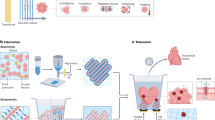
Abstract
Ultrasound-mediated gene delivery (sonoporation) is a minimally invasive, nonviral and clinically translatable method of gene therapy. This method offers a favorable safety profile over that of viral vectors and is less invasive as compared with other physical gene delivery approaches (e.g., electroporation). We have previously used sonoporation to overexpress transgenes in different skeletal tissues in order to induce tissue regeneration. Here, we provide a protocol that could easily be adapted to address various other targets of tissue regeneration or additional applications, such as cancer and neurodegenerative diseases. This protocol describes how to prepare, conduct and optimize ultrasound-mediated gene delivery in both a murine and a porcine animal model. The protocol includes the preparation of a microbubble–DNA mix and in vivo sonoporation under ultrasound imaging. Ultrasound-mediated gene delivery can be accomplished within 10 min. After DNA delivery, animals can be followed to monitor gene expression, protein secretion and other transgene-specific outcomes, including tissue regeneration. This procedure can be accomplished by a competent graduate student or technician with prior experience in ultrasound imaging or in performing in vivo procedures.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All materials are available from commercial sources or can be derived using methods described in this study. All relevant data are reported in the article.
References
Martinek, V. et al. Genetic engineering of meniscal allografts. Tissue Eng. 8, 107–117 (2002).
Jacobson, S. G. et al. Improvement and decline in vision with gene therapy in childhood blindness. N. Engl. J. Med. 372, 1920–1926 (2015).
Pierce, G. F., Lillicrap, D., Pipe, S. W. & Vandendriessche, T. Gene therapy, bioengineered clotting factors and novel technologies for hemophilia treatment. J. Thromb. Haemost. 5, 901–906 (2007).
Wang, Z. G., Wu, Z. Q., Liu, Y. & Han, W. D. New development in CAR-T cell therapy. J. Hematol. Oncol. 10, 53 (2017).
Weber, T. Do we need marker gene studies in humans to improve clinical AAV gene therapy? Gene Ther. 24, 72–73 (2017).
Sheyn, D. et al. Genetically modified cells in regenerative medicine and tissue engineering. Adv. Drug Deliv. Rev. 62, 683–698 (2010).
Kimelman Bleich, N. et al. Gene therapy approaches to regenerating bone. Adv. Drug Deliv. Rev. 64, 1320–1330 (2012).
Pelled, G. et al. Direct gene therapy for bone regeneration: gene delivery, animal models, and outcome measures. Tissue Eng. Part B Rev. 16, 13–20 (2010).
Bessis, N., GarciaCozar, F. J. & Boissier, M. C. Immune responses to gene therapy vectors: influence on vector function and effector mechanisms. Gene Ther. 11, S10–S17 (2004). (Suppl 1).
Baum, C., Kustikova, O., Modlich, U., Li, Z. & Fehse, B. Mutagenesis and oncogenesis by chromosomal insertion of gene transfer vectors. Hum. Gene Ther. 17, 253–263 (2006).
Yoon, C. S. & Park, J. H. Ultrasound-mediated gene delivery. Expert Opin. Drug Deliv. 7, 321–330 (2010).
Sheyn, D. et al. Ultrasound-based nonviral gene delivery induces bone formation in vivo. Gene Ther. 15, 257–266 (2008).
Marrero, B., Shirley, S. & Heller, R. Delivery of interleukin-15 to B16 melanoma by electroporation leads to tumor regression and long-term survival. Technol. Cancer Res. Treat. 13, 551–560 (2014).
Ayuni, E. L. et al. In vivo electroporation mediated gene delivery to the beating heart. PLoS ONE 5, e14467 (2010).
Ferrara, K. W. Driving delivery vehicles with ultrasound. Adv. Drug Deliv. Rev. 60, 1097–1102 (2008).
Sanches, P. G. et al. Ultrasound-mediated gene delivery of naked plasmid DNA in skeletal muscles: a case for bolus injections. J Control. Release 195, 130–137 (2014).
Watanabe, Y. et al. Delivery of Na/I symporter gene into skeletal muscle using nanobubbles and ultrasound: visualization of gene expression by PET. J. Nucl. Med. 51, 951–958 (2010).
Li, T., Tachibana, K., Kuroki, M. & Kuroki, M. Gene transfer with echo-enhanced contrast agents: comparison between Albunex, Optison, and Levovist in mice—initial results. Radiology 229, 423–428 (2003).
Lu, Q. L., Liang, H. D., Partridge, T. & Blomley, M. J. Microbubble ultrasound improves the efficiency of gene transduction in skeletal muscle in vivo with reduced tissue damage. Gene Ther. 10, 396–405 (2003).
Wang, X., Liang, H. D., Dong, B., Lu, Q. L. & Blomley, M. J. Gene transfer with microbubble ultrasound and plasmid DNA into skeletal muscle of mice: comparison between commercially available microbubble contrast agents. Radiology 237, 224–229 (2005).
Qin, S., Caskey, C. F. & Ferrara, K. W. Ultrasound contrast microbubbles in imaging and therapy: physical principles and engineering. Phys. Med. Biol. 54, R27–R57 (2009).
van Wamel, A., Bouakaz, A., Bernard, B., ten Cate, F. & de Jong, N. Radionuclide tumour therapy with ultrasound contrast microbubbles. Ultrasonics 42, 903–906 (2004).
De Cock, I. et al. Ultrasound and microbubble mediated drug delivery: acoustic pressure as determinant for uptake via membrane pores or endocytosis. J. Control. Release 197, 20–28 (2015).
Kimelman-Bleich, N. et al. Targeted gene-and-host progenitor cell therapy for nonunion bone fracture repair. Mol. Ther. 19, 53–59 (2011).
Shapiro, G. et al. Ultrasound-mediated transgene expression in endogenous stem cells recruited to bone injury sites. Polym. Adv. Technol. 25, 525–531 (2014).
Shapiro, G. et al. Multiparameter evaluation of in vivo gene delivery using ultrasound-guided, microbubble-enhanced sonoporation. J. Control. Release 223, 157–164 (2016).
Bez, M. et al. In situ bone tissue engineering via ultrasound-mediated gene delivery to endogenous progenitor cells in mini-pigs. Sci. Transl. Med. 9, eaal3128 (2017).
Bez, M. et al. Ultrasound-mediated gene delivery enhances tendon allograft integration in mini-pig ligament reconstruction. Mol. Ther. 26, 1746-1755 (2018).
Kilkenny, C., Browne, W. J., Cuthill, I. C., Emerson, M. & Altman, D. G. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol. 8, e1000412 (2010).
Fan, Z., Kumon, R. E. & Deng, C. X. Mechanisms of microbubble-facilitated sonoporation for drug and gene delivery. Ther. Deliv. 5, 467–486 (2014).
Delalande, A. et al. Cationic gas-filled microbubbles for ultrasound-based nucleic acids delivery. Biosci. Rep. 37, BSR20160619 (2017).
Sheyn, D. et al. PTH induces systemically administered mesenchymal stem cells to migrate to and regenerate spine injuries. Mol. Ther. 24, 318–330 (2016).
Zachos, T. A., Shields, K. M. & Bertone, A. L. Gene-mediated osteogenic differentiation of stem cells by bone morphogenetic proteins-2 or -6. J. Orthop. Res. 24, 1279–1291 (2006).
Lai, C. Y. et al. Noninvasive thermometry assisted by a dual-function ultrasound transducer for mild hyperthermia. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 57, 2671–2684 (2010).
Kheirolomoom, A. et al. Complete regression of local cancer using temperature-sensitive liposomes combined with ultrasound-mediated hyperthermia. J. Control. Release 172, 266–273 (2013).
Jingfei, L., Josquin, F., Douglas, N. S., Olivier Le, B. & Katherine, W. F. Development of a spherically focused phased array transducer for ultrasonic image-guided hyperthermia. Phys. Med. Biol. 61, 5275–5296 (2016).
Shapiro, G. et al. Multiparameter evaluation of in vivo gene delivery using ultrasound-guided, microbubble-enhanced sonoporation. J. Control. Release 223, 157–164 (2016).
Fujii, H. et al. Repeated and targeted transfer of angiogenic plasmids into the infarcted rat heart via ultrasound targeted microbubble destruction enhances cardiac repair. Eur. Heart J. 32, 2075–2084 (2011).
Leong-Poi, H. et al. Therapeutic arteriogenesis by ultrasound-mediated VEGF165 plasmid gene delivery to chronically ischemic skeletal muscle. Circ. Res. 101, 295–303 (2007).
Zolochevska, O. et al. Sonoporation delivery of interleukin-27 gene therapy efficiently reduces prostate tumor cell growth in vivo. Hum. Gene Ther. 22, 1537–1550 (2011).
Hauff, P. et al. Evaluation of gas-filled microparticles and sonoporation as gene delivery system: feasibility study in rodent tumor models. Radiology 236, 572–578 (2005).
Shimamura, M. et al. Development of efficient plasmid DNA transfer into adult rat central nervous system using microbubble-enhanced ultrasound. Gene Ther. 11, 1532–1539 (2004).
Takeuchi, D. et al. Alleviation of Abeta-induced cognitive impairment by ultrasound-mediated gene transfer of HGF in a mouse model. Gene Ther. 15, 561–571 (2008).
Nakashima, M. et al. Induction of reparative dentin formation by ultrasound-mediated gene delivery of growth/differentiation factor 11. Hum. Gene Ther. 14, 591–597 (2003).
Anderson, C. D., Moisyadi, S., Avelar, A., Walton, C. B. & Shohet, R. V. Ultrasound-targeted hepatic delivery of factor IX in hemophiliac mice. Gene Ther. 23, 510–519 (2016).
Chen, S. et al. Efficient gene delivery to pancreatic islets with ultrasonic microbubble destruction technology. Proc. Natl. Acad. Sci. USA 103, 8469–8474 (2006).
Acknowledgements
We acknowledge funding support from the California Institute for Regenerative Medicine (CIRM; (grant TR4-06713 to D.G.) and NIH grants R01EB026094 (to D.G.) and R01CA112356 (to K.W.F.).
Author information
Authors and Affiliations
Contributions
G.P., K.W.F. and D.G. designed the experiments. M.B., J.F. and G.S. performed the experiments. M.B. and G.S. analyzed the data. M.B., J.F. and G.S. created the figures. M.B., J.F. and G.S. wrote the manuscript. All authors approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
G.P. and D.G. are shareholders in GamlaStem Medical, which did not provide funds for this study. The other authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Shapiro, G. et al. J. Control. Release 223, 157–164 (2016): https://doi.org/10.1016/j.jconrel.2015.12.001
Bez, M. et al. Sci. Transl. Med. 9, eaal3128 (2017): https://doi.org/10.1126/scitranslmed.aal3128
Bez, M. et al. Mol. Ther. 26, 1746–1755 (2018): https://doi.org/10.1016/j.ymthe.2018.04.020
Supplementary information
Rights and permissions
About this article
Cite this article
Bez, M., Foiret, J., Shapiro, G. et al. Nonviral ultrasound-mediated gene delivery in small and large animal models. Nat Protoc 14, 1015–1026 (2019). https://doi.org/10.1038/s41596-019-0125-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-019-0125-y
This article is cited by
-
Cell-cycle dependent nuclear gene delivery enhances the effects of E-cadherin against tumor invasion and metastasis
Signal Transduction and Targeted Therapy (2023)
-
Enhancement of Immune Responses Elicited by Nanovaccines through a Cross-Presentation Pathway
Tissue Engineering and Regenerative Medicine (2023)
-
Cellulose nanocrystal-based enhancement of ultrasound microbubbles for increased tolerance of mechanical index values
Cellulose (2022)
-
Transdermal delivery for gene therapy
Drug Delivery and Translational Research (2022)
-
General Characteristics of Microbubble-Adenovirus Vectors Carrying Genes
Cellular and Molecular Bioengineering (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.