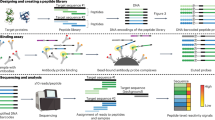
Abstract
The binding specificities of an individual’s antibody repertoire contain a wealth of biological information. They harbor evidence of environmental exposures, allergies, ongoing or emerging autoimmune disease processes, and responses to immunomodulatory therapies, for example. Highly multiplexed methods to comprehensively interrogate antibody-binding specificities have therefore emerged in recent years as important molecular tools. Here, we provide a detailed protocol for performing ‘phage immunoprecipitation sequencing’ (PhIP-Seq), which is a powerful method for analyzing antibody-repertoire binding specificities with high throughput and at low cost. The methodology uses oligonucleotide library synthesis (OLS) to encode proteomic-scale peptide libraries for display on bacteriophage. These libraries are then immunoprecipitated, using an individual’s antibodies, for subsequent analysis by high-throughput DNA sequencing. We have used PhIP-Seq to identify novel self-antigens associated with autoimmune disease, to characterize the self-reactivity of broadly neutralizing HIV antibodies, and in a large international cross-sectional study of exposure to hundreds of human viruses. Compared with alternative array-based techniques, PhIP-Seq is far more scalable in terms of sample throughput and cost per analysis. Cloning and expression of recombinant proteins are not required (versus protein microarrays), and peptide lengths are limited only by DNA synthesis chemistry (up to 90-aa (amino acid) peptides versus the typical 8- to 12-aa length limit of synthetic peptide arrays). Compared with protein microarrays, however, PhIP-Seq libraries lack discontinuous epitopes and post-translational modifications. To increase the accessibility of PhIP-Seq, we provide detailed instructions for the design of phage-displayed peptidome libraries, their immunoprecipitation using serum antibodies, deep sequencing–based measurement of peptide abundances, and statistical determination of peptide enrichments that reflect antibody–peptide interactions. Once a library has been constructed, PhIP-Seq data can be obtained for analysis within a week.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
25 October 2018
The version of this paper originally published contained typesetter-introduced errors in some of the code commands, consisting of conversion of a closing backslash (\) to a forward slash (/). These errors have been corrected in the HTML and PDF versions of the protocol.
References
Larman, H. B. et al. Autoantigen discovery with a synthetic human peptidome. Nat. Biotechnol. 29, 535–541 (2011).
Larman, H. B. et al. PhIP-Seq characterization of autoantibodies from patients with multiple sclerosis, type 1 diabetes and rheumatoid arthritis. J. Autoimmun. 43, 1–9 (2013).
Larman, H. B. et al. Cytosolic 5′-nucleotidase 1A autoimmunity in sporadic inclusion body myositis. Ann. Neurol. 73, 408–418 (2013).
Finton, K. A. et al. Ontogeny of recognition specificity and functionality for the broadly neutralizing anti-HIV antibody 4E10. PLoS Pathog. 10, e1004403 (2014).
Xu, G. J. et al. Viral immunology. Comprehensive serological profiling of human populations using a synthetic human virome. Science 348, aaa0698 (2015).
Kosuri, S. & Church, G. M. Large-scale de novo DNA synthesis: technologies and applications. Nat. Methods 11, 499–507 (2014).
Atak, A. et al. Protein microarray applications: autoantibody detection and posttranslational modification. Proteomics 16, 2557–2569 (2016).
Yu, X. et al. Multiplexed nucleic acid programmable protein arrays. Theranostics 7, 4057–4070 (2017).
Henkel, S., Wellhausen, R., Woitalla, D., Marcus, K. & May, C. Epitope mapping using peptide microarray in autoantibody profiling. Methods Mol. Biol. 1368, 209–224 (2016).
Finton, K. A. et al. Autoreactivity and exceptional CDR plasticity (but not unusual polyspecificity) hinder elicitation of the anti-HIV antibody 4E10. PLoS Pathog. 9, e1003639 (2013).
Xu, G.J. et al. Systematic autoantigen analysis identifies a distinct subtype of scleroderma with coincident cancer. Proc. Natl. Acad. Sci. USA 113, E7526-E7534 (2016).
Zhu, H., Luo, H., Yan, M., Zuo, X. & Li, Q. Z. Autoantigen microarray for high-throughput autoantibody profiling in systemic lupus erythematosus. Genomics Proteomics Bioinformatics 13, 210–218 (2015).
Miersch, S. & LaBaer, J. Nucleic acid programmable protein arrays: versatile tools for array-based functional protein studies. Curr. Protoc. Protein Sci. Chapter 27, Unit 27.2 (2011).
Zhu, J. et al. Protein interaction discovery using parallel analysis of translated ORFs (PLATO). Nat. Biotechnol. 31, 331–334 (2013).
Larman, H. B., Liang, A. C., Elledge, S. J. & Zhu, J. Discovery of protein interactions using parallel analysis of translated ORFs (PLATO). Nat. Protoc. 9, 90–103 (2014).
Jhaveri, D. T. et al. Using quantitative seroproteomics to identify antibody biomarkers in pancreatic cancer. Cancer Immunol. Res. 4, 225–233 (2016).
MacConaill, L. E. et al. Unique, dual-indexed sequencing adapters with UMIs effectively eliminate index cross-talk and significantly improve sensitivity of massively parallel sequencing. BMC Genomics 19, 30 (2018).
Acknowledgements
The development of the PhIP-Seq technology platform has been an ongoing collaboration with S.J. Elledge of the Harvard Medical School Genetics Department and the Howard Hughes Medical Institute. Special thanks to T.M. Shi (Department of Art as Applied to Medicine, Johns Hopkins School of Medicine) for creating the artwork used in Fig. 1. Recent improvements in the PhIP-Seq methodology were supported under a U24 Resource-Related Research Projects Cooperative Agreement awarded by the NIH (U24AI118633 to H.B.L. and S.J. Elledge), a grant from the Jerome L. Greene Foundation (to H.B.L. and A.N.B.), and a grant from the Sjögren’s Syndrome Foundation (to H.B.L. and A.N.B.). A.N.B. received support from NIH grant R01DE012354 (Rosen, PI).
Author information
Authors and Affiliations
Contributions
D.M., D.L.W., and B.M.S. performed experiments related to assay development and optimization. D.M. performed PhIP-Seq screening analysis of the serum samples used in this study. M.S.N. created a draft version of the peptidome design software. A.N.B. provided the Sjogren’s syndrome serum samples and disease-specific expertise. U.L. developed the pepsyn and phip-stat software packages. U.L. and H.B.L. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
1. Larman, H. B. et al. Nat. Biotech. 29, 535–541 (2011) https://doi.org/10.1038/nbt.1856
2. Larman, H. B. et al. Ann. Neurol. 73, 408–418 (2013) https://doi.org/10.1002/ana.23840
3. Xu, G. J. et al. Science 384, aaa0698 (2015) https://doi.org/10.1126/science.aaa0698
Supplementary information
Supplementary Table 1
Design of 96 ad_min_BCX_P7 PCR2 i7 indexing primers
Rights and permissions
About this article
Cite this article
Mohan, D., Wansley, D.L., Sie, B.M. et al. PhIP-Seq characterization of serum antibodies using oligonucleotide-encoded peptidomes. Nat Protoc 13, 1958–1978 (2018). https://doi.org/10.1038/s41596-018-0025-6
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-018-0025-6
This article is cited by
-
Efficient encoding of large antigenic spaces by epitope prioritization with Dolphyn
Nature Communications (2024)
-
Schistosoma mansoni vaccine candidates identified by unbiased phage display screening in self-cured rhesus macaques
npj Vaccines (2024)
-
Human antibody profiling technologies for autoimmune disease
Immunologic Research (2023)
-
Detecting antibody reactivities in Phage ImmunoPrecipitation Sequencing data
BMC Genomics (2022)
-
PepSeq: a fully in vitro platform for highly multiplexed serology using customizable DNA-barcoded peptide libraries
Nature Protocols (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.