Abstract
Pro-opiomelanocortin (POMC)-expressing neurons in the arcuate nucleus of the hypothalamus represent key regulators of metabolic homeostasis. Electrophysiological and single-cell sequencing experiments have revealed a remarkable degree of heterogeneity of these neurons. However, the exact molecular basis and functional consequences of this heterogeneity have not yet been addressed. Here, we have developed new mouse models in which intersectional Cre/Dre-dependent recombination allowed for successful labeling, translational profiling and functional characterization of distinct POMC neurons expressing the leptin receptor (Lepr) and glucagon like peptide 1 receptor (Glp1r). Our experiments reveal that POMCLepr+ and POMCGlp1r+ neurons represent largely nonoverlapping subpopulations with distinct basic electrophysiological properties. They exhibit a specific anatomical distribution within the arcuate nucleus and differentially express receptors for energy-state communicating hormones and neurotransmitters. Finally, we identify a differential ability of these subpopulations to suppress feeding. Collectively, we reveal a notably distinct functional microarchitecture of critical metabolism-regulatory neurons.
Similar content being viewed by others
Main
The melanocortin circuitry comprising agouti-related peptide (AgRP)-expressing neurons and POMC-expressing neurons in the arcuate nucleus of the hypothalamus (ARC) represents a prototypic homeostatic regulatory neurocircuit of metabolic homeostasis1. These targets of insulin, leptin and glucagon like peptide (Glp1) integrate multiple inputs to compute the energy state of the organism and adapt feeding behavior2. The POMC pro-peptide is processed to α-melanocyte-stimulating hormone, which is an activator of the melanocortin 4 receptor3 to suppress food intake4. Conversely, AgRP released from AgRP neurons acts as an inverse agonist on the melanocortin 4 receptor, thereby promoting feeding5. In addition, the melanocortin circuitry is an integrative regulator of numerous physiological functions. For instance, insulin action in AgRP neurons is required to efficiently suppress hepatic glucose production, and abrogation of insulin and leptin receptors from POMC neurons causes diabetes in mice6. Further, acute chemogenetic or optogenetic activation of AgRP neurons controls insulin sensitivity7. Thus, the melanocortin circuitry integrates the energy state of an organism to correspondingly adapt food intake as well as substrate flux across different organs8.
Although POMC neurons have been considered a homogeneous cell group, previous studies have highlighted their functional diversification. Specifically, 30% of POMC neurons increase firing in response to leptin in brain slice electrophysiology, and this response does not overlap with POMC cells in which insulin modulates firing9. Similarly, some POMC neurons express GABAergic markers and others express glutamatergic markers10. Moreover, studies using single-cell mRNA sequencing (RNA-seq) revealed a striking molecular heterogeneity of these neurons11,12. However, the molecular basis and associated functional consequences of their heterogeneity remain unknown.
Modern molecular systems neuroscience has been revolutionized through the ability to characterize neuronal subtypes on a molecular level at single-cell resolution and to functionally investigate distinct neurocircuits through employment of molecular tools for cell-type-specific manipulations. Here, Cre-loxP-mediated recombination provides a critical repertoire for cell-type-specific inactivation or activation of genes or transgenes13. These transgenes may represent genetically encoded fluorophores, expression of chemogenetically modifiable designer G-protein-coupled receptors, hM3Dq or light-regulated ion channels for remotely controlled activation or inhibition of Cre-expressing neurons in vivo14,15. To address the question of cellular heterogeneity, combinatorial recombinase-dependent targeting of distinct cell types using two different, intersectional recombinases such as Cre and FLP have been successfully developed16. Through the availability of an additional, complementary recombinase system that builds on expression of Dre-recombinase, an alternative and complementary approach has been defined17.
We have dedicated our efforts toward further developing a toolbox of transgenic mice that offers the opportunity to genetically dissect heterogeneous neuron populations, through combinatorial Cre-dependent and Dre-dependent recombination, allowing for successful labeling, three-dimensional (3D) imaging, translational profiling and functional characterization of POMC neurons expressing Lepr or Glp1r, uncovering a new organizational and functional microarchitecture of critical metabolism-regulatory neurons.
Results
Generation of POMCDre-transgenic mice
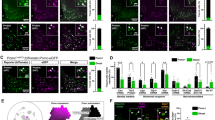
We generated mice, which express the Dre-recombinase under control of the POMC promoter (POMCDre mice; Extended Data Fig. 1a). Assessment of Dre-dependent recombination via visualization of Dre-dependent reporter expression revealed ZsGreen-positive cells in the ARC and in the anterior and intermediate lobes of the pituitary for five of seven transgenic lines (Fig. 1a–c). In particular, one line effectively labeled POMC cells in the pituitary and in the ARC (Fig. 1b,c). Double RNA in situ hybridization (ISH) revealed that 97.6% of ZsGreen-positive neurons in the ARC expressed Pomc, while only 1.9% expressed Agrp (Fig. 1d,e). Correspondingly, the proportion of ZsGreen-labeled non-Pomc-expressing cells was 2.4% and that of ZsGreen-labeled non-Agrp-expressing cells was 98.1% (Fig. 1d,e). We observed no POMCDre-dependent labeling of ZsGreen-positive neurons in the nucleus tractus solitarius (NTS; Extended Data Fig. 1b). Assessment of the bacterial artificial chromosome (BAC) transgene copy number revealed stable transgenerational copy numbers, indicating a single genomic integration (Extended Data Fig. 1c). Longitudinal assessment of Dre-dependent recombination revealed a continued increase of Dre-dependent recombination in POMC neurons from 3–15 weeks of age (Extended Data Fig. 1d). Thus, the later onset of Dre-dependent recombination in our model allows selective marking of bona fide POMC neurons without substantial recombination in the functionally antagonistic AgRP neurons as present in POMCCre transgenic mice18.
a, Schematic showing POMCDre-dependent recombination in the ROSA26rSrZsGreen reporter line. Excision of rox-flanked stop cassette leads to ZsGreen expression in POMC neurons. b, ZsGreen expression across the rostral, mid and caudal sections of the ARC in POMCDre ROSA26rSrZsGreen mice at 15 weeks of age. c, Dispersed ZsGreen expression in the intermediate and anterior lobes of the pituitary in POMCDre ROSA26rSrZsGreen mice. C1, C2 and C3 depict magnifications of the posterior, intermediate and anterior pituitary, respectively. Scale bar, 150 μm (whole image) and 50 μm (magnified images). d, RNA ISH against Pomc/ZsGreen (top) and Agrp/ZsGreen (bottom) in POMCDre ROSA26rSrZsGreen mice. Magnifications of the boxes are displayed on the right of each image. Scale bars, 50 μm (whole image) and 20 μm (magnified images). 3V, third ventricle. e, Percentage of ZsGreen-positive cells coexpressing or lacking expression of either Pomc or Agrp, quantified from RNA ISH (d). Data are represented as the mean ± s.e.m. (Pomc: 97.57 ± 0.92; Agrp: 1.88 ± 0.36; non-Pomc: 2.43 ± 0.92; non-Agrp: 98.12 ± 0.36; n = 4 mice; a minimum of 13 sections were analyzed for each group).
Next, we investigated whether insertion of the BAC transgene in this POMCDre line resulted in any metabolic phenotype. However, POMCDre mice did not exhibit any differences in body weight, food intake, locomotor activity, energy expenditure or glucose tolerance compared to their littermate controls (Extended Data Fig. 1e–n).
Combinatorial recombinase-dependent marking of heterogeneous POMC neuron populations
Leptin and Glp1 target POMC neurons to mediate at least part of their metabolism-regulatory functions4,19. Single-cell sequencing had revealed no overlap between Glp1r and Lepr expression in POMC neurons12. We therefore further examined whether Lepr and Glp1r mRNAs were indeed not coexpressed in POMC neurons. We used double fluorescence RNA ISH against Lepr and Glp1r expression in POMC neurons of wild-type mice, which revealed that while 10.2% of POMC neurons expressed mRNAs of both receptors, the larger proportion of POMC neurons expressed one receptor in the absence of the other (Fig. 2a,b).
a, Representative microscopic images of RNA ISH against Pomc, Glp1r and Lepr in C57BL/6N mice at 12 weeks of age. First image shows ISH in the ARC with nuclear counterstain (blue, DAPI). Magnifications of the dashed box (right) are shown with the indicated stainings. Pomc-positive neurons are outlined in white. Yellow and cyan arrows indicate Lepr-positive or Glp1r-positive POMC neurons, respectively. Scale bars represent 100 μm in the merged image and 25 μm in the magnifications. b, Percentage of Pomc-positive cells expressing Lepr, Glp1r or both receptors across the rostrocaudal axis. The bar graph on the right depicts the total percentage of POMC neurons coexpressing the receptors as averaged from the individual areas. Left: Lepr+Rostral: 22.18% ± 5.02%, Lepr+Mid: 27.54% ± 4.98%, Lepr+Caudal: 24.21% ± 2.49%; Glp1r+Rostral: 27.44% ± 4.73%, Glp1r+Mid: 44.16% ± 3.48%, Glp1r+Caudal: 32.73% ± 11.15%; Lepr+/Glp1r+Rostral: 8.09% ± 2.44%, Lepr+/Glp1r+Mid: 12.79% ± 3.08%, Lepr+/Glp1r+Caudal: 9.40% ± 3.96%. Right: Lepr+, 24.32% ± 3.31%; Glp1r+, 35.03% ± 3.89%; Glp1r+/Lepr+, 10.17% ± 2.10%. One-way ANOVA, F (1.656, 4.968) = 64.61, P = 0.0003, followed by Tukey’s post hoc test; Glp1r+ versus Lepr+ P = 0.0536, Glp1r+ versus Glp1r+/Lepr+ P = 0.0017, Lepr+ versus Glp1r+/Lepr+ P = 0.0129; n = 4 mice. P values were calculated on the total percentage of subpopulations using one-way repeated-measures ANOVA followed by Tukey’s test. *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001. c, Illustrations of experimental mice and schematic diagram showing Dre- and Cre-dependent recombination of ROSA26lSlrSrZsGreen reporter line. Excision of loxP- or rox-flanked stop cassettes through recombination of both Dre and Cre drivers led to ZsGreen expression in the targeted POMC population. d, Representative microscopic images of immunohistochemical staining against POMC and ZsGreen in the ARC of all resulting genotypes at 15 weeks of age. Scale bar, 50 μm. e, Percentage of ZsGreen-positive cells coexpressing or lacking expression of Pomc, quantified from RNA ISH. POMCDre LeprCre: Pomc, 99.21% ± 0.79%; non-Pomc, 0.79% ± 0.79%; POMCDre Glp1rCre: Pomc, 94.56% ± 3.43%; non-Pomc; 5.44% ± 0.3.43%; n = 3 mice per group; minimum of eight sections analyzed for each. For d and e, data are presented as mean ± s.e.m.
To genetically mark heterogeneous POMC cell populations through Cre/Dre-dependent, intersectional recombination, we established reporter mouse models carrying a cDNA encoding the fluorescent marker protein NLS-ZsGreen in the ROSA26 locus20. The expression of ZsGreen is prevented by a loxP-flanked (lSl) and an additional rox-flanked (rSr) transcriptional STOP cassette (Fig. 2c). POMCDre mice were crossed to LeprCre or Glp1rCre mice21. Resulting double transgenic POMCDre LeprCre or POMCDre Glp1rCre mice were bred with homozygous ROSA26lSlrSrZsGreen+/+ mice to yield four different genotypes. This produced mice heterozygous for the reporter (ROSA26lSlrSrZsGreen+/−) in the absence of POMCDre and of the respective Cre transgene, carrying only the POMCDre transgene, carrying only the respective Cre transgene or the combination of both; that is, POMCDre LeprCre or POMCDre Glp1rCre (Extended Data Fig. 2b). Assessment of ZsGreen expression in these mice revealed that ZsGreen immunofluorescence was absent in the ARC of mice carrying neither recombinase transgene or which only carried the POMCDre or the respective Cre transgene (LeprCre or Glp1rCre; Fig. 2d). In contrast, only triple transgenic mice exhibited ZsGreen expression in the ARC (Fig. 2d). Quantification of ZsGreen-positive neurons expressing Pomc revealed that 99.2% of ZsGreen-labeled cells in POMCDre LeprCre mice, and 94.6% of ZsGreen-labeled cells in POMCDre Glp1rCre mice expressed Pomc (Fig. 2e). Further, light-sheet fluorescence microscopy (LSFM) on cleared brain tissue revealed exclusive expression of ZsGreen-positive cells in the ARC of POMCDre LeprCre ROSA26lSlrSrZsGreen+/– mice (Extended Data Fig. 2c), indicating successful intersectional transgenic marking of selective POMC neuron subpopulations.
Distinct anatomical distribution of POMCLepr+ and POMCGlp1r+ neurons in the ARC
To obtain a holistic 3D representation of the neuronal subpopulations, we used the tissue-clearing technique uDISCO22 in combination with LSFM. The 3D images obtained via LSFM from each individual mouse were registered onto a reference atlas, that is, a grayscale Nissl volume of reconstructed brain (Allen Brain 25-μm reference atlas), for subsequent quantitative image analysis. Image registration algorithms vary based on the transformation models they use to relate the target image space to the reference image space. Thus, we used the VINCI software for this purpose23. The neuronal coordinates were subsequently extracted and plotted as an isosurface density plot using a kernel mesh fit onto the neuronal population (Fig. 3a–f). Statistical analysis of these neuronal distributions showed significant differences in the localization patterns of Lepr-expressing POMC neurons in comparison to those with Glp1r expression (Fig. 3g). To compare this distribution pattern of POMC cells endogenously expressing Lepr or Glp1r as assessed by RNA ISH (Fig. 2a), we created coronal cross sections from the 3D coordinates of transgenically labeled POMCLepr+ and POMCGlp1r+ neurons (Extended Data Fig. 2d) and compared them to the distributions of POMC neurons endogenously expressing Lepr and Glp1r in a corresponding anatomical localization (Extended Data Fig. 2e), revealing a similar, differential distribution pattern of both subpopulations in the two experimental approaches.
a–c, Representative 3D reconstruction of the entire POMC population labeled in POMCDre ROSA26rSrZsGreen mice (a), and 3D reconstruction of POMC subpopulations in POMCDre LeprCre ROSA26lSlrSrZsGreen (b) or POMCDre Glp1rCre ROSA26lSlrSrZsGreen (c) mice at 15 weeks of age. Scans were obtained using the LSFM at ×8 total magnification. n = 9 (a), n = 7 (b) and n = 8 (c) mice. d–f, Isosurface density plots of the entire POMC population (d), and the POMCLepr+ (e) and POMCGlp1r+ (f) subpopulations. Gray shaded areas in e and f depict the entire POMC population. g, Statistical representation of the differences in distribution between the POMCLepr+ and POMCGlp1r+ subpopulations using a two-tailed t-test. P values are plotted as spheres within the space occupied by the POMC neurons (background). The size and color of the spheres indicate the significance values in ranges of yellow to red (POMCLepr+) and green to blue (POMCGlp1r+). h, Representative images of 3D projection densities in POMCDre LeprCre ROSA26rSrlSltdTomato mice in the PVH, PAG, DMH, BNST and NTS. i, Quantification of 3D projection densities shown in h, normalized to the number of neurons. Data are represented as mean ± s.e.m., from n = 3 mice per group. POMCLepr+: BNST: 56,068,002.46 ± 4,157,623.02, DMH: 33,195,450.61 ± 2,618,432.01, PAG: 10,481,156.64 ± 819,984.06, PVH: 18,370,352.69 ± 1149,637.53, NTS: 45,834,835.18 ± 6,188,416.90. POMCGlp1r+; BNST: 63,188,837.7 ± 3,276,156.35, DMH: 37,050,145.65 ± 478,713.82, PAG: 11,722,976.37 ± 369,033.55, PVH: 20,851,215.3 ± 337,219.38, NTS: 5,294,0731.54 ± 3,984,858.12. BNST, Glp1r+ versus Lepr+: P = 0.249747, t = 1.345, df = 4. DMH, Glp1r+ versus Lepr+: P = 0.632027, t = 1.448, df = 4. PAG, Glp1r+ versus Lepr+: P = 0.632027, t = 1.381, df = 4. PVH, Glp1r+ versus Lepr+: P = 0.432578, t = 2.071, df = 4. NTS, Glp1r+ versus Lepr+: P = 0.632027, t = 0.9654, df = 4, unpaired Student’s t-test, Holm–Sidak correction.
Similar projection patterns of POMCLepr+ and POMCGlp1r+ neurons
To investigate, whether the Lepr- or Glp1r-expressing POMC subpopulations could target distinct projection sites, we used mice expressing tdTomato upon Cre/Dre-dependent recombination (Extended Data Fig. 3a). POMCDre LeprCre ROSA26rSrlSltdTomato+/− and POMCDre Glp1rCre ROSA26rSrlSltdTomato+/− animal samples allowed for visualization of axonal projections and dendrites of POMCLepr+ and POMCGlp1r+ neurons (Extended Data Fig. 3e)16. Sections from well-defined projection areas of melanocortin neurons in the bed nucleus of the striae terminalis (BNST), periaqueductal gray (PAG), dorsomedial nucleus of the hypothalamus (DMH) and the paraventricular nucleus of the hypothalamus (PVH) were examined for immunoreactive fiber density by the quantification of POMC expression and transgenic tdTomato labeling (Extended Data Fig. 3b,c). tdTomato immunoreactive fiber density in the investigated areas was consistently higher in sections of PomcDreLeprCreROSA26rSrlSltdTomato+/− mice compared to those of PomcDreGlp1rCreROSA26rSrlSltdTomato+/− animals (Extended Data Fig. 3d), as explained by the larger population of genetically marked POMCLepr+ compared to POMCGlp1r+ neurons.
We also analyzed the projection densities in POMC target regions via whole-brain tdTomato immunostaining and LSFM-based image acquisition followed by data processing based on co-registration onto a unified anatomical atlas. This allowed for assessment of projection densities in the different regions of interest (ROIs; Fig. 3h and Extended Data Fig. 4). At the same time, projection intensities could be normalized to the number of transgenically labeled POMC neurons of the individual animals, revealing a similar projection intensity in the different regions taking into account the differential population sizes of both POMC subpopulations (Fig. 3i).
DREADD-dependent activation of POMCLepr+ and POMCGlp1r+ neurons differentially regulates food intake
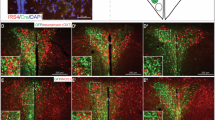
To investigate the role of distinct POMC neuron populations in energy homeostasis in vivo, we generated mice allowing for combinatorial Cre/Dre-dependent expression of the activatory DREADD (Designer Receptors Exclusively Activated by Designer Drugs) receptor hM3Dq and ZsGreen14 (Fig. 4a and Extended Data Fig. 5a,b). Male POMCDreLeprCreROSA26lSlrSrhM3Dq+/− animals were injected intraperitoneally (i.p.) with either saline or clozapine N-oxide (CNO). Activation of POMCLepr+ neurons was assessed via RNAscope ISH using probes against Pomc, Lepr, ZsGreen and Fos (Fig. 4b). The majority of ZsGreen-expressing POMC neurons were positive for Lepr expression (93.4%), supporting specific targeting of POMCLepr+ neurons (Fig. 4c). Furthermore, the proportion of ZsGreen-expressing cells that coexpressed Pomc and Lepr mRNA was assessed, revealing the expression of ZsGreen (and thus hM3Dq) in 47.3% of the cells expressing Lepr and Pomc (Fig. 4d). To investigate the efficiency of CNO-dependent cell activation, the ratio of Fos-positive cells over ZsGreen and Pomc double-positive neurons was assessed in saline-injected and CNO-injected POMCDre LeprCre ROSA26lSlrSrhM3Dq+/− animals. While only 6.9% of hM3Dq-expressing POMC neurons were positive for Fos mRNA expression in saline-injected animals, this proportion increased to 94.0% after CNO injection (Fig. 4e). Similarly, POMCDre Glp1rCre ROSA26lSlrSrhM3Dq+/− animals exhibited CNO-dependent activation of POMCGlp1r+ neurons in males (Fig. 4c–e and Extended Data Fig. 5c). Moreover, CNO-only injection elicited increased Fos expression in POMC neurons of POMCDre LeprCre ROSA26lSlrSrhM3Dq+/− and POMCDre Glp1rCre ROSA26lSlrSrhM3Dq+/− animals but not in ROSA26lSlrSrhM3Dq+/−animals lacking Dre and Cre expression or expressing either recombinase alone (Extended Data Fig. 5d,e). Moreover, more than 90% of the labeled POMC neurons in POMCDre LeprCre ROSA26lSlrSrhM3Dq+/− mice expressed Lepr, while 22.2% expressed Glp1r. In turn, only 10.9% of the labeled neurons in POMCDre Glp1rCre ROSA26lSlrSrhM3Dq+/− mice were Lepr positive and 93.5% expressed Glp1r (Extended Data Fig. 5f,g). Finally, CNO application similarly activated POMCLepr+ and POMCGlp1r+ neurons in female triple transgenic animals as observed in males (Extended Data Fig. 5h–j).
a, Illustrations of experimental mice and schematic diagram showing Dre- and Cre-dependent targeted expression of activatory hM3Dq in either POMCLepr+ or POMCGlp1r+ neurons. Excision of loxP-flanked and rox-flanked stop cassettes through recombination of both Dre and Cre drivers leads to hM3Dq expression in the targeted subpopulation. b, Representative microscopic images of RNA ISH against Pomc, Lepr, ZsGreen (in lieu of hM3Dq) and Fos in POMCDre LeprCre ROSA26lSlrSrhM3Dq males injected with saline or CNO. Images on the left show ISH in the ARC with nuclear counterstain (blue, DAPI). Magnifications of the boxes (right) are shown with the indicated stainings. Pomc-positive neurons are outlined in white. Scale bars, 50 μm (merged image) and 25 μm (magnified images). c–e, Percentage of ZsGreen-Pomc-positive cells expressing Lepr or Glp1r (c), percentage of Lepr/Glp1r-Pomc-positive cells expressing ZsGreen (d) and percentage of ZsGreen-Pomc-positive cells expressing Fos (e) in POMCDre LeprCre ROSA26lSlrSrhM3Dq or POMCDre Glp1rCre ROSA26lSlrSrhM3Dq male mice (22–26 weeks old) injected with saline or CNO. CNO, 3 mg kg−1. c: POMCLepr+: saline: 91.84% ± 1.03%, CNO: 94.87% ± 1.62%, saline versus CNO, t = 1.580, P = 0.342863: POMCGlp1r+: saline: 93.52% ± 2.04%, CNO: 96.16% ± 2.31% saline versus CNO, t = 0.8592, P = 0.438695; d: POMCLepr+: saline: 45.46% ± 6.92%, CNO: 49.19% ± 1.92%, saline versus CNO, t = 0.5191, P = 0.631109; POMCGlp1r+: saline: 36.88% ± 5.28%, CNO: 43.24% ± 1.42%, saline versus CNO, t = 1.165, P = 0.522274. e: POMCLepr+: saline: 6.87% ± 2.64%, CNO: 94.03% ± 1.66%, saline versus CNO, t = 28.0, df = 4, PuT = 0.000019; POMCGlp1r+: saline: 8.23% ± 1.13%, CNO: 87.05% ± 6.05%, saline versus CNO, t = 12.82, df = 4, PuT = 0.000214, unpaired Student’s t-test, Holm–Sidak correction; n = 3 mice. f,g, Food intake over a time course of 24 h in POMCDre LeprCre ROSA26lSlrSrhM3Dq (f) and POMCDre Glp1rCre ROSA26lSlrSrhM3Dq male mice (g) starting with the night cycle. Mice were injected with saline at 18:00 and 23:00, followed by a 1-d gap and subsequent CNO injections at 18:00 and 23:00 on the next day. Left: cumulative food intake in mice injected with saline versus CNO; right, total food intake in grams during night and day. f: n = 8; left: saline versus CNO two-way ANOVA, F(1,7) = 4.815, P = 0.0643; right: salineNight: 3.03 ± 0.39, CNONight: 2.79 ± 0.20, saline versus CNO two-way ANOVA followed by Sidak’s test, P = 0.2729, salineDay: 1.05 ± 0.15, CNODay: 1.10 ± 0.07, saline versus CNO two-way ANOVA followed by Sidak’s test, P = 0.9867. g: n = 7, left: saline versus CNO two-way ANOVA, F(1.000, 6.000) = 16.51, P = 0.0066, right: salineNight: 3.42 ± 0.38, CNONight: 2.32 ± 0.12, salineDay: 1.19 ± 0.15, CNODay: 0.98 ± 0.16; saline versus CNO two-way ANOVA followed by Sidak’s test, P = 0.0312; saline versus CNO two-way ANOVA followed by Sidak’s test, P = 0.7992. Data are represented as the mean ± s.e.m. Statistical analyses in c–e were performed by unpaired two-tailed Student’s t-test with Holm–Sidak correction for multiple comparisons. For cumulative food intake (f and g left), two-way ANOVA was used; for total food intake (f and g right), two-way ANOVA followed by Sidak’s post hoc test was used. Indices PuT: unpaired t-test. *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001, ***P ≤ 0.0001.
Next, we assessed parameters of energy homeostasis upon treating POMCDre LeprCre ROSA26lSlrSrhM3Dq+/− or POMCDre Glp1rCre ROSA26lSlrSrhM3Dq+/− animals and their respective controls with either saline or CNO, while performing an indirect calorimetry combined with continuous monitoring of food intake. Upon saline treatment feeding rates in male mice of the different genotypes did not differ (Extended Data Fig. 6a). Moreover, repeated injections of CNO in control animals did not affect food intake compared to saline-injected control animals (Extended Data Fig. 6b). In contrast, CNO injections of male POMCDre LeprCre ROSA26lSlrSrhM3Dq+/− mice at the beginning of the natural feeding cycle, reduced food intake starting at 3 h after CNO injection (Fig. 4f). This effect cumulated into a statistically nonsignificant 15.6% suppression of feeding over the duration of the dark cycle (Fig. 4f). However, chemogenetic activation of male POMCDre Glp1rCre ROSA26lSlrSrhM3Dq+/− mice resulted in an earlier (2.5 h after CNO injection) and even stronger feeding suppression, accounting for a significant 32.3% reduction in food intake over the duration of the dark cycle (Fig. 4g). In contrast, chemogenetic activation of POMCLepr+ or POMCGlp1r+ neurons in male mice did not affect energy expenditure or locomotor activity, while mildly shifting the respiratory exchange ratio toward fatty acid metabolism (Extended Data Fig. 6c–h). Interestingly, chemogenetic activation of POMCLepr+ and POMCGlp1r+ neurons in female mice did not suppress feeding.
Given the different kinetics in feeding suppression upon activation of POMCGlp1r+ neurons compared POMCLepr+ neurons in male mice, we asked whether both neuronal subpopulations might differ in their neurotransmitter characteristics. Therefore, we compared the expression of the vesicular GABA transporter (Vgat or Slc32a1) and of the vesicular glutamate transporter (Vglut2 or Slc17a6) in POMCLepr+ and POMCGlp1r+ neurons (Extended Data Fig. 7a). Quantification of the proportion of GABAergic and glutamatergic POMC neurons in both mouse lines revealed a slightly higher proportion of glutamatergic POMCLepr+ neurons. However, non-POMC neurons appeared to exhibit a stronger signal for Vglut2 expression compared to POMC neurons (Extended Data Fig. 7b).
Since POMC is also expressed not only in the ARC, but also in the anterior and intermediate lobe of the pituitary, we compared the detectability of ZsGreen-positive cells in the pituitary of POMCDre LeprCre ROSA26lSlrSrzsGreen+/− or POMCDre Glp1rCre ROSA26lSlrSrzsGreen+/− animals (Extended Data Fig. 8a,b). Although ZsGreen expression was detectable in the majority of cells in the intermediate lobe of both POMCDre LeprCre ROSA26lSlrSrzsGreen+/− and POMCDre Glp1rCre ROSA26lSlrSrzsGreen+/− animals, we did not detect ZsGreen-positive cells in the anterior lobe of the pituitary in either mouse line (Extended Data Fig. 8a,b). Activation of each subpopulation did not alter circulating corticosterone concentrations in the serum of either mouse line (Extended Data Fig. 8c,d).
Distinct translational signatures of POMCLepr+ and POMCGlp1r+ neurons
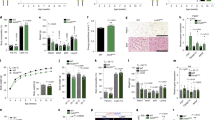
To obtain detailed transcriptional/translational profiles of POMCLepr+ and POMCGlp1r+ neurons, we generated mice enabling combinatorial Cre/Dre-dependent expression of the L10a ribosomal protein fused to enhanced green fluorescent protein (EGFP; Fig. 5a and Extended Data Fig. 9a,b). Hypothalami of resulting triple transgenic animals (POMCDre LeprCre ROSA26lSlrSrEGFPL10a+/− and POMCDre Glp1rCre ROSA26lSlrSrEGFPL10a+/− mice) were used for RNA extraction. Biological replicates for the individual lines provided input RNA samples, which were subjected to anti-EGFP immunopurification of EGFP-tagged ribosomes of POMCLepr+ or POMCGlp1r+ neurons. Having confirmed the specific expression of the EGFPL10a fusion protein using the ROSA26lSlrSrEGFPL10a mouse line (Extended Data Fig. 5c) and validated the successful, specific pulldown of RNA of subpopulations of POMC neurons, our aim was to compare the ribosome-associated transcriptome of POMCLepr+ neurons with POMCGlp1r+ neurons. Thus, RNA-seq was performed on input and immunoprecipitation (IP) RNA samples of POMCDre LeprCre ROSA26lSlrSrEGFPL10a+/− and POMCDre Glp1rCre ROSA26lSlrSrEGFPL10a+/− mice. In total, reads mapped to 17,239 genes. Principal-component analyses for the highest expressed genes showed clear separation of input from IP samples, as well as for POMCDre LeprCre ROSA26lSlrSrEGFPL10a+/− IP from POMCDre Glp1rCre ROSA26lSlrSrEGFPL10a+/− IP samples (Extended Data Fig. 9a). Next, the expression level of each IP sample was normalized to its own input sample. The normalized expression analysis of all samples from both triple transgenic mouse groups was used to visualize the differences in expression of all detected genes in both subpopulations. Almost an equal number of genes were expressed at higher levels in the POMCLepr+ compared to POMCGlp1r+ neurons and vice versa (Fig. 5b).
a, Illustration of experimental mice and schematic diagram showing Dre- and Cre-dependent targeted expression of EGFPL10a in either POMCLepr+ or POMCGlp1r+ neurons. Excision of loxP-flanked and rox-flanked stop cassettes through recombination of both Dre and Cre drivers led to EGFPL10a expression in the targeted subpopulation. b, Volcano plot of differentially ribotag-enriched transcripts in POMCLepr+ and POMCGlp1r+ neurons. Significantly differentially enriched transcripts (P ≤ 0.05) are indicated in the colored region. Yellow and cyan depict a significantly higher enrichment in POMCLepr+ and POMCGlp1r+ neurons, respectively. P values adjusted for multiple comparisons were calculated by DESeq2 (1.26.0). c, Expression of Pomc, Lepr and Glp1r in input (IN) and IP samples of each subpopulation. Statistics were analyzed using unpaired two-tailed Welch’s t-test. Pomc: Lepr IN: 39.44 ± 19.39, Lepr IP: 4,740.55 ± 618.41, IN vs IP, t = 7.6, P = 0.0047. Glp1r IN: 14.14.55 ± 0.49, Glp1r IP: 1,005.42 ± 40.19, IN vs IP, t = 24.7, P = 0. 0016. Lepr: Lepr IN: 0.92 ± 0.14, Lepr IP: 11.12 ± 2.05, IN vs IP, t = 4.95, P = 0.0155. Glp1r IN: 0.62 ± 0.07, Glp1r IP: 1.50 ± 0.40, IN vs IP, t = 2.18, P = 0.001536. Glp1r: Lepr IN: 2.97 ± 0.38, Lepr IP: 8.80 ± 1.82, IN vs IP, t = 3.15, P = 0.0458. Glp1r IN: 3.23.55 ± 0.35, Glp1r IP: 29.17 ± 2.83, IN vs IP, t = 9.08, P = 0.0108. *P ≤ 0.05, **P ≤ 0.01. d, Significantly differentially enriched genes (P ≤ 0.05) of POMCLepr+ and POMCGlp1r+ neurons, belonging to the GO term ‘neuropeptide-signaling pathways’. Vertical colored area separates higher enrichment in POMCLepr+ neurons (left) from higher enrichment in POMCGlp1r+ neurons (right). e, Overlap analysis of the publicly available single-cell RNA-seq data from mouse hypothalami with our dataset. Here, the volcano plot in b is filtered for the markers reported for each cluster. For POMCDre LeprCre ROSA26lSlrSrEGFPL10a, n = 4 samples of pooled hypothalami from N = 24 mice; for POMCDre Glp1rCre ROSA26lSlrSrEGFPL10a, n = 3 samples of pooled hypothalami from N = 36 mice. Data are represented as the mean ± s.e.m.
As expected, Pomc transcript reads were drastically (>1,000-fold) enriched in both POMCLepr+ and POMCGlp1r+ neurons compared to the global hypothalamic background (Fig. 5c). POMCLepr+ neurons exhibited a stronger enrichment for Pomc mRNA compared POMCGlp1r+ neurons, indicating a potentially differential Pomc mRNA expression (Fig. 5c). Similarly, we found clear overrepresentation for the reads of Lepr in IP samples from POMCLepr+ neurons and an enrichment of Glp1r expression in IP samples from POMCGlp1r+ neurons (Fig. 5c).
To identify which classes of transcripts differentiate POMCLepr+ and POMCGlp1r+ neurons, we subjected the differentially enriched genes of each subpopulation to a Gene Ontology (GO)-term analysis (Extended Data Fig. 9e–g). This analysis revealed that, specifically, transcripts in the GO terms neuropeptide signaling, regulation of response to nutrient levels and dendrite cytoplasm differed between both types of neurons (Extended Data Fig. 9e–g). To further investigate which neuropeptides and neuropeptide receptors are differentially expressed by POMCLepr+ and POMCGlp1r+ neurons, the relative expression levels for all genes listed in the GO term ‘neuropeptide-signaling pathway’ (GO:0007218) were analyzed. While opioid receptors (Oprd1 and Oprm1) and enrichment of neuropeptide Y (Npy) was higher in POMCGlp1r+ neurons, enrichment for prodynorphin (Pdyn), Cocaine and amphetamine regulated transcript protein (Cartpt), Neuromedin-U receptor 2 (Nmur2), and NPY-receptor (Npy1r) was higher in POMCLepr+ neurons (Fig. 5d).
Next, we aimed to investigate whether and to which extent the genes enriched in either subpopulation identified by targeted ribosomal profiling might define clusters of POMC neurons as revealed by clustering in single-cell RNA-seq. The first data source was droplet single-cell RNA-seq of 20,921 cells from the ARC/median eminence11. Here, we filtered cells expressing Pomc (4,248/20,921 cells) and clustered them using the R Seurat package24. This yielded the identification of 11 clusters of Pomc-expressing neurons (Fig. 5e and Extended Data Fig. 9h). The second data source was a single-cell mRNA-seq dataset of 163 genetically marked Pomc-expressing neurons of mice yielding four clusters of POMC neurons12 (Fig. 5e). In Fig. 5e, we filtered the volcano plot of Fig. 5b for the markers of each cluster from the aforementioned datasets. For each of these clusters, we determined the log ratio of the marker genes differentially expressed in our ribosomal profiling between POMCLepr+ and POMCGlp1r+ neurons (log ratio = log2 (n_Glp1r/n_Lepr)). We considered a cluster to be enriched for POMCGlp1r+ marker genes with a log ratio ≥ 2 and vice versa enriched for POMCLepr+ marker genes with a log ratio ≤ −2. Applying this threshold to our clustering of POMC-positive neurons from the ARC/median eminence dataset identified clusters 2 and 10 as enriched for POMCLepr+ marker genes and cluster 3 as enriched for POMCGlp1r+ marker genes (Fig. 5e). Similarly, we defined clusters 3 and 4 of the dataset of fluorescence-activated cell sorting (FACS)-purified POMC neurons enriched for POMCLepr+ and clusters 1 and 2 enriched for POMCGlp1r+ marker genes (Fig. 5e). Collectively, our translational profiles of POMCLepr+ and POMCGlp1r+ neurons allowed for successful independent identification of molecularly defined POMC clusters, which had been defined based on single-cell RNA-seq.
Verification of endogenously expressed, differentially regulated genes in POMCLepr+ and POMCGlp1r+ neurons
We then aimed to validate the mRNA expression profiles of genes characterized as differentially enriched in genetically marked POMCLepr+ and POMCGlp1r+ neurons via RNA ISH upon co-detection of endogenously expressed Lepr and Glp1r mRNA in wild-type mice (Fig. 6a). These analyses confirmed increased expression of Pomc in POMCLepr+ neurons compared to POMCGlp1r+ neurons (Fig. 6b). Similarly, Cartpt expression was increased in POMCLepr+ neurons compared to POMCGlp1r+ neurons (Fig. 6c). When we investigated the expression of exemplary genes encoding receptors for neuropeptides and energy-sensing signals, we confirmed the differential enrichment of Npy1r, Oprm1 and Nmur2 in POMCLepr+ and POMCGlp1r+ neurons (Fig. 6c), as previously indicated by our RNA-seq results (Fig. 5d).
a, Representative microscopic images of RNA ISH against Pomc, Glp1r and Lepr together with differentially expressed neuropeptidergic signaling candidate RNAs identified in the ribotag experiments, Cartpt, Npy1r, Oprm1 and Nmur2, in C57BL/6N mice at 12 weeks of age. First image shows ISH in the ARC with nuclear counterstain (blue, DAPI). Magnifications of the boxes (right) are shown with the indicated stainings. Pomc-positive neurons are outlined in white. Yellow and cyan arrows indicate Lepr-positive or Glp1r-positive POMC neurons, respectively. Scale bars represent 100 μm in the merged image and 25 μm in the magnifications. b, Violin plots showing the quantified intensity of Pomc mRNA across the rostrocaudal axis of the ARC in POMCLepr+ or POMCGlp1r+ neurons as assessed from RNA ISH (Fig. 2a). Data are from n = 4 mice, with a minimum of four sections analyzed per animal. PomcRostral, Lepr+: Q1: 7.83, Q2: 31.12, Q3: 71.31; Glp1r+: Q1: 3.92, Q2: 14.86, Q3: 37.05, unpaired Mann–Whitney U-test, Lepr+ versus Glp1r+, U = 21,051, PuT < 0.0001; PomcMid, Lepr+: Q1: 3.02, Q2: 15.71, Q3: 49.22; Glp1r+, Q1: 3.22, Q2: 13.43, Q3: 34.06, unpaired Mann–Whitney U-test, Lepr+ versus Glp1r+, U = 53,910, PuT = 0.0796; PomcCaudal, Lepr+: Q1: 9.46, Q2: 28.3, Q3: 103.5; Glp1r+ Q1: 8.03, Q2: 17.81, Q3: 91.96, unpaired Mann–Whitney U-test, Lepr+ versus Glp1r+, U = 3,467, PuT = 0.06349. a.u., arbitrary units. c, Violin plots showing quantified expression of the RNA-seq candidates measured as integrated density in POMCLepr+ or POMCGlp1r+ neurons as assessed from RNA ISH (a). Cartpt: Lepr+, Q1: 2,977, Q2: 6,729, Q3: 13,244, Glp1r+, Q1: 2,405, Q2: 4,741, Q3: 9,198, Lepr+ versus Glp1r+, UMW = 9,252, PuT = 0.0182; Npy1r: Lepr+, Q1: 1,609, Q2: 2,470, Q3: 3,887; Glp1r+, Q1: 1,154, Q2: 1,802, Q3: 3,069, Lepr+ versus Glp1r+, U = 22,352, PuT < 0.0001; Oprm1: Lepr+, Q1: 631.6, Q2: 800.8, Q3: 1,046; Glp1r+, Q1: 729.2, Q2: 1,002, Q3: 1,360, Lepr+ versus Glp1r+, UMW = 5017, PuT = 0.001; Nmur2: Lepr+, Q1: 475.4, Q2: 689.7, Q3: 1,057; Glp1r+ Q1: 407.3, Q2: 574.9, Q3: 852, Lepr+ versus Glp1r+, UMW = 8,760, PuT = 0.0029. In b and c, solid white lines represent the median (Q2) and dashed white lines represent lower and upper quartiles (Q1 and Q2, respectively). P values were calculated using the unpaired Mann–Whitney (MW) U-test. *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001, ***P ≤ 0.0001.
POMCLepr+ and POMCGlp1r+ neurons have distinct intrinsic electrophysiological properties
Next, we performed perforated patch-clamp recordings in genetically marked Lepr- and Glp1r-expressing POMC neurons in POMCDre LeprCre ROSA26lSlrSrZsGreen+/− or POMCDre Glp1rCre ROSA26lSlrSrZsGreen+/− mice. Both neuronal populations have distinct functional properties and input–output relationships. While there were no differences in certain general properties including whole-cell capacitance, spontaneous firing rate and in the proportion of spontaneously active or silent neurons (Extended Data Fig. 10a,b), we found differences in key intrinsic electrophysiological properties (Fig. 7a–t). POMCGlp1r+ neurons were more depolarized (Fig. 7a), had a higher input resistance (Fig. 7b,c) and exhibited a higher excitability (Fig. 7d–h). During prolonged depolarization, they responded with pronounced phasic activity, while the POMCLepr+ neurons were more tonically active (Fig. 7f,m,n).
a, Membrane potential (EM ) of POMCLepr+ and POMCGlp1r+ neurons (POMCLepr+, n = 38, EM = −66.72 ± 0.20 mV; POMCGlp1r+, n = 39, EM = −61.20 ± 1.29 mV; PMW = 0.001, U = 418). b,c, Input resistance. b, Mean response to 5-pA hyperpolarizing current pulses (POMCLepr+, n = 37; POMCGlp1r+, n = 28) and violin plots (c) showing the input resistance of POMCLepr+ and POMCGlp1r+ neurons (POMCLepr+, n = 37, Ri = −1.24 ± 0.09 GΩ; POMCGlp1r+, n = 28, Ri = −1.61 ± 0.12GΩ; PMW = 0.022, U = 345). d,e, Excitability I. Original recording illustrating a depolarizing ascending (asc.) and descending (desc.) current ramp protocol in POMCLepr+ and POMCGlp1r+ neurons (d) and respective spike-number ratios of the ascending and descending phase of the protocol in POMCLepr+ and POMCGlp1r+ neurons (e; 10 pA: POMCLepr+, n = 24, r = 2.33 ± 0.45; POMCGlp1r+, n = 18, r = 3.35 ± 0.33; PMW = 0.0019, U = 96.5, 15 pA: POMCLepr+, n = 29, r = 2.81 ± 0.54; POMCGlp1r+, n = 23, r = 3.64 ± 0.58; PMW = 0.0033, U = 176.5; 20 pA: POMCLepr+, n = 31, r = 3.06 ± 0.55; POMCGlp1r+, n = 24, r = 3.34 ± 0.56; PMW = 0.024, U = 239.5; 25 pA: POMCLepr+, n = 32, r = 3.28 ± 0.59; POMCGlp1r+, n = 23, r = 3.41 ± 0.68; PMW = 0.0527, U = 245.5). f–h, Excitability II. Example responses to 30-pA pulses (f). Number of action potentials (APs) as a function of current pulse amplitude (pA; g; POMCLepr+, n = 32, slope = 0.687 ± 0.073 (AP/pA); POMCGlp1r+, n = 26, slope = 0.817 ± 0.096 (AP/pA); PF = 0.0322, F = 4.62) in POMCLepr+ and POMCGlp1r+ neurons and the respective slopes (h). i,j, Post-inhibitory rebound excitation. Original recordings illustrating the responses to a depolarizing current pulse that followed a prolonged (2 s) hyperpolarizing pre-pulse (i). Mean maximal instantaneous frequency as a function of the pre-pulse potential for POMCLepr+ and POMCGlp1r+ neurons (j; −120mV: POMCLepr+, n = 32, fmax = 14.93 ± 2.97 Hz; POMCGlp1r+, n = 23, fmax = 29.12 ± 7.14 Hz; PMW = 0.0773, U = 264. −110 mV: POMCLepr+, n = 32, fmax = 15.04 ± 2.70 Hz; POMCGlp1r+, n = 23, fmax = 29.83 ± 7.16 Hz; PMW = 0.0374, U = 246. −100 mV: POMCLepr+, n = 32, fmax = 15.29 ± 2.90 Hz; POMCGlp1r+, n = 23, fmax = 31.10 ± 6.38 Hz; PMW = 0.0044, U = 203. −90 mV: POMCLepr+, n = 32, fmax = 14.05 ± 2.53 Hz; POMCGlp1r+, n = 23, fmax = 29.75 ± 6.91 Hz; PMW = 0.0374, U = 246. −80 mV: POMCLepr+, n = 32, fmax = 11.48 ± 1.78 Hz; POMCGlp1r+, n = 22, fmax = 22.14 ± 4.54 Hz; PMW = 0.017, U = 217. −70 mV: POMCLepr+, n = 29, fmax = 7.46 ± 0.54 Hz; POMCGlp1r+, n = 19, fmax = 10.25 ± 1.46 Hz; PMW = 0.2081, U = 215). k,l, Sag potentials during hyperpolarization. Original recordings illustrating the response to five consecutive hyperpolarizing current pulses adjusted to reach −120 mV (k). Violin plots illustrating the sag amplitudes at hyperpolarization to −120mV (l; POMCLepr+, n = 37, EM = −7.46 ± 0.50 mV; POMCGlp1r+, n = 28, EM = −5.69 ± 0.55 mV; PMW = 0.0057, U = 311). m,n, SFA. m, Original traces illustrating the first 5 s of a response to a 10-s depolarizing current pulse in POMCLepr+ and POMCGlp1r+ neurons. Violin plot showing SFA ratios (n) of POMCLepr+ and POMCGlp1r+ neurons (POMCLepr+, n = 30, r = 3.6 ± 0.35; POMCGlp1r+, n = 24, r = 4.66 ± 0.44; PMW = 0.0233, U = 230). o–q, Afterhyperpolarization (AHP) and afterdepolarization (ADP). o, Original traces illustrating the slow AHP after 1-s depolarizing stimuli in POMCLepr+ and POMCGlp1r+ neurons, and the ADP in POMCLepr+ neurons. p, AHP amplitude for POMCLepr+ and POMCGlp1r+ neurons as a function of the stimulus amplitude (5 pA, POMCLepr+, n = 32, ΔEM = −1.36 ± 0.19 mV; POMCGlp1r+, n = 26, ΔEM = −1.97 ± 0.30 mV; PuT = 0.084, t = 1.76, df = 56. 10 pA: POMCLepr+, n = 32, ΔEM = −0.77 ± 0.44 mV; POMCGlp1r+, n = 26, ΔEM = −2.20 ± 0.36 mV; PMW = 0.0156, U = 262. 15 pA: POMCLepr+, n = 32, ΔEM = −1.36 ± 0.50 mV; POMCGlp1r+, n = 26, ΔEM = −3.28 ± 0.51 mV; PuT = 0.0098, t = 2.675, df = 56. 20 pA: POMCLepr+, n = 32, ΔEM = −2.01 ± 0.48 mV; POMCGlp1r+, n = 26, ΔEM = −4.38 ± 0.57 mV; PMW= 0.0044, U = 236. 25 pA: POMCLepr+, n = 32, ΔEM = −3.13 ± 0.53 mV; POMCGlp1r+, n = 26, ΔEM = −4.82 ± 0.63 mV; PuT = 0.0438, t = 2.06, df = 56. 30 pA: POMCLepr+, n = 32, ΔEM = −4.06 ± 0.63 mV; POMCGlp1r+, n = 26, ΔEM = −5.82 ± 0.72 mV; PuT = 0.0688, t = 1.86, df = 56. 35 pA: POMCLepr+, n = 32, ΔEM = −4.34 ± 0.59 mV; POMCGlp1r+, n = 26, ΔEM = −6.60 ± 0.66 mV; PuT = 0.0131, t = 2.56, df = 56. 40 pA: POMCLepr+, n = 32, ΔEM = −5.07 ± 0.55 mV; POMCGlp1r+, n = 26, ΔEM = −6.86 ± 0.68 mV; PuT = 0.043, t = 2.07, df = 56. 45 pA: POMCLepr+, n = 32, ΔEM = −5.58 ± 0.67 mV; POMCGlp1r+, n = 26, ΔEM = −7.49 ± 0.73 mV; PMW = 0.0659, U = 298. 50 pA: POMCLepr+, n = 32, ΔEM = −6.29 ± 0.67 mV; POMCGlp1r+, n = 25, ΔEM = −7.66 ± 0.78 mV; PMW = 0.3579, U = 342). Inset shows amplitude of the ADP, which was predominantly observed in POMCLepr+ neurons. q, Percentage of POMCLepr+ and POMCGlp1r+ neurons revealing ADPs after the 1-s excitatory stimuli (POMCLepr+ n = 32; POMCGlp1r+ n = 26). r–t, Action potential waveform of POMCLepr+ and POMCGlp1r+ neurons. Mean action potential phase plots; the region of the dashed rectangle is shown in higher resolution on the right (r). Action potential threshold, defined as when the rate in change of EM reaches 10 mV/ms (s; POMCLepr+, n = 33, EM = −43.82 ± 0.43 mV; POMCGlp1r+, n = 30, EM = −42.28 ± 0.55 mV; PMW = 0.0212, U = 328) and depolarization rate (t; POMCLepr+, n = 33, DR = −380.6 ± 14.5 mV/ms; POMCGlp1r+, n = 30, DR = −320.5 ± 15.6 mV/ms; PuT = 0.0064, t = 2.83, df = 61). In all recordings, synaptic input was pharmacologically blocked (Methods). Error bars show ± s.e.m. ***P < 0.001; **P < 0.01; *P < 0.05. Bold lines in violin plots mark the median and light lines represent quartiles. n, number of cells recorded. DR, depolarization rate; r, ratio.
During ascending and subsequently descending current ramps (Fig. 7d,e), POMCGlp1r+ neurons tended to have a lower threshold current (Extended Data Fig. 10c) and generated more action potentials during ascending ramps than the POMCLepr+ neurons. During descending ramps, we observed the opposite; POMCLepr+ neurons generated more action potentials than POMCGlp1r+ neurons. This resulted in a higher spike-number ratio between the ascending and descending ramps in POMCGlp1r+ neurons compared to POMCLepr+ neurons, which indicates a voltage-dependent adaptation (Fig. 7e). These data suggest a phasic, adaptive excitability of POMCGlp1r+ neurons in response to sustained excitatory input, which cannot be explained simply by the differences in input resistance (Fig. 7b,c).
On a finer scale, we measured frequency–current relationships, post-inhibitory rebound, spike-frequency adaptation (SFA), and sag potentials during hyperpolarization from POMCGlp1r+ and POMCLepr+ neurons, and found higher excitability in POMCGlp1r+ neurons, which was reflected by a ‘steeper’ frequency–current relationship (Fig. 7f–h).
We also observed a stronger post-inhibitory rebound excitation in POMCGlp1r+ (Fig. 7i,j). It is remarkable that we found profound post-inhibitory rebound excitation in POMCGlp1r+ neurons, while they generated smaller sag potentials than POMCLepr+ neurons (Fig. 7k, l).
During depolarizing current pulses, POMCGlp1r+ cells underwent stronger SFA after the initial excitation (Fig. 7m,n), as reflected in a significantly higher SFA ratio (Fig. 7n). The high SFA ratio of POMCGlp1r+ neurons was accompanied by a marked afterhyperpolarization that followed depolarizations, for example, after trains of action potentials (Fig. 7o,p). This is mechanistically plausible since both SFA and afterhyperpolarization can be mediated by Ca2+-dependent K+ currents, which have been found in POMC neurons25,26. In contrast, in POMCLepr+ neurons, which have a lower SFA and a clear tonic activity component during depolarizations, a sustained train of action potentials was followed by an afterdepolarization outlasting the excitatory input (Fig. 7o–q). A spike waveform analysis (Fig. 7r–t) showed a lower action potential threshold (Fig. 7r,s), a faster depolarization rate (Fig. 7r,t) and a trend toward a faster repolarization rate (Extended Data Fig. 10d) in action potentials of POMCLepr+ neurons.
Since most active membrane properties are mediated by sets of ionic conductances, that is, multiple ion channel types, it is challenging to attribute these complex physiological properties causally to translational profiles. Nevertheless, we have identified cell-type-specific differences in the expression of ion channels (or subunits) that are considered regulators of some physiological properties that differ between POMCLepr+ and POMCGlp1r+ neurons (Extended Data Fig. 10e–j). The phasic, adaptive excitability and the accompanying greater rebound properties of the POMCGlp1r+ neurons are in line with their comparatively higher expression of a modulatory β-subunit (Cacnb4; Extended Data Fig. 10i), which can lower the activation threshold and increase the conductance of voltage-gated Ca2+ channels27. The stronger SFA and afterhyperpolarization of POMCGlp1r+ cells compared to POMCLepr+ neurons are consistent with the relatively high expression of the regulatory β4-subunit (Kcnmb4; Extended Data Fig. 10j) in POMCLepr+ neurons, which decreases the conductance of big-conductance Ca2+-activated potassium channels28. Additionally, M-currents expressed by the KCNQ gene family can contribute to SFA and are upregulated in POMCGlp1r+ cells (Extended Data Fig. 10j)29. In agreement with the relatively depolarized membrane potential of POMCGlp1r+ cells and their relatively small sag potentials during hyperpolarization, we found low expression of Na+/K+ ATPases (Atp1a1) and hyperpolarization-activated cyclic-nucleotide-gated cation channel 1 (Hcn1), respectively (Extended Data Fig. 10e, h)30.
In summary, we have revealed cell-type-specific electrophysiological differences between POMCLepr+ and POMCGlp1r+ neurons, which are paralleled by the cell-type-specific expression of ion channels and receptors for energy homeostasis-related signals.
Specific regulation of POMCLepr+ and POMCGlp1r+ neurons by energy-state-sensing signals
Most neurons showed significant but mostly subtle modulatory effects to stimulation with the cognate ligands of their defining receptors, that is, leptin (Fig. 8a–d) and Glp1 (Fig. 8e–h). The modulatory effects were often differential and not homogeneous within one cell type, and we also observed modulation upon stimulation with the non-cognate ligands (Fig. 8i–n).
a–h, Effect of leptin on POMCLepr+ neurons (a–d) and Glp1 on POMCGlp1r+ neurons (e–h). Rate histograms and respective original recordings illustrating the effect of leptin on POMCLepr+ neurons (a–c) and the effect Glp1 on POMCGlp1r+ neurons (e–g). Each figure shows a single example each of a peptide-excited, a peptide-inhibited and a nonresponsive neuron. d,h, Numbers of peptide-responsive neurons in the respective cell types. i–n, Effect of Glp1 on POMCLepr+ neurons (i–k) and leptin on POMCGlp1r+ neurons (l–n). i,j,l,m, Rate histograms and respective original recordings showing single examples of neurons that were nonresponsive or inhibited by the respective peptides. The asterisk in the rate histogram (j) reflects action potentials that were elicited by current protocols. k,n, Current-clamp recordings, in which action-potential-induced synaptic release is suppressed by TTX (1 µM). Top: original recordings. Bottom left: summary and quantification of all recordings. Red lines indicate recordings with significant changes in membrane potentials. The population responses were compared by using one-way ANOVA with Tukey’s post hoc test (k: POMCLepr+, n = 14, control (ctrl) versus Glp1 P = 0.0692, Glp1 versus wash P = 0.238, ctrl versus wash P = 0.0218, F = 6.40; n: POMCGlp1r+, n = 12, ctrl versus leptin P = 0.0067, leptin versus wash P = 0.008, ctrl versus wash P = 0.722; F = 8.72; **P < 0.01). Box plots were generated according to Tukey’s test, where ‘+’ illustrates the mean. Bottom right: numbers of peptide-responsive neurons in the respective cell types. o,p, Effect of NPY on POMCLepr+ neurons and POMCGlp1r+ neurons. o, Voltage-clamp recordings. NPY induced inward currents in POMCLepr+ neurons (n = 14; yellow) and POMCGlp1r+ neurons (n = 14; blue), shown as the mean ± s.e.m. p, Electrical charge that flowed during 10 min of NPY application (POMCLepr+, n = 14, minima = 2.73, Q1 = 4.03, Q2 = 5.68, Q3 = 9.19, maxima = 12.24, mean ± s.e.m. = 6.54 ± 0.80 nC; POMCGlp1r+, n = 14, minima = 0.78, Q1 = 2.34, Q2 = 3.274, Q3 = 4.82, maxima = 7.32, mean ± s.e.m. = 3.53 ± 0.48 nC; PuT = 0.0034, t = 3.23, df = 26, two-tailed unpaired Student’s t-test. **P < 0.01). In the box plots, a ‘+’ sign show the mean and the horizontal line is the median. The whiskers were calculated according to the Tukey method. In all recordings, synaptic input was pharmacologically blocked (Methods). Peptides were bath applied at the indicated concentrations: leptin (100 nM), Glp1 (300 nM) and NPY (100 nM). Responsiveness of individual neurons was defined by the 3-σ criterion (Methods). n, number of cells recorded. exc, excited; inh, inhibited; NR, not responsive. Q1, Q2 and Q3 represent the lower quartile, median and upper quartile, respectively.
Leptin (100 nM) excited 40% and inhibited 20% of POMCLepr+ neurons, while the remaining 40% of these cells were not responsive (Fig. 8a–d). In 60% of POMCGlp1r+ neurons, leptin did not affect the neuronal activity, while, despite the absence of Lepr expression, it inhibited 40% of these cells (Fig. 8l,m). Thus, in the presence of GABA receptor and glutamate receptor blockers, leptin modulated both neuronal populations differentially.
Correspondingly, Glp1 (300 nM) excited most (63%) POMCGlp1r+ neurons, whereas only 13% of the neurons were inhibited, and 22% were not affected by Glp1 (Fig. 8e–h). POMCLepr+ neurons, which largely do not express the Glp1r, were mostly unaffected by Glp1 (81%), and only 18% of these neurons were inhibited (Fig. 8i, j).
While the distinctive responses of both cell types to the two ligands indicate that classification of these neurons according to Lepr and Glp1r expression defines different responses to two satiety-communicating signals, the relatively high variability of the effects also raises the question of whether interactions within the energy homeostasis-regulating network contribute to the modulatory effects. Consequently, we performed recordings where, in addition to blocking GABA and glutamate receptors, action-potential-dependent signaling was suppressed by tetrodotoxin (TTX; 1 µM). Around 80% of POMCLepr+ neurons remained unaffected by Glp1, while 14% of neurons depolarized and 6% hyperpolarized (Fig. 8k). The proportion of POMCLepr+ neurons that responded to Glp1 is consistent with the proportion of these cell types expressing both Lepr and Glp1r, although lower than that previously reported to respond to Glp1, where 37% of alternatively identified POMCLepr+ neurons responded to a higher Glp1 dose (1 µM)31. Approximately 91% of POMCGlp1r+ neurons responded with a reversible hyperpolarization, while only 9% of neurons were unaffected by leptin (Fig. 8n). At first sight, these data might imply that leptin has direct effects on POMCGlp1r+ neurons (and Glp1 directly affects POMCLepr+). Alternatively, the results could indicate modulation of spontaneous release32. Moreover, these experiments indicate that the modulation of POMC neurons might lead to stronger cross-interactions between POMCLepr+ and POMCGlp1r+ neurons than originally expected.
Furthermore, consistent with a higher level of the Npy1r in POMCLepr+ neurons (Figs. 5d and 6c), NPY (100 nM) elicited larger outward currents in POMCLepr+ cells compared to POMCGlp1r+ neurons (Fig. 8o,p). Collectively, differentially expressed neuropeptide receptors, as identified via translational profiling, accurately predicted differential pharmacological responses of these cell types.
Discussion
Recent developments in single-cell RNA-seq have largely extended our view on the heterogeneity of molecularly defined neurocircuits, including those in control of energy and glucose homeostasis11. However, defining the underlying molecular heterogeneity of distinct neurocircuits, their functional organization and output remains a largely unmet territory in modern neuroscience. Here, we provide new mouse models, which allow for functional interrogation of heterogeneous populations based on combinatorial Cre- and Dre-dependent recombination in vivo. Recombinase-based gene targeting has become a valuable tool not only in neuroscience allowing for the modulation of specific cell types mostly via Cre-loxP-mediated recombination13. Alternative recombinase systems such as FLP/FRT-mediated recombination provide complementary tools, but are limited through a lower efficiency of recombination in mice despite that improved versions have been developed (FLPe and FLPo)33. While FLPe has been used for successful combinatorial recombination-based targeting of heterogeneous dopamine neurons using viral approaches, only a few examples have used FLP-based recombination for intersectional targeting in transgenic mice34.
An alternative recombinase system has been identified, that is, Dre-rox-mediated recombination17. Previous studies had indicated the feasibility of combinatorial Cre/Dre-recombinase usage for targeting specific neuronal subpopulations16. Here, we have systematically expanded the repertoire of tools and validated mice, enabling use of this system to define specific heterogeneous cell types. First, we show that Dre-based recombination allows for efficient targeting of rox-marked transgenes in single-allele configuration. Moreover, we show that Dre-dependent recombination is specific for rox-flanked DNA segments, as we have observed no cross-reactivity with loxP-flanked alleles in several lines of transgenic mice. While it had previously been reported that Dre-mediated recombination may result in background recombination of loxP-flanked DNA in a ROSA26lSlTomato reporter strain16, our extensive experiments clearly validate the usage of Cre/Dre-mediated combinatorial recombinases for complementary analyses of heterogeneous populations in addition to FLP-based approaches.
Nevertheless, Dre-dependent recombination appears to be less efficient compared to Cre-dependent recombination. Of the numerous, independent POMCDre transgenic mouse lines employing the exact same POMC BAC construct to express Dre as previously used to express Cre in these cells, the efficiency of Dre-mediated recombination is lower than Cre-mediated recombination, since the maximum rate of recombination was lower and increased over a longer period of development4. However, this represents a crucial advantage, when it allows Dre-mediated recombination to bypass narrow developmental periods, where a promoter is temporarily active during development in a subset of cells, and which thus unfaithfully marks cells upon very efficient recombinase targeting. This has been exemplified for POMC cells, where efficient POMCCre-mediated recombination during development targets cells, which later during development acquire a functionally antagonistic phenotype, that is, one of AgRP neurons18. In contrast, the lower efficiency and prolonged onset of POMCDre-mediated recombination enables circumvention of this effect and thus allows for long-term genetic marking of bona fide POMC-expressing cells.
We have successfully used combinatorial Cre/Dre-dependent recombination to genetically mark heterogeneous POMC neuron populations for fluorophore expression and translational profiling. Expression of ZsGreen in the nucleus of targeted cells allowed for efficient labeling of cell bodies and subsequent anatomical reconstruction of distribution probabilities of distinct cell types within the limited space of the ARC upon tissue clearing22. Here we have developed an image analysis pipeline that enables high-precision and high-resolution mapping of cell localization onto a standardized distribution space. Thereby, we have defined distribution maps of heterogeneous POMC cell populations within the ARC, revealing that POMCLepr+ and POMCGlp1r+ cells clearly exhibit a distinct anatomical distribution.
In addition, our experiments provide numerous new insights into the functional organization of hypothalamic POMC neurocircuits. First, the role of leptin in the regulation of POMC neurons has been subject to controversial findings over the last few years. Deletion of leptin receptors from POMC neurons throughout development causes mild obesity and hyperphagia, while the same intervention during adulthood has no effect on body weight or energy homeostasis4,35. On the other hand, POMC deficiency or selective ablation of hypothalamic POMC neurons in mice36 and POMC deficiency in humans causes massive obesity37, and re-expression of POMC in Lepr-expressing cells is sufficient to restore this effect in mice38, providing evidence for a role of POMCLepr+ neurons in the control of energy homeostasis. However, selective chemogenetic stimulation of POMCLepr+ neurons in our study only induces a minor suppression of food intake. Collectively, these data support a predominantly developmental role of Lepr-expressing POMC neurons in the control of energy homeostasis, but at the same time indicate that additional regulators other than the direct action of leptin contribute to control their activity. Recent experiments using Ca2+ imaging of POMC neuron activity in mice, have revealed that POMC neurons rapidly change their activity in response to sensory food perception, independent of changes in circulating energy-state-sensing hormones, and that this regulation contributes to the priming of liver ER adaptation upon sensory food perception39. Thus, top-down control of POMCLepr+ neurons or additional energy-state-sensing signals may also represent functionally relevant regulators of POMCLepr+ neuron activity.
In contrast, chemogenetic activation of POMCGlp1r+ neurons induces a more dramatic food intake suppression than what has previously been described, when POMCCre-expressing neurons have been activated either chemogenetically or optogenetically40. This is remarkable since transgenically targeted POMCGlp1r+ neurons represent a significantly smaller population than the POMCLepr+ neurons (data not shown). While POMC neurons were previously reported to be both GABAergic or glutamatergic, our gene expression and in situ analyses indicate low levels of Slc17a6 (Vglut2) expression in both neuronal groups.
Surprisingly, the chemogenetic activation of POMCLepr+ and POMCGlp1r+ neurons in female mice had no effect on food intake. ISH analysis showed that these neurons were also efficiently activated in females (Extended Data Fig. 5j). Thus, it is likely that the regulation of food intake in males and females is controlled differently, which is corroborated by previous reports of higher expression of Pomc mRNA and higher firing rates of POMC neurons in female mice compared to males41.
Another possibility for why selective activation of POMCGlp1r+ neurons may have a more pronounced food intake-suppressing effect than that previously reported using a global POMC population stimulation is evidenced by our electrophysiological studies. Typically, we observed differential and nonhomogeneous modulatory effects within one neuronal type, even in the presence of glutamate receptor and GABA receptor blockers. These findings raise the question whether factors other than glutamatergic and GABAergic interactions within this regulatory network might contribute to the modulation. One reason why this is a crucial question is that we have observed modulatory effects of ligands whose associated receptors are not expressed in the respective cell types. POMCGlp1r+ neurons, for example, are inhibited during leptin application, even with glutamate and GABA receptor blockers present, and when action-potential-dependent synaptic release is suppressed (Fig. 8n). Since we mainly did not detect the corresponding receptors in the majority of these neurons, this could indicate that leptin induces the release of inhibiting mediators from presynaptic neurons in a non-action-potential-dependent manner. These mediators might be released by modulation of spontaneous release32. In primary spinal afferents, activation of TRPV1 channels facilitates asynchronous synaptic release presumably by presynaptic Ca2+ influx42. Thus, it is plausible to consider that leptin mediates activation of TRP channels as previously described43 and thereby induces or increases the spontaneous release of neuronal mediators via increased Ca2+ influx.
In line with the notion of alternative regulators of POMCLepr+ and POMCGlp1r+ neuronal activity, our translational profiling has allowed the identification of numerous potentially differential regulators of these neuronal classes besides leptin and Glp1. Here, POMCLepr+ neurons in particular express numerous receptors for additional energy-state-sensing signals, which have already been validated to suppress feeding and even reduce body weight in obesity. Interestingly, our study reveals the differential expression of Npy1r and Npy5r in distinct POMC neuron populations. Given that the primary source of NPY for POMC neurons in the ARC is neighboring AgRP neurons coexpressing NPY, these findings may point to the possibility that POMCLepr+ neurons and POMCGlp1r+ neurons are under differential inhibitory control by AgRP neurons. Indeed, we have also electrophysiologically validated that NPY induces a larger (inhibitory) outward current in POMCLepr+ neurons, compared to POMCGlp1r+ neurons (Fig. 8o,p). Moreover, the surprising identification of the expression of NPY in POMCGlp1r+ neurons also warrants further study.
In summary, we have employed new models to begin to shed light on the anatomical, electrophysiological, molecular and functional heterogeneity of critical metabolism-regulatory POMC neurons. The detailed insights may also aid the development of new, rationalized strategies for the therapeutic manipulation of the melanocortin circuitry.
Methods
Animals and animal care
All animal procedures were conducted in compliance with protocols approved by local government authorities (Bezirksregierung Köln). Permission for breeding and experiments on mice was issued by the Department for Environment and Consumer Protection-Veterinary Section in Cologne ((§11) 576.1.35.2.G 07/18, 84-02.04.2017.A058). Mice were housed in individually ventilated cages at 22 –24 °C using a 12-h light/dark cycle. Animals had access to water and food ad libitum. Unless otherwise stated, animals were fed a normal chow diet (NCD; Teklad Global Rodent 2018, Harlan) containing 53.5% carbohydrate, 18.5% protein and 5.5% fat (12% of calories from fat). The high-fat diet (HFD; C1057, Altromin) consisted of 32.7% carbohydrate, 20% protein and 35.5% fat (55.2% of calories from fat).
Generation of genetically modified mouse strains
Driver lines
The LeprCre and Glp1rCre mouse lines have been previously described21,44. LeprCre mice and the Glp1rCre lines were kindly provided by M. G. Myers and F. Reimann, respectively.
The POMCDre BAC construct was generated by inserting the sequence encoding Dre recombinase together with a kanamycin/neomycin resistance cassette from the pTEDre plasmid into the start codon of the POMC gene of RP11-124K7 BAC via Red/ET recombination. Primers containing POMC-specific homology arms, 5POMC-Dre: 5′-tccctccaatcttgtttgcctctgcagagactaggcctgacacgtggaaggccaccatgggtaagaagaaga-3′ and 3POMC Dre: 5′-accagctccacacatctatggaggtctgaagcaggagggccagcaacagggaggatttaatatttctgacgc-3′, were utilized for amplification. The ATG codon from POMC was replaced by that of the Dre recombinase. The BAC was linearized with PISceI in the presence of 2,5 mM spermidine and loaded onto a self-assembled CL-4b sepharose column (Sigma, CL4B200), equilibrated with injection buffer (5 mM Tris, 0.1 mM EDTA (pH 7.6)). The flow-through fraction with the highest concentration of digested BAC was chosen for pro-nucleus injection, performed by the team of R. Naumann at the MPI for Molecular Cell Biology and Genetics in Dresden. Cre and Dre transgenic animals were bred to C57BL/6N mice for maintenance in the mouse facility of the Max Planck Institute for Metabolism Research in Germany.
ROSA26 transgenic mouse lines
Generation of ROSA26lSlrSrZsGreen (ROSA26-CAGS-lox-STOP-lox-rox-STOP-rox-ZsGreen) mice has been described in a previous study20. These mice have been crossed to a ubiquitously expressed Deleter-Cre line to obtain R26rSrZsGreen mice20. ROSA26rSrlSltdTomato (ROSA26-CAGS-rox-STOP-rox-lox-STOP-lox-tdTomato-WPRE) mice were purchased from The Jackson Laboratory.
The knock-in lines ROSA26lSlrSrZsGreen (ROSA26-CAGS-lox-STOP-lox-rox-STOP-rox-ZsGreen), ROSA26lSlrSrhM3Dq (ROSA26-CAGS-lox-STOP-lox-rox-STOP-rox-hM3Dq-2A-ZsGreen-WPRE) and ROSA26lSlrSrEGFPL10a (ROSA26-CAGS-lox-STOP-lox-rox-STOP-rox-EGFPL10a-WPRE) were generated in-house. For generation of these mouse lines, a ROSA26 locus-targeting vector (B9-36) was designed in which both a loxP-flanked STOP cassette and a rox-flanked STOP cassette prevent CAGS promoter-driven expression of the corresponding functional transgenic construct. For hM3Dq, the 5′-primer (5AscRassle) overhang used for the amplification of the transgene contained an AscI site and a Kozak consensus sequence and the 3′-primer (3AscRassle) overhang contained an AscI site and one C to stay in frame for the 2A-ZsGreen translation. For the EGFPL10a, the 5′-primer overhang used for the amplification of the transgene contained an AscI site and a Kozak consensus sequence and the 3′-primer overhang contained an XmaI. The sequence-verified knock-in sequences of hM3Dq and EGFPL10a were cloned into the AscI-digested and AscI/XmaI-digested B9-36-targeting constructs, respectively. After vector transfection into Bruce 4 embryonic stem cells, they were subsequently screened for correct integration by standard Southern blot methods. To this end, a ROSA26 probe was used45 on EcoRI-digested clonal genomic DNA that indicated homologous recombination via detection of an additional 7.1-kb band besides the 16-kb endogenous ROSA26 band. To exclude random integration, a Neo probe was used that detected one single 7.1-kb band in case of a single correct ROSA26 insertion. Correctly targeted and verified embryonic stem cell clones were chosen for blastocyst injection carried out by Taconic Biosciences to obtain chimeric animals. Resulting chimeras were backcrossed with C57BL/6N mice to obtain germline transmission on a pure C57BL/6N background.
Generation of experimental lines
For the POMCDre ROSA26rSrZsGreen mouse line, the breeding scheme consisted of mating heterozygous POMCDre mice to homozygous ROSA26rx/rx mice of the ZsGreen construct. Resulting double transgenic Dre+/− ROSA26rx/wt mice were used as experimental animals and compared to C57BL/6N mice for metabolic phenotyping. Littermates of both sexes were used for experiments.
The mouse lines POMCDre LeprCre ROSA26lSlrSrZsGreen, POMCDre Glp1rCre ROSA26lSlrSrZsGreen, POMCDre LeprCre ROSA26rSrlSltdTomato, POMCDre Glp1rCre ROSA26rSrlSltdTomato, POMCDre LeprCre ROSA26lSlrSrhM3Dq, POMCDre Glp1rCre ROSA26lSlrSrhM3Dq, POMCDre LeprCre ROSA26lSlrSrEGFPL10a and POMCDre Glp1rCre ROSA26lSlrSrEGFPL10a were generated via mating heterozygous double transgenic mice to homozygous ROSA26fl;rx/fl;rx mice of the corresponding functional transgene construct (Extended Data Fig. 2b). Resulting triple transgenic Cre+/− Dre+/− ROSA26fl;rx/wt mice were used as experimental animals and compared to genotype controls as stated in the figure legends. Littermates of both sexes were used for experiments.
The C57BL/6N mouse line was purchased from Charles River. For metabolic phenotyping, both genders were used for experiments as indicated in the figure legends.
Experimental details
Glucose tolerance test
Glucose tolerance tests were performed at 13 weeks of age with 16-h fasted mice. Body weights of mice and their basal blood glucose using a glucometer and glucose strips (Contour Next, Bayer HealthCare) were determined before the start of the experiment. Mice were injected with 20% glucose, and blood glucose was measured at 15, 30, 60 and 120 min after injection.
Nuclear magnetic resonance
Lean and fat mass were determined via nuclear magnetic resonance (NMR Analyzer Minispec mq 7.5, Bruker Optik) in live mice. Alternatively, body composition was analyzed by computed tomography (CT) in isoflurane-anesthetized mice (Dräger and Piramal Healthcare). For data acquisition on an IVIS Spectrum CT scanner (Caliper LifeScience), we used IVIS LivingImage Software V.4.3.1. Quantification of lean and fat mass contents were determined with a modification of the previously described VINCI software package 4.61.0.
Indirect calorimetry
Metabolic phenotyping and food intake were measured by an automated PhenoMaster open-circuit indirect, calorimetry system (TSE Systems). Mice were allowed to acclimatize to the experimental setup for 4 d before the start of each experiment. Food and water were available ad libitum. Data acquisition was carried out by TSE Phenomaster versions 6.2.5 and above.
Study design of DREADD animals
POMCDre LeprCre ROSA26lSlrSrhM3Dq and POMCDre Glp1rCre ROSA26lSlrSrhM3Dq mice and corresponding genotype controls were characterized at 13–15 weeks of age with ad libitum access to NCD. Saline (0.9%) or CNO injections (3 mg per kg body weight) were administered intraperitoneally. For measurements of food intake (as described under ‘Indirect calorimetry’), mice were injected with saline at 18:00 and 23:00, followed by a 1-d recovery period and subsequent CNO injections at 18:00 and 23:00 on the next day. For measurements of energy expenditure, respiratory exchange ratio and locomotion (as described under ‘Indirect calorimetry’), mice were injected with saline at 17:00, 22:00 and 07:00 followed by CNO at 17:00, 22:00 and 07:00 on the next day. Before perfusions at 22–26 weeks of age, mice were fasted for 2 h and injected with saline or CNO 1 h before perfusion. Serum for the corticosterone enzyme-linked immunosorbent assay (ELISA) was obtained from mice fasted for 2 h and injected with CNO 1 h before blood collection. Littermates of both sexes were used for experiments as indicated in text and figures.
CNO administration
CNO (Abcam, ab141704) powder was dissolved in dimethyl sulfoxide (DMSO; 100 mg ml−1) and diluted at a ratio of 1:333 in 0.9% NaCl (saline).
Study design of bacTRAP (EGFPL10a) mice
For POMCDre LeprCre ROSA26lSlrSrEGFPL10a and POMCDre Glp1rCre ROSA26lSlrSrEGFPL10a animals, 6 triple transgenic bacTRAP mice (3 females and 3 males) and 12 triple transgenic bacTRAP mice (6 females and 6 males) were pooled for each replicate, respectively, accounting for three to four replicates per POMC subpopulation. Mice were killed at 12 weeks of age in a random-fed state by decapitation. Whole hypothalami were obtained using a mouse brain slicer matrix and snap frozen in liquid nitrogen until translating ribosome affinity purification (TRAP).
Perfusion and tissue fixation
With the exception of DREADD animals, all mice were perfused in a random-fed state. Mice were deeply anesthetized and perfused transcardially with 1× PBS followed by ice-cold 4% paraformaldehyde (PFA; in 1×PBS; pH 7.4). The brain was removed from the skull and post-fixed in 4% PFA at 4 °C for approximately 24 h, and then moved to 20% sucrose solution (in 1× PBS) at 4 °C. The brains were cut at 20 μm on a sliding microtome (Leica Microsystems, SM2010R) equipped with a stage for dry ice. For immunohistochemistry, sections were either collected on slides or in bins containing anti-freeze solution (30% ethylene glycol and 20% glycerol in PBS), and subsequently stored at −20 °C until further processing. For RNA ISH, sections were mounted on SuperFrost Plus Gold slides (Thermo Fisher Scientific, 11976299) and subsequently stored at −80 °C until further processing.
Immunohistochemistry
For immunofluorescence stainings against ZsGreen, POMC, tdTomato and EGFP, all incubation steps were performed at room temperature unless otherwise stated.
Floating sections were washed once for 10 min in PBS, incubated for 10 min in 0.3% glycine, washed again for 5 min in PBS and incubated for 10 min in 0.03% SDS/PBS. Subsequently, sections were blocked for 1 h in 3% donkey serum in PBS containing 0.25% Triton X. Next, sections were incubated overnight at 4 °C in primary antibody diluted in Signal Stain (Cell Signaling, 8114). Primary antibodies and dilutions used were: rabbit anti-ZsGreen (Takara Bio Clontech, 632474; 1:100), rabbit anti-POMC (Phoenix, H-029-30; 1:1,000), rat anti-mCherry (for tdTomato; Thermo Fisher Scientific, M11217; 1:1,000) and chicken anti-GFP (Abcam, ab13970; 1:1,000) were used. The following morning, sections were washed three times for 10 min in PBS containing 0.1% Triton X and incubated for 1 h in secondary antibody in PBS containing 0.25% TritonX. Secondary antibodies and dilutions were donkey anti-rabbit Alexa Fluor 488 (Thermo Fisher, A21206; 1:500), donkey anti-rat Alexa Fluor 594 (Jackson ImmunoResearch, 712-585-153; 1:500) or goat anti-rabbit Alexa Fluor 594 (Thermo Fisher Scientific, A11012; 1:500) and goat anti-chicken Alexa Fluor 488 (Thermo Fisher Scientific, A11039; 1:500), for 1 h at room temperature. After three washing steps for 10 min in PBS containing 0.1% Triton X, sections were mounted in Vectashield DAPI-containing mounting medium (Vector Laboratories, VEC-H-1200) and stored at 4 °C in the dark until imaging.
Corticosterone ELISA
Concentrations were determined using a commercial Corticosterone ELISA kit from CrystalChem as described in the user’s manual (80556).
Imaging and quantification of immunohistochemistry
Images were captured using a confocal Leica TCS SP-8-X microscope, equipped with a ×40/1.30 oil objective with the acquisition software (Leica ASX V.3.5.5.19976). Next, z-stacks were taken with optical sections of 0.9 μm. Laser intensities were kept constant throughout all related conditions. Images were imported into FIJI where maximum intensities were projected. For representative images, adjustments in brightness and contrast for each channel were kept constant throughout all related conditions, whereas for quantifications of POMC and tdTomato signals, all channels were kept unmodified and one to four sections were quantified per mouse and area. Images were converted to 8-bit, and the threshold for signal detection for each channel was determined by visual judgment and consistently applied to all images. ROIs were defined around corresponding anatomical locations and raw integrated densities measured within ROIs for POMC and tdTomato signals.
RNA in situ hybridization
The fluorescence ISH technique (RNAscope) was used to detect mRNA of Pomc, Agrp, Lepr-tv1, Glp1r, ZsGreen, Fos, Cartpt, Vglut2, Vgat, Npy1r, Oprm1 and Nmur2. All reagents were purchased from Advanced Cell Diagnostics (ACD) if not otherwise stated. The Pomc probe (314081) contained ten oligonucleotide pairs targeting region 19–995 (NM_008895.3) of the Pomc transcript; the Agrp probe (400711) contained 16 oligonucleotide pairs targeting region 11–764 (NM_001271806.1) of the Agrp transcript; the Lepr-tv1 probe (471171) contained 19 oligonucleotide pairs targeting region 3220–4109 (NM_146146.2) of the Lepr transcript variant 1; the Glp1r probe (418851) contained 20 oligonucleotide pairs targeting region 108–1203 (NM_021332.2) of the Glp1r transcript; the ZsGreen probe (461251) contained 15 oligonucleotide pairs targeting region 980–1655 (JQ071441.1) of the ZsGreen transcript; the Fos probe (316921) contained 20 oligonucleotide pairs targeting region 407–1427 (NM_010234.2) of the Fos transcript; the Cartpt probe (432001) contained 17 oligonucleotide pairs targeting region 11–860 (NM_013732.7) of the Cartpt transcript; the Vglut2 probe (319171) contained 20 oligonucleotide pairs targeting region 1986–2998 (NM_080853.3) of the Vglut2 transcript; the Vgat probe (319191) contained 20 oligonucleotide pairs targeting region 894–2037 (NM_009508.2) of the Vgat transcript; the Npy1r probe (427021) contained 20 oligonucleotide pairs targeting region 227–1169 (NM_010934.4) of the Npy1r transcript; the Oprm1 probe (315841) contained 20 oligonucleotide pairs targeting region 1135–2162 (NM_001039652.1) of the Oprm1 transcript; and the Nmur2 probe (314111) contained 20 oligonucleotide pairs targeting region 69–1085 (NM_153079.4) of the Nmur2 transcript. RNAscope 4-plex negative (321831) and positive-control probes (321811) were processed in parallel with the target probes. All incubation steps were performed at 40 °C using the ACD HybEz hybridization system (321462) if not stated otherwise. One day before the assay, sections were mounted on SuperFrost Plus Gold slides (FT4981GLPLUS; Thermo Fisher), dried at room temperature (RT), briefly rinsed in autoclaved Millipore water, air dried and incubated at 60 °C for 4–6 h. Subsequently, slides were submerged in Target Retrieval reagent (322000) at 99.5 °C for 10 min, rinsed once in autoclaved Millipore water and dehydrated in 100% ethanol for 3 min. Slides were air dried for 5 min, a hydrophobic barrier was created around the sections using an ImmEdge hydrophobic barrier pen (310018) and slides were stored at RT until assaying. The following day, slides were incubated with Protease Plus (322330) for 25 min. The subsequent steps, that is, hybridization of the probes, amplification and detection steps, were performed according to the manufacturer’s protocol for RNAscope Fluorescent Multiplex Detection Reagent kit v2 (323110), or for more than three probes, the protocol for RNAscope 4-Plex Ancillary Kit for Multiplex Fluorescent Kit v2 (323120) was used. The probes were detected using tyramide-diluted Opal690 (1:2,000 dilution), Opal650 (1:1,500 dilution), Opal620 (1:1,000 dilution), Opal570 (1:1,000 dilution), Opal520 (1:750 dilution) or Cy3 (1:750 dilution). Sections were counterstained with DAPI and coverslipped with ProLong Gold Antifade Mountant (Thermo Fisher, P36931) and stored in the dark at 4 °C until imaged.
Imaging and quantification of RNA ISH
Images were captured using a confocal Leica TCS SP-8-X microscope, equipped with a ×40/1.30 oil objective. Next, z-stacks were taken with optical sections of 0.9 μm. Laser intensities were kept constant throughout all related conditions. Images were imported into FIJI (National Institutes of Health, version 2.0.0-rc-41/1.50d), where maximum intensities were projected. For representative images, adjustments in brightness and contrast for each channel were kept constant throughout all related conditions, whereas for quantifications, all channels were kept unmodified and approximately five to ten sections were quantified per mouse and, if required, per area. For intensity quantification of endogenous Lepr, Glp1r and Pomc expression in C57BL/6N mice, all channels were imported and fused into the Halo software (Indica Labs, V.2.2.1870). The software relies on the DAPI stain for cellular identification and calculates the cell intensity for each cell and probe (a number integrating both the fluorescence intensity and the covered probe area within the designated cell). The threshold for probe recognition was determined by visual judgment, considering approximately five or more signals per cell as positive. For generation of the distribution pattern of POMC neuronal subpopulations throughout a coronal cross section, comparable anatomical locations within the ARC were analyzed correspondingly by the Halo software and three mice per replicate were merged. For quantification of Cartpt, Npy1r, Oprm1 and Nmur2 in C57BL/6N mice, integrated density was assessed via FIJI in previously defined single-cell ROIs showing Pomc signal and five or more signals of the Lepr or Glp1r probe. Cell counting of ROSA26rSrZsGreen and ROSA26lSlrSrhM3DGq mice was performed manually, defining single-cell ROIs showing Pomc and ZsGreen signal, and considering five or more probe signals per ROI of Agrp, Lepr, Glp1r, Fos, Vglut2 or Vgat as positive.
Gene copy number assay
For determining the Pomc copy number, 20 ng of genomic DNA extracted via standard isopropanol precipitation from tail biopsies was used. Selected gene segments were amplified using TaqMan Universal PCR-Master Mix (Thermo Fisher Scientific, 4305719) according to the manufacturer’s instructions. Non-exon-spanning primers (P1: 5′-GCGACAGGGACCAAACGG-3′, P2: 5′-AGACACCCTTACCTGTCGC-3′) and probes (5′-FAM-TCAGTGGCCTCTCTTAGTCACTGC-TAMRA-3′) were designed for amplification of Pomc exon 1, while the gene expression assay for Socs3 (Thermo Fisher Scientific, Mm00545913_s1, 4331182) was purchased. The expression of Pomc exon 1 was normalized to Socs3 mRNA. Results were calculated by the ΔCt comparative method (as 2−ΔΔCt), in which fold changes were calculated by defining wild-type animals with two Pomc gene copy numbers.
uDISCO whole-brain clearing and image acquisition
The protocol was adapted from the uDISCO clearing method22. POMCDre ROSA26rSrZsGreen+/−, POMCDre LeprCre ROSA26lSlrSrZsGreen+/− and POMCDre Glp1rCre ROSA26lSlrSrZsGreen+/− male and female mice were perfused with 1× PBS (pH 7.4) for 10 min followed by 4% PFA/PBS for 10 min. The brains were post-fixed for 24 h in 4% PFA/PBS (pH 7.4) at 4 °C. The brains were dehydrated in a gradient manner via incubations in tert-Butanol diluted in distilled water: 30%, 50%, 70%, 80%, 90% and 96% (vol) for 10–16 h at 37 °C. The brains were subsequently incubated in dichloromethane for 90–120 min at RT to remove lipids. Next, BABB-D4 was used as the reagent-matching solution for 12 h at RT to complete the clearing process (BABB: benzyl alcohol + benzyl benzoate at a 1:2 ratio, respectively; BABB-D4: a mixture of BABB and diphenyl ether (DPE) at a ratio 4:1 (vol/vol)). The cleared brains were imaged with a LaVision Bio Tec Ultramicroscope II with the acquisition software Inspector Pro (V.5.0.222.0). Whole-brain and magnified scans of the ARC were obtained using ×1.6 and ×8 total magnification, respectively. For the projection density analysis, cleared brains from POMCDre LeprCre ROSA26lSlrSrtdTomato+/− and POMCDre Glp1rCre ROSA26lSlrSrtdTomato+/− mice were imaged using ×1.6 magnification with the same laser intensity across all samples.
Whole-brain immunostaining
Pretreatment before staining was as follows: the fixed brains were washed two times in PBS for 1 h, incubated in 50% methanol (in PBS) for 1 h, 80% methanol for 1 h and two times in 100% methanol for 1 h. The samples were bleached with 5% H2O2 in 20% DMSO/methanol (one vol 30% H2O2/one vol DMSO/four vol methanol) at 4 °C overnight. Subsequently, samples were transferred to methanol for 1 h twice, then in 20% DMSO/methanol for 1 h twice, then in 80% methanol for 1 h, 50% methanol for 1 h, twice in PBS for 1 h, and finally in PBS/0.2% Triton X-100 for 1 h twice before further staining procedures. Pretreated samples were incubated in PBS/0.2% Triton X-100/20% DMSO/0.3 M glycine at 37 °C overnight, then blocked in PBS/0.2% Triton X-100/10% DMSO/6% donkey serum at 37 °C overnight. This was followed by a wash in PBS/0.2% Tween-20 with 10 μg ml−1 heparin (PTwH) for 1 h twice, then incubated in rat anti-mCherry (for tdTomato; Thermo Fisher Scientific, M11217; 1:500 dilution) in PTwH/5% DMSO/3% donkey serum at 37 °C for 8 d. After a 1-d long wash in PTwH, the samples were incubated in anti-rat Alexa Fluor 594 (Thermo Fisher Scientific, A21207; 1:300 dilution) in PTwH/3% donkey serum at 37 °C for 4 d. Samples were finally washed in PTwH before starting the clearing protocol, followed by LSFM imaging.
Co-registration using VINCI
An automated workflow for co-registering each ×4-magnified scan per brain was registered to its full-brain image. Each full-brain image was subsequently registered to the Allen Brain 25-μm reference mouse brain atlas, using rigid-body correlation-coefficient and 12-parameter affine mutual information schemas in VINCI23. Originally, VINCI was developed for co-registration of clinical data but can also be utilized in animal studies46. The parameters were adapted for the multi-scale approach and preprocessing (quantile filtering) for microscopy data (VINCI versions 4.61.0 and 4.96.0). However, the size of the microscopy scans meant that further optimization was required; we needed to co-register 32-GB image files (8 × 109 voxels), while typical-use cases in positron-emission tomography, magnetic resonance imaging and computed tomography for animal and human data are in the order of 2 × 107 voxels. The processing of these large datasets was expedited through multi-threading, and the co-registration time on a dedicated system (56 cores) could be reduced from over 24 h to 0.7 h. The transformation matrix of the ×4 image to the atlas was derived as the combination of the two transformations. Quality of registration was determined based on a visual inspection of the registration result (a match of the interhemispheric fissures and the anatomical surface at the base of the hypothalamus). The scans with the best co-registration in each group were identified and the ×4 images from each mouse were aligned to the ×4 scan of the reference mouse within each group. This was performed using another specially adapted 12-parameter affine mutual information schema combined with the known transformation of the reference mouse full-brain image to the atlas. The correlation-coefficient values for the reference brain for POMC, Lepr and Glp1r were 0.85, 0.75 and 0.90, respectively. The quality of registration for the Glp1r group was not optimal so the Glp1r scans were co-registered to the ×4–×4 script but instead to the reference mouse of the POMC group.
Extraction of neuronal coordinates using Arivis and three-dimensional scatterplots
The output NIFTI files of the co-registered ×4 brain scans were uploaded into the Arivis Vision 4D software (V.3.3.0). Individual neurons were identified using the Blob Finder feature in the software and their xyz coordinates were extracted. A Python script was used to visualize the 3D scatterplots for each neuronal group, where the quality of registration was checked once again. For the coronal view of the 3D distribution, the 3D scatterplots of three mice (per group) were plotted and a coronal snapshot was created from rostral locations within the ARC comparable to that of the two-dimensional (2D) coronal distribution. The coordinates of the 2D coronal distributions were also extracted using the Arivis software from the Halo output files (‘Imaging and quantification of RNA ISH’) that had been previously aligned manually. The final distribution graph was plotted using the Prism software.
Isosurface density plots
To create the isosurface density plots, a Python script was used (V2.7.12). The probability density function of the 3D distribution of neurons was estimated for the whole POMC population, as well as the Lepr and Glp1r subpopulations. This was performed using the non-parametric kernel density estimation method. The density of neurons within the ARC was thus calculated based on the 3D neuronal coordinates to demonstrate the spatial concentration, which was then color coded to visualize the regions within the ARC with higher neuronal counts for each subpopulation.
Data analysis of three-dimensional projections
Brain scans were co-registered to an annotated atlas (https://scalablebrainatlas.incf.org/mouse/ABA12#downloads/) using VINCI. Thresholding was achieved using the threshold function on VINCI by applying a value across all scans. Using the automated calculation function of VINCI, a mask was created from the ROIs in 3D, and the intensity profile of each region was determined using the VOI define function on VINCI. Representative images in the areas of interest were rendered using the Arivis software.
Purification of mRNA from triple-positive EGFPL10a mice
The TRAP technique was performed using a modified version of a previous study on hypothalami of mice described under ‘Study design of bacTRAP (EGFPL10a) mice’47. One day before TRAP, 375 µl of Dynal Protein G magnetic beads (Invitrogen) was washed three times (1 ml) and resuspended in 275 µl of IP wash buffer (20 mM HEPES (pH 7.4), 150 mM KCl, 5 mM MgCl2 and 1% NP-40). Then, 50 µg of two monoclonal anti-GFP antibodies (HtzGFP-19C8 and HtzGFP-19F7) from the Memorial Sloan-Kettering Cancer Center was added to the magnetic beads and incubated with slow end-over-end mixing overnight at 4 °C.
On the day of TRAP, Dynal Protein G magnetic beads were washed three times (1 ml) and resuspended in 200 µl IP buffer (20 mM HEPES (pH 7.4), 150 mM KCl, 5 mM MgCl2, 1% NP-40, 0.5 mM dithiothreitol and 100 mg ml−1 cycloheximide) to remove unbound anti-GFP. Ice-cold polysome extraction buffer (20 mM HEPES (pH 7.4), 150 mM KCl, 5 mM MgCl2, 0.5 mM dithiothreitol, 100 mg ml−1 cycloheximide, protease inhibitors and 40 U ml−1 recombinant RNasin Ribonuclease inhibitor) was added to the samples containing mouse hypothalami and homogenized with a motor-driven Teflon glass homogenizer. Homogenates were centrifuged at 4 °C for 10 min at, 2000g to pellet cell debris. Supernatant was transferred to a new microcentrifuge tube and NP-40 (Applichem) and 1,2-diheptanoyl-sn-glycero-3-phosphocholine (DHPC; Avanti Polar Lipids) were added to the supernatant at a final concentration of 1% and 30 mM, respectively. After incubation on ice for 5 min, the clarified lysate was centrifuged for 10 min at 13,000g to pellet insoluble material. Next, 30 µl of supernatant (input) was snap frozen in liquid nitrogen until RNA extraction as a comparison to the immunoprecipitated sample. Then, 200 µl of anti-GFP-coated Dynal protein G magnetic beads was added to the supernatant, and the mixture was incubated at 4 °C with end-over-end rotation for 1 h. Beads were subsequently collected on a MagnaRack (Invitrogen), washed four times with high-salt polysome wash buffer (20 mM HEPES (pH 7.4), 350 mM KCl, 5 mM MgCl2, 1% NP-40, 0.5 mM dithiothreitol and 100 mg ml−1 cycloheximide). Input and IP beads were resuspended and incubated in RLT buffer (RNAeasy micro kit, Qiagen) for 5 mins at RT. Supernatant was removed from the IP beads and RNA extraction was performed according to Qiagen’s protocol, including in-column DNase digestion. RNA was resuspended in 10 μl nuclease-free water. RNA quantity and quality of input and IP were determined with a Qubit Fluorometer (Invitrogen) and Agilent 2100 Bioanalyzer.
RNA sequencing
Pre-amplification was carried out using the Ovation RNA-seq system (V2). Total RNA was used for first-strand cDNA synthesis, using both poly(T) and random primers, followed by second-strand synthesis and isothermal strand-displacement amplification. For library preparation, the Illumina Nextera XT DNA sample preparation protocol was used, with 1 ng cDNA input. After validation (Agilent 2200 TapeStation) and quantification (Invitrogen Qubit System), transcriptome libraries were pooled. The pool was quantified using the Peqlab KAPA Library Quantification Kit and the Applied Biosystems 7900HT Sequence Detection and sequenced on an Illumina HiSeq 4000 sequencing instrument with a 2 × 75-bp paired-end read length protocol.
Gene Ontology analysis
The regulated GO48 terms were derived using the clusterProfiler R package49 and visualized by mapping the percentage of regulated genes to the total GO term gene count against significant gene counts and adjusted P values per term. We visualized the GO term as a heat map, where each row represents a differentially expressed gene belonging to the term and each column represents a sample. To ensure visual differentiation between the rows not being skewed by highly expressed genes, the values of each sample were converted to z-scores with respect to the individual genes.
Overlap analysis
We used publicly available single-cell RNA-seq data from mouse hypothalami to detect overlaps with markers identified in our Glp1r and Lepr datasets. The first data source used was single-cell RNA-seq of 20,921 cells from the arcuate–median eminence complex of mice11. We filtered cells expressing POMC (4,248/20,921 cells) and clustered them using the R Seurat package24. The clustering result was visualized using the UMAP plot of Seurat. The second data source used was from the single-cell RNA-seq dataset of 163 POMC-expressing neurons in mice yielding four clusters12.
Electrophysiological experiments
Animals and brain slice preparation
Experiments were performed on brain slices from 12- to 15-week-old genetically marked (with ZsGreen) Glp1r- and Lepr-expressing POMC neurons using POMCDre LeprCre ROSA26lSlrSrZsGreen+/− or POMCDre Glp1rCre ROSA26lSlrSrZsGreen+/− male and female mice. Animals were kept under standard laboratory conditions, with tap water and chow available ad libitum, on a 12 h light/dark cycle. The animals were lightly anesthetized with isoflurane (B506; AbbVie) and decapitated. Coronal slices (270–300 µm) containing the ARC were cut with a vibration microtome (HM-650 V; Thermo Scientific) under cold (4 °C), carbogenated (95% O2 and 5% CO2), glycerol-based modified artificial cerebrospinal fluid (GaCSF)50. GaCSF contained (in mM): 244 glycerol, 2.5 KCl, 2 MgCl2, 2 CaCl2, 1.2 NaH2PO4, 10 HEPES, 21 NaHCO3 and 5 glucose, adjusted to pH 7.2 with NaOH. If not mentioned otherwise, the brain slices were continuously superfused with carbogenated aCSF at a flow rate of ~2.5 ml min−1. aCSF contained (in mM): 125 NaCl, 2.5 KCl, 2 MgCl2, 2 CaCl2, 1.2 NaH2PO4, 21 NaHCO3, 10 HEPES and 5 glucose, adjusted to pH 7.2 with NaOH. To block GABAergic and glutamatergic synaptic input, in all recordings, the aCSF contained 10−4 M picrotoxin (P1675; Sigma-Aldrich), 5 × 10−6 M CGP (CGP-54626 hydrochloride; BN0597, Biotrend), 5 × 10−5 M DL-AP5 (DL-2-amino-5-phosphonopentanoic acid; BN0086, Biotrend) and 10−5 M CNQX (6-cyano-7-nitroquinoxaline-2,3-dione; C127, Sigma-Aldrich). To suppress action-potential-dependent synaptic release, blocked voltage-dependent Na+ channels were blocked by 10−6 M TTX (T-550, Alomone).
Electrophysiology
Current-clamp and voltage-clamp recordings of ZsGreen-expressing POMC neurons were performed at ~32 °C in the perforated patch-clamp configuration. Neurons were visualized with a fixed-stage upright microscope (BX51WI, Olympus) using ×40 and ×60 water-immersion objectives (LUMplan FL/N ×40, 0.8 numerical aperture, 2 mm working distance; LUMplan FL/N ×60, 1.0 numerical aperture, 2 mm working distance, Olympus) with infrared differential interference contrast optics51 and fluorescence optics. ZsGreen-expressing POMC neurons were identified by their anatomical location in the ARC and by their ZsGreen fluorescence that was visualized with an X-Cite 120 illumination system (EXFO Photonic Solutions) in combination with a Chroma 41001 filter set (ex: HQ480/×40; bs: Q505LP; em: HQ535/50m). Electrodes with tip resistances of between 4 and 6 MΩ were fashioned from borosilicate glass (0.86-mm inner diameter; 1.5-mm outer diameter; GB150-8P, Science Products) with a vertical pipette puller (PP-830, Narishige). All recordings were performed with an EPC10 patch-clamp amplifier (HEKA) controlled by the program PatchMaster (version 2.32; HEKA) running in Windows. In parallel, data were recorded using a micro1410 data acquisition interface and Spike 2 (version 7, both from CED). Current-clamp recordings were sampled at 25 kHz and low-pass filtered at 2 kHz with a four-pole Bessel filter. Voltage-clamp recordings were sampled at 5 kHz, smoothed (𝜏 = 0.2 s) and downsampled to 0.5 Hz. The calculated liquid junction potential of 14.6 mV between intracellular and extracellular solution was compensated or subtracted offline (calculated with Patcher’s Power Tools plug-in from https://www3.mpibpc.mpg.de/groups/neher/index.php?page=software for IGOR Pro 6; Wavemetrics).
Perforated patch experiments were conducted using protocols modified from previous studies52,53. Recordings were performed with pipette solution containing (in mM): 140 K-gluconate, 10 KCl, 10 HEPES, 0.1 EGTA and 2 MgCl2, adjusted to pH 7.2 with KOH. ATP and GTP were omitted from the intracellular solution to prevent uncontrolled permeabilization of the cell membrane54. The patch pipette tip was filled with internal solution and backfilled with internal solution, which contained the ionophore to achieve perforated patch recordings and 0.02% tetramethylrhodamine-dextran (3,000 MW, D3308, Invitrogen) to monitor the stability of the perforated membrane. Amphotericin B (A4888; Sigma) was dissolved in DMSO to a concentration of 40 µg µl−1 (D8418, Sigma) following the protocols of a previous study55. The used DMSO concentration (0.1–0.3%) had no obvious effect on the investigated neurons. The ionophore was added to the modified pipette solution shortly before use. The final concentration of amphotericin B was ~120–160 µg ml−1. Amphotericin solutions were prepared from undissolved weighted samples (stored at 4 °C protected from light) on every recording day. During the perforation process, access resistance (Ra) was monitored continuously and experiments started after Ra values reached steady state (~15–20 min) and the action potential amplitude was stable.
Intrinsic electrophysiological properties
To analyze in detail the intrinsic electrophysiological properties of ZsGreen-expressing POMC neurons, a set of current-clamp protocols from a holding potential of −70 mV was applied. Cell input resistance was determined from a series of hyperpolarizing small current pulses (1 s, 2–10 pA increments) and the slope of the resulting I–V relations. Whole-cell capacitances were calculated from the membrane time constant (𝜏) and the input resistance (R): C = 𝜏/R. To analyze the IH-dependent sag potentials, the neurons were hyperpolarized with five consecutively incrementing current pulses. The increments were adjusted so that the last pulse hyperpolarized the membrane to −120 mV. The sag potential was defined as the difference between the lowest voltage reached at the beginning of the pulse and the membrane potential reached at the end of hyperpolarization. To analyze post-inhibitory rebound excitation, we used an ‘enhanced rebound protocol’, whereby the same current-step amplitudes were applied as those used for the sag-potential analysis, but this time as 2-s hyperpolarizing pre-pulses that were followed by a 1-s test pulse with the amplitude of a single increment. The maximum instantaneous frequencies during the rebound were determined and plotted over the membrane potentials of the pre-pulses. To analyze input–output relations, we applied a series of ascending and then descending current ramps (5 s each), where the ramp amplitudes were increased from 10 to 25 pA in 5-pA increments. Amplitudes were further increased if the 25-pA ramp did not elicit action potentials. Spike-number ratios were calculated by dividing the number of action potentials during the ascending ramp by the number of action potentials during the descending ramp. To further analyze excitability, that is, evoked action potential firing, a series of depolarizing current pulses (1 s; 5–50 pA in 5-pA increments) was applied. For each current pulse, the number of action potentials was determined, plotted over the current amplitude and linearly fit. Linear fits were performed for data points where action potentials were elicited. Only data points at which action potentials were triggered were considered for fit.
For SFA ratios, 10-s depolarizing stimuli were applied from a holding potential of −70 mV with initial instantaneous action potential frequencies between 30 and 40 Hz. Instantaneous frequencies were plotted (Y) over the 10-s time course, and fit to a mono-exponential decay equation with Y0 set to the initial instantaneous frequency: Y = (Y0 − plateau) × exp(−K × T) + plateau, where ‘plateau’ is the asymptotic frequency, K is the inverse time constant and T is the time. The SFA ratio is determined by dividing the maximum initial instantaneous frequency by the plateau frequency of the fit. Action potential waveform parameters were obtained from action potentials with instantaneous frequencies ≤ 5 Hz. If necessary, hyperpolarizing bias currents were used to decrease spontaneous firing.
Peptide signaling
Leptin (100 nM; L3772, Sigma-Aldrich), and Glp1 (300 nM; H-5956, Bachem AG) were bath applied for 15 or 30 min with a perfusion rate of 2.5 ml s−1. Npy (100 nM; N5071, Sigma-Aldrich) was bath applied for 10 min.
Leptin and Glp1 signaling
In line with previous studies, we found that the basic firing properties of POMC neurons and their responsiveness to leptin and Glp1 were not homogeneous. Therefore, we used the ‘three times standard deviation’ (3σ) criterion, and a neuron was considered responsive if the change in firing frequency or membrane potential induced by leptin or Glp1 was three times larger than the standard deviation. Means and respective standard deviations of spontaneous action potential firing or membrane potential were calculated from a period of 120 s, divided into 12 bins, each 10 s long. Data were taken immediately before and at the end of the peptide application.
Voltage-clamp recording of peptide-induced currents
The NPY action on POMC neurons was analyzed under voltage clamp. Based on previous studies56,57,58 and on the peptide-induced action-potential frequency modulation, which we observed in current-clamp recordings, the holding potential was set to −55 mV to optimize the recording conditions to measure inward currents. NPY (100 nM) was bath applied for 10 min after a 5-min baseline recording. The recorded peptide-induced currents were baseline subtracted and the mean (±s.e.m.) was calculated. To quantify differences in the peptide-induced currents, the area under the curve (electrical charge in nC) during the 10-min application of NPY was calculated.
Quantification and statistical analysis
Statistical analyses and reproducibility
Details on statistical analysis for Figs. 3, 5, 7 and 8 can be found in ‘Statistical analysis of 3D data’, ‘RNA-sequencing analysis workflow’ and ‘Electrophysiological data analysis and statistics’, respectively. Primary data processing and organization were performed in Microsoft Excel (2010). Statistical analyses were performed using Prism software (GraphPad, V.5.0–V.8.0). Statistical significance for two groups was determined by unpaired two-tailed Student’s t-test. In the case of unequal variance between the two groups, the unpaired Welch’s t-test or unpaired Mann–Whitney U-test was used. For determining differences between more than two groups, one-way ANOVA was applied. Depending on the scientific question, one-way ANOVA was followed by no post hoc test, by Dunnett’s post hoc test (comparing all groups to one control) or by Tukey’s post hoc test (comparing all groups among each other), as indicated in the figure legends. All remaining data (more than two groups, more than one independent factor) were analyzed with two-way ANOVA followed by Sidak’s or Tukey’s post hoc analysis. Data are expressed as the mean, and the error bars indicate the s.e.m. unless specified otherwise. In the violin plots, solid lines represent median values, and dashed lines represent lower and upper quartiles. A detailed description of statistics, including individual data points, tests, P values and further statistical parameters, are provided in the figure legends and as source data. Statistical significance was defined as: *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001 and ****P ≤ 0.0001.
No statistical methods were used to predetermine sample sizes, although sample sizes are similar to those reported in previous publications6,39,59. Data collection and analysis were carried out in a blinded format throughout the study, unless this was not possible due to the visual differences in cases of varying neuronal numbers resulting from genetic labeling as depicted in Fig. 3a–c. Data distribution was assumed to be normal, but this was not formally tested. Except for animals that died during an experiment, no data were excluded. For metabolic phenotyping, every mouse represents a replicate (n) and the number of replicates is mentioned for each experiment in the figure legend and/or source data. In this case, data were pooled from independent experiments of varying n numbers. For RNA-seq, samples of pooled hypothalami were collected from individual mice of several cohorts. For electrophysiological experiments, the sample numbers indicate the number of cells used in recordings. All measurements that did not require statistical analysis, such as representative images, were obtained from at least two animals and in most cases a minimum of three animals were used.
Statistical analysis of three-dimensional data
To carry out a statistical assessment of the differences in distribution patterns for the two subpopulations, the bounding box containing all the neuronal coordinates was divided into smaller cubes and two-sided Student’s t-test was calculated for each cube using a Python script (V.2.7.12). Due to the variance in the total neuronal counts between the Lepr and Glp1r groups, the number of neurons in each cube was corrected by introducing a density factor (number of Glp1r neurons/number of Lepr neurons). This was performed by multiplying the density factor to the total number of neurons in each group. Since the null distribution is two-sided, both null hypotheses were visualized within the same plot.
RNA-sequencing analysis workflow
The RNA-seq results of Glp1r and Lepr samples were processed using the nf-core/rnaseq pipeline (V.1.4.2)60. This involves (1) aligning the raw reads to the reference genome GRCm38.p6 using STAR (2.6.1d)61 and (2) transcript abundance estimation using Salmon (0.14.1)62 with the reference transcriptome from Ensembl (release 97)63. To normalize the ribosomal pulldown (IP) to the hypothalamic background (input) per sample, we calculated a ratio of the Salmon gene counts (IP/input). The differentially expressed genes between Glp1r and Lepr groups were derived using DESeq2 (V.1.26.0)64 where P values attained by the Wald test were corrected for multiple testing using the Benjamini–Hochberg method. This yielded genes that are regulated between the Glp1r and Lepr conditions and act as markers for these POMC populations.
Electrophysiological data analysis and statistics
Data analysis was performed with Spike2 (V.7.0; Cambridge Electronic Design), Igor Pro 6 (Wavemetrics) and GraphPad Prism (V.5.0-8.0). In violin plots, the solid line represents the median, and dashed lines represent lower and upper quartiles. In the box plots, the ‘+’ sign depicts the mean, and the horizontal line represents the median. The whiskers were calculated according to Tukey’s method. For pairwise comparisons of dependent and independent normal distributions, paired and unpaired t-tests were used, respectively. For pairwise comparisons of independent, non-normal distributions, a Mann–Whitney U-test was used. For multiple comparisons, one-way ANOVA with post hoc Tukey’s test was performed. A significance level of 0.05 was accepted for all tests. In the figures, n values are given in parentheses. Exact P values are reported if P > 0.05.
Reporting Summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
Raw RNA-seq data have been deposited in the NCBI Gene Expression Omnibus under accession code GSE153753. Other raw data is available under reasonable request to the corresponding author.
Code availability
The code for RNA-seq analysis is available at https://github.com/bruening-lab/pomc-neurons-architecture-rnaseq/ and the code for isosurface density plots and the 3D statistical analysis is available at https://github.com/bruening-lab/Heterogeneity_Scripts/. Source data are provided with this paper.
References
Gautron, L., Elmquist, J. K. & Williams, K. W. Neural control of energy balance: translating circuits to therapies. Cell 161, 133–145 (2015).
Cowley, M. A. et al. Leptin activates anorexigenic POMC neurons through a neural network in the arcuate nucleus. Nature 411, 480–484 (2001).
Boston, B. A., Blaydon, K. M., Varnerin, J. & Cone, R. D. Independent and additive effects of central POMC and leptin pathways on murine obesity. Science 278, 1641–1644 (1997).
Balthasar, N. et al. Leptin receptor signaling in POMC neurons is required for normal body weight homeostasis. Neuron 42, 983–991 (2004).
Barsh, G. et al. Neuroendocrine regulation by the Agouti/Agrp-melanocortin system. Endocr. Res. 26, 571 (2000).
Konner, A. C. et al. Insulin action in AgRP-expressing neurons is required for suppression of hepatic glucose production. Cell Metab. 5, 438–449 (2007).
Steculorum, S. M. et al. AgRP neurons control systemic insulin sensitivity via myostatin expression in brown adipose tissue. Cell 165, 125–138 (2016).
Ruud, J., Steculorum, S. M. & Bruning, J. C. Neuronal control of peripheral insulin sensitivity and glucose metabolism. Nat. Commun. 8, 15259 (2017).
Williams, K. W. et al. Segregation of acute leptin and insulin effects in distinct populations of arcuate pro-opiomelanocortin neurons. J. Neurosci. 30, 2472–2479 (2010).
Meister, B. Neurotransmitters in key neurons of the hypothalamus that regulate feeding behavior and body weight. Physiol. Behav. 92, 263–271 (2007).
Campbell, J. N. et al. A molecular census of arcuate hypothalamus and median eminence cell types. Nat. Neurosci. 20, 484–496 (2017).
Lam, B. Y. H. et al. Heterogeneity of hypothalamic pro-opiomelanocortin-expressing neurons revealed by single-cell RNA sequencing. Mol. Metab. 6, 383–392 (2017).
Sternson, S. M., Atasoy, D., Betley, J. N., Henry, F. E. & Xu, S. An emerging technology framework for the neurobiology of appetite. Cell. Metab. 23, 234–253 (2016).
Alexander, G. M. et al. Remote control of neuronal activity in transgenic mice expressing evolved G-protein-coupled receptors. Neuron 63, 27–39 (2009).
Boyden, E. S., Zhang, F., Bamberg, E., Nagel, G. & Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 8, 1263–1268 (2005).
Madisen, L. et al. Transgenic mice for intersectional targeting of neural sensors and effectors with high specificity and performance. Neuron 85, 942–958 (2015).
Anastassiadis, K. et al. Dre recombinase, like Cre, is a highly efficient site-specific recombinase in E. coli, mammalian cells and mice. Dis. Model Mech. 2, 508–515 (2009).
Padilla, S. L., Carmody, J. S. & Zeltser, L. M. Pomc-expressing progenitors give rise to antagonistic neuronal populations in hypothalamic feeding circuits. Nat. Med. 16, 403–405 (2010).
Secher, A. et al. The arcuate nucleus mediates GLP-1 receptor agonist liraglutide-dependent weight loss. J. Clin. Invest. 124, 4473–4488 (2014).
Lohr, H. et al. Diet-induced growth is regulated via acquired leptin resistance and engages a Pomc–somatostatin–growth hormone circuit. Cell Rep. 23, 1728–1741 (2018).
Leshan, R. L., Bjornholm, M., Munzberg, H. & Myers, M. G. Jr. Leptin receptor signaling and action in the central nervous system. Obesity (Silver Spring) 5, 208S–212S (2006).
Pan, C. et al. Shrinkage-mediated imaging of entire organs and organisms using uDISCO. Nat. Methods 13, 859–867 (2016).
Cizek, J. et al. Fast and robust registration of PET and MR images of human brain. Neuroimage 22, 434–442 (2004).
Butler, A., Hoffman, P., Smibert, P., Papalexi, E. & Satija, R. Integrating single-cell transcriptomic data across different conditions, technologies and species. Nat. Biotechnol. 36, 411–420 (2018).
Paeger, L. et al. Energy imbalance alters Ca2+ handling and excitability of POMC neurons. Elife 6, e25641 (2017).
Pineda, J. C., Galarraga, E., Bargas, J., Cristancho, M. & Aceves, J. Charybdotoxin and apamin sensitivity of the calcium-dependent repolarization and the afterhyperpolarization in neostriatal neurons. J Neurophysiol. 68, 287–294 (1992).
Buraei, Z. & Yang, J. The ss subunit of voltage-gated Ca2+ channels. Physiol. Rev. 90, 1461–1506 (2010).
Brenner, R. et al. BK channel β4 subunit reduces dentate gyrus excitability and protects against temporal lobe seizures. Nat. Neurosci. 8, 1752–1759 (2005).
Otto, J. F., Yang, Y., Frankel, W. N., White, H. S. & Wilcox, K. S. A spontaneous mutation involving Kcnq2 (Kv7.2) reduces M-current density and spike frequency adaptation in mouse CA1 neurons. J. Neurosci. 26, 2053–2059 (2006).
Richards, K. S., Bommert, K., Szabo, G. & Miles, R. Differential expression of Na+/K+-ATPase alpha-subunits in mouse hippocampal interneurones and pyramidal cells. J. Physiol. 585, 491–505 (2007).
He, Z. et al. Direct and indirect effects of liraglutide on hypothalamic POMC and NPY/AgRP neurons—implications for energy balance and glucose control. Mol. Metab. 28, 120–134 (2019).
Kavalali, E. T. The mechanisms and functions of spontaneous neurotransmitter release. Nat. Rev. Neurosci. 16, 5–16 (2015).
Buchholz, F., Angrand, P. O. & Stewart, A. F. Improved properties of FLP recombinase evolved by cycling mutagenesis. Nat. Biotechnol. 16, 657–662 (1998).
Poulin, J. F. et al. Mapping projections of molecularly defined dopamine neuron subtypes using intersectional genetic approaches. Nat. Neurosci. 21, 1260–1271 (2018).
Caron, A. et al. POMC neurons expressing leptin receptors coordinate metabolic responses to fasting via suppression of leptin levels. Elife 7, e33710 (2018).
Yaswen, L., Diehl, N., Brennan, M. B. & Hochgeschwender, U. Obesity in the mouse model of pro-opiomelanocortin deficiency responds to peripheral melanocortin. Nat. Med. 5, 1066–1070 (1999).
Krude, H. et al. Severe early-onset obesity, adrenal insufficiency and red hair pigmentation caused by POMC mutations in humans. Nat. Genet. 19, 155–157 (1998).
Lam, D. D. et al. Conditional expression of Pomc in the Lepr-positive subpopulation of POMC neurons is sufficient for normal energy homeostasis and metabolism. Endocrinology 156, 1292–1302 (2015).
Brandt, C. et al. Food perception primes hepatic ER homeostasis via melanocortin-dependent control of mTOR activation. Cell 175, 1321–1335 (2018).
Aponte, Y., Atasoy, D. & Sternson, S. M. AGRP neurons are sufficient to orchestrate feeding behavior rapidly and without training. Nat. Neurosci. 14, 351–355 (2011).
Wang, C. et al. TAp63 contributes to sexual dimorphism in POMC neuron functions and energy homeostasis. Nat. Commun. 9, 1544 (2018).
Peters, J. H., McDougall, S. J., Fawley, J. A., Smith, S. M. & Andresen, M. C. Primary afferent activation of thermosensitive TRPV1 triggers asynchronous glutamate release at central neurons. Neuron 65, 657–669 (2010).
Qiu, J., Fang, Y., Ronnekleiv, O. K. & Kelly, M. J. Leptin excites pro-opiomelanocortin neurons via activation of TRPC channels. J. Neurosci. 30, 1560–1565 (2010).
Richards, P. et al. Identification and characterization of GLP-1 receptor-expressing cells using a new transgenic mouse model. Diabetes 63, 1224–1233 (2014).
Belgardt, B. F., Okamura, T. & Bruning, J. C. Hormone and glucose signalling in POMC and AgRP neurons. J. Physiol. 587, 5305–5314 (2009).
Lippert, R. N. et al. Time-dependent assessment of stimulus-evoked regional dopamine release. Nat. Commun. 10, 336 (2019).
Heiman, M. et al. A translational profiling approach for the molecular characterization of CNS cell types. Cell 135, 738–748 (2008).
Ashburner, M. et al. Gene Ontology: tool for the unification of biology. Nat. Genet. 25, 25–29 (2000).
Yu, G., Wang, L. G., Han, Y. & He, Q. Y. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16, 284–287 (2012).
Ye, J. H., Zhang, J., Xiao, C. & Kong, J. Q. Patch-clamp studies in the CNS illustrate a simple new method for obtaining viable neurons in rat brain slices: glycerol replacement of NaCl protects CNS neurons. J. Neurosci. Methods 158, 251–259 (2006).
Dodt, H. U. & Zieglgansberger, W. Visualizing unstained neurons in living brain slices by infrared DIC-videomicroscopy. Brain Res. 537, 333–336 (1990).
Horn, R. & Marty, A. Muscarinic activation of ionic currents measured by a new whole-cell recording method. J. Gen. Physiol. 92, 145–159 (1988).
Akaike, A. Glutamate neurotoxicity and neuroprotective factors. Nihon Yakurigaku Zasshi 103, 193–201 (1994).
Rae, J., Cooper, K., Gates, P. & Watsky, M. Low access resistance perforated patch recordings using amphotericin B. J. Neurosci. Methods 37, 15–26 (1991).
Kyrozis, A. & Reichling, D. B. Perforated-patch recording with gramicidin avoids artifactual changes in intracellular chloride concentration. J. Neurosci. Methods 57, 27–35 (1995).
Pennock, R. L. & Hentges, S. T. Differential expression and sensitivity of presynaptic and postsynaptic opioid receptors regulating hypothalamic proopiomelanocortin neurons. J. Neurosci. 31, 281–288 (2011).
Wang, D. et al. Functional divergence of delta and mu opioid receptor organization in CNS pain circuits. Neuron 98, 90–108 (2018).
Paeger, L. et al. Antagonistic modulation of NPY/AgRP and POMC neurons in the arcuate nucleus by noradrenalin. Elife 6, e25770 (2017).
Vogt, M. C. et al. Neonatal insulin action impairs hypothalamic neurocircuit formation in response to maternal high-fat feeding. Cell 156, 495–509 (2014).
Ewels, P. A. et al. The nf-core framework for community-curated bioinformatics pipelines. Nat. Biotechnol. 38, 276–278 (2020).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Patro, R., Duggal, G., Love, M. I., Irizarry, R. A. & Kingsford, C. Salmon provides fast and bias-aware quantification of transcript expression. Nat. Methods 14, 417–419 (2017).
Yates, A. et al. The Ensembl REST API: ensembl data for any language. Bioinformatics 31, 143–145 (2015).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Acknowledgements
We thank C. Lovan, P. Scholl, N. Spenrath, C. Heilinger and J. Alber for excellent technical assistance. U. Lichtenberg, K. Schöfisch, H. Brönneke and R. Braun always provided skillful administrative help. N.B. and J.R. gratefully acknowledge financial doctoral support from the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation; 233886668/GRK1960), and I.G. also received support from the DFG (360043781). P. Kloppenburg was supported by DFG grant KL 762/7-1. This work was supported by a European Research Council (ERC) grant awarded to J.C.B. (SYNEME: 742106).
Author information
Authors and Affiliations
Contributions
Conceptualization: J.C.B. and P. Kloppenburg; methodology: N.B., P. Kloppenburg, L.P. and J.C.B.; investigation: N.B., I.G., J.R., J.S., L.P., S.C., W.C., C.W., T.K. and T.S.-H.; formal analysis, N.B., I.G., P. Klemm, J.R., J.S., L.P., M.S. and A.A.; visualization: I.G., N.B., J.R. and L.P.; writing of original draft: J.C.B.; project administration: N.B., J.S., I.G. and J.C.B.; resources: S.V., F.T.W., P. Kloppenburg and J.C.B.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Neuroscience thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 The POMCDre transgene shows stable integration into the genome and causes no metabolic phenotype as compared to wildtype controls.
a, Schematic diagram of the targeting strategy for the POMCDre BAC construct. b, RNA in situ hybridization against ZsGreen in POMCDre ROSA26rSrZsGreen mice. Scale bars represent 100 μm. ap = area postrema, NTS = nucleus tractus solitarius, DMX = dorsal motor nucleus. c, Quantification of copy number PCR of Pomc exon 1, performed on genomic DNA from the first four generations (f1-f4) of the POMCDre mouse line in comparison to wildtype (wt) animals. For wt, f1, f2, f3, f4; n = 4, 4, 3, 2, 3, respectively. For wt vs. f1 p = 0.0004, wt vs. f2 p = 0.0003, wt vs. f3 p = 0.0016, wt vs. f4 p = 0.0031. d, Quantification of ZsGreen-positive POMC neurons in the ARC of POMCDre ROSA26rSrZsGreen mice at indicated time points. For age groups 5, 10, 15, 20, 25 and 31 weeks; n = 3, 3, 6, 4, 3, 3, respectively. p = 0.0002. e-f, Body weight curve of POMCDre males (e, Control NCD n = 19, Control HFD n = 23, POMCDre NCD n = 15, POMCDre HFD n = 4) and females (f, Control NCD n = 12, Control HFD n = 8, POMCDre NCD n = 19, POMCDre HFD n = 7) versus control littermates on NCD and HFD. g-n, Glucose tolerance test (g, h), body composition (i, j), food intake (k, l), and energy expenditure (m, n) of POMCDre males and females on NCD. g, Control n = 20, POMCDre n = 16, h, Control n = 12, POMCDre n = 19, i, Control n = 17, POMCDre n = 14, j, Control n = 12, POMCDre n = 19, k, Control n = 15, POMCDre n = 16, l, Control n = 11, POMCDre n = 19, m, Control n = 17, POMCDre n = 14, n, Control n = 12, POMCDre n = 19. n indicates the number of mice. Data are represented as mean ± SEM. p-values were calculated by one-way-ANOVA (d) followed by Dunnett’s (c), two-way-ANOVA (g, h, m, n) or unpaired two-tailed Student’s t-test followed by Holm-Sidak correction for multiple comparisons (i, j, k, l). *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001. Further statistical details are given in Source data extended Fig. 1.
Extended Data Fig. 2 Schematic diagram of the B9-36 targeting, breeding scheme for experimental mice, whole brain images displaying ZsGreen expression and the comparison of coronal sections in 2D vs 3D.
a, Schematic diagram of the B9-36 targeting and Southern blot strategy for transgene expression dependent on intersectional Cre- and Dre-recombinase activity. Filled triangles: loxP sites; Filled diamonds: rox sites; SAH: short arm of homology; LAH: long arm of homology; WSS: Westphal Stop Sequence; NEO: neomycin resistance gene; CAG: chicken-β-actin promotor; WPRE: woodchuck hepatitis post-transcriptional regulatory element; 2 A: self-cleaving T2A peptide sequence. b, Breeding scheme for obtaining experimental mice, depicting the triple transgenic mice and their littermate controls. c, Left to right: Whole brain imaging of PomcDre+ LeprCre− ROSA26rSrlSlZSGreen+/−, PomcDre− GLP1RCre+ ROSA26rSrlSlZSGreen+/−, PomcDre+ LeprCre+ ROSA26rSrlSlZSGreen+/− 15-week-old mice, respectively. ZsGreen signal specific to the POMClepr+ in the ARC is displayed in the boxed area within the last image on the right. No ectopic expression was observed in only-Cre+ or only-Dre+ mice as depicted in the images on the left. Whole-brain scans were acquired using the LSFM with total magnification of 1.6X. d, Bilateral, coronal view of the distribution pattern of POMCLepr+ and POMCGlp1r+ neurons in POMCDre LeprCre ROSA26lSlrSrZsGreen and POMCDre Glp1rCre ROSA26lSlrSrZsGreen mice. Coronal cross-sections were generated by extracting the 3D coordinates of transgenically labelled POMCLepr+ and POMCGlp1r+ neurons in the given section. Data was merged from n = 3 mice/group. e, Unilateral, coronal view of the distribution pattern of POMCLepr+ and POMCGlp1r+ neurons labelled via RNA in situ hybridization. Data was merged from n = 3 mice/group.
Extended Data Fig. 3 Cre/Dre-dependent tdTomato transgenic lines used for projection analysis of POMCLepr+ and POMCGlp1r+ neurons in 2D sections.
a, Illustration of experimental mice and schematic diagram showing Dre- and Cre-dependent targeted expression of tdTomato in either POMCLepr+ or POMCGlp1r+ neurons. Excision of loxP and rox-flanked stop cassettes through recombination of both Cre and Dre drivers leads to tdTomato expression in the targeted subpopulation. b, Representative microscopic images of immunofluorescent staining against tdTomato and POMC in 12-week-old POMCDre LeprCre ROSA26rSrlSltdTomato and POMCDre Glp1rCre ROSA26rSrlSltdTomato in the anterior bed nucleus of the stria terminalis (BNST), the periaqueductal gray (PAG), the dorsomedial hypothalamic area (DMH) and the paraventricular nucleus of the hypothalamus (PVH). Scale bar represents 150 μm. 3 V = third ventricle, ac = anterior commissure, AQ = cerebral aqueduct. c-d, Quantification of POMC (c) and tdTomato fiber density (d) assessed as raw integrated density of immunofluorescent staining depicted in (b). c, n = 5 mice and a minimum of 3, 3, 2, 1section(s) per mouse were analyzed for the BNST, PVH, DMH and PAG. d, n = 5 mice and a minimum of 3,3,2,1 sections per mouse were analyzed for the BNST, PVH, DMH and PAG. In d and e, data are represented as mean ± SEM. *p ≤ 0.05. Further statistical details are given in Source data extended Fig. 2
Extended Data Fig. 4 Visualized 3D projection densities of POMCGlp1r+ neurons.
Representative images of 3D-rendered tdTomato fiber density in POMCDre Glp1rCre ROSA26lSlrSrtdTomato mice in the paraventricular nucleus of the hypothalamus (PVH), the periaqueductal gray (PAG), the dorsomedial hypothalamic area (DMH), the anterior bed nucleus of the stria terminalis (BNST) and the nucleus tractus solitarius (NTS).
Extended Data Fig. 5 Cre/Dre-dependent hM3Dq transgenic lines allow for specific neuronal activation solely in triple transgenic mice.
a-b, Southern blots of R26lSlrSrhM3Dq mouse line with the ROSA26 (a) and NEO probe (b). The asterisk indicates the clone chosen for blastocyst injection. (c) Representative microscopic images of RNA in situ hybridization against Pomc, Glp1r, ZsGreen (in lieu of hM3Dq) and Fos in POMCDre Glp1rCre ROSA26lSlrSrhM3Dq mice injected with saline (top) or CNO (bottom). Images on the left show ISH in the ARC with nuclear counterstain (blue, DAPI). Magnifications of the dashed boxes are displayed on the right showing the indicated stainings. Pomc-positive neurons have been traced with a white outline. Scale bars represent 50 μm in the merged image and 25 μm in the magnifications. 3 V = third ventricle. d-e, Percentage of Pomc-positive cells expressing Fos in the different CNO-injected genotype controls (d) and triple transgenic animals injected with saline or CNO (e) of POMCDre LeprCre ROSA26lSlrSrhM3Dq and POMCDre Glp1rCre ROSA26lSlrSrhM3Dq mice. d, n = 4 mice, e, n = 5 and 6 mice for groups with and without CNO, respectively. f-g, Percentage of Lepr+ or Glp1r+ cells in (f) POMCDre LeprCre ROSA26lSlrSrhM3Dq and (g) POMCDre Glp1rCre ROSA26lSlrSrhM3Dq male mice as quantified from RNA in situ hybridization. n = 3 mice. h-j, Percentage of ZsGreen-Pomc-positive cells expressing Lepr or Glp1r (h), percentage of Lepr/Glp1r-Pomc-positive cells expressing ZsGreen (i) and percentage of ZsGreen-Pomc-positive cells expressing Fos (j) in POMCDre LeprCre ROSA26lSlrSrhM3Dq or POMCDre Glp1rCre ROSA26lSlrSrhM3Dq females (22–26 weeks old) injected with saline or CNO. j; POMCLepr+, saline n = 3, CNO n = 4, saline vs CNO p < 0.000001, POMCGlp1r+, saline n = 3, CNO n = 3, saline vs CNO p = 0.000002. n indicates number of mice. Data are represented as mean ± SEM. CNO = 3 mg/kg. Statistical analyses on (b) were assessed by two-way-ANOVA. All other statistical analyses were performed by unpaired two-tailed Student’s t-test with (h-j) or without (e-g) Holm-Sidak correction for multiple comparisons. ***p ≤ 0.001, ****p ≤ 0.0001. Further statistical details are given in Source data extended Fig. 3
Extended Data Fig. 6 Metabolic phenotyping of Cre/Dre-dependent ROSA26lSlrSrhM3Dq in males and food intake measurements in female mice.
a-b, Cumulative food intake over a time course of 24 hours in all saline-injected genotype controls (a) and pooled saline- or CNO-injected control animals vs. saline-injected POMCDre LeprCre ROSA26lSlrSrhM3Dq and POMCDre Glp1rCre ROSA26lSlrSrhM3Dq male mice (b) starting with the night cycle. Mice were injected with saline at 18:00 and 23:00, followed by one-day gap and subsequent CNO injections at 18:00 and 23:00 on the next day. Data of all genotype controls in (a) is shown as pooled saline-injected Control in (b), data of saline-injected triple transgenic animals (a) are shown in Fig. 4f and g as control group. (a) Cre-/Dre-, Cre-/Dre+, LeprCre+/Dre-, Glp1rCre+/Dre-, n = 8, 9, 4 and 5 mice respectively. (b) Control Saline n = 25 and Control CNO n = 26 mice. c-h, Energy expenditure (c,d), RER (e,f) and locomotion (g,h) in POMCDre LeprCre ROSA26lSlrSrhM3Dq (c,e,g) and POMCDre Glp1rCre ROSA26lSlrSrhM3Dq male mice (d,f,h) vs. controls at 12–14 weeks of age. Mice were injected with saline at 17:00, 22:00 and 07:00 followed by CNO at 17:00, 22:00 and 07:00 on the next day. Scatter plots on the right show the average values during night and day as averaged from left graph. Values of control animals were pooled from all corresponding genotype controls. Sal=Saline. c and e, Control n = 29, POMCDre LeprCre n = 13. d and f, Control n = 38, POMCDre Glp1rCre n = 12. g, Control n = 31, POMCDre LeprCre n = 14. h Control n = 41, POMCDre Glp1rCre n = 12. Eright; Night: ControlSaline vs POMCLepr+CNO, p = 0.016, Day: ControlSaline vs POMCLepr+CNO, p = 0.0014, POMCLepr+Saline vs POMCLepr+CNO, p = 0.0065, Day: ControlSaline vs POMCLepr+CNO, p = 0.001. Fright; Night: ControlSaline vs POMCGlp1r+CNO, p = 0.001, ControlCNO vs POMCGlp1r+CNO, p = 0.0049, Day: ControlSaline vs POMCGlp1r+CNO, p = 0.035, POMCGlp1r+Saline vs POMCGlp1r+CNO, p = 0.028. i-j, Food intake over a time course of 24 hours in POMCDre LeprCre ROSA26lSlrSrhM3Dq (i) and POMCDre Glp1rCre ROSA26lSlrSrhM3Dq female mice (j) starting with the night cycle. Mice were injected with saline at 18:00 and 23:00, followed by one day gap and subsequent CNO injections at 18:00 and 23:00 on the next day. Left: Cumulative food intake in mice injected with saline vs. CNO. Right: Total food intake during night and day. n = 9 mice/group (i), n = 10 mice/group (j). CNO = 3 mg/kg. Data are represented as mean ± SEM. All p-values were calculated by two-way-ANOVA. For cumulative food intake (i-j, left) two-way-ANOVA was used, for total food intake (i-j, right) two-way-ANOVA followed by Sidak’s post-hoc test. In c-h significance was calculated exclusively on scatter plots and ANOVA was followed by Tukey’s post-hoc test, where relevant. *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001. Further statistical details are given in Source data extended Fig. 4
Extended Data Fig. 7 Vgat and Vglut2 expression in POMCLepr+ and POMCGlp1r+ neurons.
a, Representative microscopic images of RNA in situ hybridization against ZsGreen and Vgat or Vglut2 in POMCDre LeprCre ROSA26lSlrSrhM3Dq (left) and POMCDre Glp1rCre ROSA26lSlrSrhM3Dq (right) male mice. Scale bars represent 100 μm in the merged image and 25 μm in the magnifications. b, Quantification of the percentage overlap of Vgat or Vglut2 with ZsGreen-expressing cells. For POMCLepr+ and POMCGlp1r+ n = 4 and 5 mice, respectively. Data are represented as mean ± SEM. Statistical analyses on B were performed by two-way ANOVA with Sidak’s post-hoc test. **p ≤ 0.01. Further statistical details are given in Source data extended Fig. 5.
Extended Data Fig. 8 ZsGreen expression in pituitary of POMCDre LeprCre ROSA26lSlrSrZsGreen and POMCDre Glp1rCre ROSA26lSlrSrZsGreen mice.
a,b, Representative microscopic images of RNA in situ hybridization against ZsGreen in pituitary of POMCDre LeprCre ROSA26lSlrSrZsGreen (a) and POMCDre Glp1rCre ROSA26lSlrSrZsGreen (b) mice, respectively. Scale bars represent 150 μm in the merged image and 50 μm in the magnifications. c,d, Corticosterone levels in sera of CNO treated POMCDre LeprCre ROSA26lSlrSrhM3Dq (c) and POMCDre Glp1rCre ROSA26lSlrSrhM3Dq (d) male and female mice. Mice were fasted for 2 hours and injected with CNO 1 hour into the fast prior to serum collection. CNO = 3 mg/kg. C males; Controls n = 7, Cre+Dre+ n = 11, C females; Controls n = 8, Cre+Dre+ n = 8. D males; Controls n = 8, Cre+Dre+ n = 7, D females; Controls n = 10, Cre+Dre+ n = 11. Data are represented as mean ± SEM. Statistical analyses on (c-d) were performed by unpaired two-tailed Student’s t test with Holm-Sidak correction for multiple comparisons. Further statistical details are given in Source data extended Fig. 6
Extended Data Fig. 9 Validation of the Dre-Cre dependent ROSA26lSlrSrEGFPL10a transgenic line.
a-b, Southern blots of ROSA26lSlrSrEGFPL10a mouse line with the ROSA26 (a) and Neo probe (b). The asterisk indicates the clone chosen for blastocyst injection. c, Representative microscopic images of immunofluorescent staining against POMC and EGFP in the ARC of all resulting genotypes at 12 weeks of age. Scale bar represents 50 μm. 3 V = third ventricle. d, Principal component analysis of all RNA input and IP samples. e-g, GO term representations of the differentially enriched genes between the POMCLepr+ and POMCGlp1r+ clusters mapping the percentage of regulated genes to the total GO term gene count against significant gene count and adjusted p-value per term. h, UMAP plot of 773 POMC neurons identified in normal chow diet fed mice. Cells that are closer in position in the plot have a similar genetic profile. In (e-g) p-values were calculated using the DESeq2 1.26.0 pipeline with adjustments for multiple comparisons.
Extended Data Fig. 10 Spontaneous firing frequency, cell capacitance and significantly differentially enriched genes encoding ion channels and sub-units potentially contributing to different electrophysiological profiles of POMCLepr+ and POMCGlp1r+ neurons.
Violin plots illustrating spontaneous firing frequencies (a), whole-cell capacitance (b), threshold currents for the generation of action potentials (c), and the repolarisation rate of action potentials (d) of POMCLepr+ and POMCGlp1r+ neurons. e, Genes encoding sodium-potassium-ATPases. f, Genes encoding voltage-gated Na+ channels and subunits modulating voltage-gated Na+ channels. g, Genes encoding voltage-gated K+ channels and subunits modulating voltage-gated K+ channels. h, Gene Hcn1 hyperpolarization-activated cyclic-nucleotide-gated cation channels 1. i, Gene Cacnb4 encoding a subunit modulating L-Type voltage-gated Ca2+ channels. j, Gene Kcnq5 and Kcnmb4 encoding a M-current K+ channel and the beta-subunit 4 modulating large conductance Ca2+ activated K+ channels, respectively. Data are represented as mean ± SEM. (a-d) p-values were calculated using the unpaired Mann-Whitney-U test. e-j, Selected from differentially ribotag-enriched transcripts analyzed in Fig. 5b (p ≤ 0.05). For POMCDre LeprCre ROSA26lSlrSrEGFPL10a n = 4 hypothalami pooled from N = 24 mice, for POMCDre Glp1rCre ROSA26lSlrSrEGFPL10a n = 3 hypothalami pooled from N = 36 mice. p-values were calculated by DESeq2 1.26.0. Statistical details are given in Source data extended Fig. 7
Supplementary information
Source data
Source Data Extended Data Fig. 1
Statistical details.
Source Data Extended Data Fig. 3
Statistical details.
Source Data Extended Data Fig. 5
Statistical details.
Source Data Extended Data Fig. 6
Statistical details.
Source Data Extended Data Fig. 7
Statistical details.
Source Data Extended Data Fig. 8
Statistical details.
Source Data Extended Data Fig. 10
Statistical details.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Biglari, N., Gaziano, I., Schumacher, J. et al. Functionally distinct POMC-expressing neuron subpopulations in hypothalamus revealed by intersectional targeting. Nat Neurosci 24, 913–929 (2021). https://doi.org/10.1038/s41593-021-00854-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-021-00854-0
This article is cited by
-
Hypothalamic POMC neuron-specific knockout of MC4R affects insulin sensitivity by regulating Kir2.1
Molecular Medicine (2024)
-
Platelet-derived growth factor signaling in pericytes promotes hypothalamic inflammation and obesity
Molecular Medicine (2024)
-
Reciprocal activity of AgRP and POMC neurons governs coordinated control of feeding and metabolism
Nature Metabolism (2024)
-
Interleukin-17 as a key player in neuroimmunometabolism
Nature Metabolism (2023)
-
Mitochondria-derived peptide SHLP2 regulates energy homeostasis through the activation of hypothalamic neurons
Nature Communications (2023)