Abstract
Based on the oxidative stress theory, aging derives from the accumulation of oxidized proteins induced by reactive oxygen species (ROS) in the cytoplasm. Hydrogen peroxide (H2O2) elicits ROS that induces skin aging through oxidation of proteins, forming disulfide bridges with cysteine or methionine sulfhydryl groups. Decreased Ca2+ signaling is observed in aged cells, probably secondary to the formation of disulfide bonds among Ca2+ signaling-related proteins. Skin aging processes are modeled by treating keratinocytes with H2O2. In the present study, H2O2 dose-dependently impaired the adenosine triphosphate (ATP)-induced Ca2+ response, which was partially protected via co-treatment with β-mercaptoethanol, resulting in reduced disulfide bond formation in inositol 1, 4, 5-trisphosphate receptors (IP3Rs). Molecular hydrogen (H2) was found to be more effectively protected H2O2-induced IP3R1 dysfunction by reducing disulfide bonds, rather than quenching ROS. In conclusion, skin aging processes may involve ROS-induced protein dysfunction due to disulfide bond formation, and H2 can protect oxidation of this process.
Similar content being viewed by others
Introduction
The oxidative stress theory of aging suggests that aging results from the accumulation of aberrant proteins in the cytosol, chemical damage to macromolecules, and mitochondrial DNA changes1. This theory began as a proposal that oxygen was poisonous2, followed by the notion that reactive oxygen species (ROS) are a cause of aging, and was eventually modified as the oxidative stress theory in 19723. Among the large number of aging models proposed to date, the oxidative stress hypothesis is considered the most likely, because ROS are continuously produced in aerobic cells. Stepwise reduction of O2 produces several ROS, such as superoxide radicals \((\cdot {{\rm{O}}}_{2}^{-})\), hydrogen peroxide (H2O2), and hydroxyl radicals \((\cdot {\rm{HO}})\). ROS-induced damage of many types of cellular components is supported by a plethora of cellular and biologic data from various model systems and organisms4. Despite the enormous amount of data, however, the molecular mechanisms of aging are not clearly elucidated.
H2O2 is generally used as an instrumental ROS species despite some limitations, such as the complex effects of H2O2 on catalase. Further, H2O2 may be transported across the membrane by aquaporin channels5 and act as superoxide anions, major ROS released from the mitochondria that are converted to H2O2 by superoxide dismutase6, and the increased release of H2O2 mimics the aging process7. Interestingly, H2O2 selectively allows for the oxidization of cysteine or methionine sulfhydryl groups to sulfenic acid or disulfide bonds8, 9, inducing cytoplasm protein dysfunction with the formation of disulfide bonds10. Therefore, H2O2 impairs various physiologic processes via the oxidation of thiols, especially those in proteins. In addition, over the last 20 years, Ca2+ signaling has been identified as crucial for normal physiologic processes11,12,13. The effect of protein oxidation (or effect of aging) on Ca2+ signaling is therefore an important topic. Based on several published papers, most aged cells have decreased Ca2+ responses in the endoplasmic reticulum and decreased Ca2+ release14,15,16. These findings suggest that inositol 1, 4, 5-trisphosphate receptors (IP3Rs)-mediated Ca2+ release from ER must also be decreased14. Accordingly, the aging-related reduction of Ca2+ signaling may be mimicked by H2O2-induced disulfide bond formation.
Recently, a large number of studies have demonstrated that H2 gas selectively reduces ROS, especially hydroxyl radicals, and can strongly slow the rate of aging processes or the progression of aging-related diseases, such as ischemia, reperfusion brain injury, and Parkinson’s Disease6, 17, 18. Since the lifetime of ROS in the tissue is very short19, the effect of H2 on quenching of ROS is through to be limited. If H2 could be made to more effectively reduce the formation of disulfide bonds between SH groups, H2 could more efficiently reduce ROS-induced damage. Among human tissues, the skin is very prone to damage, thus the present study focused on the effect of treating human skin cells, keratinocytes (KC), with H2 to protect against ROS-induced damage. Specifically, the protective effect of H2 treatment against ROS-induced dysfunctional disulfide bond formation and recovery Ca2+ signaling was examined. The data demonstrated that aging processes in KC was found to be selectively oxidized IP3Rs, especially IP3R1-mediated Ca2+ signaling by inducing the formation of H2O2-mediated disulfide bonds in the skin. In addition, a major protective effect of H2 was to reduce disulfide bond formation in the protein caused by oxidative stress, and not by eliminating the generation of ROS in the skin.
Results
Reduced Ca2+ signaling in KC by H2O2
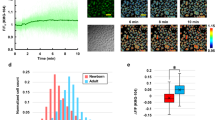
To examine the types of signaling molecules affected by aging in skin cells, different concentrations of H2O2 were used to mimic the aging process induced by ROS accumulation. Approximately 105 KC were plated onto 24-mm coverslips in 3.5-cm2 dishes, incubated for 2 days, pre-stained with 20 μM 2′,7′-dichlorofluorescein-diacetate (DCFH-DA), then treated with 5 μM, 50 μM, or 500 μM H2O2. Imaging of the DCFH-DA staining showed ROS generation in the cells, as evidenced by an increase in fluorescence intensity, and dose-dependent effects of H2O2 exposure were observed with time-lapse recordings (Fig. 1A). The cell morphology was not significantly different between groups, based on differential interference contrast imaging (Fig. 1A). Similar results were observed with 5 μM MitoSOX (Molecular Probes) staining (Fig. 1B) and quantification by flow cytometry (Fig. 1C), which showed that approximately 40% of the cells produced ROS following 500 μM H2O2 exposure, 20% after 50 μM H2O2 exposure, and 5% following 5 μM H2O2 exposure (Fig. 1C). A Ca2+-concentration calibration curve was determined (Fig. 2A). H2O2 decreased ATP (stimulator)-induced Ca2+ elevation and store-operated Ca2+ (SOC) influx in a dose-dependent manner in KC (Fig. 2B). H2O2 likely affected ATP-induced Ca2+ release via the P2Y receptor and then altered SOC channel-mediated Ca2+ entry. To identify the intracellular Ca2+ dynamic changes in detail, the ATP-induced Ca2+ signal was estimated by measuring the black area under the curve (Fig. 2C). Relative to the response to vehicle, the Ca2+ signal was reduced to 72 ± 13% in the 5 µM H2O2 group and to 54 ± 10% in the 50 µM H2O2 group, while the peak in each group was estimated to be 433 ± 43 nM for vehicle, and 383 ± 75 nM for 5 µM H2O2 treatment and 245 ± 21 nM for 50 µM H2O2 treatment (Fig. 2D,E). Interestingly, ATP stimulation did not induce Ca2+ elevation in KC treated with 500 μM H2O2 (Fig. 2C). Here the baseline corresponded to 185 ± 23 nM of Ca2+.
H2O2 dose-dependently induced oxidative stress in KC. ROS generation was stimulated by 5 μM, 50 μM, and 500 μM H2O2 for 30 min at 37 °C in humidified 5% CO2 and visualized using DCFH-DA or MitoSOX pre-staining. ROS are indicated by the fluorescent signals of (A) DCFH-DA or (B) MitoSOX, and cell morphology is shown using differential interference contrast (DIC) microscopy. Scale bars in A and B = 100 µm. (C) Quantification of ROS production from (A) was analyzed using flow cytometry after incubation with H2O2. Each group (black line) was compared with vehicle (gray line) and ROS production was quantified (lower panel) by adding all black area and subtracting all gray area within the period marked by the red line “M”, using FCS Express 4 Image Cytometry Software (De Novo Software) (**P < 0.01; ***P < 0.005).
H2O2 dose-dependently inhibited Ca2+ signaling in KC induced by ATP or IP3 uncaging. (A) Ca2+ calibration curve. Horizontal axis indicates the free concentration of Ca2+ ([Ca2+]i) in standard buffer solution. Vertical axis indicates the ratio of Fluo-4 fluorescence intensity relative to that of baseline fluorescence. (B) Effect of H2O2 on ATP-induced Ca2+ signal. Ca2+ imaging analysis of the ATP-induced Ca2+ response after 30 min pretreatment with vehicle (blue), 5 μM (green), 50 μM (red), and 500 μM (black) H2O2. Ca2+ signals represent the mean value of 20 cells. Twenty-three minutes after two applications of ATP (small black bars) at 10 min intervals in Ca2+-free BSS solution (open bar), CaCl2 was applied extracellularly (large black bar) to increase Ca2+ from 0 to 2 mM to open the store-operated Ca2+ channels (N = 5). (C) The Ca2+ signal from the first simulation with ATP in (B) is shown. (D) Relative Ca2+ signals were measured by calculating the black areas under the curves of the intracellular Ca2+ responses with or without H2O2 treatment in (C) (***P < 0.005 in D and E). (E) Quantification of the peak ATP-induced Ca2+ elevation for all groups in (C). (F) Effect of H2O2 on the IP3-induced Ca2+ response. The concentration of H2O2 used is indicated. F/F0 expresses the Fluo-4 fluorescence (F) relative to baseline fluorescence (F0), which corresponds to changes in the intracellular Ca2+ concentration. Uncaging of caged-IP3 using UV irradiation released IP3 molecules in the cell. The black area under the curve indicates the amount of Ca2+ released by uncaged IP3 (N = 5). (G) The amount of Ca2+ was calculated by quantifying the black areas under the Ca2+ curve (*P < 0.05).
As previously reported, increased phosphorylation of IP3Rs suppresses ATP-induced Ca2+ release and SOC channel-mediated Ca2+ entry20. Therefore, we examined whether H2O2 enhanced IP3Rs phosphorylation levels using caged IP3. Application of uncaged IP3 resulted in Ca2+ elevation in Ca2+-free BSS buffer without involvement of PKC activation; this Ca2+ elevation was reduced by exposure to H2O2, indicating that H2O2 interrupted IP3Rs function (Fig. 2F). The total Ca2+ response is represented by a bar chart, quantified by measuring the black areas under the Ca2+ response curves (Fig. 2G). Compared with vehicle, H2O2 dose-dependently reduced the uncaged IP3-induced Ca2+ signaling, and the Ca2+ response was almost completely blocked by 500 µM H2O2 (Fig. 2G). The rate of Ca2+ decay was similar between the ATP- and uncaged IP3-induced Ca2+ responses (Fig. 2C,F). These findings indicate that H2O2 reduced IP3Rs functionality, resulting in the suppression of Ca2+ signaling due to oxidative stress. Therefore, the possibility of increased phosphorylation of IP3Rs can be excluded as a mechanism underlying the H2O2-dependent reduction in the Ca2+ response in KC.
ATP-induced Ca2+ signaling was inhibited by disulfide bond formation in KC IP3Rs
Because the suppression of the uncaged IP3-induced Ca2+ elevation did not result from IP3Rs phosphorylation, we hypothesized a novel model to explain the findings. It is worth noting that oxidative stress can induce disulfide bond formation, thus impairing molecular chaperoning, translation, glycolysis, cytoskeletal structure, cell growth, and signal transduction10. That H2O2 can oxidize cysteine or methionine sulfhydryl groups to sulfenic acid or disulfide bonds8, 9 raised the possibility that H2O2 decreased the Ca2+ signal by eliciting IP3Rs disulfide bond formation. This possibility was examined using the reduction agent β-mercaptoethanol (2-ME; Sigma-Aldrich). ATP-induced Ca2+ elevation was completely inhibited by 500 μM H2O2 (Fig. 3A,B), but the Ca2+ response was partially recovered, increasing from 0% to 43 ± 3.5% when 10 mM 2-ME and H2O2 were applied together (500 μM H2O2 + 2-ME; Fig. 3A,B). 2-ME increased the Ca2+ peak from 198 nM (500 μM H2O2) to 248 (500 μM H2O2 + 2-ME; Fig. 3C). To further examine the effect of H2O2 on IP3Rs, caged IP3 was utilized to detect whether only IP3Rs functionality was reduced by H2O2-induced disulfide bond formation. As expected, the Ca2+ released in response to uncaged IP3 stimulation was largely recovered, increasing from 3 ± 3% to 62 ± 5% (500 μM H2O2 + 2-ME) relative to vehicle, when 2-ME was applied with H2O2 (Fig. 3D,E). When 2-ME and H2O2 were applied together, however, the H2O2-induced decrease was protected to some extent (Fig. 3D). Thus, it is possible to determine 500 μM H2O2-induced reduction of the Ca2+ signal was dependent on disulfide bond formation in IP3Rs. These results suggest that disulfide bond formation in IP3Rs induces a conformational change and obstructs IP3 binding to IP3Rs under oxidative stress. Because the aging process is associated with both oxidative stress and Ca2+ deficiency14, it is reasonable to suggest that ROS-induced protein disulfide bond formation may contribute to skin aging.
β-mercaptoethanol (2-ME) restored Ca2+ response in KC in exposure to H2O2. (A) Ca2+ signal in each group (pretreatment with vehicle, 500 μM H2O2, and 500 μM H2O2 + 2-ME) was induced by addition of ATP (black bars). (B,C) Quantification of the black areas representing ATP-induced Ca2+ signals (B) and the peak of the intracellular Ca2+ responses (C) shown in (A) (N = 3). (D) Ca2+ signal in each group (pretreatment with vehicle, 500 μM H2O2, 500 μM H2O2 + 2-ME) was stimulated by photolysis-induced uncaging of caged-IP3. (E) Quantification of the black areas in (D) represent the amount of Ca2+ released by the uncaged IP3 (*P < 0.05; **P < 0.01; ***P < 0.005).
H2 gas-containing media protected KC against H2O2-induced disulfide bond formation in IP3Rs
Recently, H2-containing medium was reported to protect against ROS-induced damage by buffering the effects of oxidative stress or superoxide formation21. H2 was further investigated to determine whether it protects against H2O2-oxidized IP3Rs function and reduction of the resulting Ca2+ signal. Preparation and how to use H2-containing BSS is described in the Materials and Methods section. The H2-containing BSS was stored in an open glass bottle, and the hydrogen concentration reduction in the media was measured every hour for 12 h using a hydrogen-sensitive electrode. The half-life of H2 was estimated to be approximately 6 h in BSS (Fig. 4A). Because the lower limitation of effective H2 concentration was estimated as 0.08 ppm18, all the experiments were designed to finish within a few hours. To examine the effect of H2-BSS on H2O2-decreased IP3Rs function, we evaluated the following four groups: vehicle, 500 μM H2O2, 500 μM H2O2 with 2-ME, and 500 μM H2O2 with H2 in BSS buffer. Ca2+ release in KC was induced with the addition of 500 μM ATP. Both the 500 μM H2O2 with 2-ME and the 500 μM H2O2 with H2 groups produced similar results, in that the Ca2+ was 65 ± 14% (500 μM H2O2 + 2-ME) or 79 ± 12% (500 μM H2O2 + H2) relative to that of the vehicle group (Fig. 4B,C) and peaked at 385 ± 45 nM (500 μM H2O2 + 2-ME) or 407 ± 73 nM (500 μM H2O2 + H2) (Fig. 4B,D). There was negligible Ca2+ response with exposure to 500 μM H2O2 alone (Fig. 4B,C). To further evaluate the effect of H2-BSS on 500 μM H2O2-induced IP3Rs dysfunction, uncaged IP3 was tested to confirm whether ROS selectively affected IP3Rs. As expected, IP3Rs function was partially protected by treatment with H2. In the focal IP3 uncaging experiment, the IP3-mediated Ca2+ signal in 2-ME treated cells was 71 ± 7% (500 μM H2O2 + 2-ME) that of vehicle and the Ca2+ signal of H2-treated cells was 80 ± 3% (500 μM H2O2 + H2; Fig. 4E,F). These results suggest that H2-containing BSS protected the ATP-induced Ca2+ signal in skin by reducing the H2O2-induced disulfide bonds in IP3Rs and had more effective than 2-ME in restoring H2O2-induced Ca2+ suppression.
H2 was more effective than 2-ME in restoring H2O2-induced Ca2+ suppression in KC. (A) Time-dependent reduction of H2 content in BSS buffer. (B) Mean Ca2+ response to ATP application in each group: vehicle, 500 μM H2O2, 500 μM H2O2 + 2-ME, and 500 μM H2O2 + H2. (C) The Ca2+ response was quantified by measuring the black areas under the Ca2+ curve. (D) The peak of ATP-induced intracellular Ca2+ signal (N = 5). (E) Protective effect of H2-BSS on H2O2-induced reduction of the uncaged IP3-stimulated Ca2+ response. Ca2+ signals are shown as black areas after uncaged IP3 stimulation. (F) Quantification of Ca2+ signals by measuring the black areas under the Ca2+ response curve (N = 5) (*P < 0.05; **P < 0.01; ***P < 0.005).
H2-BSS protected against H2O2-induced damage of IP3R1 by reducing disulfide bond formation, not by quenching ROS in KC
Since 2-ME-BSS and H2-BSS had different effects on 500 μM H2O2-induced damage of Ca2+ release with IP3Rs in KC, we performed additional experiments to make the difference between the effects of H2 and 2-ME. Briefly, cells were pretreated with 2-ME or H2 gas for 30 min and compared (Fig. 5A). Protection of the Ca2+ signaling system following pretreatment with H2-BSS (500 μM H2O2 + pre-H2) was 185 ± 19% that following pretreatment with vehicle (Fig. 5B and C), while no Ca2+ signal was observed following pretreatment with 2-ME (500 μM H2O2 + pre-2-ME) (Fig. 5B). The enormous difference between the effects of pre-treatment with H2-BSS and 2-ME may be due to the different mechanisms by which they reduce disulfide bond formation in IP3Rs. 2-ME is a strong reducing agent that reduces all proteins in the cell, resulting in considerable conformational changes and thereby dysfunction of all functional proteins. In order to confirm this possibility, we examined if pretreatment with 2-ME could reduce all proteins to the extent that no Ca2+ signal can be induced by ATP stimulation. Additional assays were performed to confirm whether H2-BSS protected against H2O2 damage by reducing disulfide bond formation. In this experiment, non-reducing sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and Western blot analysis were performed as described previously10. We focused on examining the formation of disulfide bonds of IP3Rs with three subtypes: IP3R1, IP3R2 and IP3R3 via treating the specific antibodies20, 22, 23. All IP3Rs display the molecular weight over 250 kDa. As shown in Fig. 5D, IP3R1 with disulfide bonds, as detected by IP3R1 antibody and migration in a broad band secondary to oxidized S-S formation. However, IP3R2 and IP3R3 did not present clear disulfide bonds formation based on the similar molecular weight of broad band (Fig. 5D). The formation of disulfide bonds affects the conformation and electrophoretic mobility of redox-sensitive proteins24,25,26. Proteins forming intra-molecular disulfide bonds exhibit distinct types of migration in non-reducing (without 2-ME in protein loading buffer) SDS-PAGE and reducing SDS-PAGE (with 2-ME in protein loading buffer)10. As expected, KC exposed to H2O2 had IP3R1 containing disulfide bonds, as evidenced by the lower broad band when electrophoresed under non-reducing conditions, but KC treated with a reducing agent (2-ME, H2 or pre- H2) had a higher band and lightly stained bands similar to the vehicle-treated KC (Fig. 5D). Western blot analysis also confirmed no effect on the level of IP3R1 phosphorylation (Fig. 5E). This finding suggests that the reducing agents potentially altered the conformation of the IP3R1 by breaking disulfide bonds, yet neither reducing agent completely reduced IP3R2 and IP3R3 disulfide bonds in the cell. This result explains why the IP3Rs-mediated Ca2+ signal was not completely protected with application of 2-ME or H2. These reducing agent may not be able to reach inside of protein structure, this was conjoined by prolonged treatment by H2-BSS for 30 more minutes, as shown in Fig. 5B and C. As previously established, superoxide dismutase converts superoxide anion radicals into H2O2, which is detoxified into H2O by either glutathione peroxidase or catalase27. Our examination of ROS production using flow cytometry revealed that neither H2-BSS nor 2-ME reduced ROS production induced by 500 μM H2O2 in KC (Fig. 5F and G). The finding that H2O2 was not detoxified with H2 is not similar to the results of a previous study, which showed that H2 reduces hydroxyl radicals ∙HO17, because H2O2 can be metabolized into\(\cdot {\rm{HO}}\) via catalysis28.
H2 decreased disulfide bond formation but not ROS production in H2O2-exposed KC. (A) Effect of pretreatment with 2-ME-BSS or H2-BSS on the H2O2-induced reduction of ATP-induced Ca2+ signal in KC. Experimental design for treatment in each group: vehicle, 500 μM H2O2, 500 μM H2O2 + pre-2-ME, and 500 μM H2O2 + pre-H2. Ca2+ signal was induced by ATP stimulation in each group (B) and calculated by the black-colored areas under the Ca2+ curve (N = 3) (***P < 0.005) (C). (D) Expression of IP3Rs under non-reduced and reduced conditions is shown using SDS-PAGE and Western blot. Protein extracts from each group were resolved in non-reducing (without 2-ME) SDS-PAGE (7% gel), transferred onto Hybond-P polyvinylidene fluoride membranes, and incubated with antibodies against IP3R1, IP3R2 and IP3R3 and α-Tublin. As a control, the lysates were reduced using 100 mM 2-ME (reduced; right lanes). Proteins that form disulfide bonds exhibit faster migration and thus appear as lower molecular weight bands. Quantify of IP3Rs expression in reduced gel as shown in right panel in represent the average of three independent experiments. (E) Western blot analysis of the expression of IP3R1 and phosphorylated IP3R1 (p- IP3R1) after incubation of the cells with or without 500 μM H2O2. Treatment with 500 μM H2O2 had no effect on the density of the bands for phosphorylated-IP3R1 (p-IP3R1), IP3R1 or α-Tublin. Quantify of expression in IP3R1 and p-IP3R1 as shown in right panel in represent the average of three independent experiments. (F) Flow cytometry was used to estimate the (G) ratio of ROS generation in each group (*P < 0.05; ***P < 0.005).
To detect the formation of disulfide bonds in IP3R-1, we employed liquid chromatography tandem-mass spectrometry (LC-MS/MS). The sulfhydryl residue Cys or Met, through which disulfide bridges can be produced by H2O2, is labeled with maleimide-PEO2-biotin (MPB)29. We examined whether MPB was applicable to assay H2O2-induced intermolecular disulfide bond formation in receptor protein-tyrosine phosphatase α (RPTPα). MPB-containing peptides signal located the intermolecular disulfide bond to the Cys-723 of RPTPα, and the counts of H2O2-induced MPB-containing peptides was higher than the counts in the group of vehicle or H2O2 with 2-ME (Supplementary Table S1). However, identification of the disulfide bonds formation in Cys residues with delta mass 646.24 (Cys+MPB) can’t search the MPB-modified Cys peptides. It could be either the tiny signal of MPB-modified Cys difficult detection by LC-MS/MS, or Cys is not the major residue in H2O2-induced disulfide bonds formation. Consequently, we demonstrated whether H2O2 elicited disulfide bridges formation in methionine-containing peptides. As shown in Table 1, MPB-modified residue locates in methionine and has mostly increased level of counts in H2O2 treatment. H2 partially recovered (decreased) the counts of MPB-containing peptides (Table 1). Besides, we attempted to recognize each MPB-modified methionine in IP3R1 protein via Cryo-EM and crystal structure models, which were established from Fan et al. and Seo et al. respectively30, 31. In this huge IP3R1 3D structure, most of MPB-modified methionines located in hydrogen-bonding networks not in functional domains (Fig. 6A); Met-5, Met-415 and Met-581 located in loop and sheet, but only Met-415 located in IP3 binding domain, the boundary between sheet and loop (Fig. 6B). H2 treatment restrained the level of counts in MPB-modified Mets, explained why H2 performed the Ca2+ signal significant recovery due to reduction of disulfide bonds formation.
The location of each MBP-modified methionine in IP3R1 structure. (A) Cryo-EM imaging of IP3R1 displays the sheets (yellow), helixes (red) and loops (green); each MBP-modified methionine listed Table 1 is labeled with pink spot in IP3R1. However, majority of MBP-modified Mets are constructed by hydrogen linkages (pink spots in dark empty spaces). Met-5, Met-415 and Met-581 are located in loop and sheet respectively. (B) Crystal structure of IP3 binding domain from IP3R1 shown in Met-415 (blue) is in the boundary between sheet and loop. Both Cryo-EM imaging and crystal imaging have been used to represent the information from Protein Data Bank (PDB) and the 3D structure built using PyMOL software.
Discussion
The findings of the present study reveal the potential association of the skin aging process with Ca2+ signal dysfunction via the formation of H2O2-induced disulfide bonds10 in IP3Rs that lead to selective damage of the IP3Rs. Co-treatment with 2-ME and 500 μM H2O2 decreased the disulfide bond formation, evidenced by the protection of ATP-induced Ca2+ release in KC with 2-ME. This provides clear evidence that oxidative stress-related skin damage is associated with Ca2+ signaling defects, similar to 2-ME, H2 gas in BSS decreased IP3R1 disulfide bond formation due to ROS exposure but not IP3R2 and IP3R3. The major function of H2 in BSS in skin was the protection of ATP-induced Ca2+ signaling, but not by direct quenching of ROS. ATP is thought to be an external 1st cell-to-cell communication around the other cells equipped with GPCR. Because BSS media has no Cu and Fe to make hydroxyl radical, contribution of hydroxyl radical will be smaller than usual when the cells are treated by H2O2. Previous reports indicate that the presence of reducing agents decreased number of disulfide bonds, resulting in a loss of cross-link-induced stability produced by the chemical microenvironment32. It is reasonable to assume that the skin aging process is associated with ROS accumulation in the skin, which leads to IP3Rs dysfunction by inducing conformational changes due to disulfide bond formation.
Recent study has pointed out the treatment of H2 in acute erythematous skin diseases, which are associated with large ROS accumulation in KC33. H2 improved significantly in erythema of these patients and did not affect physiological parameters and deterioration of the blood chemistry33. Although elimination of ROS produced by H2O2 is also performed in H2, our study firstly indicates H2 also reduces disulfide bond formation and restores protein function. However, our finding revealed IP3R1 but not IP3R2 and IP3R3 is recovered the function by H2 treatment. It may be possible due to the different distribution of S-S bond formation in each subtype of IP3Rs. H2O2 and H2 independently oxidize and/or reduce each subtype of IP3Rs; therefore, the exposed site (Cys or Met) of each subtype of IP3Rs could be affected by H2O2 and H2 independently or correlatively. This is reasonable to explain why IP3R1 displays more obvious in disulfide bond formation and is more efficient for H2 reduction.
Besides, H2 acts also as a reducing agent at a lower concentration, but the amount of H2 was sufficient to disrupt metabolic oxidation reducing reactions or to interrupt ROS-induced disruption of cell signaling17. H2 induces superoxide dismutases (SODs) and heat shock proteins (HSPs) activity to quench ROS production6 and may thereby completely protect the IP3Rs Ca2+ signal. Nevertheless, pretreatment with H2 failed to decrease H2O2-induced ROS production. Another possible mechanism underlying the H2-induced elimination of ROS damage of IP3Rs is the activation of glutathione/thioredoxin systems, which reduces H2O2-induced disulfide bond formation. To date, however, it is not clear whether H2 facilitates glutathione/thioredoxin system activity, although studies have reported that glutathione/thioredoxin systems are involved in the modulation of disulfide bond formation during oxidative stress10, 34.
In the skin aging process, UV irradiation increases ROS production6, 35. Superoxide radicals (∙O2 −), hydroxyl radicals (∙HO), and peroxyl radicals (LOO∙), singlet oxygen (1O2), and hydrogen peroxide (H2O2) are involved in UV-stimulated oxidative stress6. According to a previous report17, however, H2 selectively reduces the hydroxyl radical, yet it is not involved in the efficient elimination of oxidative-stress-induced damage due to UV irradiation. The results of the present study indicate that, rather, a major mechanism underlying H2 effects is to protect IP3Rs-mediated Ca2+ signaling by reducing ROS-induced disulfide bond formation. IP3Rs-mediated Ca2+ signaling has a central role in several physiologic processes, such as fertilization, proliferation, muscle contraction, cell metabolism, vesicle and fluid secretion, and information processing11. As aging-induced dysfunction of Ca2+ signaling leads to cell death or genome instability, H2 can protect skin cells from destruction by maintaining the Ca2+ signal and thus preserving normal physiologic conditions. The results of the present study clearly demonstrate the role of H2 in protecting against the aging process in skin and provide evidence to support further investigation of the clinical applicability of H2.
Methods
Cell culture
Human primary KC were isolated from foreskins obtained via routine circumcision as described previously36. The skin specimens were washed with phosphate-buffered saline (PBS), cut into small pieces, and cultured in medium containing 0.25% trypsin (Gibco BRL) overnight at 4 °C. The epidermal sheet was lifted from the dermis utilizing fine forceps. The epidermal cells were then pelleted using centrifugation (500 g, 10 min) and separated into individual cells by repeated aspiration. The KC were incubated in serum-free KC growth medium, supplemented with human recombinant epidermal growth factor and bovine pituitary extract (5 μg/mL each), insulin (5 μM/mL), and hydrocortisone (5 μg/mL; Gibco BRL) at 37 °C in humidified 5% CO2, and supplemented with KC growth medium for five generations20, 37.
Calcium imaging
The intracellular Ca2+ response was induced by application of ATP (Sigma-Aldrich), according to methods previously described20. Before the experiments, cells were stained with 1 μM Fluo-4-AM (Molecular Probes) at 37 °C for 20 min and then washed with BSS buffer (5.4 mM KCl, 5.5 mM d-glucose, 1 mM MgSO4, 130 mM NaCl, 20 mM Hepes pH 7.4, and 2 mM CaCl2). Intracellular Ca2+ concentrations were estimated based on the ratio of fluorescence intensities emitted upon excitation with consecutive 3-s pulses of 488-nm light at a resolution of 1376 × 1038 pixels using an Olympus Cell^R IX81 fluorescence microscope (Olympus) equipped with an MT 20 illumination system (Olympus) and UPLanApo 10 × objective lens. The intracellular Ca2+ concentration was estimated based on calibration curves as follows. A Ca2+ calibration curve was created using a Ca2+ Calibration Buffer kit (Molecular Probes). Intracellular Ca2+ ([Ca2+]i) was calculated from Fluo-4 excited at 488 nm and imaged using an Olympus Cell^R IX81 fluorescence microscope and UPLanApo 10 × objective lens at 20 °C. Fluo-4 signals were calibrated by measuring the fluorescence intensity from microcuvettes containing 10 mM K2-EGTA (pH 7.20) buffered to various [Ca2+] levels. Ca2+ concentration was calculated using the following formula: [Ca2+]i = KD *(F − Fmin/Fmax − F). Plotting the fluorescence intensity versus [Ca2+] yielded the calibration curve with the formula of: [Ca2+]i = KD *(F − Fmin/Fmax − F), where KD = 345 nM, F = Fluo-4 intensity, Fmax = 640, and Fmin = 21.7 for Fluo-4.
Focal uncaging
Caged compounds were uncaged to investigate modifications of IP3Rs by photolysis using UV light (300–400 nm) as described previously20. For photolytic uncaging, an Olympus FV1000 MPE multiphoton laser scanning microscope equipped with an argon laser was used to produce a collimated light beam as the principal uncaging laser line at λ = 408 nm. To detect the effect of H2O2 (Sigma-Aldrich) on IP3Rs in Ca2+ pathways, 105 primary KC were plated on 2.4-mm coverslips in a 4-cm dish. For H2O2 treatment, cells were pretreated with 1 μM caged IP3 (Molecular Probes) for 2 h38 and stained with the Ca2+ dye Fluo-4 to analyze changes in intracellular Ca2+ levels. Ca2+-induced fluorescence was observed using an Olympus FV1000 laser-scanning microscope. Caged IP3 was uncaged by illumination with UV light (λ = 408 nm) and the released IP3 molecule was immediately able to bind to the IP3Rs.
Western blot analysis
Western blot analyses were performed utilizing whole-cell lysates. Briefly, cells were lysed by incubating for 30 min on ice in M-PER Mammalian Protein Extraction Reagent (Thermo Fisher Scientific), containing proteinase and phosphatase inhibitors. Cell debris was removed via centrifugation at 10,000 g for 10 min at 4 °C. The protein concentration of cell lysates was determined using the Bradford method (Bio-Rad). Proteins (100 μg) in cell lysates were resolved using sodium dodecyl sulfate-polyacrylamide gel electrophoresis in a 7% gel with or without 2-ME and then transferred to a Hybond-P polyvinylidene fluoride membrane (Amersham Biosciences). The membrane was first incubated with primary antibodies against IP3R1 (Cell Signaling Technology), phospho-IP3R1 (Cell Signaling Technology), IP3R2 (Merck Millipore), IP3R3 (BD Biosciences) and α-Tublin (Santa Cruz Biotech), and then with horseradish peroxidase-conjugated secondary antibodies. Immunoreactive proteins were visualized using enhanced chemiluminescence reagents (Amersham Biosciences).
ROS measurement using flow cytometry
DCFH-MA (Sigma-Aldrich) staining was used to quantify ROS generation from the cells. Briefly, cells were stained with 20 μM DCFH-DA at 37 °C for 20 min and then washed with BSS buffer. The cells were collected after application of H2O2, reducing agent 2-ME, or H2 gas-containing BSS (H2-BSS) at 37 °C for 30 min. ROS production was determined using flow cytometry (LSR II, BD) with fluorescence emission at 488 nm.
Disulfide-bond labeling and in solution digestion
To investigate the intracellular disulfide bonds formation, we modified the labeling protocol from Clive Metcalfe et al. study29. Briefly, the cells were treated 2.5 mM Methyl-PEO12-maleimide (MPM, Thermo Fisher Scientific) with 0.05% TritonX-100 (Sigma-Aldrich) in PBS containing 1% bovine serum albumin (BSA, Sigma-Aldrich) at 4 °C for 30 min; MPM can entry the cell and bind the intracellular free sulfhydryl groups with TritonX-100 treatment. After washing the cells with PBS containing 1% BSA at 25 °C for three times (1 min/time), the cells were reduced with 2.5 mM tris(2-carboxyethyl)phosphine (TCEP, Sigma-Aldrich) and 10 mM dithiothreitol (DTT, Sigma-Aldrich) at 25 °C for 30 min. After washing (1% BSA in PBS) three times, cells were labelled with 2.5 mM Maleimide-PEO2-biotin (MPB, Thermo Fisher Scientific) in PBS containing 1% BSA at 4 °C for 30 min, and then cells were washed for three times. Total protein lysates were collected for in solution digestion after adding M-PER protein extraction reagent (Thermo Fisher Scientific).
We utilized nitrogen gas to blow-dry the total protein lysates to protein pellet, and suspended protein pellet with 0.1% RapiGest SF solution (Waters). Protein sample was treated with 20 mM iodoacetamide (IAA, Sigma-Aldrich) in the dark for 30 min and then desalted by using Amicon® Ultra-0.5 Centrifugal Filter Device (Merck Millipore). After incubating with trypsin for peptides digestion overnight, protein sample treated with 1% of formic acid (FA, Sigma-Aldrich) and confirmed pH <2 with a pH paper. The tryptic peptide sample was analyzed disulfide bonds labeling by use of quantitative proteomics techniques utilizing serially coupled lipid chromatography data-independent parallel fragmentation mass spectrometry (LC/MSE).
LC/MSE analysis
Quantitative analysis will be performed essentially on a Waters Xevo G2 qTof mass spectrometer (Waters). In brief, the tryptic peptide sample will be chromatographically separated on M-class UPLC separations module (Waters) incorporating 50 femtomole tryptic digested BSA as the internally spiked protein quantification standard. Peptide elution will be executed through a 75 μm × 25 cm BEH C-18 column (Waters) under gradient conditions at a flow rate of 300 nL/min over 70 min at 40 °C. The mobile phase will be composed of acetonitrile as the organic modifier and formic acid (0.1% v/v) for molecule protonation. Mass spectrometry was performed on Xevo G2 qTof (waters) instrument equipped with a nanoflow electrospray ionization (ESI) interface and operated in the data-independent collection mode (MSE). Parallel ion fragmentation will be programmed to switch between low (4 eV) and high (15–45 eV) energies in the collision cell, and data will be collected from 300 to 2000 m/z utilizing glu-fibrinopeptide B (Sigma-Aldrich, m/z 785.8426) as the separate data channel lock mass calibrant. Data will be processed with ProteinLynx GlobalServer v3.0 (waters) for qualification and Progenesis QI for proteomics (Waters) for relative quantification, respectively. Deisotoped results will be searched for protein association and modification from the Uniprot (www.uniprot.org) human protein database.
H2 gas-containing BSS and assessment of H2 content
Hydrogen-containing water (H2 water) was produced using an Aurora H2 water-making machine (Kyoyo Company, Japan). To generate H2-containing BSS, First, H2 water with one-tenth concentration of BSS was made from sterile water using the Aurora machine. Then, H2 water was mixed with BSS stock solution to make final H2-containing BSS (pH = 7.4). The H2 content was measured using a hydrogen electrode (Kyoyo Company, Japan).
Ethical approval
Human primary KC from foreskins was approved from the Institutional Review Board/Ethics Committee (IRB) in Kaohsiung Medical University Chung-Ho Memorial Hospital, number KMUH-IRB-960119. All methods were performed in accordance with the relevant guidelines and regulations. Informed consent of all participants was obtained. A total number of 32 samples (foreskins) were collected from 2007 to 2008 of which were further analyzed by isolating KC from skin.
Statistical analysis
GraphPad Prism (La Jolla, CA) was used to generate bar charts; error bars indicate standard deviations. A one-way, two-tailed analysis of variance (ANOVA) was also utilized to compare means of each group. A P-value of less than 0.05 for differences between groups was considered statistically significant.
References
Muller, F. L., Lustgarten, M. S., Jang, Y., Richardson, A. & Van Remmen, H. Trends in oxidative aging theories. Free Radic. Biol. Med. 43, 477–503, doi:10.1016/j.freeradbiomed.2007.03.034 (2007).
Cross, C. E. et al. Oxygen radicals and human disease. Ann. Intern. Med. 107, 526–545 (1987).
Polis, B. D., Grandizio, M. & Polis, E. Some in vitro and in vivo effects of a new prostaglandin derivative. Adv. Exp. Med. Biol. 33, 213–220 (1972).
Brookes, P. S., Yoon, Y., Robotham, J. L., Anders, M. W. & Sheu, S. S. Calcium, ATP, and ROS: a mitochondrial love-hate triangle. Am. J. Physiol. Cell Physiol. 287, C817–833, doi:10.1152/ajpcell.00139.2004 (2004).
Kucherenko, Y. V., Huber, S. M., Nielsen, S. & Lang, F. Decreased redox-sensitive erythrocyte cation channel activity in aquaporin 9-deficient mice. J. Membr. Biol. 245, 797–805, doi:10.1007/s00232-012-9482-y (2012).
Noda, M., Fujita, K., Lee, C. H. & Yoshioka, T. The principle and the potential approach to ROS-dependent cytotoxicity by non-pharmaceutical therapies: optimal use of medical gases with antioxidant properties. Curr. Pharm. Des. 17, 2253–2263 (2011).
Sohal, R. S. & Sohal, B. H. Hydrogen peroxide release by mitochondria increases during aging. Mech. Ageing Dev. 57, 187–202 (1991).
Thomas, J. A. & Mallis, R. J. Aging and oxidation of reactive protein sulfhydryls. Exp. Gerontol. 36, 1519–1526 (2001).
Kudryavtseva, E. V., Sidorova, M. V., Ovchinnikov, M. V. & Bespalova, Z. D. Hydrogen peroxide for disulfide bridge formation in methionine-containing peptides. Journal of peptide science: an official publication of the European Peptide Society 6, 208–216, doi:10.1002/(SICI)1099-1387(200005)6:5<208::AID-PSC241>3.0.CO;2-V (2000).
Cumming, R. C. et al. Protein disulfide bond formation in the cytoplasm during oxidative stress. The Journal of biological chemistry 279, 21749–21758, doi:10.1074/jbc.M312267200 (2004).
Berridge, M. J. Inositol trisphosphate and calcium signalling mechanisms. Biochim. Biophys. Acta 1793, 933–940, doi:10.1016/j.bbamcr.2008.10.005 (2009).
Karlstad, J., Sun, Y. & Singh, B. B. Ca(2+) signaling: an outlook on the characterization of Ca(2+) channels and their importance in cellular functions. Adv. Exp. Med. Biol. 740, 143–157, doi:10.1007/978-94-007-2888-2_6 (2012).
Lory, P., Bidaud, I. & Chemin, J. T-type calcium channels in differentiation and proliferation. Cell calcium 40, 135–146, doi:10.1016/j.ceca.2006.04.017 (2006).
Decuypere, J. P. et al. IP(3) Receptors, Mitochondria, and Ca Signaling: Implications for Aging. J. Aging Res. 2011, 920178, doi:10.4061/2011/920178 (2011).
Mattson, M. P. ER calcium and Alzheimer’s disease: in a state of flux. Sci. Signal. 3, pe10, doi:10.1126/scisignal.3114pe10 (2010).
Ermak, G. & Davies, K. J. Calcium and oxidative stress: from cell signaling to cell death. Mol. Immunol. 38, 713–721 (2002).
Ohsawa, I. et al. Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals. Nat. Med 13, 688–694, doi:10.1038/nm1577 (2007).
Fujita, K. et al. Hydrogen in drinking water reduces dopaminergic neuronal loss in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model of Parkinson’s disease. PloS one 4, e7247, doi:10.1371/journal.pone.0007247 (2009).
Novo, E. & Parola, M. Redox mechanisms in hepatic chronic wound healing and fibrogenesis. Fibrogenesis & tissue repair 1, 5, doi:10.1186/1755-1536-1-5 (2008).
Hsu, W. L. et al. Differential effects of arsenic on calcium signaling in primary keratinocytes and malignant (HSC-1) cells. Cell calcium 52, 161–169, doi:10.1016/j.ceca.2012.05.007 (2012).
Ohsawa, I. et al. Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals. Nature medicine 13, 688–694, doi:10.1038/nm1577 (2007).
Perez, P. J., Ramos-Franco, J., Fill, M. & Mignery, G. A. Identification and functional reconstitution of the type 2 inositol 1,4,5-trisphosphate receptor from ventricular cardiac myocytes. The Journal of biological chemistry 272, 23961–23969 (1997).
Maranto, A. R. Primary structure, ligand binding, and localization of the human type 3 inositol 1,4,5-trisphosphate receptor expressed in intestinal epithelium. The Journal of biological chemistry 269, 1222–1230 (1994).
Cumming, R. C. et al. Fanconi anemia group C protein prevents apoptosis in hematopoietic cells through redox regulation of GSTP1. Nat. Med 7, 814–820, doi:10.1038/89937 (2001).
Delaunay, A., Pflieger, D., Barrault, M. B., Vinh, J. & Toledano, M. B. A thiol peroxidase is an H2O2 receptor and redox-transducer in gene activation. Cell 111, 471–481 (2002).
Manalo, D. J., Lin, Z. & Liu, A. Y. Redox-dependent regulation of the conformation and function of human heat shock factor 1. Biochemistry 41, 2580–2588 (2002).
Turrens, J. F. Mitochondrial formation of reactive oxygen species. J. Physiol. 552, 335–344, doi:10.1113/jphysiol.2003.049478 (2003).
Petersen, A. B., Gniadecki, R., Vicanova, J., Thorn, T. & Wulf, H. C. Hydrogen peroxide is responsible for UVA-induced DNA damage measured by alkaline comet assay in HaCaT keratinocytes. Journal of photochemistry and photobiology. B, Biology 59, 123–131 (2000).
Metcalfe, C., Cresswell, P., Ciaccia, L., Thomas, B. & Barclay, A. N. Labile disulfide bonds are common at the leucocyte cell surface. Open biology 1, 110010, doi:10.1098/rsob.110010 (2011).
Fan, G. et al. Gating machinery of InsP3R channels revealed by electron cryomicroscopy. Nature 527, 336–341, doi:10.1038/nature15249 (2015).
Seo, M. D. et al. Structural and functional conservation of key domains in InsP3 and ryanodine receptors. Nature 483, 108–112, doi:10.1038/nature10751 (2012).
Keten, S., Chou, C. C., van Duin, A. C. & Buehler, M. J. Tunable nanomechanics of protein disulfide bonds in redox microenvironments. J. Mech. Behav. Biomed. Mater. 5, 32–40, doi:10.1016/j.jmbbm.2011.08.017 (2012).
Ono, H. et al. Hydrogen(H2) treatment for acute erythymatous skin diseases. A report of 4 patients with safety data and a non-controlled feasibility study with H2 concentration measurement on two volunteers. Medical gas research 2, 14, doi:10.1186/2045-9912-2-14 (2012).
Aon, M. A. et al. Glutathione/thioredoxin systems modulate mitochondrial H2O2 emission: an experimental-computational study. J. Gen. Physiol. 139, 479–491, doi:10.1085/jgp.201210772 (2012).
Rittie, L. & Fisher, G. J. UV-light-induced signal cascades and skin aging. Ageing Res. Rev. 1, 705–720 (2002).
Lee, C. H. et al. Mechanistic correlations between two itch biomarkers, cytokine interleukin-31 and neuropeptide beta-endorphin, via STAT3/calcium axis in atopic dermatitis. Br. J. Dermatol. 167, 794–803, doi:10.1111/j.1365-2133.2012.11047.x (2012).
Hsu, W. L. et al. Derinat Protects Skin against Ultraviolet-B (UVB)-Induced Cellular Damage. Molecules 20, 20297–20311, doi:10.3390/molecules201119693 (2015).
Tertyshnikova, S. & Fein, A. Inhibition of inositol 1,4,5-trisphosphate-induced Ca2+ release by cAMP-dependent protein kinase in a living cell. Proc. Natl. Acad. Sci. USA. 95, 1613–1617 (1998).
Acknowledgements
We thank the Center for Research Resources and Development at Kaohsiung Medical University for providing the use of the confocal microscope, Flow Cytometry and Olympus Cell^R IX81 fluorescence microscope. This work was supported by the slow aging program, “Aim for the Top Universities Grant”, KMU-TP104D04 and KMU-TP105D02 (Tohru Yoshioka received) at Kaohsiung Medical University, kmtth-104-010 (Ching-Ying Wu received) at Kaohsiung Municipal Ta-Tung Hospital, CLFHR10509 at Chi Mei Medical Center (Jui-Lin Liang received), and the Ministry of Science and Technology of Taiwan, MOST, 104-2314-B-037-060 and 105-2628-B-037-006-MY3 (Ching-Ying Wu received). A part of this funding was supported by Glyen-Po Chen (Tohru Yoshioka received).
Author information
Authors and Affiliations
Contributions
C.Y.W., W.L.H., M.H.T., J.L.L., C.Y.L., S.J.Y., and T.Y. designed the experiments. H.S.Y. prepared the foreskins and J.H.L. and C.J.Y. completed the keratinocyte isolation from the foreskins and performed the experiments. M.H.T. performed the statistical analysis using GraphPad Prism. M.N. and C.H.C. provided advice on the project. C.Y.W., W.L.H., J.L.L., M.N., C.H.C., S.J.Y., and T.Y. wrote the manuscript. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wu, CY., Hsu, WL., Tsai, MH. et al. Hydrogen gas protects IP3Rs by reducing disulfide bridges in human keratinocytes under oxidative stress. Sci Rep 7, 3606 (2017). https://doi.org/10.1038/s41598-017-03513-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-03513-2
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.