Abstract
Sleep is beneficial for learning. However, it remains unclear whether learning is facilitated by non-rapid eye movement (NREM) sleep or by REM sleep, whether it results from plasticity increases or stabilization, and whether facilitation results from learning-specific processing. Here, we trained volunteers on a visual task and measured the excitatory and inhibitory (E/I) balance in early visual areas during subsequent sleep as an index of plasticity. The E/I balance increased during NREM sleep irrespective of whether pre-sleep learning occurred, but it was associated with post-sleep performance gains relative to pre-sleep performance. In contrast, the E/I balance decreased during REM sleep but only after pre-sleep training, and the decrease was associated with stabilization of pre-sleep learning. These findings indicate that NREM sleep promotes plasticity, leading to performance gains independent of learning, while REM sleep decreases plasticity to stabilize learning in a learning-specific manner.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The datasets generated and/or analyzed in the current study are included in this published article as source data and Supplementary Table 14. Source data are provided with this paper.
Code availability
The computer code that was used to generate results central to the conclusions of this study is available from the corresponding author upon request.
References
Rechtschaffen, A. & Kales, A. A Manual of Standardized Terminology, Techniques, and Scoring System for Sleep Stages of Human Subjects (Public Health Service, US Government Printing Office, 1968).
Gais, S., Plihal, W., Wagner, U. & Born, J. Early sleep triggers memory for early visual discrimination skills. Nat. Neurosci. 3, 1335–1339 (2000).
Tamaki, M. et al. Enhanced spontaneous oscillations in the supplementary motor area are associated with sleep-dependent offline learning of finger-tapping motor-sequence task. J. Neurosci. 33, 13894–13902 (2013).
Tononi, G. & Cirelli, C. Sleep and the price of plasticity: from synaptic and cellular homeostasis to memory consolidation and integration. Neuron 81, 12–34 (2014).
Maquet, P. et al. Experience-dependent changes in cerebral activation during human REM sleep. Nat. Neurosci. 3, 831–836 (2000).
Karni, A., Tanne, D., Rubenstein, B. S., Askenasy, J. J. & Sagi, D. Dependence on REM sleep of overnight improvement of a perceptual skill. Science 265, 679–682 (1994).
Diekelmann, S. & Born, J. The memory function of sleep. Nat. Rev. Neurosci. 11, 114–126 (2010).
Giuditta, A. et al. The sequential hypothesis of the function of sleep. Behav. Brain Res. 69, 157–166 (1995).
Stickgold, R., James, L. & Hobson, J. A. Visual discrimination learning requires sleep after training. Nat. Neurosci. 3, 1237–1238 (2000).
Fischer, S., Hallschmid, M., Elsner, A. L. & Born, J. Sleep forms memory for finger skills. Proc. Natl Acad. Sci. USA 99, 11987–11991 (2002).
Ellenbogen, J. M., Hulbert, J. C., Stickgold, R., Dinges, D. F. & Thompson-Schill, S. L. Interfering with theories of sleep and memory: sleep, declarative memory, and associative interference. Curr. Biol. 16, 1290–1294 (2006).
Albouy, G. et al. Cerebral activity associated with transient sleep-facilitated reduction in motor memory vulnerability to interference. Sci. Rep. 6, 34948 (2016).
Sonni, A. & Spencer, R. M. Sleep protects memories from interference in older adults. Neurobiol. Aging 36, 2272–2281 (2015).
Dudai, Y. The neurobiology of consolidations, or, how stable is the engram? Annu. Rev. Psychol. 55, 51–86 (2004).
Stickgold, R. Sleep-dependent memory consolidation. Nature 437, 1272–1278 (2005).
Puentes-Mestril, C. & Aton, S. J. Linking network activity to synaptic plasticity during sleep: hypotheses and recent data. Front. Neural Circuits 11, 61 (2017).
Dosher, B. A. & Lu, Z. L. Hebbian reweighting on stable representations in perceptual learning. Learn. Percept. 1, 37–58 (2009).
Watanabe, T., Nanez, J. E. & Sasaki, Y. Perceptual learning without perception. Nature 413, 844–848 (2001).
Hensch, T. K. Critical period plasticity in local cortical circuits. Nat. Rev. Neurosci. 6, 877–888 (2005).
Bang, J. W. et al. Consolidation and reconsolidation share behavioral and neurochemical mechanisms. Nat. Hum. Behav. 2, 507–513 (2018).
Shibata, K. et al. Overlearning hyperstabilizes a skill by rapidly making neurochemical processing inhibitory-dominant. Nat. Neurosci. 20, 470–475 (2017).
Shadmehr, R. & Holcomb, H. H. Neural correlates of motor memory consolidation. Science 277, 821–825 (1997).
Seitz, A. R. et al. Task-specific disruption of perceptual learning. Proc. Natl Acad. Sci. USA 102, 14895–14900 (2005).
Karni, A. & Sagi, D. Where practice makes perfect in texture discrimination: evidence for primary visual cortex plasticity. Proc. Natl Acad. Sci. USA 88, 4966–4970 (1991).
Yotsumoto, Y., Chang, L. H., Watanabe, T. & Sasaki, Y. Interference and feature specificity in visual perceptual learning. Vis. Res. 49, 2611–2623 (2009).
Mednick, S. C., Cai, D. J., Shuman, T., Anagnostaras, S. & Wixted, J. T. An opportunistic theory of cellular and systems consolidation. Trends Neurosci. 34, 504–514 (2011).
Rothman, D. L., Petroff, O. A., Behar, K. L. & Mattson, R. H. Localized 1H NMR measurements of gamma-aminobutyric acid in human brain in vivo. Proc. Natl Acad. Sci. USA 90, 5662–5666 (1993).
Sanacora, G. et al. Increased cortical GABA concentrations in depressed patients receiving ECT. Am. J. Psychiatry 160, 577–579 (2003).
Barron, H. C. et al. Unmasking latent inhibitory connections in human cortex to reveal dormant cortical memories. Neuron 90, 191–203 (2016).
Stagg, C. J. et al. Polarity-sensitive modulation of cortical neurotransmitters by transcranial stimulation. J. Neurosci. 29, 5202–5206 (2009).
Stanley, J. A. et al. Functional dynamics of hippocampal glutamate during associative learning assessed with in vivo 1H functional magnetic resonance spectroscopy. NeuroImage 153, 189–197 (2017).
Floyer-Lea, A., Wylezinska, M., Kincses, T. & Matthews, P. M. Rapid modulation of GABA concentration in human sensorimotor cortex during motor learning. J. Neurophysiol. 95, 1639–1644 (2006).
Dinse, H. R., Ragert, P., Pleger, B., Schwenkreis, P. & Tegenthoff, M. Pharmacological modulation of perceptual learning and associated cortical reorganization. Science 301, 91–94 (2003).
Diering, G. H. et al. Homer1a drives homeostatic scaling-down of excitatory synapses during sleep. Science 355, 511–515 (2017).
Chung, S. et al. Identification of preoptic sleep neurons using retrograde labelling and gene profiling. Nature 545, 477–481 (2017).
Saper, C. B. & Fuller, P. M. Wake–sleep circuitry: an overview. Curr. Opin. Neurobiol. 44, 186–192 (2017).
Lee, S. H. et al. Activation of specific interneurons improves V1 feature selectivity and visual perception. Nature 488, 379–383 (2012).
Li, W., Ma, L., Yang, G. & Gan, W. B. REM sleep selectively prunes and maintains new synapses in development and learning. Nat. Neurosci. 20, 427–437 (2017).
Wilson, M. A. & McNaughton, B. L. Reactivation of hippocampal ensemble memories during sleep. Science 265, 676–679 (1994).
Riedel, G., Platt, B. & Micheau, J. Glutamate receptor function in learning and memory. Behav. Brain Res 140, 1–47 (2003).
Niethard, N., Burgalossi, A. & Born, J. Plasticity during sleep is linked to specific regulation of cortical circuit activity. Front. Neural Circuits 11, 65 (2017).
McDevitt, E. A., Duggan, K. A. & Mednick, S. C. REM sleep rescues learning from interference. Neurobiol. Learn. Mem. 122, 51–62 (2015).
Bang, J. W., Khalilzadeh, O., Hamalainen, M., Watanabe, T. & Sasaki, Y. Location specific sleep spindle activity in the early visual areas and perceptual learning. Vis. Res. 99, 162–171 (2014).
Tamaki, M., Wang, Z., Watanabe, T. & Sasaki, Y. Trained-feature-specific offline learning by sleep in an orientation detection task. J. Vis. 19, 12 (2019).
Tamaki, M. et al. Reward does not facilitate visual perceptual learning until sleep occurs. Proc. Natl Acad. Sci. USA 117, 959–968 (2020).
Tononi, G. & Cirelli, C. Sleep and synaptic homeostasis: a hypothesis. Brain Res. Bull. 62, 143–150 (2003).
Boyce, R., Glasgow, S. D., Williams, S. & Adamantidis, A. Causal evidence for the role of REM sleep theta rhythm in contextual memory consolidation. Science 352, 812–816 (2016).
Nishida, M., Pearsall, J., Buckner, R. L. & Walker, M. P. REM sleep, prefrontal theta, and the consolidation of human emotional memory. Cereb. Cortex 19, 1158–1166 (2009).
Faul, F., Erdfelder, E., Lang, A. G. & Buchner, A. G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 39, 175–191 (2007).
Tamaki, M., Bang, J. W., Watanabe, T. & Sasaki, Y. The first-night effect suppresses the strength of slow-wave activity originating in the visual areas during sleep. Vis. Res. 99, 154–161 (2014).
Tamaki, M., Bang, J. W., Watanabe, T. & Sasaki, Y. Night watch in one brain hemisphere during sleep associated with the first-night effect in humans. Curr. Biol. 26, 1190–1194 (2016).
Roenneberg, T., Wirz-Justice, A. & Merrow, M. Life between clocks: daily temporal patterns of human chronotypes. J. Biol. Rhythms 18, 80–90 (2003).
Green, C. S. & Bavelier, D. Action video game modifies visual selective attention. Nature 423, 534–537 (2003).
Berard, A. V., Cain, M. S., Watanabe, T. & Sasaki, Y. Frequent video game players resist perceptual interference. PLoS ONE 10, e0120011 (2015).
Horne, J. A. & Ostberg, O. A self-assessment questionnaire to determine morningness–eveningness in human circadian rhythms. Int. J. Chronobiol. 4, 97–110 (1976).
Buysse, D. J., Reynolds, C. F. III, Monk, T. H., Berman, S. R. & Kupfer, D. J. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 28, 193–213 (1989).
Lubin, A., Hord, D. J., Tracy, M. L. & Johnson, L. C. Effects of exercise, bedrest and napping on performance decrement during 40 hours. Psychophysiology 13, 334–339 (1976).
Hoddes, E., Vincent, Z. & Dement, W. C. The development and use of the Stanford sleepiness scale (SSS). Psychophysiology 9, 150 (1972).
Hoddes, E., Zarcone, V., Smythe, H., Phillips, R. & Dement, W. C. Quantification of sleepiness: a new approach. Psychophysiology 10, 431–436 (1973).
Dinges, D. F. & Powell, J. W. Microcomputer analyses of performance on a portable, simple visual RT task during sustained operations. Behav. Res. Methods Instrum. Comput. 17, 652–655 (1985).
Tamaki, M., Nittono, H. & Hori, T. The first-night effect occurs at the sleep-onset period regardless of the temporal anxiety level in healthy students. Sleep Biol. Rhythms 3, 92–94 (2005).
Agnew, H. W. Jr., Webb, W. B. & Williams, R. L. The first night effect: an EEG study of sleep. Psychophysiology 2, 263–266 (1966).
Carskadon, M. A. & Dement, W. C. Cumulative effects of sleep restriction on daytime sleepiness. Psychophysiology 18, 107–113 (1981).
Schwartz, S., Maquet, P. & Frith, C. Neural correlates of perceptual learning: a functional MRI study of visual texture discrimination. Proc. Natl Acad. Sci. USA 99, 17137–17142 (2002).
Yotsumoto, Y. et al. Location-specific cortical activation changes during sleep after training for perceptual learning. Curr. Biol. 19, 1278–1282 (2009).
Brainard, D. H. The Psychophysics Toolbox. Spat. Vis. 10, 433–436 (1997).
Pelli, D. G. The VideoToolbox software for visual psychophysics: transforming numbers into movies. Spat. Vis. 10, 437–442 (1997).
Machizawa, M., Patey, R., Kim, D. & Watanabe, T. Different aspects of training on a texture discrimination task (TDT) improves different attentional abilities. J. Vis. 14, 951 (2014).
Wichmann, F. A. & Hill, N. J. The psychometric function: I. Fitting, sampling, and goodness of fit. Percept. Psychophys. 63, 1293–1313 (2001).
Luce, R. D. in Human Memory and Cognitive Capabilities (eds Klix, F. & Hagendorf, H.) 109-121 (Elsevier, 1986).
Lim, J. & Dinges, D. F. Sleep deprivation and vigilant attention. Ann. NY Acad. Sci. 1129, 305–322 (2008).
Iber, C., Ancoli-Israel, S., Chesson, A. & Quan, S. F. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology, and Technical Specification (American Academy of Sleep Medicine, 2007).
Sharbrough, F. C. G. E., Lesser, R. P., Lüders, H., Nuwer, M. & Picton, T. W. American electroencephalographic society guidelines for standard electrode position nomenclature. J. Clin. Neurophysiol. 8, 200–202 (1991).
Mescher, M., Merkle, H., Kirsch, J., Garwood, M. & Gruetter, R. Simultaneous in vivo spectral editing and water suppression. NMR Biomed. 11, 266–272 (1998).
Edden, R. A. & Barker, P. B. Spatial effects in the detection of γ-aminobutyric acid: improved sensitivity at high fields using inner volume saturation. Magn. Reson. Med. 58, 1276–1282 (2007).
Hu, Y., Chen, X., Gu, H. & Yang, Y. Resting-state glutamate and GABA concentrations predict task-induced deactivation in the default mode network. J. Neurosci. 33, 18566–18573 (2013).
Muthukumaraswamy, S. D., Edden, R. A., Jones, D. K., Swettenham, J. B. & Singh, K. D. Resting GABA concentration predicts peak gamma frequency and fMRI amplitude in response to visual stimulation in humans. Proc. Natl Acad. Sci. USA 106, 8356–8361 (2009).
Stagg, C. J., Bachtiar, V. & Johansen-Berg, H. The role of GABA in human motor learning. Curr. Biol. 21, 480–484 (2011).
Robertson, C. E., Ratai, E. M. & Kanwisher, N. Reduced GABAergic action in the autistic brain. Curr. Biol. 26, 80–85 (2016).
Henry, M. E., Lauriat, T. L., Shanahan, M., Renshaw, P. F. & Jensen, J. E. Accuracy and stability of measuring GABA, glutamate, and glutamine by proton magnetic resonance spectroscopy: a phantom study at 4 Tesla. J. Magn. Reson. 208, 210–218 (2011).
Stagg, C. J. et al. Neurochemical effects of theta burst stimulation as assessed by magnetic resonance spectroscopy. J. Neurophysiol. 101, 2872–2877 (2009).
Stagg, C. J. et al. Local GABA concentration is related to network-level resting functional connectivity. eLife 3, e01465 (2014).
Tkác, I., Starcuk, Z., Choi, I. Y. & Gruetter, R. In vivo 1H NMR spectroscopy of rat brain at 1 ms echo time. Magn. Reson. Med. 41, 649–656 (1999).
Rothman, D. L., Behar, K. L., Hetherington, H. P. & Shulman, R. G. Homonuclear 1H double-resonance difference spectroscopy of the rat brain in vivo. Proc. Natl Acad. Sci. USA 81, 6330–6334 (1984).
Klose, U. In vivo proton spectroscopy in presence of eddy currents. Magn. Reson. Med. 14, 26–30 (1990).
Oeltzschner, G., Schnitzler, A., Wickrath, F., Zollner, H. J. & Wittsack, H. J. Use of quantitative brain water imaging as concentration reference for J-edited MR spectroscopy of GABA. Magn. Reson. Imaging 34, 1057–1063 (2016).
Gasparovic, C. et al. Use of tissue water as a concentration reference for proton spectroscopic imaging. Magn. Reson. Med. 55, 1219–1226 (2006).
Provencher, S. W. Estimation of metabolite concentrations from localized in vivo proton NMR spectra. Magn. Reson. Med. 30, 672–679 (1993).
Provencher, S. W. Automatic quantitation of localized in vivo 1H spectra with LCModel. NMR Biomed. 14, 260–264 (2001).
Lunghi, C., Emir, U. E., Morrone, M. C. & Bridge, H. Short-term monocular deprivation alters GABA in the adult human visual cortex. Curr. Biol. 25, 1496–1501 (2015).
Zeinali-Rafsanjani, B. et al. MRS shimming: an important point which should not be ignored. J. Biomed. Phys. Eng. 8, 261–270 (2018).
Kreis, R. Issues of spectral quality in clinical 1H-magnetic resonance spectroscopy and a gallery of artifacts. NMR Biomed. 17, 361–381 (2004).
Cohen, J. Statistical Power Analysis for the Behavioral Sciences (Lawrence Erlbaum and Associates, 1988).
Acknowledgements
This work was supported by the NIH (R21EY028329, R01EY019466, R01EY027841, T32EY018080 and T32MH115895) and by BSF2016058. Part of this research was also supported by the Center for Vision Research, Brown University.
Author information
Authors and Affiliations
Contributions
M.T., T.W. and Y.S. designed the research. M.T., Z.W., T.B.-D., D.G., A.V.B. and E.W. performed the experiments. M.T., E.W., Z.W. and T.B.-D. analyzed the data. M.T., Z.W., T.B.-D., D.G., A.V.B., E.W., T.W. and Y.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Neuroscience thanks Penelope Lewis, Bryce Mander and Caroline Robertson for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Example structural MRS image indicating the voxel located in early visual areas.
Based on the measured anatomical structure, the voxel of interest was manually placed on the most posterior part of the occipital lobe, covering the calcarine sulci that corresponds to early visual areas bilaterally21.
Extended Data Fig. 2 Example spectra from the voxel located in early visual areas.
The measured spectrum is shown in the top row. “Fit” in the second row represents the spectrum fitted with the LCModel (see the section “MRS acquisition” in the Methods for details). “Residual” in the bottom row represents the residual remaining after the fitting. The remaining rows indicate individual fits for all metabolites that can be detected by a given acquisition. Macromolecular and lipid signals were used for the baseline correction. NAA, NAAG, GSH, Glu, Gln, and GABA represent N-acetylaspartate, N-acetylaspartylglutamate, glutathione, glutamate, glutamine, and gamma-aminobutyric acid, respectively. Glx is obtained by adding glutamine and glutamate in the LCModel. The same procedure was repeated 272 times independently with similar results.
Extended Data Fig. 3 The experimental design and results of Experiment 2.
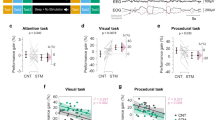
a, Design. The first texture discrimination task (TDT) training (task A) was conducted with background A, and the second training (task B) was conducted with background B. Test sessions were conducted before the first training (pretest) and after the second training (post-test) to measure performance gains on task A. b, Boxplots for the performance change (%) for task A from the pretest session to the post-test session in the NREM + REM group (red box, n = 23 subjects) and the NREM-only group (gray box, n = 15 subjects). Significant off-line performance gains were observed among subjects who had both NREM sleep and REM sleep (**** two-sided one sample t-test against 0, t22 = 5.48, p < 0.001, d = 1.14, 95% CI [15.86, 35.17]), whereas the subjects who had only NREM sleep did not show any significant off-line performance gains (two-sided one sample t-test against 0, t14 = 0.39, p = 0.699). Furthermore, off-line performance gains were significantly different between the groups (**** two-sided independent-samples t-test, t36 = 4.02, p < 0.001, d = 1.33, 95% CI [13.49, 41.01]). For each boxplot, the bottom and top of the box correspond to the 25th and 75th percentiles (the lower and upper quartiles), respectively. The inner thick horizontal line represents the median, and the plus mark represents the mean. The whiskers show the maximum and minimum of the data. Individual data (dots) are overlaid. Grubbs’ test showed no outliers (Alpha = .05, two-sided).
Extended Data Fig. 4 Mean raw spectra in experiment 1 (a, n = 19 subjects, red plots) and in experiment 3 (b, n = 19 subjects, blue plots).
The value below each plot shows the mean full-width-at-half-maximum linewidth for NAA in Hz (mean ± SEM, Experiment 1, 8.0 ± 0.07 Hz; Experiment 3, 8.0 ± 0.05 Hz).
Extended Data Fig. 5 Correlation between EEG features, performance changes, and E/I balance changes during NREM and REM sleep.
We investigated whether sigma-band (13–16 Hz) and delta-band (1–4 Hz) activities during NREM sleep and theta-band (5–7 Hz) activity during REM sleep were involved in off-line performance gain or resilience to interference. We focused on these oscillatory activities because they are implicated in learning and memory43,44,45,46,47,48. First, a fast-Fourier transformation was applied to the EEG data in 5-sec epochs, and the data were smoothed with a tapered cosine window44 to compute brain activities. Second, six epochs were used to yield the mean spectral for 30 s. Third, we calculated the power for each frequency band during both sleep and wakefulness in the trained region of early visual areas using a set of O1, PO3 and PO7 EEG channels or a set of O2, PO4, and PO8 EEG channels, depending on whether the target appeared in the right or left upper visual field. Fourth, we calculated the power for each frequency band during both sleep and wakefulness in the MT region, which was considered to be a control region according to a previous paper44, using P7 and P8 EEG channels. Fifth, we normalized the power for each frequency band in both the trained region and the control region by subtracting the power for each frequency during sleep from that during wakefulness and dividing that value by that during wakefulness for each region to obtain the power of each frequency during sleep. Finally, we obtained the trained-region specific power during sleep for each frequency band by subtracting the normalized power in the control region from the normalized power in the trained region. Because NREM sleep was associated with off-line performance gains (see Fig. 1c, d), we measured the Pearson’s correlation coefficients for sigma power during NREM sleep and off-line performance gain and for delta power during NREM sleep and off-line performance gain. Analogously, because REM sleep was associated with resilience to interference (see Fig. 1e, f), we measured the Pearson’s correlation for theta-band power during REM sleep and resilience to interference. First, sigma power during NREM sleep was mildly correlated with off-line performance gains (a, n = 19 subjects, Pearson’s r17 = 0.458, two-sided t-test, p = 0.048, 95% CI [0.01, 0.76], with a Bonferroni adjusted alpha level of 0.025 (0.05/2)) and E/I balance during NREM sleep (c, n = 19 subjects, Pearson’s r17 = 0.397, two-sided t-test, p = 0.093, 95% CI [−0.070, 0.721]). Second, theta power during REM sleep was mildly correlated with resilience to interference (b, n = 10 subjects, Pearson’s r8 = 0.632, two-sided t-test, p = 0.050, with a Bonferroni adjusted alpha level of 0.025 (0.05/2), 95% CI [0.003, 0.902]) and E/I balance during REM sleep (d, n = 10 subjects, Pearson’s r8 = −0.68, two-sided t-test, p = 0.031, 95% CI [−0.916, −0.084]). Finally, delta power during NREM sleep was not significantly correlated with performance gains (n = 19 subjects, Pearson’s r17 = 0.23, two-sided t-test, p = 0.334) or E/I balance (n = 19 subjects, Pearson’s r17 = 0.38, two-sided t-test, p = 0.113) during NREM sleep. These results suggest that EEG power is only mildly correlated with off-line performance gains or with resistance to retrograde interference. However, the PSG data were obtained in a strong magnetic environment, which may not be suitable for performing a detailed power analysis.
Extended Data Fig. 6 An illustration of how to co-register MRS data and PSG data.
a, Sleep stage as a function of time (min). There are 9 segments for each 10-min MRS segment. In each segment, there are 20 epochs (each 30 sec) of sleep stage scoring. Each blue dot represents the sleep stage score for 30 sec. b, Simplified sleep stage as a function of time. The sleep stage score is simplified as 3 PSG states: Wake (W), NREM sleep (N; stage N1, N2 and N3 combined), and REM sleep (R). c, Splitting of two 10-min MRS segments (original #1 and #8) into five 2-min segments. This was because there were 2 or 3 PSG states within one original 10-min segment. In a “New” segments row, a gray section indicates Wake, a pink section indicates NREM sleep, and a blue section indicates REM sleep. d, The E/I balance during NREM sleep and REM sleep for each MRS segment was obtained by normalizing them to that during Wake. e, Time course of the concentrations of Glx (red) and GABA (cyan) underlying the E/I balance. Between the transition from e to d, there were several steps, as described in “Calculation of the mean E/I balance for NREM and REM sleep” in the section “Co-registration of MRS data and sleep stages” in the Methods.
Supplementary information
Supplementary Information
Supplementary Tables 1–13.
Supplementary Table 14
This file contains source data for Supplementary Tables 1–13.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 5
Statistical source data.
Rights and permissions
About this article
Cite this article
Tamaki, M., Wang, Z., Barnes-Diana, T. et al. Complementary contributions of non-REM and REM sleep to visual learning. Nat Neurosci 23, 1150–1156 (2020). https://doi.org/10.1038/s41593-020-0666-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-020-0666-y
This article is cited by
-
Auditory stimulation during REM sleep modulates REM electrophysiology and cognitive performance
Communications Biology (2024)
-
Multiple timescales of learning indicated by changes in evidence-accumulation processes during perceptual decision-making
npj Science of Learning (2023)
-
A data assimilation method to track excitation-inhibition balance change using scalp EEG
Communications Engineering (2023)
-
The phase of plasticity-induced neurochemical changes of high-frequency repetitive transcranial magnetic stimulation are different from visual perceptual learning
Scientific Reports (2023)
-
Temperature-robust rapid eye movement and slow wave sleep in the lizard Laudakia vulgaris
Communications Biology (2022)