Abstract
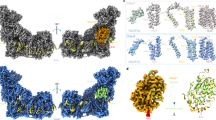
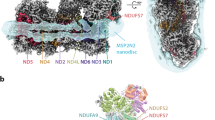
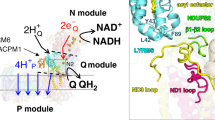
Mitochondrial complex I powers ATP synthesis by oxidative phosphorylation, exploiting the energy from ubiquinone reduction by NADH to drive protons across the energy-transducing inner membrane. Recent cryo-EM analyses of mammalian and yeast complex I have revolutionized structural and mechanistic knowledge and defined structures in different functional states. Here, we describe a 2.7-Å-resolution structure of the 42-subunit complex I from the yeast Yarrowia lipolytica containing 275 structured water molecules. We identify a proton-relay pathway for ubiquinone reduction and water molecules that connect mechanistically crucial elements and constitute proton-translocation pathways through the membrane. By comparison with known structures, we deconvolute structural changes governing the mammalian ‘deactive transition’ (relevant to ischemia–reperfusion injury) and their effects on the ubiquinone-binding site and a connected cavity in ND1. Our structure thus provides important insights into catalysis by this enigmatic respiratory machine.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hirst, J. Mitochondrial complex I. Annu. Rev. Biochem. 82, 551–575 (2013).
Parey, K., Wirth, C., Vonck, J. & Zickermann, V. Respiratory complex I—structure, mechanism and evolution. Curr. Opin. Struct. Biol. 63, 1–9 (2020).
Kaila, V. R. I. Long-range proton-coupled electron transfer in biological energy conversion: towards mechanistic understanding of respiratory complex I. J. R. Soc. Interface 15, 20170916 (2018).
Fassone, E. & Rahman, S. Complex I deficiency: clinical features, biochemistry and molecular genetics. J. Med. Genet. 49, 578–590 (2012).
Fiedorczuk, K. & Sazanov, L. A. Mammalian mitochondrial complex I structure and disease-causing mutations. Trends Cell Biol. 28, 835–867 (2018).
Balaban, R. S., Nemoto, S. & Finkel, T. Mitochondria, oxidants, and aging. Cell 120, 483–495 (2005).
Babot, M., Birch, A., Labarbuta, P. & Galkin, A. Characterisation of the active/de-active transition of mitochondrial complex I. Biochim. Biophys. Acta Bioenerg. 1837, 1083–1092 (2014).
Chouchani, E. T. et al. A unifying mechanism for mitochondrial superoxide production during ischemia-reperfusion injury. Cell Metab. 23, 254–263 (2016).
Stroud, D. A. et al. Accessory subunits are integral for assembly and function of human mitochondrial complex I. Nature 538, 123–126 (2016).
Zhu, J., Vinothkumar, K. R. & Hirst, J. Structure of mammalian respiratory complex I. Nature 536, 354–358 (2016).
Morgner, N. et al. Subunit mass fingerprinting of mitochondrial complex I. Biochim. Biophys. Acta Bioenerg. 1777, 1384–1391 (2008).
Kmita, K. & Zickermann, V. Accessory subunits of mitochondrial complex I. Biochem. Soc. Trans. 41, 1272–1279 (2013).
Kerscher, S., Dröse, S., Zwicker, K., Zickermann, V. & Brandt, U. Yarrowia lipolytica, a yeast genetic system to study mitochondrial complex I. Biochim. Biophys. Acta Bioenerg. 1555, 83–91 (2002).
Tocilescu, M. A., Fendel, U., Zwicker, K., Kerscher, S. & Brandt, U. Exploring the ubiquinone binding cavity of respiratory complex I. J. Biol. Chem. 282, 29514–29520 (2007).
Varghese, F., Atcheson, E., Bridges, H. R. & Hirst, J. Characterization of clinically identified mutations in NDUFV1, the flavin-binding subunit of respiratory complex I, using a yeast model system. Hum. Mol. Genet. 24, 6350–6360 (2015).
Cabrera-Orefice, A. et al. Locking loop movement in the ubiquinone pocket of complex I disengages the proton pumps. Nat. Commun. 9, 4500 (2018).
Galemou Yoga, E. et al. Mutations in a conserved loop in the PSST subunit of respiratory complex I affect ubiquinone binding and dynamics. Biochim. Biophys. Acta Bioenerg. 1860, 573–581 (2019).
Zickermann, V. et al. Mechanistic insight from the crystal structure of mitochondrial complex I. Science 347, 44–49 (2015).
Parey, K. et al. High-resolution cryo-EM structures of respiratory complex I: mechanism, assembly, and disease. Sci. Adv. 5, eaax9484 (2019).
Agip, A. A. et al. Cryo-EM structures of complex I from mouse heart mitochondria in two biochemically defined states. Nat. Struct. Mol. Biol. 25, 548–556 (2018).
Blaza, J. N., Vinothkumar, K. R. & Hirst, J. Structure of the deactive state of mammalian respiratory complex I. Structure 26, 312–319 (2018).
Sharma, V. et al. Redox-induced activation of the proton pump in the respiratory complex I. Proc. Natl Acad. Sci. USA 112, 11571–11576 (2015).
Brandt, U. A two-state stabilization-change mechanism for proton-pumping complex I. Biochim. Biophys. Acta Bioenerg. 1807, 1364–1369 (2011).
Warnau, J. et al. Redox-coupled quinone dynamics in the respiratory complex I. Proc. Natl Acad. Sci. USA 115, E8413–E8420 (2018).
Fedor, J. G., Jones, A. J. Y., Di Luca, A., Kaila, V. R. I. & Hirst, J. Correlating kinetic and structural data on ubiquinone binding and reduction by respiratory complex I. Proc. Natl Acad. Sci. USA 114, 12737–12742 (2017).
Baradaran, R., Berrisford, J. M., Minhas, G. S. & Sazanov, L. A. Crystal structure of the entire respiratory complex I. Nature 494, 443–448 (2013).
Teixeira, M. H. & Arantes, G. M. Balanced internal hydration discriminates substrate binding to respiratory complex I. Biochim. Biophys. Acta Bioenerg. 1860, 541–548 (2019).
Efremov, R. G. & Sazanov, L. A. Structure of the membrane domain of respiratory complex I. Nature 476, 414–421 (2011).
Jones, A. J. Y., Blaza, J. N., Varghese, F. & Hirst, J. Respiratory complex I in Bos taurus and Paracoccus denitrificans pumps four protons across the membrane for every NADH oxidized. J. Biol. Chem. 292, 4987–4995 (2017).
Galkin, A. S., Grivennikova, V. G. & Vinogradov, A. D. →H+/2e– stoichiometry in NADH-quinone reductase reactions catalyzed by bovine heart submitochondrial particles. FEBS Lett. 451, 157–161 (1999).
Di Luca, A., Gamiz-Hernandez, A. P. & Kaila, V. R. I. Symmetry-related proton transfer pathways in respiratory complex I. Proc. Natl Acad. Sci. USA 114, E6314–E6321 (2017).
Di Luca, A., Mühlbauer, M. E., Saura, P. & Kaila, V. R. I. How inter-subunit contacts in the membrane domain of complex I affect proton transfer energetics. Biochim. Biophys. Acta Bioenerg. 1859, 734–741 (2018).
Kotlyar, A. B. & Vinogradov, A. D. Slow active/inactive transition of the mitochondrial NADH-ubiquinone reductase. Biochim. Biophys. Acta Bioenerg. 1019, 151–158 (1990).
Meyerson, J. R. et al. Self-assembled monolayers improve protein distribution on holey carbon cryo-EM supports. Sci. Rep. 4, 7084 (2014).
Parey, K. et al. Cryo-EM structure of respiratory complex I at work. Elife 7, e39213 (2018).
Letts, J. A., Fiedorczuk, K., Degliesposti, G., Skehel, M. & Sazanov, L. A. Structures of respiratory supercomplex I+III2 reveal functional and conformational crosstalk. Mol. Cell 75, 1131–1146 (2019).
Abdrakhmanova, A., Dobrynin, K., Zwicker, K., Kerscher, S. & Brandt, U. Functional sulfurtransferase is associated with mitochondrial complex I from Yarrowia lipolytica, but is not required for assembly of its iron-sulfur clusters. FEBS Lett. 579, 6781–6785 (2005).
D’Imprima, E. et al. Cryo-EM structure of respiratory complex I reveals a link to mitochondrial sulfur metabolism. Biochim. Biophys. Acta Bioenerg. 1857, 1935–1942 (2016).
Fontecilla-Camps, J. C., Volbeda, A., Cavazza, C. & Nicolet, Y. Structure/function relationships of [NiFe]- and [FeFe]-hydrogenases. Chem. Rev. 107, 4273–4303 (2007).
Szőri-Dorogházi, E. et al. Analyses of the large subunit histidine-rich motif expose an alternative proton transfer pathway in [NiFe] hydrogenases. PLoS ONE 7, e34666 (2012).
Ogata, H. et al. Activation process of [NiFe] hydrogenase elucidated by high-resolution X-ray analyses: conversion of the ready to the unready state. Structure 13, 1635–1642 (2005).
Haapanen, O. & Sharma, V. Role of water and protein dynamics in proton pumping by respiratory complex I. Sci. Rep. 7, 7747 (2017).
Djurabekova, A., Haapanen, O. & Sharma, V. Proton motive function of the terminal antiporter-like subunit in respiratory complex I. Biochim. Biophys. Acta Bioenerg. 1861, 148185 (2020).
Guo, R., Zong, S., Wu, M., Gu, J. & Yang, M. Architecture of human mitochondrial respiratory megacomplex I2III2IV2. Cell 170, 1247–1257 (2017).
Gu, J. et al. The architecture of the mammalian respirasome. Nature 537, 639–643 (2016).
Laughlin, T. G., Bayne, A. N., Trempe, J. F., Savage, D. F. & Davies, K. M. Structure of the complex I-like molecule NDH of oxygenic photosynthesis. Nature 566, 411–414 (2019).
Tian, W., Chen, C., Lei, X., Zhao, J. & Liang, J. CASTp 3.0: computed atlas of surface topography of proteins. Nucleic Acids Res. 46, W363–W367 (2018).
Galkin, A. et al. Identification of the mitochondrial ND3 subunit as a structural component involved in the active/deactive enzyme transition of respiratory complex I. J. Biol. Chem. 283, 20907–20913 (2008).
Gavrikova, E. V. & Vinogradov, A. D. Active/de-active state transition of the mitochondrial complex I as revealed by specific sulfhydryl group labeling. FEBS Lett. 455, 36–40 (1999).
Di Luca, A. & Kaila, V. R. I. Global collective motions in the mammalian and bacterial respiratory complex I. Biochim. Biophys. Acta Bioenerg. 1859, 326–332 (2018).
Banba, A., Tsuji, A., Kimura, H., Murai, M. & Miyoshi, H. Defining the mechanism of action of S1QELs, specific suppressors of superoxide production in the quinone-reaction site in mitochondrial complex I. J. Biol. Chem. 294, 6550–6561 (2019).
Matias, P. M. et al. [NiFe] hydrogenase from Desulfovibrio desulfuricans ATCC 27774: gene sequencing, three-dimensional structure determination and refinement at 1.8 Å and modelling studies of its interaction with the tetrahaem cytochrome c3. J. Biol. Inorg. Chem. 6, 63–81 (2001).
Zivanov, J., Nakane, T. & Scheres, S. H. W. Estimation of high-order aberrations and anisotropic magnification from cryo-EM data sets in RELION−3.1. IUCrJ 7, 253–267 (2020).
Zivanov, J. et al. New tools for automated high-resolution cryo-EM structure determination in RELION-3. Elife 7, e42166 (2018).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Zhang, K. Gctf: real-time CTF determination and correction. J. Struct. Biol. 193, 1–12 (2016).
Vilas, J. L. et al. MonoRes: automatic and accurate estimation of local resolution for electron microscopy maps. Structure 26, 337–344 (2018).
Ramírez-Aportela, E. et al. Automatic local resolution-based sharpening of cryo-EM maps. Bioinformatics 36, 765–772 (2019).
de la Rosa-Trevín, J. M. et al. Scipion: a software framework toward integration, reproducibility and validation in 3D electron microscopy. J. Struct. Biol. 195, 93–99 (2016).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
Abdrakhmanova, A. et al. Subunit composition of mitochondrial complex I from the yeast Yarrowia lipolytica. Biochim. Biophys. Acta Bioenerg. 1658, 148–156 (2004).
Angerer, H. et al. A scaffold of accessory subunits links the peripheral arm and the distal proton-pumping module of mitochondrial complex I. Biochem. J. 437, 279–288 (2011).
Fukasawa, Y. et al. MitoFates: improved prediction of mitochondrial targeting sequences and their cleavage sites. Mol. Cell. Proteom. 14, 1113–1126 (2015).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Croll, T. I. ISOLDE: a physically realistic environment for model building into low-resolution electron-density maps. Acta Crystallogr. D Struct. Biol. 74, 519–530 (2018).
Goddard, T. D. et al. UCSF ChimeraX: meeting modern challenges in visualization and analysis. Protein Sci. 27, 14–25 (2018).
Waterhouse, A. et al. SWISS-MODEL: homology modelling of protein structures and complexes. Nucleic Acids Res. 46, W296–W303 (2018).
Ploegman, J. H., Drent, G., Kalk, K. H. & Hol, W. G. J. Structure of bovine liver rhodanese. J. Mol. Biol. 123, 557–594 (1978).
Amunts, A. et al. Structure of the yeast mitochondrial large ribosomal subunit. Science 343, 1485–1489 (2014).
Afonine, P. V. et al. New tools for the analysis and validation of cryo-EM maps and atomic models. Acta Crystallogr. D Struct. Biol. 74, 814–840 (2018).
Bridges, H. R., Mohammed, K., Harbour, M. E. & Hirst, J. Subunit NDUFV3 is present in two distinct isoforms in mammalian complex I. Biochim. Biophys. Acta Bioenerg. 1858, 197–207 (2017).
Kumar, P. & Bansal, M. Identification of local variations within secondary structures of proteins. Acta Crystallogr. D Biol. Crystallogr. 71, 1077–1086 (2015).
Kabsch, W. & Sander, C. Dictionary of protein secondary structure: pattern recognition of hydrogen-bonded and geometrical features. Biopolymers 22, 2577–2637 (1983).
The PyMOL Molecular Graphics System v.2.2.3 (Schrödinger, 2019).
Acknowledgements
We thank K. Dent and the staff at the UK National Electron Bio-Imaging Centre (eBIC) at the Diamond Light Source, proposal EM-17057–27, funded by the Wellcome Trust, MRC and BBSRC, for assistance with cryo-EM data collection; Z. Yin (MBU Cambridge) and D. Chirgadze (University of Cambridge cryo-EM facility) for assistance with cryo-EM grid preparation and screening; S. Ding and I. Fearnley (MBU Cambridge) for mass spectrometry analyses; and M. Hartley and A. Raine (MBU Cambridge) for IT support. This work was supported by the Medical Research Council (MC_U105663141 and MC_UU_00015/2 to J.H.).
Author information
Authors and Affiliations
Contributions
D.N.G. carried out all experimental and cryo-EM work, processed the cryo-EM data and built the models. D.N.G. and J.H. analyzed and interpreted the models and wrote the manuscript. J.H. directed the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Peer reviewer reports are available. Inês Chen was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 2 Resolution estimates of the cryo-EM maps for Y. lipolytica complex I.
a, the global map of Y. lipolytica complex I; b, the map from focused refinement of the sub-stoichiometric ST1 subunit. The masks used are indicated by outlines and local resolutions were estimated using Relion. The resolution estimates from the masked Fourier shell correlation curves, FSCfinal, are 2.7 and 3.5 Å for the global and ST1 maps at FSC = 0.143 (dotted lines) and 3.2 and 4.3 Å at FSC = 0.5 (solid lines). Model–map FSC curves (FSCmodel) are in magenta. Overfitting analyses71,72 were performed by refining the model with the atoms displaced by 0.5 Å against one half map, followed by FSC map–model validation (FSCwork). The output was used with the second half map for FSC map–model validation (FSCtest). For the global model the curves match well beyond the 2.7 Å data used for refinement. The ST1 model, which was generated from a homology model, shows evidence of overfitting. Residue side chains were removed in low resolution regions to reduce the overfitting, while the overall secondary structures and architecture of the homology model were retained.
Extended Data Fig. 3 The local resolution of sequential focused refinements of the ST1 subunit, and the partial ST1 subunit model docked onto the complex.
a, The improvement in map quality at the position of the ST1 subunit after focused refinement. b, The partially built ST1 subunit model (pink) docked onto the full Y. lipolytica complex I model. The model was built into its focus-refined map (lower left inset, surface coloured by local resolution), docked onto the model for the whole complex, then adjusted locally. ST1–subunit interactions are indicated.
Extended Data Fig. 4 Sequence alignments of the π1 bulges defined in Supplementary Table 2.
The residues that comprise the detected π bulges are highlighted in blue. When the exact position of the π bulge varied between the two detection algorithms, the π bulge that best corresponded to the π bulges detected in the other organisms was used. For mouse, the π bulge detected in the ND6 subunit is from the deactive state. ClustalW symbols (*:.) are used to indicate perfect alignment, strong similarity, and weak similarity, respectively. The initial residue numbering is displayed for the Y. lipolytica sequences.
Extended Data Fig. 5 Comparison of the π bulges in ND4-TMH8 of complex I from Y. lipolytica and T. thermophilus (PDB 4HEA)26 and ND6-TMH3 of complex I from Y. lipolytica and M. musculus (PDB 6G2J and PDB 6G72)20.
a, The backbones of both ND4 subunits are shown in ribbon with equivalent residues in sticks. The T. thermophilus backbone deviates substantially from α-helical structure around the conserved Lys235 residue, shifting its orientation, with a π bulge observed only in the π2 position. b, The conserved proline on the loop between TMHs 7 and 8 is shifted along the sequence by one residue, resulting in a markedly different entry geometry to the π bulge helix. c, ND6 is in teal and ND3 is in green. Homologous residues are labelled. The orientations of backbone groups at the π bulge in the Y. lipolytica and deactive mouse models agree closely. The active mouse model no longer adopts the π bulge conformation; the helix is rotated and the conserved glycine residue (Gly61) in the π bulge hydrogen-bonding network is displaced. See also Supplementary Video 4.
Extended Data Fig. 6 Lipid interactions with the conserved π bulge of ND1 and the amphipathic helix of NDUFS8 in Y. lipolytica.
a, The ND1 subunit and the amphipathic helix of NDUFS8 are shown in orange and blue cartoon, respectively. Lipids (PE: phosphatidylethanolamine and CDL: cardiolipin) and relevant side chains are shown in black sticks, along with the conserved π1 bulge. The view in a is from the matrix and is the same as that in Fig. 5a. The in-membrane view in b) highlights the membrane-embedded amphipathic helix of NDUFS8 and shows how the sandwiched PE molecule helps to bridge the vertical and horizontal helices of ND1 and NDUFS8, respectively.
Extended Data Fig. 7 A comparison of residues in the ND1 subunit of our Y. lipolytica complex I with a DDM (n-dodecyl β-d-maltoside) molecule bound, the Y. lipolytica complex I with ubiquinone-9 bound (PDB 6RFR)19, and the active mouse model (PDB 6G2J)20 with nothing observed in the ubiquinone-binding site.
a, b and c show the hydrogen-bonding networks (dashed lines) present at the hydrophilic kink region of the ubiquinone-binding channel. The ND1-bound DDM molecule is displayed in orange sticks for Y. lipolytica and as an outline in the active mouse enzyme as a reference. The ubiquinone-9 molecule is shown similarly. Residues of ND1 and NDUFS7 are coloured orange and purple sticks, respectively. Water molecules are in red spheres. Equivalent residues where displayed are as follows: Q34(MmQ32), R36(MmR34), D99(MmD78), D101(MmD80), R27(MmR25), T23(MmT21), D53(MmD51), W77(MmW56), Q113(MmQ92), R108(MmR87), Y232(MmY228), E206(MmE202), E231(MmE227), E208(MmE204), D203(MmD199), R199(MmR195), R297(MmR274), R302(MmR279), E26 (MmE24).
Supplementary information
Supplementary Information
Supplementary Tables 1–4.
Supplementary Video 1
Hydration of complex I and the antiporter-like (ND) subunits of complex I.
Supplementary Video 2
Differences between ND1, ND3 and ND6 NDUFS2 of our DDM-bound Yarrowia lipolytica model and the active mouse model (PDB 6G2J).
Supplementary Video 4
Movement of ND6-π1 in the transition from our DDM-bound Yarrowia lipolytica model to that of the active mouse model (PDB 6G2J).
Rights and permissions
About this article
Cite this article
Grba, D.N., Hirst, J. Mitochondrial complex I structure reveals ordered water molecules for catalysis and proton translocation. Nat Struct Mol Biol 27, 892–900 (2020). https://doi.org/10.1038/s41594-020-0473-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41594-020-0473-x
This article is cited by
-
The antipsychotic medications aripiprazole, brexpiprazole and cariprazine are off-target respiratory chain complex I inhibitors
Biology Direct (2023)
-
H2O2 selectively damages the binuclear iron-sulfur cluster N1b of respiratory complex I
Scientific Reports (2023)
-
Mechanism of rotenone binding to respiratory complex I depends on ligand flexibility
Scientific Reports (2023)
-
The assembly, regulation and function of the mitochondrial respiratory chain
Nature Reviews Molecular Cell Biology (2022)
-
Cryo-EM structures define ubiquinone-10 binding to mitochondrial complex I and conformational transitions accompanying Q-site occupancy
Nature Communications (2022)