before applying hPSCs to cell therapy. An important safety concern of hPSCs is their tumorigenic risk because these cells can form teratomas after in vivo injections at ectopic sites2, 3. Thousands of undifferentiated hPSCs residing in millions of differentiated cells are sufficient to induce teratomas in a mouse model4. Thus, it is critical to remove all or most of the residue-undifferentiated hPSCs that have teratoma potential before clinical applications using hPSC-derived cells.
There are several strategies to selectively remove hPSCs. These methods include the use of cytotoxic antibodies5, 6, specific antibody cell sorting7,8,9, genetic manipulations10,11,12, and pharmacological approaches13,14,15,16. However, each method has certain disadvantages, such as a high cost (cytotoxic antibodies and specific antibody cell sorting), variation among different lots (cytotoxic antibodies and specific antibody cell sorting)17, 18, non-specific binding (cytotoxic antibodies)18,19,20, requirement of genetic manipulation and stable integration of toxic genes (genetic manipulation), and time-consuming procedures (genetic manipulation, specific antibody cell sorting and cytotoxic antibodies). Although many studies have attempted to prevent or block teratoma formation in residual hPSCs, a clinically applicable strategy to eliminate teratoma formation remains to be developed2, 21.
In contrast, small molecule approaches have several advantages as follows: these approaches are robust, efficient, fast, simple, and inexpensive, and there is no need to insert genes into cells. Certain small molecules have been shown to inhibit teratoma formation in hPSCs. The inhibitor of stearoyl-CoA desaturase PluriSin #1 prevented teratoma formation15. Stearoyl-CoA desaturase is a key enzyme in the biosynthesis of mono-saturated fatty acids and is required for hPSC survival15. The N-benzylnonanamide JC011 induced ER stress through the PERK/AT4/DDIT3 pathway22. Chemical inhibitors of survivin, such as quercetin and YM155, induced selective cell death and efficiently inhibited teratoma formation14. However, neither of these drugs is well defined or approved by the FDA.
In this study, we investigated the roles of cardiac glycosides in human PSCs. Cardiac glycosides (CGs) (also named cardiotonic steroids, CSs) belong to a large family of compounds that can be derived from nature products. Although these compounds have diverse structures, they share a common structural motif. These compounds are specific inhibitors of the transmembrane sodium pump (Na+/K+-ATPase). CGs inhibit the Na+/K+-ATPase and then increase the intracellular concentrations of calcium ions23. These compounds act as positive inotropic agents, and members of this group have been used in the treatment of heart failure for more than 200 years. One member of this family, digoxin, is still in clinical use24. Furthermore, CGs are currently considered to have a potential therapeutic role in cancer therapy25. Several studies have reported that CGs play important roles in inducing cell death in several cancer cells23. Cancer cells show more susceptibility than cells in normal tissues. The molecular mechanism may be the overexpression of specific alpha subunits of Na+/K+-ATPase in cancerous cells26. These studies indicate that CGs are selective according to the cell type and distinguish between normal cells and transformed cells.
Although cardiac glycosides act as multiple signal transducers, no studies have investigated whether these drugs can eliminate undifferentiated PSCs while sparing their progeny or differentiated cells. In this study, we used digoxin, lanatoside C, bufalin, and proscillaridin A to investigate whether CGs can target hESCs and selectively induce cell death in pluripotent cells. Of these drugs, digoxin and lanatoside C are both FDA approved. Surprisingly, we found that these four drugs efficiently induced cell death in hESCs, but not in differentiated cells or hESC-derived mesenchymal stem cells (MSCs). The in vivo experiments also showed that digoxin and lanatoside C successfully prevented teratoma formation.
Results
Differential expression of the alpha subunit of Na+/K+-ATPase in hESCs and hBMMSCs
Because not all cancer signals overlap with hESC signals, we determined the expression levels of cardiac glycosides target genes, Na+/K+-ATPase, to evaluate whether they can eliminate the undifferentiated hESCs. It has previously been reported that cardiac glycosides have anti-cancer effects by targeting Na+/K+-ATPase25, 26. Via a western blot analysis, we found that the hESCs expressed Na+/K+-ATPase more abundantly than adult stem cells, such as human bone marrow mesenchymal stem cells (hBMMSCs) (Fig. 1a). This finding suggests that hESCs may be more sensitive to cardiac glycosides than hBMMSCs due to their differential expression of Na+/K+-ATPase.
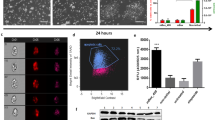
Cardiac glycosides induced cytotoxic effect of in hESCs but not in hMSCs. (a) Protein expression levels of the Na+/K+-ATPase alpha1 and alpha2 subunit in hESCs and hMSCs were detected by western blotting. All unprocessed blot images are presented in Supplementary Fig. S7a. (b,d) Cell colony and morphology of hESCs and hMSCs under bright field. hESCs and MSCs were treated with DMSO solvent control, 2.5 μM digoxin or 2.5 μM lanatoside C (for 12 hours and 24 hours). Scale bar: 500 μm. (c,e) LDH release was measured in hESCs and hMSCs to investigate the cytotoxic effect in 96-well culture dishes. DMSO, 2.5 μM digoxin, or 2.5 μM lanatoside C was used to treat the hESCs of hMSCs for 24 hours, and the supernatant was harvested for the LDH detection. The samples were normalized to the DMSO-treated hESCs. ***P < 0.001; **P < 0.01; n.s. not significant. Data are shown as the mean ± SD.
Digoxin and Lanatoside C-induced cell death in hESCs but not in hBMMSCs
We investigated whether cardiac glycosides affected the viability of hESCs or other cell types. First, we treated undifferentiated hESCs with digoxin and lanatoside C for 12 hours and 24 hours, respectively. Both digoxin (2.5 μM) and lanatoside C (2.5 μM) induced dramatic cell death in the hESCs (Fig. 1b). To investigate the cytotoxic effect of the cardiac glycosides, we measured the release of LDH in the culture supernatants after the hESCs were treated with digoxin or Lanatoside C for 24 hours (Fig. 1c). Both drugs significantly induced a cytotoxic effect in the hESCs (Fig. 1c). Consistently, in another hESC line, i.e., HUES6, both digoxin (2.5 μM) and lanatoside C (2.5 μM) induced cell death (Fig. S1a) and cytotoxicity (Fig. S1b).
In contrast, digoxin and lanatoside C did not affect the survival of hBMMSCs (Fig. 1d). Both drugs had no cytotoxic effects on the hBMMSCs as measured by the LDH cytotoxic assay (Fig. 1e).
Cardiac glycosides can be divided into two subgroups based on the natural structure of their lactone moiety23, 26. Digoxin and lanatoside C belong to a cardenolides subgroup that has butyrolactone23. We choose two drugs in the bufadienolides subgroup that have a pyrone ring23. We used bufalin or proscillaridin A to treat the hESCs and hBMMSCs. The results were similar to the results of the digoxin- and lanatoside C-treated cells in which bufalin or proscillaridin A induced cytotoxicity in the hESCs but not in the hBMMSCs (Fig. S2).
To determine whether the cytotoxic effect of the cardiac glycosides is selective to hESCs, we also performed a PI/Annexin flow cytometry analysis. After treating the cells for 24 hours, the cell death reached 70% following the digoxin treatment and 82% following the lanatoside C treatment (Fig. 2a). In contrast, more than 98% of the cells were alive in the digoxin- or lanatoside C-treated hBMMSCs (Fig. 2b). In addition, we observed increases in the cleaved form of PARP, caspase-3, and caspase-7 in the digoxin- and lanatoside C-treated hESCs (Fig. 2c). In contrast, no cleaved form of the apoptosis markers was detected in the hBMMSCs treated with digoxin or lanatoside C (Fig. 2c). In addition to the induction of cell death, the tumorigenic potential of the remaining of hESCs was abolished upon cell differentiation. After the hESCs were treated with digoxin or lanatoside C for 12 hours, the protein levels of Nanog were downregulated (Fig. 2d). Nanog is a part of the core transcriptional regulatory networks in ESCs. Loss of Nanog in the hESCs can induce extraembryonic lineage differentiation27. Nanog has been reported to play important roles in hESC pluripotency and self-renewal28. These results suggested that cardiac glycosides induce cell death in hESCs but not in hBMMSCs.
Cell death markers were upregulated in the cardiac glycoside-treated hESCs but not in the hBMMSCs. Cell death was analyzed using hESCs or MSCs. (a) hESCs were treated with DMSO, 1.25 μM digoxin, or 2.5 μM lanatoside C for 24 hour. The bar graph represents the statistical results of the FASC data. *P < 0.05; Data are shown as the mean ± SD. Live cells are represented by PI−/Annexin V−; dead cells are represented by PI−/Annexin V+, PI+/Annexin−, and PI+/Annexin+. The bar graph represents the statistical results of the FASC data. n.s. not significant. Data are shown as the mean ± SD. (b) hMSCs were treated with DMSO, 2.5 μM digoxin, or 2.5 μM lanatoside C for 24 hours. Cells were stained with PI and Annexin V and subjected to flow cytometry analysis. The bar graph represents the statistical results of the FASC data. n.s. not significant. Data are shown as the mean ± SD. (c,d) Protein levels of the cleaved form of the apoptotic markers and pluripotent stem cell markers were detected by western blotting. hESCs and hMSCs were treated with DMSO, 2.5 μM digoxin or 2.5 μM lanatoside C for 12 hours. Cells were harvested, and the cleaved and uncleaved forms of PARP, caspase7, and caspase3 were detected. For the detection of the pluripotent stem cell markers, the cells were harvested, and Nanog and Oct4 were detected. All unprocessed blot images are presented in Supplementary Fig. S7b,c.
Differentiation abilities of hBMMSCs into three lineages are not affected by the digoxin or lanatoside C treatment
We demonstrated that the cardiac glycosides did not affect the survival of hBMMSCs. MSCs are multipotent cells that are promising for regenerative medicine. MSCs can be specifically induced into osteoblasts, adipocytes, and cartilage cells29. To determine whether the differentiation ability of hBMMSCs is affected by digoxin or lanatoside C, we performed a differentiation assay. Digoxin or lanatoside C was removed after treating the hBMMSCs for 24 hours, and the cells were differentiated into three lineages. Notably, neither digoxin nor lanatoside C affected the differentiation ability of the hBMMSCs into osteoblasts (Fig. 3a), adipocytes (Fig. 3b), and chondrocytes (Fig. 3c). Based on these results, cardiac glycosides do not influence the multipotency of hBMMSCs.
Cardiac glycoside treatment of hBMMSCs did not affect the differentiation abilities. hBMMSCs were treated with DMSO, 2.5 μM digoxin, or 2.5 μM lanatoside C for 24 hours, and the drugs were removed for further differentiation. (a) Osteogenic differentiation. Mineralization was stained with Alizarin Red S, and the quantification was performed at O.D 570 nm. (b) Adipogenic differentiation. Lipid drop was stained with oil red, and quantification was performed at O.D 510 nm. (c) Chondrogenic differentiation. Glycosaminoglycan was stained with Alcian blue, and quantification was performed at O.D 650 nm. Scale bar: 500 μm. n.s. not significant. Data are shown as the mean ± SD.
Digoxin or Lanatoside C did not induce cytotoxic effects in hESC-derived MSCs
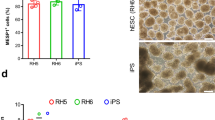
To further validate the effects of the cardiac glycosides in hESCs and hESC-derived cell types, we first choose hESC-derived MSCs (hESC-MSCs) as our model. Dr. Xu and colleagues provided a simple and fast method to induce hESCs into hMSCs using a two-step approach30 (Fig. S3a). We differentiated the H9 hESCs into MSCs and examined whether the cardiac glycosides affected the viability of the hESC-MSCs. The hESC-MSCs were treated with digoxin and lanatoside C for 12 hours and 24 hours, respectively. Neither digoxin (2.5 μM) nor lanatoside C (2.5 μM) induced cell death in the H9 hESC-MSCs (Fig. 4a). To investigate the cytotoxic effect of the cardiac glycosides, we measured the release of LDH in the culture supernatants after treating the H9 hESC-MSCs with digoxin or Lanatoside C for 24 hours. Neither Digoxin nor lanatoside C affected the survival of the H9 hESC-MSCs (Fig. 4b). In addition to digoxin and lanatoside C, we also found that bufalin or proscillaridin A do not induce cytotoxicity (Fig. S3d). This result is consistent with the effect observed in the hBMMSCs (Fig. S2b).
Cardiac glycosides did not affect the cell viability of H9 hESC-derived MSCs. (a) Cell morphology of H9 hESC-MSCs under bright field. hESC-MSCs were treated with DMSO solvent control, 2.5 μM digoxin or 2.5 μM lanatoside C for 12 hours and 24 hours respectively. Scale bar: 500 μm. (b) LDH release in hESC-MSCs was measured to investigate the cytotoxicity effect in 96-well culture plates. Cells were treated with DMSO or the drugs for 24 hours, and then, the supernatant was harvested for the LDH detection. The samples were normalized against the DMSO-treated hESCs. (c) Flow cytometry for the cell death analysis. hESC-MSCs were treated with DMSO, 2.5 μM digoxin or 2.5 μM lanatoside C for 24 hours. Cells were stained with PI and Annexin V, and the cells were analyzed by flow cytometry. The bar graph represents the statistical results of the FASC data. n.s. not significant. Data are shown as the mean ± SD. (d) hESCs and hESC-MSCs were treated with DMSO, 2.5 μM digoxin or 2.5 μM lanatoside C for 12 hours. Cells were harvested, and the cleaved and uncleaved forms of PARP, caspase3 and caspase7 were detected by western blotting. (e) Protein expression levels of the Na+/K+-ATPase alpha1 and alpha2 subunits in the hESCs and hESC-MSCs were detected by western blotting. All unprocessed blot images in (d) and (e) are presented in Supplementary Fig. S8.
To investigate whether the cardiac glycosides induced cell death in the H9 hESC-MSCs, PI/Annexin flow cytometry and western blot analyses were performed. After treating the hESC-MSCs with digoxin or lanatoside C for 24 hours, the cell death was less than 2% (Fig. 4c). Consistently, no cleaved form of the apoptosis markers was detected in the hESC-MSCs treated with digoxin or lanatoside C (Fig. 4d). These results suggested that the cardiac glycosides did not induce cell death in the H9 hESC-MSCs. In addition, Na+/K+-ATPase was abundantly expressed in the undifferentiated hESCs but not in the hESC-MSCs (Fig. 4e), which might demonstrate that the toxicity of the cardiac glycosides is limited to the undifferentiated hESCs.
Digoxin and lanatoside C did not or slightly induce cell death in hPSCs derived endothelium cells, neurons, or hepatocyte endoderm
We next tested whether digoxin and lanatoside C affected other hPSC-differentiated cell types. The mesoderm lineage of CD34+/CD144+ hiPSC-derived endothelial cells was obtained from Dr. Chiang and his colleagues (National Cheng Kung University, Tainan, Taiwan) (Fig. S4a). Undifferentiated hiPSCs and hiPSCs-derived endothelial cells were exposed to digoxin or lanatoside C for 24 hours. After the treatment, digoxin and lanatoside C induced cytotoxicity in the hiPSCs (Fig. S4b), but the hiPSC-derived endothelial cells remained alive (Fig. S4c). We differentiated the H9 hESCs into TUJ1-positive neurons that belong to the ectoderm (Fig. S4d) and then treated these hESC-neurons with digoxin or lanatoside C for 24 hours. Digoxin and lanatoside C did not induce cytotoxicity in the neuronal cells (Fig. S4e). In addition, we differentiated the hESCs into AFP-expressing hepatocyte endoderm, which belongs to the endoderm (Fig. S4f). The results showed that the drugs slightly, if at all, induced any cell death in the hepatocyte endoderm cells (less than 10%) (Fig. S4g). Based on the above mentioned results, digoxin and lanatoside C might specifically induce cell death in undifferentiated hPSCs but not in their differentiated progeny.
Digoxin and lanatoside C prevent teratoma formation in NSG mice
To investigate whether hESCs treated with cardiac glycosides lose their ability to form teratomas, hESCs were treated with DMSO, digoxin or lanatoside C and were transplanted into NSG mice individually for xenograft. We found that the tumor weight was significantly decreased in the cardiac glycoside drug-treated group (Fig. 5a and b). Thus, digoxin and lanatoside C severely hampered most of the tumor formation ability in the hESCs. Teratoma formation in the DMSO-, digoxin-, or lanatoside C-treated hESCs was shown to contain all three germ layers (Figs 5c and S5). These results demonstrate the pluripotent ability of these hESCs. We demonstrated that the cardiac glycosides efficiently prevented tumor formation in vivo.
Cardiac glycosides treatment prevented teratoma formation in hESCs in NSG mice. Tumors from NSG mice transplanted with DMSO-, digoxin-, or lanatoside C-treated cells for two months (n = 8 in each group). Scale bar: 10 mm. (b) Quantification of the teratoma weight. Dot: DMSO; Square: digoxin; Triangle: lanatoside C. ****P < 0.0001. Data are shown as the mean ± SD. (c) These images show a DMSO-treated hESC-derived teratoma. These teratoma paraffin sections were stained with H&E staining (top panel) and IHC staining of three lineage markers (bottom panel). AFP (alpha-fetoprotein): endoderm marker. SMA (smooth muscle actin): mesoderm marker. Tuj1 (beta-III tubulin): ectoderm marker. Scale bar: 50 μm.
Finally, to investigate whether digoxin- or lanatoside C-treated hBMMSCs remain in vivo, we constructed GFP overexpressing hBMMSCs. The GFP overexpressing hBMMSCs did not form tumors in the NSG mice under the kidney capsules (Fig. S6a). The GFP-hBMMSCs remained at the graft site, which was observed by GFP IHC staining (Fig. S6a). Then, a mixture of hESCs and hBMMSCs that were treated with the drugs was injected into NSG mice under the kidney capsules. The tumor area was also significantly inhibited in the digoxin- or lanatoside C-treated groups (Fig. S6b), and we still observed GFP-positive hBMMSCs (Fig. S6c). These results demonstrated that digoxin- and lanatoside C-treated hBMMSCs remained in the NSG mice under the kidney capsules.
Discussion
In cell therapy, residual undifferentiated ESCs or iPSCs in their differentiated progenies raise concerns regarding the safety (teratoma) of using PSC-derived cells. The tumorigenic ability of undifferentiated PSCs is lost upon terminal differentiation. However, residual undifferentiated PSCs must be removed prior to the application of ESC and iPSC cell therapy31. In this study, our data demonstrated the cytotoxicity effect of cardiac glycosides in hESCs (Figs 1b,c, 2a,c and S1). This phenomenon was not observed in the hBMMSCs (Figs 1d,e and 2b,c). Most importantly, these drugs did not affect the stem cells differentiation abilities (Fig. 3). A similar effect of cardiac glycosides was shown in the hESC-derived progeny. The viability of the hESC-MSCs, hESC-neurons and hiPSC-endothelial cells were also not affected by digoxin and lanatoside C (Figs 4 and S4). Furthermore, the in vivo experiments showed that digoxin and lanatoside C efficiently prevented teratoma formation (Fig. 5). For the first time, our work described the cytotoxic effect and tumor prevention capabilities of cardiac glycosides in hESCs. Digoxin and lanatoside C are also the first FDA approved drugs that have been shown to have cytotoxicity in hESCs.
Cardiac glycosides are rich in many plants, such as the Digitalis species, and they are also extensively found in animal species, mainly in toads25. There are only a few reports describing the differentiation roles of cardiac glycosides in PSCs or PSC-derived cells. A high-throughput screening assay of small compound libraries revealed that cardiac glycosides, i.e., cymarin (CYM) and sarmentogenin (SRM), promoted the early differentiation, but not cytotoxicity, of hESCs32. Treating hESCs with CYM and SRM inhibited OCT4 expression and induced SOX17 expression32. A previous report has shown that digoxin and lanatoside C may reduce TDP-43 protein aggregation in iPSC-derived neurons in sporadic amyotrophic lateral sclerosis (sALS) patients33. Change in TDP-43 protein expression and subcellular localization is the most important pathology in sALS34. Therefore, these compounds have potential neuroprotective properties. In our present work, we further demonstrated that cardiac glycosides, such as digoxin and lanatoside C, are also involved in the cytotoxic effect and pluripotency.
Cardiac glycosides have been used in the clinic for the treatment of cardiac diseases for a long time. The anti-cancer role of cardiac glycosides is rather novel23, 35. Digoxin was found to inhibit several types of cancer cells, such as breast cancer, lymphoma, melanoma, myeloma, and small cell lung cancer36,37,38. Lanatoside C was also demonstrated to inhibit tumor growth, such as colorectal cancer and human hepatocellular carcinoma39,40,41. For synergistic cancer therapy, lanatoside C combined with TRAIL-secreted neural stem cells can target glioblastoma42. Thus, cardiac glycosides induce cell death in cancer cells. Cardiac glycosides are well known as a group of Na+/K+-ATPase specific inhibitors. Changes in the expression levels of Na+/K+-ATPase subunits were shown in various cancers23. The Na+/K+-ATPase alpha1 subunit was overexpressed in non-small cell lung cancer (NSCLC) cell lines43. Inhibiting the expression of the alpha1 subunit impaired proliferation and migration in NSCLC cell lines43. A recent study has shown that human NSCLC cells overexpressed the alpha1, alpha2, or alpha3 subunits of Na+/K+-ATPase and were induced cell death by cardiac glycosides (i.e., digoxin and ouabain)44. In our data, the expression levels of the alpha1 and alpha2 subunits of Na+/K+-ATPase were significantly higher in the hESCs than in the hMSCs or hESC-derived progeny (Figs 1a and 4e). Cardiac glycosides may induce cytotoxicity in hESCs through Na+/K+-ATPase, which is similar to cancer. More details need to be investigated.
Another possible mechanism of cardiac glycosides is a BCL-2 family anti-apoptotic protein, MCL-1. MCL-1 was demonstrated to be an essential and universal target of cardiac glycosides, and cardiac glycosides cause a downregulation of the MCL-1 protein in various human adherent and non-adherent cancer cells45. In another paper, MCL-1 was reported to be more highly expressed in undifferentiated hESCs than in differentiated cells46. The authors demonstrated that the loss of Mcl-1 induced cell death in H9 hESCs and suggested that Mcl-1 was critical for hESC survival. Thus, it is also possible that cardiac glycosides induced cell death in the H9 cells by downregulating MCL-1.
To overcome the risk of teratoma formation in regenerative medicine, several strategies have been proposed47, 48. Antibody-sorting and cytotoxic antibody strategies may be simple, but their efficiency is limited due to single-cell dissociation requirements or antibody batch variations7, 17, 18, 49. The cost of these approaches is also high. Another strategy is based on the genetic manipulation of traceable target cells10, 12, 50, 51, but these methods are laborious and expensive. Most importantly, insertion mutagenesis is a biosafety threat in the clinical use of such genetically altered cells48. Chemical ablation strategies are rapid, robust and efficient, and they are also the most cost-effective methods. Chemical approaches do not require cell sorting and any genetic manipulation.
In addition to small molecule approaches, a study has used metabolic selection to enrich PSC-derived cardiomyocytes13. The authors provided an interesting method to purify cardiomyocytes from PSCs using glucose-depleted and lactate-rich culture conditions. This method is very suitable for generating high purity cardiomyocytes. However, other cell types might need more tests to determine the cell type specific metabolism. This strategy is attractive but can be applied in only a very few cell types. We used CG drugs to eliminate undifferentiated hESCs, and the drugs did not affect the survival of several different cell types (i.e., MSCs, endothelium cells, neurons). Since CGs is easy to purchase and are cost efficient, this method may be convenient for applications in the future. Digoxin and lanatoside C are potent small molecules that inhibit tumorigenic hESCs in culture as shown by the teratoma formation assay (Fig. 5). The expression levels of the Na+/K+-ATPase subunits were different between the cancer and normal cells or tissues23. Furthermore, our data suggested that the expression levels of the Na+/K+-ATPase subunits were also different between the undifferentiated and differentiated cells (Figs 1a and 4e). In this study, we revealed a novel application of cardiac glycosides that may improve the major concern of hPSCs cell therapy by preventing teratoma formation.
Materials and Methods
All methods were performed in accordance with the relevant guidelines and regulations. All experiments were approved by Human Subject Research Ethics (AS-IRB02-106069) and Institutional Animal Care & Utilization Committee (IACUC, 14-03-684), Academia Sinica (Taipei, Taiwan). All culture medium and supplements unless otherwise specified, were obtained from ThermoFisher Scientific (Wilmington, DE, USA). All chemicals unless otherwise specified, were brought from Sigma (St. Louis, MO, USA).
Cell lines and culture conditions
The hESC line H9 was purchased from WiCells (Madison, WI, USA)52. Another hESC line, HUES6, was kindly provided by Dr. Douglas A. Melton (Harvard University, Boston, MA, USA)53. Cells were maintained on the feeder cells and cultured in Dulbecco’s modified Eagle’s medium (DMEM)/F12 supplemented with 20% knockout serum replacement, 2 mM L-glutamine, 1% nonessential amino acids, 4 ng/mL human bFGF, and 0.1 mM 2-mercaptoethanol. For the feeder cells culture, C57BL/6 mouse embryonic fibroblasts (MEF) were cultured in the DMEM with 10% FBS and treated cells with 0.01 mg/ml mitomycin C 2 hours for inactivation. For the feeder-free culture, hESCs were seeded on the Matrigel Matrix (BD Biosciences, San Jose, CA, USA) coated culture plates and maintained by conditioned medium of MEF (C57BL/6). hBMMSCs were cultured in mesenPRO RSTM kit (Life Technologies, Camarillo, CA, USA). All cells were cultured in a humidified atmosphere containing 5% CO2 at 37 °C.
Lactate dehydrogenase (LDH) Cytotoxicity assay
The supernatants of cells treated with digoxin 2.5 μM, lanatoside C 2.5 μM, or DMSO solvent control for 24 hours were harvested. The released LDH was measured using CytoTox 96 Non-Cytotoxicity assay according to the manufacture’s instruction (Promega, Southampton, UK). The supernatants and reagents were incubated at room temperature for 20 minutes and then the reaction was stopped by Stop Solution. The absorbance at 490 nm was measured using a plate-reading spectrophotometer (Benchmark Plus Microplate Spectrophotometer System, BIO-RAD, Hercules, CA, USA).
Western blot analysis
RIPA buffer was used to harvest cell lysates (RIPA buffer: NaCl 150 mM, Tris pH 8.0 50 mM, EDTA pH 8.0 5 mM, NP-40 1.0%, SDS 0.5%, sodium deoxycholate 0.1%). Western blot analyses were performed as previously described with different types of primary antibodies54. The primary antibody includes anti-β-actin (A5441; Sigma), anti-Oct4 (sc-9081; Santa Cruz Biotechnology, Santa Cruz, CA, USA), anti-Nanog (3580; Cell Signaling Technology, Danvers, MA, USA), apoptosis antibody kit (9915; Cell Signaling Technology), ATP1A1 (3010; Cell Signaling Technology), and ATP1A2 (16836-1-AP; proteintechTM) were used. After reaction at 4 °C overnight, the blots were incubated with goat anti-mouse- or goat anti-rabbit antibody conjugated with horseradish peroxidase (Jackson ImmunoResearch Inc). The chemiluminescent substrate (WBKLS0500; Millipore, Darmstadt, Germany) was used to detect the blots. Fujifilm LAS-4000 (FUJIFILM, Tokyo, Japan) was used to take the images.
Flow cytometry
For cell death assay, Propidium iodine/Annexin V assay was performed according to the manufacturer’s instruction (Alexa Fluor® 488 Annexin V/Dead Cell Apoptosis Kit; Life Technologies). In brief, live cells were dissociated with trypsin and incubated with Annexin V antibody and PI working solution for 15 minutes. We added 400 μl 1X annexin binding buffer and analyzed the stained cells by FACSCantoTM (Becton Dickinson, Franklin Lakes, NJ, USA). For analyzing the phenotypic signature of MSCs we used a StemflowTM hMSC analysis kit (BD Biosciences) and FACSCantoTM. All flow data was analyzed by FACSDivaTM software (BD Biosciences) and FlowJoTM (FlowJo, LLC, Ashland, USA).
hESC-derive MSCs
According to the report from Dr. Xu and colleagues30, hESCs were cultured with 10 ng/ml BMP4 and 1 μM of A8301 for 5 days. Next, the cells were passaged on the gelatin-coated plate and the culture medium was switched to MSC culture medium [Minimum Essential Medium Eagle Alpha Modification (αMEM) medium supplemented with 20% fetal bovine serum (FBS), l-glutamine (Gluta- MAX), 1x nonessential amino acids]. Cells were expanded within passage 5, and CD73+ (11-0793, ThermoFisher Scientific) and CD105+ (12–1057, ThermoFisher Scientific) double positive cells were sorted.
Cell sorting
hESC-derived MSCs were trypsinized and washed with PBS. Then cells were incubated with anti-human CD73 FITC (ThermoFisher Scientific) and anti-human CD105 PE (ThermoFisher Scientific) for 15 minutes at 4 °C. Cells were washed cells with PBS for three times. CD73+/CD105+ cells were sorted with the cell sorter (BD FACSAria II, BD Biosciences). Sorted cells were maintained in MSC culture medium.
Osteogenic differentiation and Alizarin Red S staining
BMMSCs were treated with digoxin (2.5 μM), lanatoside C (2.5 μM), or DMSO solvent control for 24 hours. Drugs were removed, and the cells were washed with PBS and cultured in MSCgo™ Osteogenic Differentiation medium (Biological Industries, Kibbutz Beit-Haemek, Israel)55. The media changed twice per week for 14–21 days. Next, the cells were fixed with ice-cold 70% ethanol at −20 °C for 1 hour. After washing with water for three times, the cells were then stained with 40 mM Alizarin Red S (ARS) (pH 4.2) at room temperature for 10 minutes. The cells were then washed with PBS for five times. An Olympus CK-40 microscope was used to take the images. For quantification, the dye was extracted with 10% (w/v) cetylpyridinium chloride (Sigma 0732) in sodium phosphate buffer (pH 7.0) for 15 minutes, and the O.D. at 570 nm was measured.
Adipogenesis and Oil Red O assay
hBMMSCs were treated with digoxin (2.5 μM), lanatoside C (2.5 μM), or DMSO solvent control for 24 hours. Next, drugs were removed, and the cells were cultured in MSCgo™ Adipogenic Differentiation Medium (Biological Industries)55. The media was changed every 3–4 days for 8–12 days. After the adipogenic induction, we replaced the MSCgo™ Adipogenic complete medium with MSC maintenance medium for 6–9 days. The cells were carefully fixed with 4% formaldehyde for 1 hour at room temperature, washed with 60% isopropanol, and air-dried. The lipid drops were stained with Oil Red O working solution (30 ml 0.35% oil red solution in isopropanol diluted with 20 ml of distilled water) for 10 minutes. Next, the cells were washed with water 4 times. For quantification, Oil Red O stain was extracted with 100% isopropanol, and the absorbance at 510 nm was detected.
Chondrogenic induction and Alcian blue assay
BMMSCs were treated with digoxin (2.5 μM), lanatoside C (2.5 μM), or DMSO solvent control for 24 hours. Next, drugs were removed, and 1 × 105 cells were seeded in 96-well U-bottom culture plated. After 24 hours, we changed the complete MSCgo™ Chondrogenic medium (Biological Industries) for 21 days. The media were changed every 3–4 days. Next, the pelleted cells were fixed with 4% formaldehyde for 1 hours, washed twice with ddH2O, and stained with a 1% Alcian blue solution in 0.1 N HCl for 30 min. For Alcian Blue elution, we added 8 M Guanidine HCL solution and incubated overnight at RT. The absorbance at 650 nm was detected.
In vivo tumorigenicity assay and immunohistochemistry
hESCs were treated with digoxin (2.5 μM), lanatoside C (2.5 μM), or DMSO control for 24 hours. Approximately 106 treated cells were mixed with 105 MEFs to promote teratoma formation in 50 μl PBS56. The cells mixture and 1x Matrigel Matrix was mixed well and the cells were subcutaneously injected into NOD scid gamma mice (NSG mice) for 8 weeks. After 8 weeks, animals were sacrificed and teratoma was removed, fixed in 10% formalin, embedded in paraffin and stained with hematoxylin and eosin. H&E stain protocol was modified from previous study57. For immunohistochemistry, teratoma sections were blocked using 5% milk for 1 hour, and stained with primary antibody at 4 °C overnight, follow by secondary antibody (Dako, Santa Clara, CA, USA) for 1 hour at RT and DAB enhancer (Dako). Primary antibody: anti-human alpha-1-fetoprotein (A0008, Dako) for endoderm lineage; anti-human smooth muscle actin, clone 1A4 (M0851, Dako) for mesoderm lineage; anti-Tuj1 (MAB1637, EMD Millipore, Darmstadt, Germany) for ectoderm.
Statistical analysis
All statistical data are presented as the mean ± standard deviation (S.D.) of at least three biological replicates. Statistically significant differences were assessed by t test or One-Way ANOVA, where p-value < 0.05 was considered a significant difference.
References
Ben-David, U., Kopper, O. & Benvenisty, N. Expanding the boundaries of embryonic stem cells. Cell Stem Cell 10, 666–677, doi:10.1016/j.stem.2012.05.003 (2012).
Knoepfler, P. S. Deconstructing stem cell tumorigenicity: a roadmap to safe regenerative medicine. Stem cells (Dayton, Ohio) 27, 1050–1056, doi:10.1002/stem.37 (2009).
Ben-David, U. & Benvenisty, N. The tumorigenicity of human embryonic and induced pluripotent stem cells. Nat Rev Cancer 11, 268–277, doi:10.1038/nrc3034 (2011).
Lee, A. S. et al. Effects of cell number on teratoma formation by human embryonic stem cells. Cell Cycle 8, 2608–2612, doi:10.4161/cc.8.16.9353 (2009).
Tan, H. L., Fong, W. J., Lee, E. H., Yap, M. & Choo, A. mAb 84, a cytotoxic antibody that kills undifferentiated human embryonic stem cells via oncosis. Stem cells (Dayton, Ohio) 27, 1792–1801, doi:10.1002/stem.109 (2009).
Choo, A. B. et al. Selection against undifferentiated human embryonic stem cells by a cytotoxic antibody recognizing podocalyxin-like protein-1. Stem cells (Dayton, Ohio) 26, 1454–1463, doi:10.1634/stemcells.2007-0576 (2008).
Tang, C. et al. An antibody against SSEA-5 glycan on human pluripotent stem cells enables removal of teratoma-forming cells. Nat Biotechnol 29, 829–834, doi:10.1038/nbt.1947 (2011).
Ben-David, U., Nudel, N. & Benvenisty, N. Immunologic and chemical targeting of the tight-junction protein Claudin-6 eliminates tumorigenic human pluripotent stem cells. Nat Commun 4, 1992, doi:10.1038/ncomms2992 (2013).
Fong, C. Y., Peh, G. S., Gauthaman, K. & Bongso, A. Separation of SSEA-4 and TRA-1-60 labelled undifferentiated human embryonic stem cells from a heterogeneous cell population using magnetic-activated cell sorting (MACS) and fluorescence-activated cell sorting (FACS). Stem Cell Rev 5, 72–80, doi:10.1007/s12015-009-9054-4 (2009).
Blum, B., Bar-Nur, O., Golan-Lev, T. & Benvenisty, N. The anti-apoptotic gene survivin contributes to teratoma formation by human embryonic stem cells. Nat Biotechnol 27, 281–287, doi:10.1038/nbt.1527 (2009).
Menendez, S. et al. Increased dosage of tumor suppressors limits the tumorigenicity of iPS cells without affecting their pluripotency. Aging cell 11, 41–50, doi:10.1111/j.1474-9726.2011.00754.x (2012).
Schuldiner, M., Itskovitz-Eldor, J. & Benvenisty, N. Selective ablation of human embryonic stem cells expressing a “suicide” gene. Stem cells (Dayton, Ohio) 21, 257–265, doi:10.1634/stemcells.21-3-257 (2003).
Tohyama, S. et al. Distinct metabolic flow enables large-scale purification of mouse and human pluripotent stem cell-derived cardiomyocytes. Cell Stem Cell 12, 127–137, doi:10.1016/j.stem.2012.09.013 (2013).
Lee, M. O. et al. Inhibition of pluripotent stem cell-derived teratoma formation by small molecules. Proceedings of the National Academy of Sciences of the United States of America 110, E3281–3290, doi:10.1073/pnas.1303669110 (2013).
Ben-David, U. et al. Selective elimination of human pluripotent stem cells by an oleate synthesis inhibitor discovered in a high-throughput screen. Cell Stem Cell 12, 167–179, doi:10.1016/j.stem.2012.11.015 (2013).
Dabir, D. V. et al. A small molecule inhibitor of redox-regulated protein translocation into mitochondria. Developmental cell 25, 81–92, doi:10.1016/j.devcel.2013.03.006 (2013).
Baker, M. Reproducibility crisis: Blame it on the antibodies. Nature 521, 274–276, doi:10.1038/521274a (2015).
Prassas, I. & Diamandis, E. P. Translational researchers beware! Unreliable commercial immunoassays (ELISAs) can jeopardize your research. Clin Chem Lab Med 52, 765–766, doi:10.1515/cclm-2013-1078 (2014).
Egelhofer, T. A. et al. An assessment of histone-modification antibody quality. Nat Struct Mol Biol 18, 91–93, doi:10.1038/nsmb.1972 (2011).
Michel, M. C., Wieland, T. & Tsujimoto, G. How reliable are G-protein-coupled receptor antibodies? Naunyn-Schmiedeberg’s archives of pharmacology 379, 385–388, doi:10.1007/s00210-009-0395-y (2009).
Blum, B. & Benvenisty, N. The tumorigenicity of human embryonic stem cells. Adv Cancer Res 100, 133–158, doi:10.1016/S0065-230X(08)00005-5 (2008).
Richards, M. et al. A new class of pluripotent stem cell cytotoxic small molecules. PLoS One 9, e85039, doi:10.1371/journal.pone.0085039 (2014).
Prassas, I. & Diamandis, E. P. Novel therapeutic applications of cardiac glycosides. Nat Rev Drug Discov 7, 926–935, doi:10.1038/nrd2682 (2008).
Gheorghiade, M., Adams, K. F. Jr. & Colucci, W. S. Digoxin in the management of cardiovascular disorders. Circulation 109, 2959–2964, doi:10.1161/01.CIR.0000132482.95686.87 (2004).
Mijatovic, T. et al. Cardiotonic steroids on the road to anti-cancer therapy. Biochim Biophys Acta 1776, 32–57, doi:10.1016/j.bbcan.2007.06.002 (2007).
Diederich, M., Muller, F. & Cerella, C. Cardiac glycosides: From molecular targets to immunogenic cell death. Biochem Pharmacol. doi:10.1016/j.bcp.2016.08.017 (2016).
Hyslop, L. et al. Downregulation of NANOG induces differentiation of human embryonic stem cells to extraembryonic lineages. Stem cells (Dayton, Ohio) 23, 1035–1043, doi:10.1634/stemcells.2005-0080 (2005).
Wang, Z., Oron, E., Nelson, B., Razis, S. & Ivanova, N. Distinct lineage specification roles for NANOG, OCT4, and SOX2 in human embryonic stem cells. Cell Stem Cell 10, 440–454, doi:10.1016/j.stem.2012.02.016 (2012).
Pittenger, M. F. et al. Multilineage potential of adult human mesenchymal stem cells. Science 284, 143–147 (1999).
Wang, X. et al. Immune modulatory mesenchymal stem cells derived from human embryonic stem cells through a trophoblast-like stage. Stem cells (Dayton, Ohio) 34, 380–391, doi:10.1002/stem.2242 (2016).
Nishikawa, S., Goldstein, R. A. & Nierras, C. R. The promise of human induced pluripotent stem cells for research and therapy. Nat Rev Mol Cell Biol 9, 725–729, doi:10.1038/nrm2466 (2008).
Desbordes, S. C. et al. High-throughput screening assay for the identification of compounds regulating self-renewal and differentiation in human embryonic stem cells. Cell Stem Cell 2, 602–612, doi:10.1016/j.stem.2008.05.010 (2008).
Burkhardt, M. F. et al. A cellular model for sporadic ALS using patient-derived induced pluripotent stem cells. Mol Cell Neurosci 56, 355–364, doi:10.1016/j.mcn.2013.07.007 (2013).
Toyoshima, Y. & Takahashi, H. TDP-43 pathology in polyglutamine diseases: with reference to amyotrphic lateral sclerosis. Neuropathology 34, 77–82, doi:10.1111/neup.12053 (2014).
Patel, S. Plant-derived cardiac glycosides: Role in heart ailments and cancer management. Biomed Pharmacother 84, 1036–1041, doi:10.1016/j.biopha.2016.10.030 (2016).
Johansson, S. et al. Cytotoxicity of digitoxin and related cardiac glycosides in human tumor cells. Anticancer Drugs 12, 475–483 (2001).
Bielawski, K., Winnicka, K. & Bielawska, A. Inhibition of DNA topoisomerases I and II, and growth inhibition of breast cancer MCF-7 cells by ouabain, digoxin and proscillaridin A. Biol Pharm Bull 29, 1493–1497 (2006).
Eskiocak, U. et al. Synergistic effects of ion transporter and MAP kinase pathway inhibitors in melanoma. Nat Commun 7, 12336, doi:10.1038/ncomms12336 (2016).
Durmaz, I. et al. Liver cancer cells are sensitive to Lanatoside C induced cell death independent of their PTEN status. Phytomedicine 23, 42–51, doi:10.1016/j.phymed.2015.11.012 (2016).
Kang, M. A. et al. Lanatoside C suppressed colorectal cancer cell growth by inducing mitochondrial dysfunction and increased radiation sensitivity by impairing DNA damage repair. Oncotarget 7, 6074–6087, doi:10.18632/oncotarget.6832 (2016).
Chao, M. W. et al. Lanatoside C, a cardiac glycoside, acts through protein kinase Cdelta to cause apoptosis of human hepatocellular carcinoma cells. Sci Rep 7, 46134, doi:10.1038/srep46134 (2017).
Teng, J., Hejazi, S., Badr, C. E. & Tannous, B. A. Systemic anticancer neural stem cells in combination with a cardiac glycoside for glioblastoma therapy. Stem cells (Dayton, Ohio) 32, 2021–2032, doi:10.1002/stem.1727 (2014).
Mijatovic, T. et al. The alpha1 subunit of the sodium pump could represent a novel target to combat non-small cell lung cancers. J Pathol 212, 170–179, doi:10.1002/path.2172 (2007).
Cherniavsky Lev, M., Karlish, S. J. & Garty, H. Cardiac glycosides induced toxicity in human cells expressing alpha1-, alpha2-, or alpha3-isoforms of Na-K-ATPase. Am J Physiol Cell Physiol 309, C126–135, doi:10.1152/ajpcell.00089.2015 (2015).
Cerella, C. et al. Early downregulation of Mcl-1 regulates apoptosis triggered by cardiac glycoside UNBS1450. Cell Death Dis 6, e1782, doi:10.1038/cddis.2015.134 (2015).
Huskey, N. E. et al. CDK1 inhibition targets the p53-NOXA-MCL1 axis, selectively kills embryonic stem cells, and prevents teratoma formation. Stem Cell Reports 4, 374–389, doi:10.1016/j.stemcr.2015.01.019 (2015).
Ben-David, U. & Benvenisty, N. Chemical ablation of tumor-initiating human pluripotent stem cells. Nat Protoc 9, 729–740, doi:10.1038/nprot.2014.050 (2014).
Rashin, M., Amir, A. H. & Javad, V. a. A., S.-H. Safe transplantation of pluripotent stem cell by preventing teratoma formation. Journal of Stem Cell Research & Therapy 4, doi:10.4172/2157-7633.1000212 (2014).
Schriebl, K. et al. Selective removal of undifferentiated human embryonic stem cells using magnetic activated cell sorting followed by a cytotoxic antibody. Tissue Eng Part A 18, 899–909, doi:10.1089/ten.TEA.2011.0311 (2012).
Chung, S. et al. Genetic selection of sox1GFP-expressing neural precursors removes residual tumorigenic pluripotent stem cells and attenuates tumor formation after transplantation. J Neurochem 97, 1467–1480, doi:10.1111/j.1471-4159.2006.03841.x (2006).
Huber, I. et al. Identification and selection of cardiomyocytes during human embryonic stem cell differentiation. FASEB J 21, 2551–2563, doi:10.1096/fj.05-5711com (2007).
Thomson, J. A. et al. Embryonic stem cell lines derived from human blastocysts. Science 282, 1145–1147 (1998).
Cowan, C. A. et al. Derivation of embryonic stem-cell lines from human blastocysts. N Engl J Med 350, 1353–1356, doi:10.1056/NEJMsr040330 (2004).
Wang, C. H. et al. A shRNA functional screen reveals Nme6 and Nme7 are crucial for embryonic stem cell renewal. Stem cells (Dayton, Ohio) 30, 2199–2211, doi:10.1002/stem.1203 (2012).
Lai, P. L. et al. Efficient Generation of Chemically Induced Mesenchymal Stem Cells from Human Dermal Fibroblasts. Sci Rep 7, 44534, doi:10.1038/srep44534 (2017).
Hentze, H. et al. Teratoma formation by human embryonic stem cells: evaluation of essential parameters for future safety studies. Stem Cell Res 2, 198–210, doi:10.1016/j.scr.2009.02.002 (2009).
Fischer, A. H., Jacobson, K. A., Rose, J. & Zeller, R. Hematoxylin and eosin staining of tissue and cell sections. CSH Protoc 2008, pdb prot4986, doi:10.1101/pdb.prot4986 (2008).
Acknowledgements
We thank Dr. Douglas A. Melton (Harvard University, Boston, MA, USA), Dr. Joanne Jeou-Yuan Chen (Academia Sinica, Taipei, Taiwan), Dr. Han-Chung Wu (Academia Sinica, Taipei, Taiwan), Dr. and Dr. Ching Hwa Tsai (National Taiwan University, Taipei, Taiwan) for their assistance in this study. We are grateful for the technical support of flow cytometry facility of the GRC flow core (Genomic Research Center, Academia Sinica, Taipei, Taiwan). We thank the Human Disease iPSC Service Consortium (MOST 106-2319-B-001-003), the Taiwan Ministry of Science and Technology (MOST), for the iPSC generation and technical support. This work was funded by Academia Sinica, the National Health Research Institute (NHRI) (NHRI-EX106-10415SI), and the Taiwan MOST (102-2311-B-002-041-MY3, 103-2321-B-001-064, 104-2320-B-001-005-, 104-0210-01-09-02, 105-0210-01-13-01, 105-2320-B-001 -026 -MY2, 106-0210-01-15-02).
Author information
Authors and Affiliations
Contributions
Y.T.L. performed most of the work, analyzed the data, wrote the manuscript manuscript; C.K.W. and S.C.Y. assisted the tissue culture; H.L., F.P.C. and T.C.K. assisted data collection; S.C.H. and F.L.L. discussed the data and contributed to manuscript writing; C.N.C. discussed the data and provided reagents; M.H. and P.M.C. provided reagents and discussed the data; J.L. directed the project, discussed the data, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lin, YT., Wang, CK., Yang, SC. et al. Elimination of undifferentiated human embryonic stem cells by cardiac glycosides. Sci Rep 7, 5289 (2017). https://doi.org/10.1038/s41598-017-05616-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-05616-2
This article is cited by
-
Monovalent ions and stress-induced senescence in human mesenchymal endometrial stem/stromal cells
Scientific Reports (2022)
-
Prevention of tumor risk associated with the reprogramming of human pluripotent stem cells
Journal of Experimental & Clinical Cancer Research (2020)
-
Human iPSC banking: barriers and opportunities
Journal of Biomedical Science (2019)
-
MiR-499 Responsive Lethal Construct for Removal of Human Embryonic Stem Cells after Cardiac Differentiation
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.