Abstract
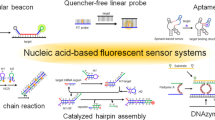
Sensors that detect specific molecules of interest in a living organism can be useful tools for studying biological functions and diseases. Here, we provide a protocol for the construction of nanosensors that can noninvasively detect biologically important targets with magnetic resonance imaging (MRI). The key operating principle of these sensors is magnetic resonance tuning (MRET), a distance-dependent phenomenon occurring between a superparamagnetic quencher and a paramagnetic enhancer. The change in distance between the two magnetic components modulates the longitudinal (T1) relaxivity of the enhancer. In this MRET sensor, distance variation is achieved by interactive linkers that undergo binding, cleavage, or folding/unfolding upon their interaction with target molecules. By the modular incorporation of suitable linkers, the MRET sensor can be applied to a wide range of targets. We showcase three examples of MRET sensors for enzymes, nucleic acid sequences, and pH. This protocol comprises three stages: (i) chemical synthesis and surface modification of the quencher, (ii) conjugation with interactive linkers and enhancers, and (iii) MRI sensing of biological targets. The entire procedure takes up to 3 d.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Sonnichsen, C., Reinhard, B. M., Liphardt, J. & Alivisatos, A. P. A molecular ruler based on plasmon coupling of single gold and silver nanoparticles. Nat. Biotechnol. 23, 741–745 (2005).
Yun, C. S. et al. Nanometal surface energy transfer in optical rulers, breaking the FRET barrier. J. Am. Chem. Soc. 127, 3115–3119 (2005).
Medintz, I. L. et al. Self-assembled nanoscale biosensors based on quantum dot FRET donors. Nat. Mater. 2, 630–638 (2003).
Rizzo, M. A., Springer, G. H., Granada, B. & Piston, D. W. An improved cyan fluorescent protein variant useful for FRET. Nat. Biotechnol. 22, 445–449 (2004).
Ntziachristos, V. Going deeper than microscopy: the optical imaging frontier in biology. Nat. Methods 7, 603–614 (2010).
Glenn, D. R. et al. Single-cell magnetic imaging using a quantum diamond microscope. Nat. Methods 12, 736–U161 (2015).
Choi, J. S. et al. Distance-dependent magnetic resonance tuning as a versatile MRI sensing platform for biological targets. Nat. Mater. 16, 537–542 (2017).
Tu, C., Osborne, E. A. & Louie, A. Y. Activatable T 1 and T 2 magnetic resonance imaging contrast agents. Ann. Biomed. Eng. 39, 1335–1348 (2011).
Davies, G. L., Kramberger, I. & Davis, J. J. Environmentally responsive MRI contrast agents. Chem. Commun. 49, 9704–9721 (2013).
Stuber, M. et al. Positive contrast visualization of iron oxide-labeled stem cells using inversion-recovery with on-resonant water suppression (IRON). Magn. Reson. Med. 58, 1072–1077 (2007).
Kobayashi, H. & Choyke, P. L. Target-cancer-cell-specific activatable fluorescence imaging probes: rational design and in vivo applications. Acc. Chem. Res. 44, 83–90 (2011).
Shi, H. et al. Activatable aptamer probe for contrast-enhanced in vivo cancer imaging based on cell membrane protein-triggered conformation alteration. Proc. Natl. Acad. Sci. USA 108, 3900–3905 (2011).
Vogel, S. S., van der Meer, B. W. & Blank, P. S. Estimating the distance separating fluorescent protein FRET pairs. Methods 66, 131–138 (2014).
Ozerdem, U. & Hargens, A. R. A simple method for measuring interstitial fluid pressure in cancer tissues. Microvasc. Res. 70, 116–120 (2005).
Arami, H., Khandhar, A., Liggitt, D. & Krishnan, K. M. In vivo delivery, pharmacokinetics, biodistribution and toxicity of iron oxide nanoparticles. Chem. Soc. Rev. 44, 8576–8607 (2015).
Wei, H. et al. Exceedingly small iron oxide nanoparticles as positive MRI contrast agents. Proc. Natl. Acad. Sci. USA 114, 2325–2330 (2017).
Shin, T. H., Choi, Y., Kim, S. & Cheon, J. Recent advances in magnetic nanoparticle-based multi-modal imaging. Chem. Soc. Rev. 44, 4501–4516 (2015).
Caravan, P., Ellison, J. J., McMurry, T. J. & Lauffer, R. B. Gadolinium(iii) chelates as MRI contrast agents: structure, dynamics, and applications. Chem. Rev. 99, 2293–2352 (1999).
Shin, T. H. et al. T 1 and T 2 dual-mode MRI contrast agent for enhancing accuracy by engineered nanomaterials. ACS Nano 8, 3393–3401 (2014).
Choi, J.-s et al. Self-confirming “AND” logic nanoparticles for fault-free MRI. J. Am. Chem. Soc. 132, 11015–11017 (2010).
House, M. J. et al. Correlation of proton transverse relaxation rates (R2) with iron concentrations in postmortem brain tissue from alzheimer’s disease patients. Magn. Reson. Med. 57, 172–180 (2007).
Jang, J. T. et al. Critical enhancements of mri contrast and hyperthermic effects by dopant-controlled magnetic nanoparticles. Angew. Chem. Int. Ed. Engl. 48, 1234–1238 (2009).
Lee, H., Shin, T. H., Cheon, J. & Weissleder, R. Recent developments in magnetic diagnostic systems. Chem. Rev. 115, 10690–10724 (2015).
Song, H. T. et al. Surface modulation of magnetic nanocrystals in the development of highly efficient magnetic resonance probes for intracellular labeling. J. Am. Chem. Soc. 127, 9992–9993 (2005).
Lee, J.-H. et al. Artificially engineered magnetic nanoparticles for ultra-sensitive molecular imaging. Nat. Med. 13, 95–99 (2007).
Huh, Y. M. et al. In vivo magnetic resonance detection of cancer by using multifunctional magnetic nanocrystals. J. Am. Chem. Soc. 127, 12387–12391 (2005).
Hagberg, G. E. & Scheffler, K. Effect of r1 and r2 relaxivity of gadolinium-based contrast agents on the T 1-weighted MR signal at increasing magnetic field strengths. Contrast Media Mol. Imaging. 8, 456-465, (2013).
Rockwood, C. A., Green, D. P. & Bucholz, R. W. Rockwood and Green’s Fractures in Adults 7th edn, 443 (Wolters Kluwer Health/Lippincott Williams & Wilkins, Philadelphia, 2010).
Bonevich, J. E. & Wolfgang, K. H. NIST-NCL Joint Assay Protocol, pcc-7: Measuring the Size of Nanoparticles Using Transmission Electron Microscopy (National Institute of Standards and Technology, Gaithersburg, MD, 2010).
Rowlands, N. & Burgess, S. Energy dispersive analysis in the TEM. Mater. Today 12, 46–48 (2010).
Qiao, L. et al. Standardizing size- and shape-controlled synthesis of monodisperse magnetite (Fe3O4) nanocrystals by identifying and exploiting effects of organic impurities. ACS Nano 11, 6370–6381 (2017).
Jonsson, A., Hjalmarsson, C., Falk, P. & Ivarsson, M. L. Levels of matrix metalloproteinases differ in plasma and serum—aspects regarding analysis of biological markers in cancer. Br. J. Cancer 115, 703–706 (2016).
Kim, B. H. et al. Large-scale synthesis of uniform and extremely small-sized iron oxide nanoparticles for high-resolution T 1 magnetic resonance imaging contrast agents. J. Am. Chem. Soc. 133, 12624–12631 (2011).
Estelrich, J., Sanchez-Martin, M. J. & Busquets, M. A. Nanoparticles in magnetic resonance imaging: from simple to dual contrast agents. Int. J. Nanomed. 10, 1727–1741 (2015).
Dormann, J. L., Fiorani, D. & Tronc, E. On the models for interparticle interactions in nanoparticle assemblies: comparison with experimental results. J. Magn. Magn. Mater. 202, 251–267 (1999).
Kim, J. W. et al. Single-cell mechanogenetics using monovalent magnetoplasmonic nanoparticles. Nat. Protoc. 12, 1871–1889 (2017).
Acknowledgements
We thank S. J. Kim. for helpful discussions and support. This work was supported by grants from the Institute for Basic Science (IBS-R026-D1) and the Korea Healthcare Technology R&D Project, Ministry for Health & Welfare Affairs (HI08C2149).
Author information
Authors and Affiliations
Contributions
J.C. conceived and designed the project; T.-H.S., S.K., S.P., and J.-s.C. developed protocols for MRET sensor syntheses; P.K.K. contributed methods for MRI measurements; T.-H.S., S.K., J.-s.C., and J.C. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Choi, J.-s. et al. Nat. Mater. 16, 537–542 (2017): https://doi.org/10.1038/nmat4846
Shin, T. H. et al. ACS Nano 8, 3393–3401 (2014): https://doi.org/10.1021/nn405977t
Choi, J.-s. et al. J. Am. Chem. Soc. 132, 11015–11017 (2010): https://doi.org/10.1021/ja104503g
Lee, J.-H. et al. Nat. Med. 13, 95–99 (2007): https://doi.org/10.1038/nm1467
Supplementary information
Rights and permissions
About this article
Cite this article
Shin, TH., Kang, S., Park, S. et al. A magnetic resonance tuning sensor for the MRI detection of biological targets. Nat Protoc 13, 2664–2684 (2018). https://doi.org/10.1038/s41596-018-0057-y
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-018-0057-y
This article is cited by
-
PDGFB-targeted functional MRI nanoswitch for activatable T1–T2 dual-modal ultra-sensitive diagnosis of cancer
Journal of Nanobiotechnology (2023)
-
High-resolution T1 MRI via renally clearable dextran nanoparticles with an iron oxide shell
Nature Biomedical Engineering (2021)
-
Activatable MRI probes for the specific detection of bacteria
Analytical and Bioanalytical Chemistry (2021)
-
Early stratification of radiotherapy response by activatable inflammation magnetic resonance imaging
Nature Communications (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.