Abstract
Neddylation is the post-translational protein modification most closely related to ubiquitination. Whereas the ubiquitin-like protein NEDD8 is well studied for its role in activating cullin−RING E3 ubiquitin ligases, little is known about other substrates. We developed serial NEDD8-ubiquitin substrate profiling (sNUSP), a method that employs NEDD8 R74K knock-in HEK293 cells, allowing discrimination of endogenous NEDD8- and ubiquitin-modification sites by MS after Lys-C digestion and K-εGG-peptide enrichment. Using sNUSP, we identified 607 neddylation sites dynamically regulated by the neddylation inhibitor MLN4924 and the de-neddylating enzyme NEDP1, implying that many non-cullin proteins are neddylated. Among the candidates, we characterized lysine 112 of the actin regulator cofilin as a novel neddylation event. Global inhibition of neddylation in developing neurons leads to cytoskeletal defects, altered actin dynamics and neurite growth impairments, whereas site-specific neddylation of cofilin at K112 regulates neurite outgrowth, suggesting that cofilin neddylation contributes to the regulation of neuronal actin organization.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
MS raw data files were deposited at UCSD MassIVE with the data set identifier MSV000084683. Source data for Figs. 2 and 4–7 and Extended Data Figs. 1, 3 and 5–8 are available with the paper online. All other data supporting the findings of this study are available from the corresponding authors upon reasonable request.
Change history
26 February 2020
A Correction to this paper has been published: https://doi.org/10.1038/s41594-020-0395-7
References
Kamitani, T. K., Kito, H. P., Nguyen, E. & Yeh, T. H. Characterization of NEDD8, a developmentally down-regulated ubiquitin-like protein. J. Biol. Chem. 272, 28557–28562 (1997).
Liakopoulos, D., Büsgen, T., Brychzy, A., Jentsch, S. & Pause, A. Conjugation of the ubiquitin-like protein NEDD8 to cullin-2 is linked to von Hippel-Lindau tumor suppressor function. Proc. Natl Acad. Sci. USA 96, 5510–5515 (1999).
Kumar, S., Tomooka, Y. & Noda, M. Identification of a set of genes with developmentally down-regulated expression in the mouse brain. Biochem. Biophys. Res. Commun. 185, 1155–1161 (1992).
Enchev, R. I., Schulman, Ba & Peter, M. Protein neddylation: beyond cullin–RING ligases. Nat. Rev. Mol. Cell Biol. 16, 30–44 (2015).
Xirodimas, D. P. Novel substrates and functions for the ubiquitin-like molecule NEDD8. Biochem. Soc. Trans. 36, 802–806 (2008).
Lyapina, S. et al. Promotion of NEDD8-CUL1 conjugate cleavage by COP9 signalosome. Science 292, 1382–1385 (2001).
Cope, G. A. et al. Role of predicted metalloprotease motif of Jab1/Csn5 in cleavage of Nedd8 from cul1. Science 298, 608–611 (2002).
Schwechheimer, C. et al. Interactions of the COP9 Signalosome with the E3 ubiquitin ligase SCFTIR1 in mediating auxin response. Science 292, 1379–1382 (2001).
Mendoza, H. M. et al. NEDP1, a highly conserved cysteine protease that deNEDDylates cullins. J. Biol. Chem. 278, 25637–25643 (2003).
Wu, K. et al. DEN1 is a dual function protease capable of processing the C terminus of Nedd8 and deconjugating hyper-neddylated CUL1. J. Biol. Chem. 278, 28882–28891 (2003).
Gan-Erdene, T. et al. Identification and characterization of DEN1, a deneddylase of the ULP family. J. Biol. Chem. 278, 28892–28900 (2003).
Gong, L., Kamitani, T., Millas, S. & Yeh, E. T. H. Identification of a novel isopeptidase with dual specificity for ubiquitin- and NEDD8-conjugated proteins. J. Biol. Chem. 275, 14212–14216 (2000).
Deshaies, R. J. & Joazeiro, C. A. P. RING domain E3 ubiquitin ligases. Annu. Rev. Biochem. 78, 399–434 (2009).
Xu, G., Paige, J. S. & Jaffrey, S. R. Global analysis of lysine ubiquitination by ubiquitin remnant immunoaffinity profiling. Nat. Biotechnol. 28, 868–873 (2010).
Bustos, D., Bakalarski, C. E., Yang, Y., Peng, J. & Kirkpatrick, D. S. Characterizing ubiquitination sites by peptide based immunoaffinity enrichment. Mol. Cell. Proteomics 11, 1529–1540 (2012).
Keuss, M. J. et al. Unanchored tri‐NEDD8 inhibits PARP‐1 to protect from oxidative stress‐induced cell death. EMBO J. 38, e100024 (2019).
Coleman, K. E. et al. SENP8 limits aberrant neddylation of NEDD8 pathway components to promote cullin-RING ubiquitin ligase function. Elife 6, e24325 (2017).
Leidecker, O., Matic, I., Mahata, B., Pion, E. & Xirodimas, D. P. The ubiquitin E1 enzyme Ube1 mediates NEDD8 activation under diverse stress conditions. Cell Cycle 11, 1142–1150 (2012).
Soucy, T. A. et al. An inhibitor of NEDD8-activating enzyme as a new approach to treat cancer. Nature 458, 732–736 (2009).
Kim, W. et al. Systematic and quantitative assessment of the ubiquitin-modified proteome. Mol. Cell 44, 325–340 (2011).
Wagner, S. A. et al. A proteome-wide, quantitative survey of in vivo ubiquitylation sites reveals widespread regulatory roles. Mol. Cell. Proteomics 10, M111.013284 (2011).
Tammsalu, T. et al. Proteome-wide identification of SUMO2 modification sites. Sci. Signal. 7, rs2 (2014).
Lumpkin, R. J. et al. Site-specific identification and quantitation of endogenous SUMO modifications under native conditions. Nat. Commun. 8, 1171 (2017).
Becker, J. et al. Detecting endogenous SUMO targets in mammalian cells and tissues. Nat. Struct. Mol. Biol. 20, 525–531 (2013).
Cope, G. A. & Deshaies, R. J. Targeted silencing of Jab1/Csn5 in human cells downregulates SCF activity through reduction of F-box protein levels. BMC Biochem. 7, 1 (2006).
Mergner, J., Heinzlmeir, S., Kuster, B. & Schwechheimer, C. DENEDDYLASE1 deconjugates NEDD8 from non-cullin protein substrates in Arabidopsis thaliana. Plant Cell 27, 741–753 (2015).
Mergner, J., Kuster, B. & Schwechheimer, C. DENEDDYLASE1 counters automodification of neddylating enzymes to maintain NEDD8 homeostasis in arabidopsis. J. Biol. Chem. 292, 3854–3865 (2017).
Chan, Y. et al. DEN1 deneddylates non-cullin proteins in vivo. J. Cell Sci. 121, 3218–3223 (2008).
Bailly, A. P. et al. The balance between mono- and NEDD8-chains controlled by NEDP1 upon DNA damage is a regulatory module of the HSP70 ATPase activity. Cell Reports 29, 212–224 (2019).
Rose, C. M. et al. Highly multiplexed quantitative mass spectrometry analysis of ubiquitylomes. Cell Syst. 3, 395–403.e4 (2016).
Udeshi, N. D., Mertins, P., Svinkina, T. & Carr, S. A. Large-scale identification of ubiquitination sites by mass spectrometry. Nat. Protoc. 8, 1950–1960 (2013).
Fischer, E. S. et al. The molecular basis of CRL4 DDB2/CSA ubiquitin ligase architecture, targeting, and activation. Cell 147, 1024–1039 (2011).
Clough, T., Thaminy, S., Ragg, S., Aebersold, R. & Vitek, O. Statistical protein quantification and significance analysis in label-free LC-MS experiments with complex designs. BMC Bioinformatics 13, S16 (2012).
Pan, Z.-Q., Kentsis, A., Dias, D. C., Yamoah, K. & Wu, K. Nedd8 on cullin: building an expressway to protein destruction. Oncogene 23, 1985–1997 (2004).
Gareau, J. R. & Lima, C. D. The SUMO pathway: emerging mechanisms that shape specificity, conjugation and recognition. Nat. Rev. Mol. Cell Biol. 11, 861–871 (2010).
Rodriguez, M. S., Dargemont, C. & Hay, R. T. SUMO-1 conjugation in vivo requires both a consensus modification motif and nuclear targeting. J. Biol. Chem. 276, 12654–12659 (2001).
Vogl, A. M. et al. Neddylation inhibition impairs spine development, destabilizes synapses and deteriorates cognition. Nat. Neurosci. 18, 239–251 (2015).
Akimov, V. et al. UbiSite approach for comprehensive mapping of lysine and N-terminal ubiquitination sites. Nat. Struct. Mol. Biol. 25, 631–640 (2018).
Flynn, K. C. et al. ADF/cofilin-mediated actin retrograde flow directs neurite formation in the developing brain. Neuron 76, 1091–1107 (2012).
Bellenchi, G. C. et al. N-cofilin is associated with neuronal migration disorders and cell cycle control in the cerebral cortex. Genes Dev. 21, 2347–2357 (2007).
Tedeschi, A. et al. ADF/cofilin-mediated actin turnover promotes axon regeneration in the adult CNS. Neuron 103, 1073–1085.e6 (2019).
Coles, C. H. & Bradke, F. Coordinating neuronal actin-microtubule dynamics. Curr. Biol. 25, R677–R691 (2015).
Witte, H., Neukirchen, D. & Bradke, F. Microtubule stabilization specifies initial neuronal polarization. J. Cell Biol. 180, 619–632 (2008).
Bernstein, B. W. & Bamburg, J. R. ADF/cofilin: a functional node in cell biology. Trends Cell Biol. 20, 187–195 (2010).
Hotulainen, P. Actin-depolymerizing factor and cofilin-1 play overlapping roles in promoting rapid F-actin depolymerization in mammalian nonmuscle cells. Mol. Biol. Cell 16, 649–664 (2004).
Jayabalan, A. K. et al. NEDDylation promotes stress granule assembly. Nat. Commun. 7, 12125 (2016).
Jones, J. et al. A targeted proteomic analysis of the ubiquitin-like modifier Nedd8 and associated proteins. J. Proteome Res. 7, 1274–1287 (2008).
Xirodimas, D. P. et al. Ribosomal proteins are targets for the NEDD8 pathway. EMBO Rep. 9, 280–286 (2008).
Maghames, C. M. et al. NEDDylation promotes nuclear protein aggregation and protects the ubiquitin proteasome system upon proteotoxic stress. Nat. Commun. 9, 4376 (2018).
Emanuele, M. J. et al. Global identification of modular cullin-RING ligase substrates. Cell 147, 459–474 (2011).
Udeshi, N. D. et al. Methods for quantification of in vivo changes in protein ubiquitination following proteasome and deubiquitinase inhibition. Mol. Cell. Proteomics 11, 148–159 (2012).
Starita, L. M., Lo, R. S., Eng, J. K., von Haller, P. D. & Fields, S. Sites of ubiquitin attachment in Saccharomyces cerevisiae. Proteomics 12, 236–240 (2012).
Elia, A. E. H. et al. Quantitative proteomic atlas of ubiquitination and acetylation in the DNA damage response. Mol. Cell 59, 867–881 (2015).
Wagner, S. A. et al. Proteomic analyses reveal divergent ubiquitylation site patterns in murine tissues. Mol. Cell. Proteomics 11, 1578–1585 (2012).
Cappadocia, L. & Lima, C. D. Ubiquitin-like protein conjugation: structures, chemistry, and mechanism. Chem. Rev. 118, 889–918 (2017).
Tanaka, K. et al. Structural basis for cofilin binding and actin filament disassembly. Nat. Commun. 9, 1860 (2018).
Lappalainen, P., Fedorov, E. V., Fedorov, A. A., Almo, S. C. & Drubin, D. G. Essential functions and actin-binding surfaces of yeast cofilin revealed by systematic mutagenesis. EMBO J. 16, 5520–5530 (1997).
Rappsilber, J., Mann, M. & Ishihama, Y. Protocol for micro-purification, enrichment, pre-fractionation and storage of peptides for proteomics using StageTips. Nat. Protoc. 2, 1896–1906 (2007).
Bingol, B. et al. The mitochondrial deubiquitinase USP30 opposes parkin-mediated mitophagy. Nature 510, 370–375 (2014).
Eng, J. K., Jahan, T. A. & Hoopmann, M. R. Comet: an open-source MS/MS sequence database search tool. Proteomics 13, 22–24 (2013).
Käll, L., Canterbury, J. D., Weston, J., Noble, W. S. & MacCoss, M. J. Semi-supervised learning for peptide identification from shotgun proteomics datasets. Nat. Methods 4, 923–925 (2007).
Beausoleil, S. A., Villén, J., Gerber, S. A., Rush, J. & Gygi, S. P. A probability-based approach for high-throughput protein phosphorylation analysis and site localization. Nat. Biotechnol. 24, 1285–1292 (2006).
Kirkpatrick, D. S. et al. Phosphoproteomic characterization of DNA damage response in melanoma cells following MEK/PI3K dual inhibition. Proc. Natl Acad. Sci. USA 110, 19426–19431 (2013).
Choi, M. et al. MSstats: an R package for statistical analysis of quantitative mass spectrometry-based proteomic experiments. Bioinformatics 30, 2524–2526 (2014).
Mi, H. et al. PANTHER version 11: expanded annotation data from gene ontology and reactome pathways, and data analysis tool enhancements. Nucleic Acids Res. 45, D183–D189 (2016).
Bailey, T. L. et al. MEME Suite: tools for motif discovery and searching. Nucleic Acids Res. 37, 202–208 (2009).
Refojo, D. et al. Glutamatergic and dopaminergic neurons mediate anxiogenic and anxiolytic effects of CRHR1. Science 333, 1903–1907 (2011).
Nakahira, E. & Yuasa, S. Neuronal generation, migration, and differentiation in the mouse hippocampal primoridium as revealed by enhanced green fluorescent protein gene transfer by means of in utero electroporation. J. Comp. Neurol. 483, 329–340 (2005).
Barabas, F. M. et al. Automated quantification of protein periodic nanostructures in fluorescence nanoscopy images: abundance and regularity of neuronal spectrin membrane-associated skeleton. Sci. Reports 7, 16029 (2017).
Schönle, A. Imspector image acquisition & analysis software, v0.1 http://www.imspector.de. (2006).
Dotti, C. G., Sullivan, C. A. & Banker, G. A. The establishment of polarity by hippocampal neurons in culture. J. Neurosci. 8, 1454–1468 (1988).
Acknowledgements
PTMscan studies were performed at Genentech under a limited-use license from Cell Signaling Technology. We thank A. Attorresi, C. Pollak and S. Romano for helping with confocal and live-imaging studies. We also thank J. Cupp and members of the FACS lab for assistance. This work was supported by the German Science Foundation Collaborative Research Centre (CRC) 870 (W.W.) and by the Network Fund of the Helmholtz Association (AMPro project, to W.W.). This work was also supported by the DFG SPP1365 grant (D.R.), the Max Planck Society (D.R. and F.D.S.), the Volkswagen Stiftung (D.R.), the Agencia Nacional de Promoción Científica y Tecnológica, Argentina (D.R. and F.D.S.), the Alexander von Humboldt Foundation (F.D.S.) and the fondo para la convergencia estructural del mercosur-FOCEM (D.R.).
Author information
Authors and Affiliations
Contributions
A.M.V. designed the study, performed experiments, and analyzed results. L.P., T.H., E.V., and D.S.K. designed, performed and analyzed proteomic experiments. R.B. and S.G. performed and analyzed cultured neuron and in utero electroporation experiments. M.D.B. and F.D.S. set up and performed super-resolution microscopy. P.Y. performed consensus site analysis. A.H. designed CRISPR constructs. M.A. performed and analyzed imaging experiments. C.C.H. helped analyze cytoskeleton experiments. W.W. directed the generation of Nedp1–/– mice. M.S. and D.R. directed the study. A.M.V., D.S.K., D.R. and M.S. wrote the manuscript. All authors read and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
A.M.V., L.P., E.V., T.H., M.A., A.H., C.C.H., D.S.K. and M.S. are employees and shareholders of Genentech, Inc., a member of the Roche group. The authors declare no additional competing financial interests.
Additional information
Peer review information Katarzyna Marcinkiewicz was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 NEDD8R74K knock-in (KI) and NEDP1 knockout (KO) strategy for sNUSP.
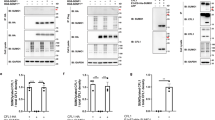
a, Representative immunoblotting of total cell lysates of Nedp1WT and Nedp1KO CRISPR HEK293 cell clones (KO #1: sgRNA 1 + 4; KO #2: sgRNA 2 + 5), with anti-NEDD8, anti-NEDP1, anti-tubulin, and anti-actin antibodies. b, Rescue of neddylation patterns in Nedp1KO cells by re-expression of NEDP1WT. Nedp1WT and Nedp1KO HEK293 cell clones were transiently transfected with pcDNA3.1EMPTY, NEDP1WT-HA, NEDP1C163A-HA, or CSN5WT-FLAG constructs, and treated with DMSO or 2 μM MLN4924 for 24 h, as indicated. Representative immunoblotting of total cell lysates with anti-NEDD8, anti-NEDP1, anti-tubulin, anti-actin, and anti-GAPDH antibodies. c, Analysis of neddylation and ubiquitination patterns of CRISPR Nedd8WT or R74K Nedp1WT or KO HEK293 cell clones. Representative immunoblotting of total cell lysates from indicated cell clones, treated with DMSO or 3 μM MLN4924 for 24 h, with anti-NEDD8, anti-ubiquitin (FK2), anti-tubulin, anti-actin, and anti-GAPDH antibodies. Uncropped blot images are provided as source data. Experiments were repeated at least three times with similar results. Molecular mass is indicated in kilodaltons.
Extended Data Fig. 2 MLN4924- and NEDP1-regulated NEDD8R74K K-εGG proteins from sNUSP.
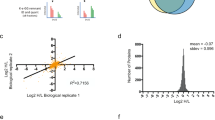
a–c, Tables show all significantly MLN4924- and NEDP1-regulated NEDD8R74K K-εGG proteins for key comparisons of DMSO vs. MLN4924 in Nedp1WT cells (a), DMSO vs. MLN4924 in Nedp1KO cells (b), and Nedp1KO (DMSO) vs. Nedp1WT (DMSO) cells (c) with log2 fold change >1.0 and –log10 adjusted P value >1.3 (P < 0.05). Experiment was repeated twice.
Extended Data Fig. 3 Analysis of neddylation substrates identified by sNUSP.
a, Cullin NEDD8R74K-specific and ubiquitin-specific K-εGG sites detected in sNUSP experiments. Asterisks mark known and experimentally confirmed canonical neddylation sites of cullins. b, Regulation of NEDD8R74K and ubiquitin K-εGG sites of CUL3 by MLN4924 (downregulation in red) and NEDP1 loss (downregulation in blue). c, Regulation of cullin neddylation by MLN4924 and NEDP1 at the protein level. d, Gene Ontology (GO) term enrichment analysis of neddylated proteins identified in NEDD8R74K K-εGG MS experiments (PANTHER, http://www.pantherdb.org/, overrepresentation test, P values defined by Fisher’s exact test). Protein class categories with positive fold enrichment and P values <0.01 are shown, and the number of hits compared to the category size is indicated after each GO term. e, Number of NEDD8R74K-modified (pink) and ubiquitin-modified (blue) K-εGG sites per protein. On average, 2.03 neddylation sites (1,327 sites/609 proteins) compared to 3.85 ubiquitination sites (12,599 sites/3,270 proteins) per protein; large majority of NEDD8 substrates with only one or two neddylation sites per protein (491 of 609 proteins), and one to five ubiquitination sites per protein for ubiquitinated substrates (2,658 of 3,270 proteins). Data for graphs in d,e are available as Source data.
Extended Data Fig. 4 Analysis of neddylation and ubiquitination events on substrate proteins.
a–g, Regulation of NEDD8R74K and ubiquitin K-εGG sites of (a) UBC12, (b) UBA3, (c) NEDD8, (d) PARP1, (e) CAND1, (f) LMNA and (g) LMNB1 by MLN4924 (downregulation in red) and Nedp1KO (upregulation in red).
Extended Data Fig. 5 Validation of cofilin as NEDD8 and NEDP1 substrate.
a, Structure of cofilin (green) and actin (purple and cyan) (modified from PDB 5YU8) with the cofilinK112 neddylation site (dark blue). b, Lysine (K) to arginine (R) mutagenesis of neddylated lysine residues identified by NEDD8R74K-specific K-εGG MS confirms K112 as the main neddylation site of cofilin. Nedp1KO HEK293 cells were transiently transfected with the different COF1BIO-HA constructs and treated with DMSO or 3 μM MLN4924 for 24 h, as indicated. Representative immunoblotting of denaturing biotin-streptavidin pulldown samples (COF1) and lysates for endogenous NEDD8 with anti-NEDD8 antibodies and COF1BIO-HA with anti-HA antibodies. Note, the neddylated COF1 band is absent in all samples with K112R mutations. c, Increased cofilin neddylation in Nedp1KO cells. Nedp1KO and Nedp1WT HEK293 cells were transiently transfected with COF1WT-BIO-HA constructs and treated with DMSO or 2 μM MLN4924 for 24 h, as indicated. Representative immunoblotting of denaturing biotin-streptavidin pulldown samples (COF1) and lysates for endogenous NEDD8 with anti-NEDD8 antibodies and COF1BIO-HA with anti-HA antibodies. Uncropped blot images for b and c are available as Source data. Experiments in b,c were repeated at least three times with similar results. Molecular mass is indicated in kilodaltons.
Extended Data Fig. 6 Regulation of the neuronal actin cytoskeleton by neddylation.
a, Neddylation-deficient neurons form enlarged axonal growth cones. Mouse hippocampal neurons treated with DMSO or 1 μM MLN4924 at DIV2 for 24 h, fixed at DIV3 and stained for microtubules (tyr-tubulin) and F-actin (phalloidin). Measurement of axonal growth cone area and actin/microtubule ratio in stage 3 neurons (unpaired, two-tailed t tests, growth cone area, t(22) = 3.25, **P < 0.01 (P = 0.0037); actin/microtubule ratio, t(22) = 2.413, *P < 0.05 (P = 0.0246), n = 11 control, 13 MLN4924 neurons, from one culture). Scale bar, 2 μm. b, Neddylation inhibition increases actin dynamics and retrograde flow. Spinning disk confocal microscopy (every 2 s for 5 min) of axon growth cones of DIV2 rat hippocampal neurons, nucleofected with LifeAct-GFP and treated with DMSO or 1 μM MLN4924 for 3−5 h. Measurement of relative changes in LifeAct-GFP pixel intensity (unpaired, two-tailed t test, t(72) = 2.511, *P < 0.05 (P = 0.0143), n = 38 DMSO and 36 MLN4924 growth cones, from four neuron cultures) and retrograde flow (unpaired, two-tailed t test, t(8) = 3.304, *P < 0.05 (P = 0.0108), each data point represents a single growth cone in which the retrograde flow was measured and averaged in kymographs of three ROIs, each for six events, n = 5 growth cones per condition, from two cultures). Scale bars, 10 μm. Related to Supplementary Video 2. Data for graphs are available as Source data. Data are presented as mean ± s.e.m.
Extended Data Fig. 7 Neddylation regulates neurite outgrowth in developing neurons.
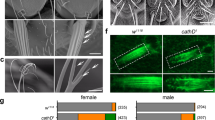
a, Representative images of time lapse imaging (DIV1–5) of mouse hippocampal neurons, separately nucleofected with ControlIRES-mRFP (red) or UBC12C111S-IRES-GFP (green) and mixed before plating. Quantification of neurons in developmental stages 1–3 at DIV1 and 3 (two-way ANOVA, treatment x stage interaction, F(6,219) = 66.42, ***P < 0.001; Tukey’s multiple comparisons test, comparing stages 1, 2 and 3 for control vs. UBC12C111S, *P < 0.05, ***P < 0.001; n = 12−27 fields of view per group, from two cultures). Arrow marks growing axon, arrowhead marks UBC12C111S-expressing cell. Scale bar, 25 μm. b, Representative images of rat hippocampal neurons, nucleofected with pCAGGS-GFP, treated with DMSO or 0.5 μM MLN4924 at DIV1 and 2, imaged at DIV3 (24 h after last treatment). Quantification of total neurite outgrowth per cell (unpaired, two-tailed t test, t(6) = 8.002, ***P < 0.001 (P = 0.0002); n = 4 areas with >1,000 neurons each, from two cultures). Scale bar, 100 μm. Data for graphs are available as Source data. Data are presented as mean ± s.e.m.
Extended Data Fig. 8 General neddylation inhibition and specific blockade of NEDD8 conjugation to K112 reduce cofilin phosphorylation in neurons.
a, Neddylation inhibition does not change neuronal cofilin protein levels. Representative immunoblotting of total cell lysates of cultured rat cortical neurons, treated with DMSO, 1 μM MLN4924 or 1 μM MG132 for 2, 4, 6 and 24 h at DIV14–15, with anti-cofilin, anti-tubulin, anti-actin, anti-GAPDH, anti-NEDD8 and anti-ubiquitin (FK2) antibodies. Cofilin levels were measured and normalized to tubulin (Kruskal-Wallis tests; 2 h, *P = 0.0107, Dunn’s multiple comparison test, *P < 0.05 (DMSO vs. MG132); 4 h, P = 0.1965; 6 h, P = 0.5403; 24 h, P = 0.5474; n = 4 samples per treatment for 2, 4 and 6 h, n = 8 wells per treatment for 24 h, from two cultures). b, MLN4924 treatment decreases cofilin Ser3 phosphorylation. Immunoblotting of total cell lysates of cultured rat hippocampal neurons, treated with DMSO or 1 μM MLN4924 for 24, 12 or 8 h from DIV1–2 (representative blot) and DIV5–6, with anti-P-cofilin (Ser3), anti-cofilin, anti-tubulin, anti-actin, and anti-GAPDH antibodies. Quantification of P-cofilin/total cofilin levels (one-way ANOVA, F(5,35) = 14.3, ***P < 0.001; Sidak’s multiple comparisons (DMSO vs. MLN4924), *P < 0.05, ***P < 0.001; n = 6 DMSO and n = 7 or 8 MLN4924, from three neuron cultures). c, Reduced phosphorylation of non-neddylatable cofilinK112R. Immunoblotting of total cell lysates of cultured rat hippocampal neurons, nucleofected with cofilinBIO-HA WT or K112R constructs from DIV0–7, with anti-P-cofilin (Ser3), anti-cofilin, anti-tubulin, and anti-actin antibodies. Quantification of P-cofilin/total cofilin levels (unpaired, two-tailed t test, t(6) = 4.622, **P < 0.01 (P = 0.0036), n = 4 samples per group, from two cultures). Uncropped blot images and data for graphs are available as Source data. Molecular mass is indicated in kilodalton. Data are presented as mean ± s.e.m.
Supplementary information
Supplementary Information
Supplementary Notes, Supplementary Figs. 1–8 and Supplementary Table 3.
Supplementary Table 1
Quantitative sNUSP NEDD8R74K-specific K-εGG mass spectrometry data
Supplementary Table 2
Quantitative sNUSP ubiquitin K-εGG mass spectrometry data
Supplementary Video 1
Transient formation of ectopic lamellipodia structures by neddylation inhibition. Differential interference contrast (DIC) time lapse imaging of a DIV2 mouse hippocampal neuron before, during, and after 1 μM MLN4924 treatment. Experiment was repeated twice with similar results and with 10–15 cells imaged per experiment.
Supplementary Video 2
Increased actin dynamics and retrograde flow in axonal growth cones of MLN4924-treated neurons. Rat hippocampal neurons, nucleofected with LifeAct-GFP before plating and treated with DMSO or 1 μM MLN4924 at DIV2 (~43 h after plating). Axonal growth cones imaged 3–5 h later on a spinning disk confocal microscope (every 2 s for 5 min). Scale bar, 10 μm. Experiment was repeated four times with similar results. Related to Extended Data Fig. 6b.
Source data
Source Data Fig. 2
Source Data for graphs
Source Data Fig. 4
Unprocessed western blots
Source Data Fig. 5
Unprocessed western blots
Source Data Fig. 6
Source Data for graphs
Source Data Fig. 7
Source Data for graphs
Source Data Extended Data Fig. 1
Unprocessed western blots
Source Data Extended Data Fig. 3
Source Data for graphs
Source Data Extended Data Fig. 5
Unprocessed western blots
Source Data Extended Data Fig. 6
Source Data for graphs
Source Data Extended Data Fig. 7
Source Data for graphs
Source Data Extended Data Fig. 8
Source Data for graphs
Source Data Extended Data Fig. 8
Unprocessed western blots
Rights and permissions
About this article
Cite this article
Vogl, A.M., Phu, L., Becerra, R. et al. Global site-specific neddylation profiling reveals that NEDDylated cofilin regulates actin dynamics. Nat Struct Mol Biol 27, 210–220 (2020). https://doi.org/10.1038/s41594-019-0370-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41594-019-0370-3
This article is cited by
-
Protein neddylation and its role in health and diseases
Signal Transduction and Targeted Therapy (2024)
-
Noncanonical assembly, neddylation and chimeric cullin–RING/RBR ubiquitylation by the 1.8 MDa CUL9 E3 ligase complex
Nature Structural & Molecular Biology (2024)
-
UBE3B promotes breast cancer progression by antagonizing HIF-2α degradation
Oncogene (2023)
-
Activity-based profiling of cullin–RING E3 networks by conformation-specific probes
Nature Chemical Biology (2023)
-
N-terminal α-amino SUMOylation of cofilin-1 is critical for its regulation of actin depolymerization
Nature Communications (2023)