Abstract
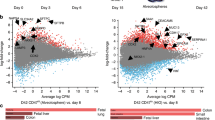
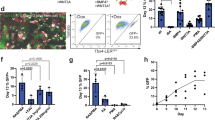
Development of visceral organs such as the esophagus, lung, liver and stomach are coordinated by reciprocal signaling interactions between the endoderm and adjacent mesoderm cells in the fetal foregut. Although the recent successes in recapitulating developmental signaling in vitro has enabled the differentiation of human pluripotent stem cells (hPSCs) into various types of organ-specific endodermal epithelium, the generation of organ-specific mesenchyme has received much less attention. This is a major limitation in ongoing efforts to engineer complex human tissue. Here, we describe a protocol to differentiate hPSCs into different types of organ-specific mesoderm, leveraging signaling networks and molecular markers elucidated from single-cell transcriptomics of mouse foregut organogenesis. Building on established methods, hPSC-derived lateral plate mesoderm treated with either retinoic acid (RA) or RA together with a Hedgehog (HH) agonist generates posterior or anterior foregut splanchnic mesoderm, respectively, after 4-d cultures. These are directed into organ-specific mesenchyme lineages by the combinatorial activation or inhibition of WNT, BMP, RA or HH pathways from days 4 to 7 in cultures. By day 7, the cultures are enriched for different types of mesoderm with distinct molecular signatures: 60–90% pure liver septum transversum/mesothelium-like, 70–80% pure liver-like fibroblasts and populations of ~35% respiratory-like mesoderm, gastric-like mesoderm or esophageal-like mesoderm. This protocol can be performed by anyone with moderate experience differentiating hPSCs, provides a novel platform to study human mesoderm development and can be used to engineer more complex foregut tissue for disease modeling and regenerative medicine.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The main data discussed in this protocol were generated as part of the studies published in the supporting primary research papers14. The raw datasets of qRT-PCR and immunostaining have been deposited in the online open access figshare repository (https://doi.org/10.6084/m9.figshare.19723189). The published mouse foregut scRNA-seq data used to infer the signaling roadmap is available from the Gene Expression Omnibus database under accession code GSE136689. The scRNA-seq data can also be explored at https://research.cchmc.org/ZornLab-singlecell/.
References
Zorn, A. M. & Wells, J. M. Vertebrate endoderm development and organ formation. Annu. Rev. Cell Dev. Biol. 25, 221–251 (2009).
Kraus, M. R. & Grapin-Botton, A. Patterning and shaping the endoderm in vivo and in culture. Curr. Opin. Genet. Dev. 22, 347–353 (2012).
Kechele, D. O. & Wells, J. M. Recent advances in deriving human endodermal tissues from pluripotent stem cells. Curr. Opin. Cell Bio. l 61, 92–100 (2019).
Iwasawa, K. & Takebe, T. Organogenesis in vitro. Curr. Opin. Cell Biol. 73, 84–91 (2021).
McCracken, K. W. et al. Wnt/β-catenin promotes gastric fundus specification in mice and humans. Nature 541, 182–187 (2017).
McCracken, K. W. et al. Modelling human development and disease in pluripotent stem-cell-derived gastric organoids. Nature 516, 400–404 (2014).
Múnera, J. O. et al. Differentiation of human pluripotent stem cells into colonic organoids via transient activation of BMP signaling. Cell Stem Cell 21, 51–64.e6 (2017).
Spence, J. R. et al. Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature 470, 105–109 (2011).
Trisno, S. L. et al. Esophageal organoids from human pluripotent stem cells delineate Sox2 functions during esophageal specification. Cell Stem Cell 23, 501–515.e7 (2018).
Zhang, Y. et al. 3D modeling of esophageal development using human PSC-derived basal progenitors reveals a critical role for notch signaling. Cell Stem Cell 23, 516–529.e5 (2018).
He, S. et al. Single-cell transcriptome profiling of an adult human cell atlas of 15 major organs. Genome Biol. 21, 294 (2020).
Han, X. et al. Construction of a human cell landscape at single-cell level. Nature 581, 303–309 (2020).
Yu, Q. et al. Charting human development using a multi-endodermal organ atlas and organoid models. Cell 184, 3281–3298.e22 (2021).
Han, L. et al. Single cell transcriptomics identifies a signaling network coordinating endoderm and mesoderm diversification during foregut organogenesis. Nat. Commun. 11, 4158 (2020).
Ferretti, E. & Hadjantonakis, A. K. Mesoderm specification and diversification: from single cells to emergent tissues. Curr. Opin. Cell Biol. 61, 110–116 (2019).
Tam, P. P. & Loebel, D. A. Gene function in mouse embryogenesis: get set for gastrulation. Nat. Rev. Genet. 8, 368–381 (2007).
Sweetman, D., Wagstaff, L., Cooper, O., Weijer, C. & Münsterberg, A. The migration of paraxial and lateral plate mesoderm cells emerging from the late primitive streak is controlled by different Wnt signals. BMC Dev. Biol. 8, 63 (2008).
Barnes, R. M., Firulli, B. A., Conway, S. J., Vincentz, J. W. & Firulli, A. B. Analysis of the Hand1 cell lineage reveals novel contributions to cardiovascular, neural crest, extra-embryonic, and lateral mesoderm derivatives. Dev. Dyn. 239, 3086–3097 (2010).
Tang, X. et al. Transcriptome regulation and chromatin occupancy by E2F3 and MYC in mice. Sci. Data 3, 160008 (2016).
Zhang, Y., Bailey, D., Yang, P., Kim, E. & Que, J. The development and stem cells of the esophagus. Development 148, dev193839 (2021).
Kishimoto, K. et al. Bidirectional Wnt signaling between endoderm and mesoderm confers tracheal identity in mouse and human cells. Nat. Commun. 11, 4159 (2020).
Loh, K. M. et al. Efficient endoderm induction from human pluripotent stem cells by logically directing signals controlling lineage bifurcations. Cell Stem Cell 14, 237–252 (2014).
Loh, K. M. et al. Mapping the pairwise choices leading from pluripotency to human bone, heart, and other mesoderm cell types. Cell 166, 451–467 (2016).
Protze, S. I., Lee, J. H. & Keller, G. M. Human pluripotent stem cell-derived cardiovascular cells: from developmental biology to therapeutic applications. Cell Stem Cell 25, 311–327 (2019).
Edwards, N. A. et al. Developmental basis of trachea-esophageal birth defects. Dev. Biol. 477, 85–97 (2021).
Kishimoto, K. & Morimoto, M. Mammalian tracheal development and reconstruction: insights from in vivo and in vitro studies. Development 148, dev198192 (2021).
Eicher, A. K. et al. Functional human gastrointestinal organoids can be engineered from three primary germ layers derived separately from pluripotent stem cells. Cell Stem Cell 29, 36–51.e6 (2021).
Takebe, T. et al. Massive and reproducible production of liver buds entirely from human pluripotent stem cells. Cell Rep. 21, 2661–2670 (2017).
Coll, M. et al. Generation of hepatic stellate cells from human pluripotent stem cells enables in vitro modeling of liver fibrosis. Cell Stem Cell 23, 101–113.e7 (2018).
Koui, Y. et al. An in vitro human liver model by iPSC-derived parenchymal and non-parenchymal cells. Stem Cell Rep. 9, 490–498 (2017).
Dye, B. R. et al. In vitro generation of human pluripotent stem cell derived lung organoids. Elife 4, e05098 (2015).
Chen, Y. W. et al. A three-dimensional model of human lung development and disease from pluripotent stem cells. Nat. Cell Biol. 19, 542–549 (2017).
Dye, B. R. et al. A bioengineered niche promotes in vivo engraftment and maturation of pluripotent stem cell derived human lung organoids. Elife 5, e19732 (2016).
Snowball, J., Ambalavanan, M., Whitsett, J. & Sinner, D. Endodermal Wnt signaling is required for tracheal cartilage formation. Dev. Biol. 405, 56–70 (2015).
Asahina, K., Zhou, B., Pu, W. T. & Tsukamoto, H. Septum transversum-derived mesothelium gives rise to hepatic stellate cells and perivascular mesenchymal cells in developing mouse liver. Hepatology 53, 983–995 (2011).
Thomson, J. A. et al. Embryonic stem cell lines derived from human blastocysts. Science 282, 1145–1147 (1998).
Acknowledgements
We thank P. Chaturvedi for generating plots from the mouse scRNA-seq data. We thank all members in the Zorn, Takebe, Wells and Morimoto laboratories for reagents and feedback. We also thank C. Mayhew and A. Pitstick from the Pluripotent Stem Cell Facility as well as M. Kofron and E. Meyer from the Confocal Imaging Core at Cincinnati Children’s Hospital for constant support and guidance. This work was supported by grant NICHD P01HD093363 to A.M.Z. and J.M.W., the NIH Director’s New Innovator Award (DP2 DK128799-01) (to T.T.), the New York Stem Cell Foundation (to T.T.) and by a CCHMC CURE award to A.M.Z., J.M.W. and T.T. K.K. is supported by a Uehara Memorial Foundation postdoctoral fellowship and a fund for the Promotion of Joint International Research (A) (18KK0423). K.I. is supported by the Japan Society for the Promotion of Science Overseas Research Fellowship.
Author information
Authors and Affiliations
Contributions
K.K., L.H., J.M.W., T.T. and A.M.Z. conceived the project. K.K. and A.M.Z. wrote the manuscript with support from K.I., M.M., J.M.W. and T.T. K.K., K.I., A.S. and C.F.-H. performed experiments, and K.K. and K.I. analyzed data. A.M.Z. supervised the project.
Corresponding author
Ethics declarations
Competing interests
K.K., L.H., M.M. and A.M.Z. have filed a patent application on this protocol. All other authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks Jonaa Fowler, Kyle Loh and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Han, L. et al. Nat. Commun. 11, 4158 (2020): https://doi.org/10.1038/s41467-020-17968-x
Eicher, A. K. et al. Cell Stem Cell 29, 36–51.e6 (2022): https://doi.org/10.1016/j.stem.2021.10.010
Supplementary information
Supplementary Information
Supplementary Figs. 1–3.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kishimoto, K., Iwasawa, K., Sorel, A. et al. Directed differentiation of human pluripotent stem cells into diverse organ-specific mesenchyme of the digestive and respiratory systems. Nat Protoc 17, 2699–2719 (2022). https://doi.org/10.1038/s41596-022-00733-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-022-00733-3
This article is cited by
-
Directed differentiation of mouse pluripotent stem cells into functional lung-specific mesenchyme
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.