Abstract
The long-range GABAergic input from the ventral tegmental area (VTA) to the nucleus accumbens (NAc) is relatively understudied, and therefore its role in reward processing has remained unknown. In the present study, we show, in both male and female mice, that long-range GABAergic projections from the VTA to the ventral NAc shell, but not to the dorsal NAc shell or NAc core, are engaged in reward and reinforcement behavior. We show that this GABAergic projection exclusively synapses on to cholinergic interneurons (CINs) in the ventral NAc shell, thereby serving a specialized function in modulating reinforced reward behavior through the inhibition of ventral NAc shell CINs. These findings highlight the diversity in the structural and functional topography of VTA GABAergic projections, and their neuromodulatory interactions across the dorsoventral gradient of the NAc shell. They also further our understanding of neuronal circuits that are directly implicated in neuropsychiatric conditions such as depression and addiction.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The raw data that support the findings of the present study are available from the corresponding authors upon request.
Code availability
Custom MATLAB code used for data analysis is available at https://github.com/BruchasLab.
Change history
09 September 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41593-021-00928-z
References
Schultz, W. Dopamine reward prediction-error signalling: a two-component response. Nat. Rev. Neurosci. 17, 183–195 (2016).
Hamid, A. A. et al. Mesolimbic dopamine signals the value of work. Nat. Neurosci. 19, 117–126 (2016).
Salamone, J. D. & Correa, M. The mysterious motivational functions of mesolimbic dopamine. Neuron 76, 470–485 (2012).
Watabe-Uchida, M., Eshel, N. & Uchida, N. Neural circuitry of reward prediction error. Annu. Rev. Neurosci. 40, 373–394 (2017).
Koob, G. F. & Le Moal, M. Addiction and the brain antireward system. Annu Rev. Psychol. 59, 29–53 (2008).
Nestler, E. J. & Carlezon, W. A. Jr. The mesolimbic dopamine reward circuit in depression. Biol. Psychiatry 59, 1151–1159 (2006).
Ikemoto, S. & Bonci, A. Neurocircuitry of drug reward. Neuropharmacology 76, 329–341 (2014).
Morales, M. & Margolis, E. B. Ventral tegmental area: cellular heterogeneity, connectivity and behaviour. Nat. Rev. Neurosci. 18, 73–85 (2017).
Buck, S. A., Torregrossa, M. M., Logan, R. W. & Freyberg, Z. Roles of dopamine and glutamate co-release in the nucleus accumbens in mediating the actions of drugs of abuse. FEBS J. 288, 1462–1474 (2021).
Creed, M. C., Ntamati, N. R. & Tan, K. R. VTA GABA neurons modulate specific learning behaviors through the control of dopamine and cholinergic systems. Front. Behav. Neurosci. 8, 8 (2014).
Van Bockstaele, E. J. & Pickel, V. M. GABA-containing neurons in the ventral tegmental area project to the nucleus accumbens in rat brain. Brain Res. 682, 215–221 (1995).
Brown, M. T. C. et al. Ventral tegmental area GABA projections pause accumbal cholinergic interneurons to enhance associative learning. Nature 492, 452–456 (2012).
Breton, J. M. et al. Relative contributions and mapping of ventral tegmental area dopamine and GABA neurons by projection target in the rat. J. Comp. Neurol. 527, 916–941 (2019).
Beier, K. T. et al. Topological organization of ventral tegmental area connectivity revealed by viral–genetic dissection of input–output relations. Cell Rep. 26, 159–167.e6 (2019).
Lee, J., Finkelstein, J., Choi, J. Y. & Witten, I. B. Linking cholinergic interneurons, synaptic plasticity, and behavior during the extinction of a cocaine–context association. Neuron 90, 1071–1085 (2016).
Badrinarayan, A. et al. Aversive stimuli differentially modulate real-time dopamine transmission dynamics within the nucleus accumbens core and shell. J. Neurosci. 32, 15779–15790 (2012).
Al-Hasani, R. et al. Distinct subpopulations of nucleus accumbens dynorphin neurons drive aversion and reward. Neuron 87, 1063–1077 (2015).
Castro, D. C. & Bruchas, M. R. A motivational and neuropeptidergic hub: anatomical and functional diversity within the nucleus accumbens shell. Neuron 102, 529–552 (2019).
Reed, S. J. et al. Coordinated reductions in excitatory input to the nucleus accumbens underlie food consumption. Neuron 99, 1260–1273.e4 (2018).
Castro, D. C. & Berridge, K. C. Opioid hedonic hotspot in nucleus accumbens shell: mu, delta, and kappa maps for enhancement of sweetness ‘liking’ and ‘wanting’. J. Neurosci. 34, 4239–4250 (2014).
de Jong, J. W. et al. A neural circuit mechanism for encoding aversive stimuli in the mesolimbic dopamine system. Neuron 101, 133–151.e7 (2019).
Gunaydin, L. A. et al. Natural neural projection dynamics underlying social behavior. Cell 157, 1535–1551 (2014).
Soden, M. E. et al. Anatomic resolution of neurotransmitter-specific projections to the VTA reveals diversity of GABAergic inputs. Nat. Neurosci. 23, 968–980 (2020).
Pontieri, F. E., Tanda, G. & Di Chiara, G. Intravenous cocaine, morphine, and amphetamine preferentially increase extracellular dopamine in the ‘shell’ as compared with the ‘core’ of the rat nucleus accumbens. Proc. Natl Acad. Sci. USA 92, 12304–12308 (1995).
Chen, B. T., Hopf, F. W. & Bonci, A. Synaptic plasticity in the mesolimbic system: therapeutic implications for substance abuse. Ann. NY Acad. Sci. 1187, 129–139 (2010).
Nestler, E. J. & Aghajanian, G. K. Molecular and cellular basis of addiction. Science 278, 58–63 (1997).
Hangya, B., Ranade, S. P., Lorenc, M. & Kepecs, A. Central cholinergic neurons are rapidly recruited by reinforcement feedback. Cell 162, 1155–1168 (2015).
Witten, I. B. et al. Cholinergic interneurons control local circuit activity and cocaine conditioning. Science 330, 1677–1681 (2010).
Jing, M. et al. An optimized acetylcholine sensor for monitoring in vivo cholinergic activity. Nat. Methods 17, 1139–1146 (2020).
Klapoetke, N. C. et al. Independent optical excitation of distinct neural populations. Nat. Methods 11, 338–346 (2014).
Berridge, K. C. & Robinson, T. E. What is the role of dopamine in reward: hedonic impact, reward learning, or incentive salience? Brain Res. Rev. 28, 309–369 (1998).
van Zessen, R., Phillips, J. L., Budygin, E. A. & Stuber, G. D. Activation of VTA GABA neurons disrupts reward consumption. Neuron 73, 1184–1194 (2012).
Rizzi, G., Li, Z., Hogrefe, N. & Tan, K. R. Lateral ventral tegmental area GABAergic and glutamatergic modulation of conditioned learning. Cell Rep. 34, 108867 (2021).
Lowes, D. C. et al. Ventral tegmental area GABA neurons mediate stress-induced blunted reward-seeking in mice. Nat. Commun. 12, 1–13 (2021).
Aosaki, T. et al. Responses of tonically active neurons in the primate’s striatum undergo systematic changes during behavioral sensorimotor conditioning. J. Neurosci. 14, 3969–3984 (1994).
Apicella, P. Leading tonically active neurons of the striatum from reward detection to context recognition. Trends Neurosci. 30, 299–306 (2007).
Collins, A. L. & Saunders, B. T. Heterogeneity in striatal dopamine circuits: form and function in dynamic reward seeking. J. Neurosci. Res. 98, 1046–1069 (2020).
Shin, J. H., Adrover, M. F., Wess, J. & Alvarez, V. A. Muscarinic regulation of dopamine and glutamate transmission in the nucleus accumbens. Proc. Natl Acad. Sci. USA 112, 8124–8129 (2015).
Dorst, M. C. et al. Polysynaptic inhibition between striatal cholinergic interneurons shapes their network activity patterns in a dopamine-dependent manner. Nat. Commun. 11, 5113 (2020).
Lewis, R. G. et al. Dopaminergic control of striatal cholinergic interneurons underlies cocaine-induced psychostimulation. Cell Rep. 31, 107527 (2020).
Shin, J. H., Adrover, M. F. & Alvarez, V. A. Distinctive modulation of dopamine release in the nucleus accumbens shell mediated by dopamine and acetylcholine receptors. J. Neurosci. 37, 11166–11180 (2017).
Chuhma, N., Mingote, S., Moore, H. & Rayport, S. Dopamine neurons control striatal cholinergic neurons via regionally heterogeneous dopamine and glutamate signaling. Neuron 81, 901–912 (2014).
Zhang, Y.-F. & Cragg, S. J. Pauses in striatal cholinergic interneurons: what is revealed by their common themes and variations? Front. Syst. Neurosci. 11, 80 (2017).
Crespo, J. A., Stöckl, P., Zorn, K., Saria, A. & Zernig, G. Nucleus accumbens core acetylcholine is preferentially activated during acquisition of drug- vs food-reinforced behavior. Neuropsychopharmacology 33, 3213–3220 (2008).
Aitta-aho, T. et al. Accumbal cholinergic interneurons differentially influence motivation related to satiety signaling. eNeuro https://doi.org/10.1523/ENEURO.0328-16.2017 (2017).
Pratt, W. E. & Kelley, A. E. Nucleus accumbens acetylcholine regulates appetitive learning and motivation for food via activation of muscarinic receptors. Behav. Neurosci. 118, 730–739 (2004).
Nougaret, S. & Ravel, S. Modulation of tonically active neurons of the monkey striatum by events carrying different force and reward information. J. Neurosci. 35, 15214–15226 (2015).
Collins, A. L. et al. Dynamic mesolimbic dopamine signaling during action sequence learning and expectation violation. Sci. Rep. 6, 20231 (2016).
Cachope, R. et al. Selective activation of cholinergic interneurons enhances accumbal phasic dopamine release: setting the tone for reward processing. Cell Rep. 2, 33–41 (2012).
London, T. D. et al. Coordinated ramping of dorsal striatal pathways preceding food approach and consumption. J. Neurosci. 38, 3547–3558 (2018).
Threlfell, S. et al. Striatal dopamine release is triggered by synchronized activity in cholinergic interneurons. Neuron 75, 58–64 (2012).
Picciotto, M. R., Higley, M. J. & Mineur, Y. S. Acetylcholine as a neuromodulator: cholinergic signaling shapes nervous system function and behavior. Neuron 76, 116–129 (2012).
Dautan, D. et al. A major external source of cholinergic innervation of the striatum and nucleus accumbens originates in the brainstem. J. Neurosci. 34, 4509–4518 (2014).
Tashakori-Sabzevar, F. & Ward, R. D. Basal forebrain mediates motivational recruitment of attention by reward-associated cues. Front. Neurosci. 12, 786 (2018).
Ballinger, E., Ananth, M., Talmage, D. A. & Role, L. Basal forebrain cholinergic circuits and signaling in cognition and cognitive decline. Neuron 91, 1199–1218 (2016).
Koob, G. F. & Volkow, N. D. Neurocircuitry of addiction. Neuropsychopharmacology 35, 217–238 (2010).
Saunders, B. T. & Janak, P. H. Nucleus accumbens plasticity underlies multifaceted behavioral changes associated with addiction. Biol. Psychiatry 75, 92–93 (2014).
Scofield, M. D. et al. The nucleus accumbens: mechanisms of addiction across drug classes reflect the importance of glutamate homeostasis. Pharm. Rev. 68, 816–871 (2016).
Di Chiara, G. Nucleus accumbens shell and core dopamine: differential role in behavior and addiction. Behav. Brain Res 137, 75–114 (2002).
Hikida, T., Kitabatake, Y., Pastan, I. & Nakanishi, S. Acetylcholine enhancement in the nucleus accumbens prevents addictive behaviors of cocaine and morphine. Proc. Natl Acad. Sci. USA 100, 6169–6173 (2003).
Gonzales, K. K. & Smith, Y. Cholinergic interneurons in the dorsal and ventral striatum: anatomical and functional considerations in normal and diseased conditions. Ann. NY Acad. Sci. 1349, 1–45 (2015).
Warner-Schmidt, J. L. et al. Cholinergic interneurons in the nucleus accumbens regulate depression-like behavior. Proc. Natl Acad. Sci. USA 109, 11360–11365 (2012).
Cheng, J., Umschweif, G., Leung, J., Sagi, Y. & Greengard, P. HCN2 channels in cholinergic interneurons of nucleus accumbens shell regulate depressive behaviors. Neuron 101, 662–672.e5 (2019).
Sparta, D. R. et al. Construction of implantable optical fibers for long-term optogenetic manipulation of neural circuits. Nat. Protoc. 7, 12–23 (2012).
Parker, K. E. et al. A paranigral VTA nociceptin circuit that constrains motivation for reward. Cell 178, 653–671.e19 (2019).
McCall, J. G. et al. CRH engagement of the locus coeruleus noradrenergic system mediates stress-induced anxiety. Neuron 87, 605–620 (2015).
Siuda, E. R. et al. Spatiotemporal control of opioid signaling and behavior. Neuron 86, 923–935 (2015).
Kim, T. et al. Injectable, cellular-scale optoelectronics with applications for wireless optogenetics. Science 340, 211–216 (2013).
Acknowledgements
We thank all at M.R.B.’s lab and R.A.-H.’s lab, and J. McCall for their helpful insight and discussion throughout the preparation of the manuscript, and in particular C. Stander, L. Lawson, D. Blumenthal and A. Suko for their technical support. We thank our funding sources: NIH/NIDA K99/R00 DA038725 (to R.A.-H.), NIH/NIDA F31 DA051124 (to C.E.P), Addictions, Drug and Alcohol Institute (ADAI) Fellowship (to D.J.M), SCAN Design Fellowship (to S.E.S), Mary Gates Fellowship (to A.E.J), NARSAD Young Investigator Grant from the Brain and Behavior Research Foundation, grant no. 28243 (to R.A.-H.), NIH/NIMH K99/R00 MH109627 (to J.C.L) and NIH/NIDA R37 DA033396, P30 DA048736 (to M.R.B).
Author information
Authors and Affiliations
Contributions
R.A.-H., G.P.S., R.G. and M.R.B. conceptualized and designed the experiments. R.A.-H., R.G., G.P.S., T.E.H., S.E.S., A.J.E. and J.C.L. performed the surgeries. J.C.L. and D.J.M. performed the electrophysiology. R.G., S.E.S. and C.E.P. performed the fiber photometry. R.G. and C.E.P. recorded the ITI. R.A.-H., R.G., A.J.E., T.E.H., G.P.S. and J.C.L. carried out the immunohistochemistry. R.G. did the uncued reward delivery. R.A.-H., R.G. and G.P.S. performed the operant self-stimulation and real-time place testing. R.G., C.E.P. and S.E.S. carried out the operant behavior paradigm (photometry) and the pavlovian behavior paradigm (photometry). Y.L. and M.J. provided the sensor resources. R.A.-H., R.G., G.P.S., C.E.P., J.C.L., V.A.A. and M.R.B. wrote the original draft, and reviewed and edited it. R.A.-H. and M.R.B. acquired funds. R.A.-H. and M.R.B. administered the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Neuroscience thanks William Howe and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
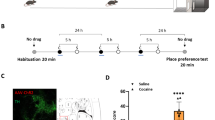
Extended Data Fig. 1 VTA GABAergic terminals in the vNAcSh are preferentially engaged during reward reinforcement.
(a) Coronal brain cartoon of fiber placements in the dNAcSh (light blue; n = 5) or vNAcSh (dark blue; n = 7) of VGAT-Cre+ mice. (b) Inter-trial interval (ITI) head entries for Pavlovian training, vNAcSh (n = 7 mice; P1 vs. P5 paired t test p = 0.0008, t = 6.274, df=6). (c) Mean vNAcSh recorded calcium activity during pellet retrieval averaged across all trials from day 1-5 of Pavlovian conditioning (n = 7 mice). Data represented as mean +/− SEM. (d) Mean vNAcSh recorded calcium activity during unrewarded, ITI head entries averaged across all trials from day 1-5 of Pavlovian conditioning (n = 7 mice). Data represented as mean +/− SEM. (e) Mean vNAcSh recorded calcium activity averaged across all trials from day 3 of Pavlovian conditioning (n = 7 mice). Data represented as mean +/− SEM. (f) Mean dNAcSh recorded calcium activity averaged across all trials from day 3 of Pavlovian conditioning (n = 5 mice). Data represented as mean +/− SEM. (g) Mean vNAcSh recorded calcium activity averaged across all trials from day 7 of Pavlovian conditioning extinction (n = 7 mice). Data represented as mean +/− SEM. (h) Mean dNAcSh recorded calcium activity averaged across all trials from day 7 of Pavlovian conditioning extinction (n = 5 mice). Data represented as mean +/− SEM. (i) Operant training schedule with photometry recordings, as indicated. (j) Top – vNAcSh active nosepokes for Operant training days (n = 7 mice; O1 vs. O3 paired t test p > 0.0001, t = 14.24, df=6). Bottom – dNAcSh active nosepokes for Operant training days (n = 5 mice; O1 vs. O3 paired t test p = 0.0035, t = 6.180, df=4). Data represented as mean +/− SEM. (k) Peak fluorescence during cue period (0-6 s) for Operant training days. Top – vNAcSh (n = 7 mice; O1 vs. O3 paired t test p = 0.5002, t = 0.6759, df=142). Bottom – dNAcSh (n = 5 mice; O1 vs. O3 paired t test p = 0.0047, t = 2.871, df=146). Data represented as mean +/− SEM. (l) Peak fluorescence during reward period (6-30 s) for Operant training days. Top – vNAcSh (n = 7 mice; O1 vs. O3 paired t test p = 0.9724, t = 03463, df=141). Bottom – dNAcSh (n = 5 mice; O1 vs. O3 vs. P5 paired t test p = 0.0003, t = 3.702, df=146). Data represented as mean +/− SEM. (m) Top – Peak fluorescence during cue period (0-6 s) comparing vNAcsh (n = 7 mice) vs. dNAcSh (n = 5 mice) for Operant training (unpaired t test, p = 0.6729, t = 0.4222, df=1038). Bottom - Peak fluorescence during reward period (6-30 s) comparing vNAcsh vs. dNAcSh for Operant training (unpaired t test, p < 0.0001, t = 9.513, df=1038). Data represented as mean +/− SEM. (n) Mean vNAcSh recorded calcium activity averaged across all trials from day 1-5 of Operant conditioning (n = 7 mice). Data represented as mean +/− SEM. (o) Heatmap raster plot of vNAcSh recorded calcium activity for every trial of Operant conditioning across all days. Trials are displayed in ascending order by average activity following pellet delivery (n = 7 mice). (p) Mean dNAcSh recorded calcium activity averaged across all trials from day 1-5 of Operant conditioning (n = 5 mice). Data represented as mean +/− SEM. (q) Heatmap raster plot of vNAcSh recorded calcium activity for every trial of Operant conditioning across all days. Trials are displayed in ascending order by average activity following pellet delivery (n = 5 mice).
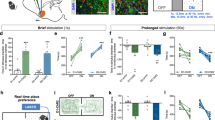
Extended Data Fig. 2 Photo-activation of VTA GABAergic projections to the vNAcSh promotes positive reinforcement.
(a) Cartoon of ventral NAcSh fiber optic placement. (b) Representative examples of real-time mouse heatmap raster plots following no stimulation, 2 Hz, 10 Hz, 20 Hz, 40 Hz, and 60 Hz ventral NAcSh photostimulation. (c) Hit map showing fiber optic placements of each individual mouse included in the ventral NAcSh ChR2 group. (d) Frequency response curve showing photostimulation of vGAT terminal in vNAcSh at no stimulation, 2 Hz, 5 Hz, 10 Hz, 20 Hz, 40 Hz, and 60 Hz; 20 ms pulse width. Photostimulation shows significant preference at 40 and 60 Hz. Unpaired Student’s t Test, two-tailed, Control vs ChR2, *p < 0.05; Unpaired Student’s t Test, two-tailed, ChR2 group 0 vs 40 Hz,**p < 0.01 (data represented as mean +/− SEM, n = 6 mice/Control,7 mice/ChR2). (e) Cartoon of dorsal NAcSh fiber optic placement. (f) Representative examples of real-time mouse heatmap raster plots following no stimulation, 2 Hz, 10 Hz, 20 Hz, 40 Hz, and 60 Hz dorsal NAcSh photostimulation. (g) Hit map showing fiber optic placements of each individual mouse included in the dorsal NAcSh ChR2 group. (h) Data showing number of nose pokes during fixed-ratio 1 testing versus fixed-ratio 3 testing (data represented as mean +/− SEM, n = 6 mice/FR-3, 9 mice/FR-1). (i) Data showing number of nose pokes during progressive ratio testing with no significant differences between control and experimental groups ((data represented as mean +/− SEM, n = 6 mice/Control, 9 micee/Experimental). (j) Frequency response curve showing photostimulation of vGAT terminal in dNAcSh at no stimulation, 2 Hz, 5 Hz, 10 Hz, 20 Hz, 40 Hz, and 60 Hz; 20 ms pulse width. Photostimulation shows no significant preference. Control vs ChR2 (data represented as mean + - SEM, n = 6 mice/Control, n = 10 mice/ChR2). (k) Cartoon of NAcc fiber optic placement. (l) Hit map showing fiber optic placements of each individual mouse included in the NAcC ChR2 group. (m) Data showing number of nose pokes during fixed-ratio 1 testing versus fixed-ratio 3 testing (data represented as mean + - SEM, n = 7 mice/FR-3, 11 mice/FR-1). (n) Data showing number of nose pokes during progressive ratio testing with no significant differences between control and experimental groups ((data represented as mean + - SEM, n = 7 mice/Control, 11 mice/Experimental). (o) Frequency response curve showing photostimulation of vGAT terminal in NAcC at no stimulation, 2 Hz, 5 Hz, 10 Hz, 20 Hz, 40 Hz, and 60 Hz; 20 ms pulse width. Photostimulation shows no significant preference. Control vs ChR2 (data represented as mean + - SEM, n = 6 mice/Control, 8 mice/ChR2). (p) Total head entries performed in matched, counterbalanced sessions comparing laser on vs. off (pulse − 2 s on, 2 s ramp down, 1 s off) in the JAWS group (n = 7 mice; Off vs. On paired t test p = 0.0269, t = 2.913, df=6). Right – YFP group (n = 7 mice; Off vs. On paired t test p = 0.2739, t = 1.204, df=6). Data represented as mean +/− SEM. (q) Total head entries performed in matched, counterbalanced sessions comparing laser on vs. off (pulse − 2 s on, 2 s ramp down, 1 s off) in the YFP group (n = 7 mice; Off vs. On paired t test p = 0.2739, t = 1.204, df=6). Data represented as mean +/− SEM.
Extended Data Fig. 3 VTA GABAergic terminals project to and inhibit cholinergic interneurons in the NAc.
(a) Top – Saggital brain cartoon depicting rostral-caudal position. Bottom − 20X confocal image of rostral VTA (left; −2.9 A/P relative to bregma, scalebar – 100 𝜇m), middle VTA (middle; −3.4 A/P relative to bregma, scalebar – 100 𝜇m) and caudal VTA (right; −3.8 A/P relative to bregma, scalebar – 100 𝜇m) showing expression of ChR2 (green) and DAPI (blue). (b) Table comparing electrophysiological parameters for CINs vs. MSNs – Rm: unpaired t-test: t = 3.000, p = 0.0056, n = 12 (MSNs)−18(CINs), Cm: unpaired t-test: t = 0.3852, p = 0.7030, n = 12-18, sIPSC frequency: unpaired t-test: t = 3.617, p = 0.0040, n = 8 (CINs), 5(MSNs), sIPSC amplitude: unpaired t-test: t = 0.5700, p = 0.5802, n = 8,5, Holding current: 2-way RM ANOVA, time x cell type interaction: F1,18 = 5.031, p = 0.0377; main effect of cell type: F1,18 = 28.29, p < 0.0001, post-hoc Sidak t-test – baseline vs. gabazine: CINs: p = 0.02, MSNs: p = 0.8194). Data represented as mean +/− SEM. (c) oIPSC amplitude comparing 5 Hz vs. 40 Hz stimulation (5 Hz: 269 ± 64 pA; 40 Hz: 280 ± 62 pA; paired t-test, t = 0.7367, p = 0.4755, n = 13 cells/4 mice). Data represented as mean +/− SEM. (d) oIPSC area (nA*s) over 250 ms period comparing 5 Hz vs. 40 Hz stimulation (5 Hz: 8 ± 3 nA*s; 40 Hz: 18 ± 4 nA*s; paired t-test, t = 3.804, p = 0.0025, n = 13 cells/4 mice). Data represented as mean +/− SEM. (e) Image showing relative position of patch pipette for recordings in the vNAcSh. (f) Image showing relative position of patch pipette for recordings in the dNAcSh.
Extended Data Fig. 4 Photo-activation of vNAcSh cholinergic interneuron activity does not impact reinforcement.
(a) Cartoon of ventral NAcSh virus and fiber optic placement. (b) Placement map showing fiber optic placements of each individual mouse included in the vNAcSh ChR2 group. (c) Data showing number of nose pokes during FR-1 self stimulation with no significant differences between control and experimental groups (data represented as mean +/− SEM, n = 7 each, unpaired t test, p = 0.4165, t = 0.8416, df=12). (d) Cartoon of bilateral ventral NAcSh virus and fiber optic placement. (e) Representative examples of real-time mouse heatmap raster plots following no stimulation, 2 s phasic stimulation, and constant ventral NAcSh photostimulation. (f) Cartoon of bilateral ventral NAcSh virus and fiber optic placement. (g) No aversion real-time behavior following phasic photo-inhibition of ChAT neurons in ventral NAcSh (data represented as mean +/− SEM, n = 8 mice/Control, 12 mice/Halo). (h) Total head entries performed in matched, counterbalanced sessions comparing laser on vs. off (pulse − 2 s on, 2 s ramp down, 1 s off). Left – ChR2 group (n = 9 mice; Off vs. On paired t test p = 0.1328, t = 1.673, df=8). Right – YFP group (n = 6 mice; Off vs. On paired t test p = 0.1629, t = 1.635, df=5). Data represented as mean +/− SEM.
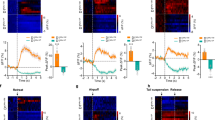
Extended Data Fig. 5 Cholinergic interneuron activity and acetylcholine release in the vNAcSh are inhibited during reward reinforcement.
(a) Coronal brain cartoon of fiber placements in the vNAcSh for VGAT-Cre+ mice injected with GRAB Ach3.0 (violet; n = 6). (b) Inter-trial interval (ITI) head entries for Pavlovian training, vNAcSh GRAB Ach3.0 (n = 6 mice; P1 vs. P5 paired t test p = 0.0053, t = 4.713, df=5). (c) Mean vNAcSh GRAB Ach3.0 recorded calcium activity during pellet retrieval averaged across all trials from day 1-5 of Pavlovian conditioning (n = 6 mice). Data represented as mean +/− SEM. (d) Mean vNAcSh GRAB Ach3.0 recorded calcium activity during unrewarded, ITI head entries averaged across all trials from day 1-5 of Pavlovian conditioning (n = 6 mice). Data represented as mean +/− SEM. (e) Coronal brain cartoon of fiber placements in the vNAcSh for CHAT-Cre+ mice injected with GCaMP6s (majenta; n = 5). (f) Inter-trial interval (ITI) head entries for Pavlovian training, vNAcSh GCaMP6s (n = 5 mice; P1 vs. P5 paired t test p = 0.0034, t = 6.20, df=4). (g) Mean vNAcSh GCaMP6s recorded calcium activity during pellet retrieval averaged across all trials from day 1-5 of Pavlovian conditioning (n = 5 mice). Data represented as mean +/− SEM. (h) Mean vNAcSh GCaMP6s recorded calcium activity during unrewarded, ITI head entries averaged across all trials from day 1-5 of Pavlovian conditioning (n = 5 mice). Data represented as mean +/− SEM. (i) Mean vNAcSh GRAB Ach3.0 recorded calcium activity averaged across all trials from day 3 of Pavlovian conditioning (n = 6 mice). Data represented as mean +/− SEM. (j) Mean vNAcSh GCaMP6s recorded calcium activity averaged across all trials from day 3 of Pavlovian conditioning (n = 5 mice). (k) Mean vNAcSh GRAB Ach3.0 recorded calcium activity averaged across all trials from day 7 of Pavlovian conditioning extinction (n = 6 mice). (l) Mean vNAcSh GCaMP6s recorded calcium activity averaged across all trials from day 7 of Pavlovian conditioning extinction (n = 5 mice). (m) Top – vNAcSh GRAB Ach3.0 active nosepokes for Operant training days (n = 6 mice; O1 vs. O3 paired t test p = 0.0287, t = 2.457, df=5). Bottom – vNAcSh GCaMP6s active nosepokes for Operant training days (n = 5 mice; O1 vs. O3 paired t test p = 0.0129, t = 4.274, df=4). Data represented as mean +/− SEM. (n) Peak fluorescence during cue period (0-6 s) for Operant training days. Top – vNAcSh GRAB Ach3.0 (n = 6 mice; O1 vs. O3 paired t test p = 0.0364, t = 1.810, df=263). Bottom – vNAcSh GCaMP6s (n = 5 mice; O1 vs. O3 paired t test p = 0.0197, t = 2.070, df=299). Data represented as mean +/− SEM. (o) Peak fluorescence during reward period (6-30 s) for Operant training days. Top – vNAcSh GRAB Ach3.0 (n = 6 mice; O1 vs. O3 paired t test p = 0.1010, t = 1.658, df=263). Bottom – vNAcSh GCaMP6s (n = 5 mice; O1 vs. O3 paired t test p = 0.1423, t = 1.471, df=299). Data represented as mean +/− SEM. (p) Minimum fluorescence during reward period (6-30 s) for Operant training days. Top – vNAcSh GRAB Ach3.0 (n = 6 mice; O1 vs. O3 paired t test p = 0.0005, t = 3.608, df=263). Bottom – vNAcSh GCaMP6s (n = 5 mice; O1 vs. O3 paired t test p = 0.5695, t = 0.5702, df=299). Data represented as mean +/− SEM. (q) Mean vNAcSh GRAB Ach3.0 recorded calcium activity averaged across all trials from day 1-5 of Operant conditioning (n = 6 mice). Data represented as mean +/− SEM. (r) Heatmap raster plot of vNAcSh GRAB Ach3.0 recorded calcium activity for every trial of Operant conditioning across all days. Trials are displayed in ascending order by average activity following pellet delivery (n = 7 mice). (s) Mean vNAcSh GCaMP6s recorded calcium activity averaged across all trials from day 1-5 of Operant conditioning (n = 5 mice). Data represented as mean +/− SEM. (t) Heatmap raster plot of vNAcSh GCaMP6s recorded calcium activity for every trial of Operant conditioning across all days. Trials are displayed in ascending order by average activity following pellet delivery (n = 5 mice).
Extended Data Fig. 6 Acetylcholine release in the dNAcSh is unchanged during reward reinforcement.
(a) Coronal brain cartoon of fiber placements in the dNAcSh for WT mice injected with GRAB Ach3.0 (plum; n = 5). (b) Top – dNAcSh GRAB Ach3.0 latency to eat for Pavlovian training days (n = 5 mice; P1 vs. P5 paired t test p = 0.0284, t = 2.654, df=4). Bottom – dNAcSh GRAB Ach3.0 active nosepokes for Operant training days (n = 5 mice; O1 vs. O3 paired t test p = 0.0057, t = 5.395, df=4). Data represented as mean +/− SEM. (c) Peak fluorescence during cue period (0-6 s) for Pavlovian or Operant training days. Top – dNAcSh GRAB Ach3.0 Pavlovian days (n = 5 mice; P1 vs. P5 paired t test p = 0.6406, t = 0.4683, df=95). Bottom – dNAcSh GRAB Ach3.0 Operant days (n = 5 mice; O1 vs. O3 paired t test p = 0.7091, t = 0.3753, df=46). Data represented as mean +/− SEM. (d) Peak fluorescence during reward period (6-30 s) for Pavlovian or Operant training days. Top – dNAcSh GRAB Ach3.0 Pavlovian days (n = 5 mice; P1 vs. P5 paired t test p = 0.3495, t = 0.9401, df=95). Bottom – dNAcSh GRAB Ach3.0 Operant days (n = 5 mice; O1 vs. O3 paired t test p = 0.3006, t = 1.047, df=46). Data represented as mean +/− SEM. (e) Minimum fluorescence during reward period (6-30 s) for Pavlovian or Operant training days. Top – dNAcSh GRAB Ach3.0 Pavlovian days (n = 5 mice; P1 vs. P5 paired t test p = 0.0016, t = 3.241, df=95). Bottom – dNAcSh GRAB Ach3.0 Operant days (n = 5 mice; O1 vs. O3 paired t test p = 0.7585, t = 0.3093, df=46). Data represented as mean +/− SEM. (f) Top – Minimum fluorescence during reward period (6-30 s) comparing vNAcsh (n = 6 mice) vs. dNAcSh (n = 5 mice) GRAB Ach3.0 for Pavlovian training (unpaired t test, p < 0.0001, t = 9.760, df=422). Bottom – Minimum fluorescence during reward period (6-30 s) comparing vNAcsh vs. dNAcSh GRAB Ach3.0 for Operant training (unpaired t test, p < 0.0001, t = 5.470, df=278). Data represented as mean +/− SEM. (g) Mean dNAcSh GRAB Ach3.0 recorded calcium activity averaged across all trials from day 1-5 of Pavlovian conditioning (n = 5 mice). Data represented as mean +/− SEM. (h) Heatmap raster plot of dNAcSh GRAB Ach3.0 recorded calcium activity for every trial of Operant conditioning across all days. Trials are displayed in ascending order by average activity following pellet delivery (n = 5 mice). (i) Mean dNAcSh GRAB Ach3.0 recorded calcium activity averaged across all trials from day 1-5 of Operant conditioning (n = 5 mice). Data represented as mean +/− SEM. (j) Heatmap raster plot of dNAcSh GRAB Ach3.0 recorded calcium activity for every trial of Operant conditioning across all days. Trials are displayed in ascending order by average activity following pellet delivery (n = 5 mice).
Supplementary information
Rights and permissions
About this article
Cite this article
Al-Hasani, R., Gowrishankar, R., Schmitz, G.P. et al. Ventral tegmental area GABAergic inhibition of cholinergic interneurons in the ventral nucleus accumbens shell promotes reward reinforcement. Nat Neurosci 24, 1414–1428 (2021). https://doi.org/10.1038/s41593-021-00898-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-021-00898-2
This article is cited by
-
Updating the striatal–pallidal wiring diagram
Nature Neuroscience (2024)
-
Deep tissue super-resolution imaging with adaptive optical two-photon multifocal structured illumination microscopy
PhotoniX (2023)
-
Dopamine and glutamate regulate striatal acetylcholine in decision-making
Nature (2023)
-
AAV11 enables efficient retrograde targeting of projection neurons and enhances astrocyte-directed transduction
Nature Communications (2023)
-
Neuropeptides Modulate Feeding via the Dopamine Reward Pathway
Neurochemical Research (2023)