Abstract
Axon degeneration is a hallmark of many neurodegenerative disorders. The current assumption is that the decision of injured axons to degenerate is cell-autonomously regulated. Here we show that Schwann cells (SCs), the glia of the peripheral nervous system, protect injured axons by virtue of a dramatic glycolytic upregulation that arises in SCs as an inherent adaptation to axon injury. This glycolytic response, paired with enhanced axon–glia metabolic coupling, supports the survival of axons. The glycolytic shift in SCs is largely driven by the metabolic signaling hub, mammalian target of rapamycin complex 1, and the downstream transcription factors hypoxia-inducible factor 1-alpha and c-Myc, which together promote glycolytic gene expression. The manipulation of glial glycolytic activity through this pathway enabled us to accelerate or delay the degeneration of perturbed axons in acute and subacute rodent axon degeneration models. Thus, we demonstrate a non-cell-autonomous metabolic mechanism that controls the fate of injured axons.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Source data are provided with this paper. Additional source data underlying Figs. 1–8 and the results presented in the extended data and supplementary figures are available from the corresponding author upon reasonable request.
Code availability
PhosphoSitePlus v.6.5.9.2 powered by Cell Signaling Technology is available at https://www.phosphosite.org.
Change history
16 February 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41593-020-00731-2
References
Coleman, M. P. & Höke, A. Programmed axon degeneration: from mouse to mechanism to medicine. Nat. Rev. Neurosci. 21, 183–196 (2020).
Figley, M. D. & DiAntonio, A. The SARM1 axon degeneration pathway: control of the NAD+ metabolome regulates axon survival in health and disease. Curr. Opin. Neurobiol. 63, 59–66 (2020).
Krauss, R., Bosanac, T., Devraj, R., Engber, T. & Hughes, R. O. Axons matter: the promise of treating neurodegenerative disorders by targeting SARM1-mediated axonal degeneration. Trends Pharmacol. Sci. 41, 281–293 (2020).
Yang, J. et al. Pathological axonal death through a MAPK cascade that triggers a local energy deficit. Cell 160, 161–176 (2015).
Walker, L. et al. MAPK signaling promotes axonal degeneration by speeding the turnover of the axonal maintenance factor NMNAT2.eLife 6, e22540 (2017).
Gerdts, J., Brace, E. J., Sasaki, Y., DiAntonio, A. & Milbrandt, J. SARM1 activation triggers axon degeneration locally via NAD+ destruction. Science 348, 453–457 (2015).
Essuman, K. et al. The SARM1 Toll/interleukin-1 receptor domain possesses intrinsic NAD+ cleavage activity that promotes pathological axonal degeneration. Neuron 93, 1334–1343.e5 (2017).
Yamagishi, Y. & Tessier-Lavigne, M. An atypical SCF-like ubiquitin ligase complex promotes Wallerian degeneration through regulation of axonal Nmnat2. Cell Rep. 17, 774–782 (2016).
Babetto, E., Beirowski, B., Russler, E. V., Milbrandt, J. & DiAntonio, A. The Phr1 ubiquitin ligase promotes injury-induced axon self-destruction. Cell Rep. 3, 1422–1429 (2013).
Babetto, E. et al. Targeting NMNAT1 to axons and synapses transforms its neuroprotective potency in vivo. J. Neurosci. 30, 13291–13304 (2010).
Gilley, J. & Coleman, M. P. Endogenous Nmnat2 is an essential survival factor for maintenance of healthy axons. PLoS Biol. 8, e1000300 (2010).
Wang, J. et al. A local mechanism mediates NAD-dependent protection of axon degeneration. J. Cell Biol. 170, 349–355 (2005).
Yahata, N., Yuasa, S. & Araki, T. Nicotinamide mononucleotide adenylyltransferase expression in mitochondrial matrix delays Wallerian degeneration. J. Neurosci. 29, 6276–6284 (2009).
Guertin, A. D., Zhang, D. P., Mak, K. S., Alberta, J. A. & Kim, H. A. Microanatomy of axon/glial signaling during Wallerian degeneration. J. Neurosci. 25, 3478–3487 (2005).
Yang, D. P. et al. p38 MAPK activation promotes denervated Schwann cell phenotype and functions as a negative regulator of Schwann cell differentiation and myelination. J. Neurosci. 32, 7158–7168 (2012).
Wong, K. M., Babetto, E. & Beirowski, B. Axon degeneration: make the Schwann cell great again. Neural Regen. Res. 12, 518–524 (2017).
Arthur-Farraj, P. J. et al. c-Jun reprograms Schwann cells of injured nerves to generate a repair cell essential for regeneration. Neuron 75, 633–647 (2012).
Parrinello, S. et al. EphB signaling directs peripheral nerve regeneration through Sox2-dependent Schwann cell sorting. Cell 143, 145–155 (2010).
Vaquié, A. et al. Injured axons instruct Schwann cells to build constricting actin spheres to accelerate axonal disintegration. Cell Rep. 27, 3152–3166.e7 (2019).
Catenaccio, A. et al. Molecular analysis of axonal-intrinsic and glial-associated co-regulation of axon degeneration. Cell Death Dis. 8, e3166 (2017).
Gomez-Sanchez, J. A. et al. Schwann cell autophagy, myelinophagy, initiates myelin clearance from injured nerves. J. Cell Biol. 210, 153–168 (2015).
Beirowski, B. et al. Metabolic regulator LKB1 is crucial for Schwann cell-mediated axon maintenance. Nat. Neurosci. 17, 1351–1361 (2014).
Saab, A. S. et al. Oligodendroglial NMDA receptors regulate glucose import and axonal energy metabolism. Neuron 91, 119–132 (2016).
Lee, Y. et al. Oligodendroglia metabolically support axons and contribute to neurodegeneration. Nature 487, 443–448 (2012).
Jha, M. K. et al. Monocarboxylate transporter 1 in Schwann cells contributes to maintenance of sensory nerve myelination during aging. Glia 68, 161–177 (2020).
Babetto, E. A Schwann cell–neuron coculture system to study neuron–glia interaction during axon degeneration. Methods Mol. Biol. 2143, 97–110 (2020).
Beirowski, B. et al. The progressive nature of Wallerian degeneration in wild-type and slow Wallerian degeneration (WldS) nerves. BMC Neurosci. 6, 6 (2005).
Doherty, J. R. & Cleveland, J. L. Targeting lactate metabolism for cancer therapeutics. J. Clin. Invest. 123, 3685–3692 (2013).
Kim, J. W., Tchernyshyov, I., Semenza, G. L. & Dang, C. V. HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab. 3, 177–185 (2006).
Domènech-Estévez, E. et al. Distribution of monocarboxylate transporters in the peripheral nervous system suggests putative roles in lactate shuttling and myelination. J. Neurosci. 35, 4151–4156 (2015).
Pérez-Escuredo, J. et al. Monocarboxylate transporters in the brain and in cancer. Biochim. Biophys. Acta 1863, 2481–2497 (2016).
Beirowski, B. Measuring bioenergetic signatures of peripheral nerve segments by extracellular flux analysis. Methods Mol. Biol. 2143, 191–203 (2020).
Düvel, K. et al. Activation of a metabolic gene regulatory network downstream of mTOR complex 1. Mol. Cell 39, 171–183 (2010).
Sun, Q. et al. Mammalian target of rapamycin up-regulation of pyruvate kinase isoenzyme type M2 is critical for aerobic glycolysis and tumor growth. Proc. Natl Acad. Sci. USA 108, 4129–4134 (2011).
Cairns, R. A., Harris, I. S. & Mak, T. W. Regulation of cancer cell metabolism. Nat. Rev. Cancer 11, 85–95 (2011).
DeBerardinis, R. J., Lum, J. J., Hatzivassiliou, G. & Thompson, C. B. The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell Metab. 7, 11–20 (2008).
Dang, C. V., Kim, J.-W., Gao, P. & Yustein, J. The interplay between MYC and HIF in cancer. Nat. Rev. Cancer 8, 51–56 (2008).
Donnelly, R. P. & Finlay, D. K. Glucose, glycolysis and lymphocyte responses. Mol. Immunol. 68, 513–519 (2015).
Hardie, D. G., Ross, F. A. & Hawley, S. A. AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat. Rev. Mol. Cell Biol. 13, 251–262 (2012).
Sherman, D. L. et al. Arrest of myelination and reduced axon growth when Schwann cells lack mTOR. J. Neurosci. 32, 1817–1825 (2012).
Beirowski, B., Wong, K. M., Babetto, E. & Milbrandt, J. mTORC1 promotes proliferation of immature Schwann cells and myelin growth of differentiated Schwann cells. Proc. Natl Acad. Sci. USA 114, E4261–E4270 (2017).
Figlia, G., Camilla Norrmén, C., Pereira, J. A., Gerber, D. & Suter, U. Dual function of the PI3K-Akt-mTORC1 axis in myelination of the peripheral nervous system.eLife 6, e29241 (2017).
Ko, M. H., Chen, W. P., Lin-Shiau, S. Y. & Hsieh, S. T. Age-dependent acrylamide neurotoxicity in mice: morphology, physiology, and function. Exp. Neurol. 158, 37–46 (1999).
Keswani, S. C. et al. Nitric oxide prevents axonal degeneration by inducing HIF-1-dependent expression of erythropoietin. Proc. Natl Acad. Sci. USA 108, 4986–4990 (2011).
Norrmén, C. et al. mTORC1 is transiently reactivated in injured nerves to promote c-Jun elevation and Schwann cell dedifferentiation. J. Neurosci. 38, 4811–4828 (2018).
Stassart, R. M. et al. A role for Schwann cell-derived neuregulin-1 in remyelination. Nat. Neurosci. 16, 48–54 (2013).
Napoli, I. et al. A central role for the ERK-signaling pathway in controlling Schwann cell plasticity and peripheral nerve regeneration in vivo. Neuron 73, 729–742 (2012).
Norrmén, C. et al. mTORC1 controls PNS myelination along the mTORC1-RXRγ-SREBP-lipid biosynthesis axis in Schwann cells. Cell Rep. 9, 646–660 (2014).
Peters, O. M. et al. Loss of Sarm1 does not suppress motor neuron degeneration in the SOD1G93A mouse model of amyotrophic lateral sclerosis. Hum. Mol. Genet. 27, 3761–3771 (2018).
Vande Velde, C., Garcia, M. L., Yin, X., Trapp, B. D. & Cleveland, D. W. The neuroprotective factor WldS does not attenuate mutant SOD1-mediated motor neuron disease. Neuromolecular Med. 5, 193–203 (2004).
Mallon, B. S., Shick, H. E., Kidd, G. J. & Macklin, W. B. Proteolipid promoter activity distinguishes two populations of NG2-positive cells throughout neonatal cortical development. J. Neurosci. 22, 876–885 (2002).
Young, C. D. et al. Modulation of glucose transporter 1 (GLUT1) expression levels alters mouse mammary tumor cell growth in vitro and in vivo. PLoS ONE 6, e23205 (2011).
Viollet, B. et al. The AMP-activated protein kinase α2 catalytic subunit controls whole-body insulin sensitivity. J. Clin. Invest. 111, 91–98 (2003).
de Alboran, I. M. et al. Analysis of C-MYC function in normal cells via conditional gene-targeted mutation. Immunity 14, 45–55 (2001).
Hernandez, O., Way, S., McKenna, J. 3rd & Gambello, M. J. Generation of a conditional disruption of the Tsc2 gene. Genesis 45, 101–106 (2007).
Feltri, M. L. et al. P0-Cre transgenic mice for inactivation of adhesion molecules in Schwann cells. Ann. N. Y. Acad. Sci. 883, 116–123 (1999).
Laranjeira, C. et al. Glial cells in the mouse enteric nervous system can undergo neurogenesis in response to injury. J. Clin. Invest. 121, 3412–3424 (2011).
Sasaki, Y., Vohra, B. P., Lund, F. E. & Milbrandt, J. Nicotinamide mononucleotide adenylyl transferase-mediated axonal protection requires enzymatic activity but not increased levels of neuronal nicotinamide adenine dinucleotide. J. Neurosci. 29, 5525–5535 (2009).
Poitelon, Y. et al. Spatial mapping of juxtacrine axo-glial interactions identifies novel molecules in peripheral myelination. Nat. Commun. 6, 8303 (2015).
Vroemen, M. & Weidner, N. Purification of Schwann cells by selection of p75 low affinity nerve growth factor receptor expressing cells from adult peripheral nerve. J. Neurosci. Methods 124, 135–143 (2003).
Meyer, N. et al. Oligodendrocytes in the mouse corpus callosum maintain axonal function by delivery of glucose. Cell Rep. 22, 2383–2394 (2018).
Yin, Y. et al. Glucose oxidation is critical for CD4+ T cell activation in a mouse model of systemic lupus erythematosus. J. Immunol. 196, 80–90 (2016).
Du, J. et al. Inhibition of mitochondrial pyruvate transport by zaprinast causes massive accumulation of aspartate at the expense of glutamate in the retina. J. Biol. Chem. 288, 36129–36140 (2013).
Quanz, M. et al. Preclinical efficacy of the novel monocarboxylate transporter 1 inhibitor BAY-8002 and associated markers of resistance. Mol. Cancer Ther. 17, 2285–2296 (2018).
Benjamin, D. et al. Dual inhibition of the lactate transporters MCT1 and MCT4 is synthetic lethal with metformin due to NAD+ depletion in cancer cells. Cell Rep. 25, 3047–3058.e4 (2018).
Gamage, K. K. et al. Death receptor 6 promotes Wallerian degeneration in peripheral axons. Curr. Biol. 27, 890–896 (2017).
Shin, J. E. et al. Dual leucine zipper kinase is required for retrograde injury signaling and axonal regeneration. Neuron 74, 1015–1022 (2012).
Cho, Y. et al. Activating injury-responsive genes with hypoxia enhances axon regeneration through neuronal HIF-1α. Neuron 88, 720–734 (2015).
Miller, A. et al. Exploring metabolic configurations of single cells within complex tissue microenvironments. Cell Metab. 26, 788–800.e6 (2017).
Van Noorden, C. J. F. & Frederiks, W. M. Enzyme Histochemistry: a Laboratory Manual of Current Methods (Oxford Univ. Press, Royal Microscopical Society, 1992).
Fazal, S. V. et al. Graded elevation of c-Jun in Schwann cells in vivo: gene dosage determines effects on development, re-myelination, tumorigenesis, and hypomyelination. J. Neurosci. 37, 12297–12313 (2017).
Beirowski, B. et al. Non-nuclear WldS determines its neuroprotective efficacy for axons and synapses in vivo. J. Neurosci. 29, 653–668 (2009).
Mack, T. G. et al. Wallerian degeneration of injured axons and synapses is delayed by a Ube4b/Nmnat chimeric gene. Nat. Neurosci. 4, 1199–1206 (2001).
Conforti, L. et al. NAD+ and axon degeneration revisited: Nmnat1 cannot substitute for WldS to delay Wallerian degeneration. Cell Death Differ. 14, 116–127 (2007).
Morrison, B. M. et al. Deficiency in monocarboxylate transporter 1 (MCT1) in mice delays regeneration of peripheral nerves following sciatic nerve crush. Exp. Neurol. 263, 325–338 (2015).
Bogdanik, L. P. et al. Loss of the E3 ubiquitin ligase LRSAM1 sensitizes peripheral axons to degeneration in a mouse model of Charcot–Marie–Tooth disease. Dis. Model. Mech. 6, 780–792 (2013).
Gold, B. G., Griffin, J. W. & Price, D. L. Somatofugal axonal atrophy precedes development of axonal degeneration in acrylamide neuropathy. Arch. Toxicol. 66, 57–66 (1992).
Von Burg, R., Penney, D. P. & Conroy, P. J. Acrylamide neurotoxicity in the mouse: a behavioral, electrophysiological and morphological study. J. Appl. Toxicol. 1, 227–233 (1981).
Lehning, E. J., Persaud, A., Dyer, K. R., Jortner, B. S. & LoPachin, R. M. Biochemical and morphologic characterization of acrylamide peripheral neuropathy. Toxicol. Appl. Pharmacol. 151, 211–221 (1998).
Acknowledgements
We thank R. Loren, T. Bierschenk, the animal husbandry staff of the Laboratory Animal Shared Resources at the Roswell Park Comprehensive Cancer Center for technical assistance with mouse colony maintenance, C. Deppmann for help with microfluidic devices, L. Feltri for help with rat SC cultures, S. James for assistance with digital droplet PCR analysis, E.D. Abel for the conditional GLUT1 mice, F. Alt for the conditional c-Myc mice, M. Gambello for the conditional Tsc2 mice, V. Pachnis for the iSox10Cre mice and the Michigan Regional Comprehensive Metabolomics Resource Core for help with the metabolomics analysis. This study was supported by Muscular Dystrophy Association grant nos. 577844 (to B.B.) and 292306 (to E.B.), GBS/CIDP Foundation International grant no 81463 (to E.B.), and start-up funding for B.B. provided through Empire State Development Corporation for Hunter James Kelly Research Institute grant nos. W753 and U446 and the Hunter’s Hope Foundation. This work was also supported by a National Institutes of Health/National Cancer Institute grant no. P30CA016056.
Author information
Authors and Affiliations
Contributions
B.B. conceived and supervised the study. B.B., E.B. and K.M.W. designed and planned the experiments. E.B. performed the experiments and analyzed and interpreted the data for Figs. 1, 4b,f,g, 5a and 8c, and Extended Data Fig. 5 and Supplementary Fig. 1. B.B. and K.M.W. performed the experiments and analyzed and interpreted the data for Figs. 2, 3, 4a,c–e, 5b–j, 6, 7 and 8a,b,d–i, and Extended Data Figs. 1–4, 6–10 and Supplementary Figs. 2–5. B.B. and E.B. supervised K.M.W. B.B. and E.B. prepared the figures. B.B. and E.B. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Glycolytic and fermentative enzyme expression in SCs after nerve injury.
a–i, Representative immunofluorescence for the indicated metabolic components on longitudinal frozen sections from uninjured control nerve segments and axotomized distal sciatic nerve stumps at the shown post-injury times. HK2: Hexokinase 2 (a). GPI: Glucose-6-phosphate isomerase (b). ALDA: Aldolase A (c). GAPDH: Glyceraldehyde-3-phosphate dehydrogenase (d). PGK1: Phosphoglycerate kinase 1 (e). PGAM1: Phosphoglycerate mutase 1 (f). ENO1: Enolase 1 (g). PKM1: Pyruvate kinase M1 (h). LDHB: Lactate dehydrogenase B (i). Arrows depict colocalization in SCs. Scale bars: 50μm. The experiments for each component were reproduced three times independently with similar results.
Extended Data Fig. 2 Enzymes driving mitochondrial glucose catabolism in SCs are not upregulated upon nerve lesion.
a, b, Representative immunofluorescence using wide-field fluorescence microscopy (a) and confocal microscopy (b, merged z-projections) for the indicated mitochondrial enzymes on longitudinal frozen sections from uninjured control nerve segments and axotomized distal sciatic nerve stumps at the shown post-injury times. PDH: Pyruvate dehydrogenase complex. CS: Citrate synthase. IDH/IDH3a: Isocitrate dehydrogenase catalytic subunit α. α-KGDH/OGDH: Alpha-ketoglutarate dehydrogenase also known as 2-oxoglutarate dehydrogenase E1 component, mitochondrial. Note prominent CS, IDH, and α-KGDH axonal staining in uninjured control nerves, in addition to the SC-derived signals. The axonal staining weakens in injured preparations due to axonal degeneration. Red: respective mitochondrial enzyme. Green: PLP-EGFP. Blue: DAPI. Scale bars: 50μm (a), 100μm (b). The experiments for each mitochondrial enzyme were reproduced three times independently with similar results. c, d, Left: Representative images of longitudinal sections from uninjured control nerve segments and axotomized distal sciatic nerve stumps stained for the activity of isocitrate dehydrogenase (IDH, c) and succinate dehydrogenase (SDH, d) (formazan formation) with superimposition of DAPI signals (cyan). Scale bars: 50μm. Right: Densitometric quantification of formazan intensity representing IDH or SDH enzyme activity, respectively, on nerve sections. Note markedly reduced IDH and SDH enzyme activities following nerve injury, in contrast to increased glycolytic enzyme activities (Error bars represent s.e.m. n = 3 mice for each graph). Statistical evaluation in c and d was performed using Student’s t-test, unpaired, two-tailed.
Extended Data Fig. 3 Nerve lactate metabolism following nerve injury.
a, Model for axonal consumption of lactate released by SCs in the nerve stump distal to site of axotomy. b, Lactate concentrations in lysates of uninjured control nerve segments and distal sciatic nerve stumps following axotomy (Error bars represent s.e.m. n = 3 mice per condition for 6, 12, 24, 36, and 72 h after axotomy, n = 4 mice per condition for 48 h after axotomy, *P = 0.0097, **P = 0.0446, ***P = 0.0057). c, Left: Scheme for measuring extracellular monocarboxylate release of uninjured and injured peripheral nerve segments by extracellular flux analysis in Seahorse islet capture microplates. Right: ECAR traces of control uninjured and axotomized nerve segments at the indicated post-injury times (Error bars represent s.e.m. n = 3 mice for uninjured nerve segments and n = 4 mice for injured for 24 h after axotomy, n = 7 mice for uninjured nerve segments and n = 8 mice for injured for 48 h after axotomy, n = 9 mice for uninjured and injured nerve segments for 72 h after axotomy). d, Relative glucose injection–induced maximum ECARs in uninjured control and axotomized nerve segments reflecting extracellular concentrations of glucose-derived monocarboxylates. Note extracellular accumulation of glucose-derived monocarboxylates as axonal degeneration proceeds. (Error bars represent s.e.m. n = 3 mice for uninjured nerve segments and n = 4 mice for injured for 24 h after axotomy, n = 7 mice for uninjured nerve segments and n = 8 mice for injured for 48 h after axotomy, n = 9 mice for uninjured and injured nerve segments for 72 h after axotomy). Statistical evaluation in b and d was performed using multiple Student’s t-tests, unpaired, two-tailed.
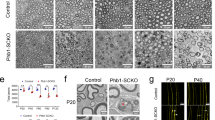
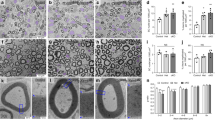
Extended Data Fig. 4 Conditional mutant mice lacking key glucose metabolism regulators in SCs show overtly normal nerve structure.
a–c, Western blot analysis (cropped blot images) of sciatic nerve lysates from indicated 8-weeks-old control and mutant mice with the indicated genotypes probed with the shown antibodies (Error bars represent s.e.m. n = 3 mice per genotype for each graph. Each dot represents measurement from sciatic nerve lysate from one mouse). d–f, Representative electron micrographs of transverse sciatic nerve sections from 8-weeks-old control and mutant mice with the indicated genotypes. Scale bars: 2μm. g–i, Quantification of myelinated axons in sciatic nerves from indicated 8-weeks-old control and mutant mice (Error bars represent s.e.m. n = 4 mice per genotype for each graph). j–l, Quantification of g ratios (left: scatter plots show g ratios of individual myelinated axons as function of axon diameter, right: corresponding cumulative g ratios per animal) in sciatic nerves from indicated 8-weeks-old control and mutant mice (Error bars represent s.e.m. n = 4 mice per genotype for j and n = 3 mice per genotype for k, l). m, Representative immunofluorescence for the indicated glycolytic regulators on longitudinal frozen sections from control uninjured nerves and axotomized distal sciatic nerve stumps of mice with the indicated genotypes 36 h after nerve transection injury (blue: DAPI). Scale bars: 50μm. The experiments for each glycolytic regulator were reproduced three times independently with similar results. Statistical evaluation in a–c and g–l was performed using Student’s t-test, unpaired, two-tailed.
Extended Data Fig. 5 Additional analysis of MCT inhibitors in co-cultures.
a, b, Representative immunofluorescence (confocal (a) and wide-field fluorescence (b)) microscopy of control uninjured axons under the indicated conditions. Note normal appearance of axon structure after 24 h of treatment with the indicated MCT inhibitors. Scale bars: 50μm. The experiments for each condition were reproduced three times independently with similar results. c, Box and whiskers plots (maximum, 25th percentile, median, 75th percentile, minimum) of axon survival 24 h following axotomy under the indicated conditions (BAY-8002: β-III-tub and NF-H, Neurons: n = 37 DRG neurite preparations, Neurons+SCs DMSO: n = 44 DRG neurite preparations, Neurons+SCs BAY-8002: n = 38 DRG neurite preparations, all DRG neurite preparations from four experimental sets performed on different days. UK-5099: β-III-tub, Neurons: n = 53 DRG neurite preparations, Neurons+SCs DMSO: n = 57 DRG neurite preparations, Neurons+SCs UK-5099: n = 38 DRG neurite preparations, all DRG neurite preparations from five experimental sets performed on different days (four experimental sets performed on different days for Neurons+SCs UK-5099). NF-H, Neurons: n = 53 DRG neurite preparations, Neurons+SCs DMSO: n = 49 DRG neurite preparations, Neurons+SCs UK-5099: n = 36 DRG neurite preparations, all DRG neurite preparations from five experimental sets performed on different days (four experimental sets performed on different days for Neurons+SCs UK-5099)). Statistical evaluation was performed using One-way-ANOVA and Sidak’s multiple comparisons tests.
Extended Data Fig. 6 Upregulation of AMPK activity in injury-activated SCs, and analysis of nerves from conditional mutant mice lacking AMPK or mTOR activity in SCs.
a, Western blot analysis (cropped blot images) of lysates from uninjured control nerve segments and axotomized distal sciatic nerve stumps from C57Bl/6 J mice showing AMPK activity at different times following nerve transection. Note marked AMPK activation as reflected by increased p-AMPKα phosphorylation at Thr172 already 10 min after nerve injury. Individual lanes represent pooled data from at least three mice. b, Representative immunofluorescence for p-AMPKα (Thr172) on longitudinal frozen section from axotomized distal sciatic nerve stump 12 h after nerve injury. Arrows depict colocalization in SCs. Scale bar: 20μm. The experiment was reproduced three times independently with similar results. c, f, Quantification of myelinated axons in sciatic nerves from indicated 8-weeks-old control and mutant mice (Error bars represent s.e.m. n = 4 mice per genotype for each graph). d, g, Quantification of g ratios (left: scatter plots show g ratios of individual myelinated axons as function of axon diameter, right: corresponding cumulative g ratios per animal) in sciatic nerves from indicated 8-weeks-old control and mutant mice (Error bars represent s.e.m. n = 4 mice per genotype for d and n = 3 mice per genotype for g). e, Quantitative analysis of relative axon survival in distal sciatic nerve stumps 36 h after axotomy in mice with the indicated genotypes (Error bars represent s.e.m. n = 4 mice per genotype). h, Quantification of SC nuclei in sciatic nerve cross sections in 8-weeks-old mice with the indicated genotypes (Error bars represent s.e.m. n = 4 mice per genotype). i, j, Representative semithin and electron micrographs (last panel with pseudocoloring) of transverse sciatic nerve sections of distal nerve stumps from mice with the indicated genotypes at different time points after sciatic nerve transection (i) with corresponding quantifications of relative axon survival (j). Electron micrographs show pseudocoloring of intact (turquoise) and degenerated (magenta) myelinated fibers. Note accelerated axonal degeneration in the mTORfl/fl; P0Cre mutants (Error bars represent s.e.m. n = 3 mice for mTORfl/fl for 0 and 36 h after axotomy, n = 4 mice for mTORfl/fl for 24 and 48 h after axotomy, n = 3 mice for mTORfl/fl; P0Cre for 0 h after axotomy, n = 4 mice for mTORfl/fl; P0Cre for 24 and 36 h after axotomy, n = 6 mice for mTORfl/fl; P0Cre for 48 h after axotomy, *P = 0.003, ** P < 0.0001). Scale bars: 10μm. Statistical evaluation in c–h was performed using Student’s t-test, unpaired, two-tailed, and in j using multiple Student’s t-tests, unpaired, two-tailed.
Extended Data Fig. 7 Analysis of nerves from mutant mice lacking key mTOR components in SCs.
a, Western blot mTORC1 activity (reflected by S6 phosphorylation at Ser240/244) analysis of sciatic nerve lysates (cropped blot images) from control and mTORfl/fl; iSox10Cre mice (30 days after last tamoxifen administration) probed with the indicated antibodies (Error bars represent s.e.m. n = 3 mice per genotype. Each dot represents measurement from sciatic nerve lysate from one mouse). b, Representative semithin micrographs of transverse sciatic nerve sections from 12-weeks-old control and mTORfl/fl; iSox10Cre mice 30 days following last tamoxifen administration. Note indistinguishable nerve structure between control and mutant mice. Scale bar: 50μm. c, Quantification of SC nuclei in sciatic nerve cross sections from mice with the indicated genotypes 30 days following tamoxifen administration (Error bars represent s.e.m. n = 5 mice for mTORfl/fl and n = 7 mice for mTORfl/fl; iSox10Cre). d, Quantification of g ratios (left: scatter plots show g ratios of individual myelinated axons as function of axon diameter, right: corresponding cumulative g ratios per animal) in sciatic nerves from mice with the indicated genotypes 30 days following tamoxifen administration (Error bars represent s.e.m. n = 3 mice per genotype). e, f, Western blot analysis (cropped blot images) of sciatic nerve lysates from indicated control and mutant mice (age 5 days in e and 8 weeks in f) probed with the shown antibodies (Error bars represent s.e.m. n = 3 mice per genotype in e and n = 4 mice per genotype in f. Each dot represents measurement from sciatic nerve lysate from one mouse). g, Quantification of myelinated axons in sciatic nerves from indicated 8-weeks-old control and mutant mice (Error bars represent s.e.m. n = 4 mice per genotype for each graph). h, Representative immunofluorescence using the indicated markers on longitudinal frozen sections of distal sciatic nerve stumps from control and Rictorfl/fl; P0Cre mutant mice 36 h after nerve transection injury. Note normal induction of mTORC1 activity in SCs of Rictor-deficient mice as reflected by indistinguishable p-S6 (Ser240/244) immunoreactivity. Scale bar: 50μm. The experiment was reproduced three times independently with similar results. Statistical evaluation in a, c–g was performed using Student’s t-test, unpaired, two-tailed.
Extended Data Fig. 8 Nerves from mutant mice with depletion of c-Myc and/or Hif1α in SCs show no abnormalities of myelinated axons.
a, Representative immunofluorescence using the indicated antibodies on longitudinal sections of axotomized sciatic nerve stumps from control and c-Mycfl/fl; iSox10Cre mutant mice (30 days after last tamoxifen administration) 36 h after nerve transection injury. Note largely abolished induction of c-Myc expression in SCs (S100 + ) of c-Mycfl/fl; iSox10Cre mice. Scale bar: 50μm. The experiment was reproduced three times independently with similar results. b, Quantification of myelinated axons in sciatic nerves from 12-weeks-old mice with the indicated genotypes 30 days following tamoxifen administration (Error bars represent s.e.m. n = 4 mice per genotype). c, Quantification of g ratios (left: scatter plots show g ratios of individual myelinated axons as function of axon diameter, right: corresponding cumulative g ratios per animal) in sciatic nerves from 12-weeks-old mice with the indicated genotypes 30 days following tamoxifen administration (Error bars represent s.e.m. n = 3 mice per genotype). d, Western blot analysis (cropped blot images) of sciatic nerve lysates from 8-weeks-old mice with the indicated genotypes probed with the shown antibodies (Error bars represent s.e.m. n = 3 mice per genotype for each graph. Each dot represents measurement from sciatic nerve lysate from one mouse). e, Representative immunofluorescence using the indicated markers on longitudinal frozen sections of axotomized sciatic nerve stumps from control and Hif1αfl/fl; P0Cre mutant mice 36 h after nerve transection injury. Note largely abolished induction of Hif1α expression in Hif1αfl/fl; P0Cre mice. Scale bar: 50μm. The experiment was reproduced three times independently with similar results. f, Representative electron micrographs of transverse sciatic nerve sections from 8-weeks-old control and Hif1αfl/fl; P0Cre mice. Note indistinguishable nerve ultrastructure between control and mutant mice. Scale bar: 2μm. g, Quantification of myelinated axons in sciatic nerves from 8-weeks-old mice with the indicated genotypes (Error bars represent s.e.m. n = 4 mice per genotype for each graph). h, Quantification of g ratios (left: scatter plots show g ratios of individual myelinated axons as function of axon diameter, right: corresponding cumulative g ratios per animal) in sciatic nerves from 8-weeks-old mice with the indicated genotypes (Error bars represent s.e.m. n = 3 mice per genotype). i, Representative semithin (left) and electron micrographs (right) of transverse sciatic nerve sections from distal nerve stumps of mice with the indicated genotypes 36 h after sciatic nerve transection with pseudocoloring of intact (turquoise) and degenerated (magenta) myelinated fibers. Scale bars: 10μm. The experiment was reproduced three times independently with similar results. j, Representative electron micrographs of transverse sciatic nerve sections from 12-weeks-old control and Hif1αfl/fl; c-Myc fl/fl; iSox10Cre mice 30 days following the last tamoxifen administration. Note indistinguishable nerve ultrastructure between control and mutant mice. Scale bar: 2μm. k, Quantification of myelinated axons in sciatic nerves from 12-weeks-old mice with the indicated genotypes 30 days following tamoxifen administration (Error bars represent s.e.m. n = 4 mice per genotype). l, Quantification of g ratios (left: scatter plots show g ratios of individual myelinated axons as function of axon diameter, right: corresponding cumulative g ratios per animal) in sciatic nerves from 12-weeks-old mice with the indicated genotypes 30 days following tamoxifen administration (Error bars represent s.e.m. n = 3 mice per genotype). Statistical evaluation in b–d, g, h, k, l was performed using Student’s t-test, unpaired, two-tailed.
Extended Data Fig. 9 Nerves from mutant mice with depletion of TSC2 in SCs show no abnormalities of myelinated axons.
a, Representative semithin (left) and electron micrographs (right) of transverse sciatic nerve sections from 12-weeks-old control and TSC2fl/fl; iSox10Cre mice 30 days following tamoxifen administration. Scale bars: 10μm. b, Quantification of myelinated axons in sciatic nerves from mice with the indicated genotypes 30 days following tamoxifen administration (Error bars represent s.e.m. n = 4 mice per genotype). c, Quantification of g ratios (left: scatter plots show g ratios of individual myelinated axons as function of axon diameter, right: corresponding cumulative g ratios per animal) in sciatic nerves from mice with the indicated genotypes 30 days following tamoxifen administration (Error bars represent s.e.m. n = 3 mice per genotype). Statistical evaluation in b and c was performed using Student’s t-test, unpaired, two-tailed.
Extended Data Fig. 10 Additional behavioral, electrophysiological, and structural analysis of ACR-treated mice.
a, Behavioral analysis of C57Bl/6 J mice over the course of ACR intoxication (or control treatment) for 14 days. Note accentuated deterioration of motor performance (rotarod, hanging wire, grip strength) relative to sensory performance (tail flick, hot and cold plate) (Error bars represent s.e.m. n = 8 mice per group and time point, except n = 7 mice for control treatment 0 days tail flick, *P = 0.0024, **P < 0.0001 for rotarod, *P < 0.0001 for hanging wire, *P = 0.0049, **P = 0.0130, ***P < 0.0001 for grip strength). b, Weight analysis of C57Bl/6 J mice over the course of ACR intoxication (or control treatment) for 14 days (Error bars represent s.e.m. n = 5 female and n = 3 male control mice, n = 4 female and male ACR-treated mice, *P = 0.0299, **P = 0.0346). c, Analysis of CMAP amplitudes recorded in gastrocnemius muscles evoked after sciatic nerve stimulation of C57Bl/6 J mice following 14d of ACR treatment, or control treatment (Error bars represent s.e.m. n = 8 mice per group). d, Representative electron micrographs of transverse tibial nerve sections from C57Bl/6 J mice following 14d of ACR admininistration (or control treatment), with pseudocoloring of intact (turquoise) and degenerated (magenta) myelinated fiber profiles. Degenerated profiles appeared as collapsed myelinated axons with little axoplasm or axons with segregation of the axoplasm by segments of the myelin sheath (d1), deranged fibers with axoplasm constriction due to myelin infoldings and convolution (d2), and myelinated fibers with accumulation of membrane like material, multivesicular structures, and dense bodies in the axoplasm (d3). Scale bars: 2μm. e, Densities of degenerated axon profiles in tibial nerves from C57Bl/6 J mice following control treatment or 14d of ACR admininistration (Error bars represent s.e.m. n = 8 mice per group). f, Representative electron micrographs of transverse tibial nerve sections from C57Bl/6 J mice following control treatment or 14d of ACR admininistration show normal ultrastructure of unmyelinated axons in Remak bundles (‘N’ depicts nuclei of SCs forming Remak bundles). Scale bar: 2μm. The experiment was reproduced three times independently with similar results. g, Weight analysis of ACR-treated control TSC2fl/fl and mutant TSC2fl/fl; iSox10Cre mice (Error bars represent s.e.m. n = 4 female and n = 7 male mice (except n = 6 male mice for 14 days treatment time point) with genotype TSC2fl/fl, n = 8 female and n = 6 male mice with genotype TSC2fl/fl; iSox10Cre). Statistical evaluation in a, b, and g was performed using multiple Student’s t-tests, unpaired, two-tailed, and in c and e using Student’s t-test, unpaired, two-tailed.
Supplementary information
Supplementary Information
Supplementary Figs. 1–5.
Supplementary Table
Supplementary Table 1. List of antibodies with validation details
Source data
Source Data Fig. 1
Statistical Source Data
Source Data Fig. 2
Statistical Source Data
Source Data Fig. 3
Statistical Source Data
Source Data Fig. 3
Unprocessed western blots
Source Data Fig. 4
Statistical Source Data
Source Data Fig. 5
Statistical Source Data
Source Data Fig. 5
Unprocessed western blots
Source Data Fig. 6
Statistical Source Data
Source Data Fig. 7
Statistical Source Data
Source Data Fig. 8
Statistical Source Data
Source Data Fig. 8
Unprocessed western blots
Source Data Extended Data Fig. 2
Statistical Source Data
Source Data Extended Data Fig. 3
Statistical Source Data
Source Data Extended Data Fig. 4
Statistical Source Data
Source Data Extended Data Fig. 4
Unprocessed western blots
Source Data Extended Data Fig. 5
Statistical Source Data
Source Data Extended Data Fig. 6
Statistical Source Data
Source Data Extended Data Fig. 6
Unprocessed western blots
Source Data Extended Data Fig. 7
Statistical Source Data
Source Data Extended Data Fig. 7
Unprocessed western blots
Source Data Extended Data Fig. 8
Statistical Source Data
Source Data Extended Data Fig. 8
Unprocessed western blots
Source Data Extended Data Fig. 9
Statistical Source Data
Source Data Extended Data Fig. 10
Statistical Source Data
Source Dataset
Supporting data for Supplementary Fig. 2
Rights and permissions
About this article
Cite this article
Babetto, E., Wong, K.M. & Beirowski, B. A glycolytic shift in Schwann cells supports injured axons. Nat Neurosci 23, 1215–1228 (2020). https://doi.org/10.1038/s41593-020-0689-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-020-0689-4
This article is cited by
-
Mechanosensitive channel of large conductance enhances the mechanical stretching-induced upregulation of glycolysis and oxidative metabolism in Schwann cells
Cell Communication and Signaling (2024)
-
TREM2 deficiency impairs the energy metabolism of Schwann cells and exacerbates peripheral neurological deficits
Cell Death & Disease (2024)
-
Intercellular communication atlas reveals Oprm1 as a neuroprotective factor for retinal ganglion cells
Nature Communications (2024)
-
Bioenergetics of Axon Integrity and Its Regulation by Oligodendrocytes and Schwann Cells
Molecular Neurobiology (2024)
-
Next-generation RNA sequencing elucidates transcriptomic signatures of pathophysiologic nerve regeneration
Scientific Reports (2023)