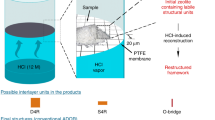
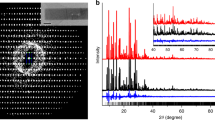
Abstract
High-silica zeolites, some of the most important and widely used catalysts in industry, have potential for application across a wide range of traditional and emerging technologies. The many structural topologies of zeolites have a variety of potential uses, so a strong drive to create new zeolites exists. Here, we present a protocol, the assembly–disassembly–organization–reassembly (ADOR) process, for a relatively new method of preparing these important solids. It allows the synthesis of new high-silica zeolites (Si/Al >1,000), whose synthesis is considered infeasible with traditional (solvothermal) methods, offering new topologies that may find novel applications. We show how to identify the optimal conditions (e.g., duration of reaction, temperature, acidity) for ADOR, which is a complex process with different possible outcomes. Following the protocol will allow researchers to identify the different products that are possible from a reaction without recourse to repetitive and time-consuming trial and error. In developing the protocol, germanium-containing UTL zeolites were subjected to hydrolysis conditions using both water and hydrochloric acid as media, which provides an understanding of the effects of temperature and pH on the disassembly (D) and organization (O) steps of the process that define the potential products. Samples were taken from the ongoing reaction periodically over a minimum of 8 h, and each sample was analyzed using powder X-ray diffraction to yield a time course for the reaction at each set of conditions; selected samples were analyzed using transmission electron microscopy and solid-state NMR spectroscopy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Data supporting this publication are available from the corresponding author upon request.
References
Sartbaeva, A. & Wells, S. A. Framework flexibility and rational design of new zeolites for catalysis. Appl. Petrochemical Res. 2, 69–72 (2012).
Martínez, C. & Corma, A. Inorganic molecular sieves: preparation, modification and industrial application in catalytic processes. Coord. Chem. Rev. 255, 1558–1580 (2011).
Mazur, M. et al. Synthesis of ‘unfeasible’ zeolites. Nat. Chem 8, 58–62 (2015).
Mazur, M., Chlubná-Eliášová, P., Roth, W. J. & Čejka, J. Intercalation chemistry of layered zeolite precursor IPC-1P. Catal. Today 227, 37–44 (2014).
Wheatley, P. S. et al. Zeolites with continuously tuneable porosity. Angew. Chem. Int. Ed. 53, 13210–13214 (2014).
Roth, W. J. et al. Postsynthesis transformation of three-dimensional framework into a lamellar zeolite with modifiable architecture. J. Am. Chem. Soc. 133, 6130–6133 (2011).
Roth, W. J. et al. A family of zeolites with controlled pore size prepared using a top-down method. Nat. Chem. 5, 628–633 (2013).
Morris, R. E. & Čejka, J. Exploiting chemically selective weakness in solids as a route to new porous materials. Nat. Chem. 7, 381–388 (2015).
Eliášová, P. et al. The ADOR mechanism for the synthesis of new zeolites. Chem. Soc. Rev. 44, 7177–206 (2015).
Cundy, C. S. & Cox, P. A. The hydrothermal synthesis of zeolites: history and development from the earliest days to the present time. Chem. Rev. 103, 663–702 (2003).
Davis, M. E. & Lobo, R. F. Zeolite and molecular sieve synthesis. Chem. Mater. 4, 756–768 (1992).
Gutiérrez-Sevillano, J. J. et al. Critical role of dynamic flexibility in Ge-containing zeolites: impact on diffusion. Chemistry 22, 10036–10043 (2016).
Paillaud, J.-L., Harbuzaru, B., Patarin, J. & Bats, N. Extra-large-pore zeolites with two-dimensional channels formed by 14 and 12 rings. Science 304, 990–992 (2004).
Millini, R., Bellussi, G., Smeets, S., Xiaodong, Z. & Strohmaier, K. Zeolites in Catalysis: Properties and Applications Vol. 28, 4 (Royal Society of Chemistry, Cambridge, 2017).
Liu, X., Ravon, U., Bosselet, F., Bergeret, G. & Tuel, A. Probing Ge distribution in zeolite frameworks by post-synthesis introduction of fluoride in as-made materials. Chem. Mater. 24, 3016–3022 (2012).
Johnson, G. M., Tripathi, A. & Parise, J. B. novel routes for the preparation of a range of germanium containing zeolites. Chem. Mater. 11, 10–12 (1999).
Verheyen, E. et al. Design of zeolite by inverse sigma transformation. Nat. Mater. 11, 1059–1064 (2012).
Blasco, T. et al. Preferential location of Ge in the double four-membered ring units of ITQ-7 zolite. J. Phys. Chem. B 106, 2634–2642 (2002).
Shamzhy, M. et al. Germanosilicate precursors of ADORable zeolites obtained by disassembly of ITH, ITR, and IWR zeolites. Chem. Mater. 26, 5789–5798 (2014).
Chlubná-Eliášová, P. et al. The assembly-disassembly-organization-reassembly mechanism for 3D-2D-3D transformation of germanosilicate IWW zeolite. Angew. Chem. 126, 7168–7172 (2014).
Kasneryk, V. et al. Consecutive interlayer disassembly–reassembly during alumination of UOV zeolites: insight into the mechanism. J. Mater. Chem. A 5, 22576–22587 (2017).
Kasneryk, V. et al. Expansion of the ADOR strategy for the synthesis of zeolites: the synthesis of IPC-12 from zeolite UOV. Angew. Chem. Int. Ed. 56, 4324–4327 (2017).
Firth, D. S. et al. Assembly-disassembly-organization-reassembly synthesis of zeolites based on cfi-type layers. Chem. Mater. 29, 5605–5611 (2017).
Morris, S. A. et al. Combined PDF and Rietveld studies of ADORable zeolites and the disordered intermediate IPC-1P. Dalton Trans. 45, 14124–14130 (2016).
Morris, S. A. et al. In situ solid-state NMR and XRD studies of the ADOR process and the unusual structure of zeolite IPC-6. Nat. Chem. 9, 1012–1018 (2017).
Henkelis, S. E. et al. Monitoring the assembly–disassembly–organisation–reassembly process of germanosilicate UTL through in situ pair distribution function analysis. J. Mater. Chem. A 6, 17011–17018 (2018).
Bignami, G. P. M. et al. Synthesis, isotopic enrichment, and solid-state NMR characterization of zeolites derived from the assembly, disassembly, organization, reassembly process. J. Am. Chem. Soc. 139, 5140–5148 (2017).
Wheatley, P. S., Čejka, J. & Morris, R. E. Synthesis of zeolites using the ADOR (assembly-disassembly-organization-reassembly) route. J. Vis. Exp. 2016, e53463 (2016).
Corma, A., Díaz-Cabañas, M. J., Rey, F., Nicolopoulus, S. & Boulahya, K. ITQ-15: the first ultralarge pore zeolite with a bi-directional pore system formed by intersecting 14- and 12-ring channels, and its catalytic implications. Chem. Commun. 0, 1356–1357 (2004).
Massiot, D. et al. Modelling one- and two-dimensional solid-state NMR spectra. Magn. Reson. Chem. 40, 70–76 (2002).
Acknowledgements
The authors thank the EPSRC (grants: EP/K025112/1, EP/K005499/1, EP/K503162/1, EP/N509759/1) for funding. R.E.M., J.C. and M.M. acknowledge OP VVV ‘Excellent Research Teams’, project no. CZ.02.1.01/0.0/0.0/15_003/0000417-CUCAM. S.E.A. thanks the Royal Society and the Wolfson Foundation for a merit award. J.C. acknowledges the Czech Science Foundation (P106/12/G015). We thank O. Morris for the animation of the process that is available as Supplementary Video 1.
Author information
Authors and Affiliations
Contributions
S.E.H. completed the development of the protocol and collected the synthesis data. M.M. completed the electron microscopy; and C.M.R., G.P.M.B. and S.E.A. collected the solid-state NMR data. P.S.W., J.Č. and R.E.M. initiated the project. All authors checked the protocol and contributed to the writing of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Mazur, M. et al. Nat. Chem. 8, 58–62 (2015): https://www.nature.com/articles/nchem.2374
Wheatley, P. S. et al. Angew. Chem. Int. Ed. 53, 13210–13214 (2014): https://onlinelibrary.wiley.com/doi/abs/10.1002/anie.201407676
Roth, W. J. et al. Nat. Chem. 5, 628–633 (2013): https://www.nature.com/articles/nchem.1662
Eliášová, P. et al. Chem. Soc. Rev. 44, 7177–7206 (2015): https://pubs.rsc.org/-/content/articlepdf/2015/cs/c5cs00045a?page=search
Integrated supplementary information
Supplementary Figure 1 1H MAS NMR spectra (9.4 T, 10 kHz MAS) showing the changes in 1H environments in Ge-UTL occurring during hydrolysis at 100 °C in water.
3 p.p.m. – Sharp peak from non-coordinated water; 4-10 p.p.m. - Unresolved hydroxyls. No proton signals other than minimal probe background is observed for calcined Ge-UTL.
Supplementary Figure 2 1H MAS NMR spectra (9.4 T, 10 kHz MAS) showing the different proton environments in hydrolyzed Ge-UTL (5 min; water; 18 °C), utilizing different drying methods.
Drying in the oven was chosen for this study as it produces a relatively dry sample (compared to air drying) suitable for PXRD collection and NMR acquisition, with confirmed presence of hydroxyls. Although the samples produced via vacuum drying and argon loading are drier, due to the high-throughput nature of the PXRD area of this study, oven drying, which takes only 10 minutes, was deemed the most suitable method to proceed with. All reactions involved hydrolyzing 50 mg of calcined Ge-UTL in 50 mg of distilled water. Air drying was carried out at 18 °C for 10 minutes, with oven drying taking place at 110 °C for the same amount of time. Vacuum drying was carried out using Schlenk apparatus at 110 °C overnight to give a vacuum approaching 10–5 Torr. Samples dried in this manner were cooled to room temperature, flushed with argon and then flame sealed under argon. Spectra are scaled according to proton peak intensity (Vacuum: Oven: Air = 1: 2.7: 8.7).
Supplementary Figure 3 TEM images of the samples of Ge-UTL hydrolyzed in water at 100 °C.
a = Parent Ge-UTL; b = after 1 min; c = after 1 hr; d = after 4 hr. FFT images are shown as insets. d spacings can be measured using standard software.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 and Supplementary Table 1
Supplementary Video 1
ADOR protocol video: animation of the ADOR protocol.
Rights and permissions
About this article
Cite this article
Henkelis, S.E., Mazur, M., Rice, C.M. et al. A procedure for identifying possible products in the assembly–disassembly–organization–reassembly (ADOR) synthesis of zeolites. Nat Protoc 14, 781–794 (2019). https://doi.org/10.1038/s41596-018-0114-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-018-0114-6
This article is cited by
-
Synthesis and interlayer structure reconstruction of a new layered zeolitic aluminosilicate
Journal of Porous Materials (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.