Abstract
Cerebellar dysfunction has been demonstrated in autism spectrum disorders (ASDs); however, the circuits underlying cerebellar contributions to ASD-relevant behaviors remain unknown. In this study, we demonstrated functional connectivity between the cerebellum and the medial prefrontal cortex (mPFC) in mice; showed that the mPFC mediates cerebellum-regulated social and repetitive/inflexible behaviors; and showed disruptions in connectivity between these regions in multiple mouse models of ASD-linked genes and in individuals with ASD. We delineated a circuit from cerebellar cortical areas Right crus 1 (Rcrus1) and posterior vermis through the cerebellar nuclei and ventromedial thalamus and culminating in the mPFC. Modulation of this circuit induced social deficits and repetitive behaviors, whereas activation of Purkinje cells (PCs) in Rcrus1 and posterior vermis improved social preference impairments and repetitive/inflexible behaviors, respectively, in male PC-Tsc1 mutant mice. These data raise the possibility that these circuits might provide neuromodulatory targets for the treatment of ASD.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The authors confirm that all relevant non-MRI data are included in the paper and/or its supplementary information files. Raw MRI data that support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
All code is publicly available on GitHub (https://github.com/Mouse-Imaging-Centre/RMINC) or is available upon reasonable request.
References
D’Mello, A. M. & Stoodley, C. J. Cerebro-cerebellar circuits in autism spectrum disorder. Front. Neurosci. 9, 408 (2015).
Fatemi, S. H. et al. Consensus paper: pathological role of the cerebellum in autism. Cerebellum 11, 777–807 (2012).
Mosconi, M. W., Wang, Z., Schmitt, L. M., Tsai, P. & Sweeney, J. A. The role of cerebellar circuitry alterations in the pathophysiology of autism spectrum disorders. Front. Neurosci. 9, 296 (2015).
Tsai, P. T. Autism and cerebellar dysfunction: evidence from animal models. Semin. Fetal Neonatal Med. 21, 349–355 (2016).
Tsai, P. T. et al. Autistic-like behaviour and cerebellar dysfunction in Purkinje cell Tsc1 mutant mice. Nature 488, 647–651 (2012).
Pierce, K. & Courchesne, E. Evidence for a cerebellar role in reduced exploration and stereotyped behavior in autism. Biol. Psychiatry 49, 655–664 (2001).
D’Mello, A. M., Crocetti, D., Mostofsky, S. H. & Stoodley, C. J. Cerebellar gray matter and lobular volumes correlate with core autism symptoms. Neuroimage Clin. 7, 631–639 (2015).
Stoodley, C. J. et al. Altered cerebellar connectivity in autism and cerebellar-mediated rescue of autism-related behaviors in mice. Nat. Neurosci. 20, 1744–1751 (2017).
Krienen, F. M. & Buckner, R. L. Segregated fronto-cerebellar circuits revealed by intrinsic functional connectivity. Cereb. Cortex 19, 2485–2497 (2009).
Middleton, F. A. & Strick, P. L. Cerebellar projections to the prefrontal cortex of the primate. J. Neurosci. 21, 700–712 (2001).
Van Overwalle, F. & Marien, P. Functional connectivity between the cerebrum and cerebellum in social cognition: a multi-study analysis. Neuroimage 124, 248–255 (2015).
Watson, T. C., Becker, N., Apps, R. & Jones, M. W. Back to front: cerebellar connections and interactions with the prefrontal cortex. Front. Syst. Neurosci. 8, 4 (2014).
Steriade, M. Two channels in the cerebellothalamocortical system. J. Comp. Neurol. 354, 57–70 (1995).
Hazlett, H. C. et al. Early brain development in infants at high risk for autism spectrum disorder. Nature 542, 348–351 (2017).
Dichter, G. S. Functional magnetic resonance imaging of autism spectrum disorders. Dialogues Clin. Neurosci. 14, 319–351 (2012).
Gilbert, S. J., Meuwese, J. D., Towgood, K. J., Frith, C. D. & Burgess, P. W. Abnormal functional specialization within medial prefrontal cortex in high-functioning autism: a multi-voxel similarity analysis. Brain 132, 869–878 (2009).
Rogers, T. D. et al. Connecting the dots of the cerebro-cerebellar role in cognitive function: neuronal pathways for cerebellar modulation of dopamine release in the prefrontal cortex. Synapse 65, 1204–1212 (2011).
Rogers, T. D. et al. Reorganization of circuits underlying cerebellar modulation of prefrontal cortical dopamine in mouse models of autism spectrum disorder. Cerebellum 12, 547–556 (2013).
Buckner, R. L., Krienen, F. M., Castellanos, A., Diaz, J. C. & Yeo, B. T. The organization of the human cerebellum estimated by intrinsic functional connectivity. J. Neurophysiol. 106, 2322–2345 (2011).
Gilbert, S. J., Bird, G., Brindley, R., Frith, C. D. & Burgess, P. W. Atypical recruitment of medial prefrontal cortex in autism spectrum disorders: an fMRI study of two executive function tasks. Neuropsychologia 46, 2281–2291 (2008).
Pirone, A. et al. Social stimulus causes aberrant activation of the medial prefrontal cortex in a mouse model with autism-like behaviors. Front. Synaptic Neurosci. 10, 35 (2018).
Kobayashi, M., Hayashi, Y., Fujimoto, Y. & Matsuoka, I. Decreased parvalbumin and somatostatin neurons in medial prefrontal cortex in BRINP1-KO mice. Neurosci. Lett. 683, 82–88 (2018).
Yizhar, O. et al. Neocortical excitation/inhibition balance in information processing and social dysfunction. Nature 477, 171–178 (2011).
Selimbeyoglu, A. et al. Modulation of prefrontal cortex excitation/inhibition balance rescues social behavior in CNTNAP2-deficient mice. Sci. Transl. Med. 9, eaah6733 (2017).
Alexander-Bloch, A., Giedd, J. N. & Bullmore, E. Imaging structural co-variance between human brain regions. Nat. Rev. Neurosci. 14, 322–336 (2013).
Baribeau, D. A. et al. Structural neuroimaging correlates of social deficits are similar in autism spectrum disorder and attention-deficit/hyperactivity disorder: analysis from the POND. Netw. Transl. Psychiatry 9, 72 (2019).
Jansen, J. & Brodal, A.Experimental studies on the intrinsic fibers of the cerebellum II. The cortico-nuclear projection. Cerebellum 10, 126–180 (1940).
Zingg, B. et al. AAV-mediated anterograde transsynaptic tagging: mapping corticocollicular input-defined neural pathways for defense behaviors. Neuron 93, 33–47 (2017).
Kuramoto, E. et al. Ventral medial nucleus neurons send thalamocortical afferents more widely and more preferentially to layer 1 than neurons of the ventral anterior-ventral lateral nuclear complex in the rat. Cereb. Cortex 25, 221–235 (2015).
Mitchell, B. D. & Cauller, L. J. Corticocortical and thalamocortical projections to layer I of the frontal neocortex in rats. Brain Res. 921, 68–77 (2001).
Urbain, N., Fourcaud-Trocme, N., Laheux, S., Salin, P. A. & Gentet, L. J. Brain-state-dependent modulation of neuronal firing and membrane potential dynamics in the somatosensory thalamus during natural sleep. Cell Rep. 26, 1443–1457 (2019).
Badura, A. et al. Normal cognitive and social development require posterior cerebellar activity. eLife. 7, e36401 (2018).
Srivastava, S. et al. Volumetric analysis of the basal ganglia and cerebellar structures in patients with Phelan–McDermid syndrome. Pediatr. Neurol. 90, 37–43 (2019).
Courville, J. & Diakiw, N. Cerebellar corticonuclear projection in the cat. The vermis of the anterior and posterior lobes. Brain Res. 110, 1–20 (1976).
Groenewegen, H. J. Organization of the afferent connections of the mediodorsal thalamic nucleus in the rat, related to the mediodorsal-prefrontal topography. Neuroscience 24, 379–431 (1988).
Haroian, A. J., Massopust, L. C. & Young, P. A. Cerebellothalamic projections in the rat: an autoradiographic and degeneration study. J. Comp. Neurol. 197, 217–236 (1981).
Rubenstein, J. L. & Merzenich, M. M. Model of autism: increased ratio of excitation/inhibition in key neural systems. Genes Brain Behav. 2, 255–267 (2003).
Takarae, Y. & Sweeney, J. Neural hyperexcitability in autism spectrum disorders. Brain Sci. 7, 129 (2017).
Contractor, A., Klyachko, V. A. & Portera-Cailliau, C. Altered neuronal and circuit excitability in fragile X syndrome. Neuron 87, 699–715 (2015).
Sohal, V. S., Zhang, F., Yizhar, O. & Deisseroth, K. Parvalbumin neurons and gamma rhythms enhance cortical circuit performance. Nature 459, 698–702 (2009).
Mouginot, D. & Gahwiler, B. H. Characterization of synaptic connections between cortex and deep nuclei of the rat cerebellum in vitro. Neuroscience 64, 699–712 (1995).
Ryu, Y. H. et al. Perfusion impairments in infantile autism on technetium-99m ethyl cysteinate dimer brain single-photon emission tomography: comparison with findings on magnetic resonance imaging. Eur. J. Nucl. Med. 26, 253–259 (1999).
Carta, I., Chen, C. H., Schott, A. L., Dorizan, S. & Khodakhah, K. Cerebellar modulation of the reward circuitry and social behavior. Science 363, eaav0581 (2019).
Bicks, L. K., Koike, H., Akbarian, S. & Morishita, H. Prefrontal cortex and social cognition in mouse and man. Front. Psychol. 6, 1805 (2015).
Popa, L. S. & Ebner, T. J. Cerebellum, predictions and errors. Front. Cell Neurosci. 12, 524 (2018).
Carmi, L. et al. Clinical and electrophysiological outcomes of deep TMS over the medial prefrontal and anterior cingulate cortices in OCD patients. Brain Stimulation 11, 158–165 (2018).
van Dun, K., Bodranghien, F., Manto, M. & Marien, P. Targeting the cerebellum by noninvasive neurostimulation: a review. Cerebellum 16, 695–741 (2017).
Parker, K. L. et al. Delta-frequency stimulation of cerebellar projections can compensate for schizophrenia-related medial frontal dysfunction. Mol. Psychiatry 22, 647–655 (2017).
Brady, R. O., Jr. et al. Cerebellar-prefrontal network connectivity and negative symptoms in schizophrenia. Am. J. Psychiatry 176, 512–520 (2019).
Tsai, P. T. et al. Sensitive periods for cerebellar-mediated autistic-like behaviors. Cell Rep. 25, 357–367 (2018).
Barski, J. J., Dethleffsen, K. & Meyer, M. Cre recombinase expression in cerebellar Purkinje cells. Genesis 28, 93–98 (2000).
Kwiatkowski, D. J. et al. A mouse model of TSC1 reveals sex-dependent lethality from liver hemangiomas, and up-regulation of p70S6 kinase activity in Tsc1 null cells. Hum. Mol. Genet. 11, 525–534 (2002).
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci. 13, 133–140 (2009).
Paxinos, G. & Franklin, K. Paxinos and Franklin’s the Mouse Brain in Stereotaxic Coordinates 4th edn (Academic Press, 2012).
Watson, C., Paxinos, G. & Puelles, L. The Mouse Nervous System (Academic Press, 2011).
Armbruster, B. N., Li, X., Pausch, M. H., Herlitze, S. & Roth, B. L. Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand. Proc. Natl Acad. Sci. USA 104, 5163–5168 (2007).
Cahill, L. S. et al. Preparation of fixed mouse brains for MRI. Neuroimage 60, 933–939 (2012).
Lerch, J. P., Sled, J. G. & Henkelman, R. M. MRI phenotyping of genetically altered mice. Methods Mol. Biol. 711, 349–361 (2011).
Nieman, B. J. et al. MR technology for biological studies in mice. NMR Biomed. 20, 291–303 (2007).
Nieman, B. J. et al. Magnetic resonance imaging for detection and analysis of mouse phenotypes. NMR Biomed. 18, 447–468 (2005).
Nieman, B. J., Flenniken, A. M., Adamson, S. L., Henkelman, R. M. & Sled, J. G. Anatomical phenotyping in the brain and skull of a mutant mouse by magnetic resonance imaging and computed tomography. Physiological Genomics 24, 154–162 (2006).
Dazai, J. et al. Multiple mouse biological loading and monitoring system for MRI. Magn. Reson. Med. 52, 709–715 (2004).
Dazai, J., Spring, S., Cahill, L. S. & Henkelman, R. M. Multiple-mouse neuroanatomical magnetic resonance imaging. J. Vis. Exp. 48, 2497 (2011).
Thomas, D. L. et al. High-resolution fast spin echo imaging of the human brain at 4.7T: implementation and sequence characteristics. Magn. Reson. Med. 51, 1254–1264 (2004).
Nieman, B. J. et al. Fast spin-echo for multiple mouse magnetic resonance phenotyping. Magn. Reson. Med. 54, 532–537 (2005).
Dorr, A. E., Lerch, J. P., Spring, S., Kabani, N. & Henkelman, R. M. High resolution three-dimensional brain atlas using an average magnetic resonance image of 40 adult C57Bl/6J mice. Neuroimage 42, 60–69 (2008).
Richards, K. et al. Segmentation of the mouse hippocampal formation in magnetic resonance images. Neuroimage 58, 732–740 (2011).
Steadman, P. E. et al. Genetic effects on cerebellar structure across mouse models of autism using a magnetic resonance imaging atlas. Autism Res. 7, 124–137 (2014).
Ullmann, J. F., Watson, C., Janke, A. L., Kurniawan, N. D. & Reutens, D. C. A segmentation protocol and MRI atlas of the C57BL/6J mouse neocortex. Neuroimage 78, 196–203 (2013).
Chakravarty, M. M. et al. Performing label-fusion-based segmentation using multiple automatically generated templates. Hum. Brain Mapp. 34, 2635–2654 (2013).
Park, M. T. et al. Derivation of high-resolution MRI atlases of the human cerebellum at 3T and segmentation using multiple automatically generated templates. Neuroimage 95, 217–231 (2014).
Kim, J. S. et al. Automated 3-D extraction and evaluation of the inner and outer cortical surfaces using a Laplacian map and partial volume effect classification. Neuroimage 27, 210–221 (2005).
Lerch, J. P. & Evans, A. C. Cortical thickness analysis examined through power analysis and a population simulation. Neuroimage 24, 163–173 (2005).
Lyttelton, O., Boucher, M., Robbins, S. & Evans, A. An unbiased iterative group registration template for cortical surface analysis. Neuroimage 34, 1535–1544 (2007).
Reardon, P. K. et al. Normative brain size variation and brain shape diversity in humans. Science 360, 1222–1227 (2018).
Roth, B. L. DREADDs for neuroscientists. Neuron 89, 683–694 (2016).
Deisseroth, K. Optogenetics: 10 years of microbial opsins in neuroscience. Nat. Neurosci. 18, 1213–1225 (2015).
Holmes, A. et al. Behavioral characterization of dopamine D5 receptor null mutant mice. Behav. Neurosci. 115, 1129 (2001).
Buitrago, M. M., Schulz, J. B., Dichgans, J. & Luft, A. R. Short and long-term motor skill learning in an accelerated rotarod training paradigm. Neurobiol. Learn. Mem. 81, 211–216 (2004).
Yang, M., Silverman, J. L. & Crawley, J. N. Automated three-chambered social approach task for mice. Curr. Protoc. Neurosci. 56, 8.26.21–28.26.16 (2011).
Silverman, J. L. et al. Sociability and motor functions in Shank1 mutant mice. Brain Res. 1380, 120–137 (2011).
Yang, M. & Crawley, J. N. Simple behavioral assessment of mouse olfaction. Curr. Protoc. Neurosci. 48, 8.24.1–8.24.12 (2009).
Bednar, I. et al. Selective nicotinic receptor consequences in APPSWE transgenic mice. Mol. Cell. Neurosci. 20, 354–365 (2002).
Acknowledgements
P.T.T. acknowledges support from the National Institute of Neurologic Disorders and Stroke (NS083733), the National Institute of Mental Health (MH116882), the Tuberous Sclerosis Alliance and the Department of Defense. E.K. acknowledges support from National Institute of Neurologic Disorders and Stroke (NS107004) and Autism Speaks. H.F. and S.d.L. acknowledge support from the National Institute of Neurologic Disorders and Stroke (NS095232 and NS105039). F. Morgado, J.E. and J.P.L. acknowledge support from the Canadian Institute for Health Research and the Ontario Brain Institute. L.C.R and C.J.S. acknowledge support from the National Institute for Health (MH106957). M.A.B. acknowledges support from the Medical Research Council (MR/K022377/1). N.K. acknowledges support from the National Institute of Mental Health (MH094268) and declares that he is a paid consultant for Rescindo Therapeutics, although this does not provide a competing interest with this study. P.T.T. and E.K. acknowledge support from V. Jakkamsetti and J. Pascual for support with in vivo extracellular and acute slice recordings recordings and analysis; S. Birnbaum for assistance in behavioral studies; G. Konopka, C. Powell, L. Osburne, J. Foster, J. Lai, K. Rilett, E. Kim and A. Raznahan for generous provision of animal models; and J. Chadwick for graphics assistance.
Author information
Authors and Affiliations
Contributions
E.K., F. Meng, H. F., J.P.L., S.d.L. and P.T.T. formulated experiments and analysis. E.K., F. Meng, H.F., Y.K., C.O.E., J.M.G., S.S., C.R., D.J., R.P., T.T. and B.E.P. performed experiments and analysis. F. Morgado, J.E. and J.P.L. carried out the mouse structural imaging experiments and analysis. F. Morgado, J.E., M.J.T, C.H., E.A. and J.P.L. carried out the human structural imaging experiments and analysis. L.C.R. and C.J.S. performed functional imaging in humans and analysis of these studies. M.A.B., R.D.B., S.D., C.G., M.K.H., N.K., D.M.R., J.L.S, K.K.S. and R.W. provided critical reagents. E.K., H.F., J.P.L., S.d.L. and P.T.T. prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Neuroscience thanks Ted Abel, Sarah Ferri and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
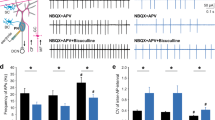
Extended Data Fig. 1 mPFC Gi DREADDs in PC-Tsc1 mutant mice.
a, Sample of injection site locations from PC-Tsc1 mutants injected with Gi (inhibitory) DREADDs/GFP into left prelimbic (PRL) medial prefrontal cortex (mPFC). b, Awake in vivo single unit recordings in the left PRL of control or PC- Tsc1 mutant mice. c, Three chambered social approach assay; time spent sniffing novel animal (NA), or novel object (NO). d, Three chambered social novelty testing; time spent sniffing NA or familiar animal (FA). e, Time in the open arm and distance traveled in the elevated plus maze assay. f, Time in the center of the open field and g, distance traveled in the open field. h, Latency to fall in accelerating rotarod test. Box line denoted median/whiskers denoted 5–95%. n ≥ 10 for all experiments. All behavioral tests were analyzed with two or three-way ANOVA and Sidak post test and single unit recordings were analyzed with Mann-Whitney test, shown as mean SEM. ****P < 0.0001; ***P < 0.001; **P < 0.01; *P < 0.05. P values and complete animal numbers can be found in Supplementary Table 2.
Extended Data Fig. 2 Left Prelimbic Synaptic Properties in PC- Tsc1 Mutant Mice.
a, mini EPSC (mEPSC) representative traces with zoomed in views below initial traces as noted; b, mEPSC frequency; c, mEPSC amplitude; d, mEPSC rise time; e, mEPSC decay time; f, representative mIPSC traces with zoomed in views blow initial traces as noted; g, mIPSC frequency; h, mIPSC amplitude; i, mIPSC rise time; j, and mIPSC decay time in PC-Tsc1 mutant mice and control mice. Groups had 12–15 cells from 5 animals in each. Students t-test, ****P < 0.0001; ***P < 0.001; **P < 0.01; *P < 0.05.
Extended Data Fig. 3 Impact of chemogenetic inhibition of mPFC on Rcrus1 PC inhibition-regulated behaviors.
a, Sample injection site locations in area Rcrus1. b, Awake in vivo single unit recordings in prelimbic cortex (PRL) in mice with Rcrus1 Gi DREADD inhibition (CNO) or vehicle treatment. c, In vivo single unit recordings in motor cortex of anesthetized mice with Rcrus1 Gi DREADDs inhibition compared to baseline. No significant change identified. d, In vivo single unit recordings in right PRL of anesthetized mice with Rcrus1 Gi DREADD inhibition compared to baseline. No significant change identified. e, In vivo single unit recordings in left PRL of anesthetized mice with Rcrus1 Gi DREADD inhibition and either GFP or Gi injection in the mPFC. f, Three chambered social approach assay; time spent sniffing novel animal (NA), or novel object (NO). g, Three chambered social novelty testing; time spent sniffing NA, or familiar animal (FA). h, Time in the open arm and distance traveled in the elevated plus maze assay. i, Time in the center of the open field and j, distance traveled in the open field. k, Latency to fall in accelerating rotarod test. All behavioral tests were analyzed with two or three-way ANOVA and Sidak post test. Single unit recordings were analyzed with Mann-Whitney test, shown as mean SEM. All raw values for frequency of spiking that are shown in figures as normalized values can be found in Supplementary Fig. 12. Box line denoted median/whiskers denoted 5–95%. n ≥ 10 for all behavioral experiments. ****P < 0.0001; ***P < 0.001; **P < 0.01; *P < 0.05. P values and complete animal numbers can be found in Supplementary Table 2.
Extended Data Fig. 4 Inhibition of LN in PC-Tsc1 mutant mice.
a, Sample injection site locations and injection site pictures of Gi DREADDs injected into LN of PC-Tsc1 mutant mice. b, Single unit activity in the right lateral nucleus (LN) with chemogenetic inhibition (Gi) or control GFP injection. c, Three chambered social approach assay; time spent sniffing novel animal (NA), or novel object (NO). d, Three chambered social novelty testing; time spent sniffing NA, or familiar animal (FA). e, Time in the open arm and distance traveled in the elevated plus maze assay. f, Time in the center of the open field and g, distance traveled in the open field. h, Latency to fall in accelerating rotarod test. All behavioral tests were analyzed with two or three-way ANOVA and Sidak post hoc testing. Recordings were analyzed with Mann-Whitney test, shown as mean SEM. For frequency of spiking shown in figures as normalized values, raw values can be found in Supplementary Fig. 12. Box line denoted median/whiskers denoted 5-95%. n ≥ 10 for all behavioral experiments. ****P < 0.0001; ***P < 0.001; **P < 0.01; *P < 0.05. P values and complete animal numbers can be found in Supplementary Table 2.
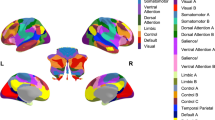
Extended Data Fig. 5 Thalamic and layer 1 cortical areas targeted by the medial and lateral cerebellar nuclei.
a, Thalamic nuclei with monosynaptic inputs from the right medial nucleus (MN) and lateral nucleus (LN) identified via AAV1-mediated anterograde transsynaptic tracing. The number of the trans-synaptically labeled thalamic neurons in each nucleus indicated as + + (high), + (low), and - (none). b, Disynaptic inputs from the right MN and LN to distinct cortical regions of the left cerebral cortex, identified via transsynaptic tracing. Labeled input density to layer 1 of each cortical area is indicated as + + (high), + (low), and - (none). c, Representative tracing results of disynaptic inputs from the right MN and LN to distinct areas in the left cerebral cortex. Axons and terminals of thalamic neurons trans-synaptically labeled from injections to MN and LN are indicated in black. Results from injections to MN and LN are in the left and right, respectively, of each panel. Top and bottom parts of each panel correspond to layer 1 and 6. Inputs to layer 1 originate predominantly from the VM thalamus. Arrowheads indicate layer 1. Scale bar applies to all panels. d, representative injection site in the LN and e, representative injection site in the MN. Scale bar applies to all panels. Abbreviations; Aud, auditory cortex; Cg, cingulate cortex; CL, centrolateral thalamic nucleus; FrA, frontal association cortex; IL, infralimbic cortex; LD, laterodorsal thalamic nucleus; LO, lateral orbital cortex; LP, lateroposterior thalamic nucleus; M1 and M2, primary and secondary motor cortex; MD, mediodorsal thalamic nucleus; MO, medial orbital cortex; PF, parafascicular thalamic nucleus; Po, posterior thalamic group; PrL, prelimbic cortex; Ptl, parietal association cortex; Rhi, ecto-/peri-/ento-entorhinal cortex; Rsp, retrosplenial cortex; S1 and S2, primary and secondary sensory cortex; Tem, temporal association cortex; VL, ventrolateral thalamic nucleus; VM, ventromedial thalamic nucleus; VO, ventral orbital cortex.
Extended Data Fig. 6 ChR2 activation of VM thalamus-PRL mPFC circuit.
a, Sample injection site locations for ChR2/Arch injection into the left VM thalamus. b, To test for possible antidromic activation, in vivo anesthetized single unit recordings in the VM thalamus-targeted parietal association cortex (PAC) were performed upon mPFC laser stimulation of ChR2 or GFP VM-thalamus terminals at 20 Hz or 4 Hz. Image of TdTomato positive terminals in PAC from LN AAV-1 tracing injection (top left). c, Three chambered social approach assay; time spent sniffing novel animal (NA), or novel object (NO) with 20 Hz (left) or 4 Hz (right) stimulation. d, Three chambered social novelty testing; time spent sniffing NA, or familiar animal (FA) with 20 Hz (left) or 4 Hz (right) stimulation. e, Time in the open arm and distance traveled in the elevated plus maze assay. f, Distance traveled in the open field and g, time in the center of the open field. All behavioral tests were analyzed with two or three-way ANOVA and Sidak post test and recordings were analyzed with two way ANOVA (unmarked = not significant). For frequency of spiking shown in figures as normalized values (mean SEM), raw values can be found in Supplementary Fig. 12. Box line denoted median/whiskers denoted 5-95%. n ≥ 10 for all behavioral experiments. ****P < 0.0001; ***P < 0.001; **P < 0.01; *P < 0.05. P values and complete animal numbers can be found in Supplementary Table 2.
Extended Data Fig. 7 Arch inhibition of VM thalamus-PRL mPFC circuit on PC-Tsc1 mutant mice.
a, Three chambered social approach assay; time spent sniffing novel animal (NA), or novel object (NO) with 20 Hz (left) or 4 Hz (right) stimulation. b, Three chambered social novelty testing; time spent sniffing NA, or familiar animal (FA) with 20 Hz (left) or 4 Hz (right) stimulation. c, Time in the open arm and distance traveled in the elevated plus maze assay. d, Time in the center of the open field and distance traveled in the open field. All behavioral tests were analyzed with two or three-way ANOVA and Sidak post test. Box line denoted median/whiskers denoted 5-95%. N ≥ 10 for all behavioral experiments. ****P < 0.0001; ***P < 0.001; **P < 0.01; *P < 0.05. P values and complete animal numbers can be found in Supplementary Table 2.
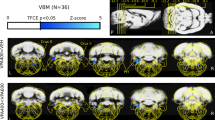
Extended Data Fig. 8 Structural covariance MRI of cerebellar vermis and mPFC.
Structural covariance MRI in mouse models of ASD vs. controls. Comparisons between vermis lobule VII (top), VIII (middle) or IX (bottom) and mPFC (left) or Prelimbic (PRL) mPFC (right). q values are stated. Each dot represents single imaged brain. Additional demographic information can be found in Supplementary Table 3.
Extended Data Fig. 9 Gi DREADDs inhibition of PCs in the posterior vermis and Gq DREADDs activation of posterior vermis in PC-Tsc1 mutant mice.
a, Injection sites from MN and LN tracing to VM thalamus (top) and sample locations of Gi (inhibitory) or Gq (excitatory) DREADD injections into posterior vermis of PC-Tsc1 mice (bottom). b, Three chambered social approach assay; time spent sniffing novel animal (NA), or novel object (NO). c, Three chambered social novelty testing; time spent sniffing NA, or familiar animal (FA). d, Time in the open arm and e, distance traveled in the elevated plus maze assay. f, Time in the center of the open field and g, distance traveled in the open field. h, Latency to fall in accelerating rotarod test. i, Three chambered social approach assay; time spent sniffing novel animal (NA), or novel object (NO). j, Three chambered social novelty testing; time sniffing novel animal (NA), or familiar animal (FA). k, Time in the open arm and distance traveled in the elevated plus maze assay. l, Time in the center of the open field and distance traveled in the open field. m, Latency to fall in accelerating rotarod test. All behavioral tests were analyzed with two or three-way ANOVA and Sidak post test. Box line denoted median/whiskers denoted 5-95%. n ≥ 10 for all behavioral tests. ****P < 0.0001; ***P < 0.001; **P < 0.01; *P < 0.05. P values and complete animal numbers can be found in Supplementary Table 2.
Extended Data Fig. 10 Raw frequency values for all normalized single unit graphs.
a, Frequency of firing of single units recorded in the left medial prefrontal cortex (mPFC) with control or inhibitory (Gi) DREADDs. b, Frequency of firing recorded in the left mPFC or motor cortex, with Rcrus1 Gi inhibition. c, Spike frequency in the mPFC with Rcrus1 Gi and mPFC Gi inhibition. d, Right mPFC spike frequency with Rcrus1 Gi. e, Spike Frequency in the Lateral Nucleus (LN) with LN Gi inhibition. f, mPFC spike frequency with LN Gi DREADDs. g, mPFC spike frequency with VM thalamic-mpfc Channel Rhodopsin (ChR2) activation of 4 Hz or h, 20 Hz. i, mPFC spike frequency with VM thalamus-mPFC Archaerhodopsin (Arch) inhibition at 4 Hz or j, 20 Hz. k, Parietal Association Cortex (PAC) firing with VM thalamic-mPFC ChR2 stimulation at 4 Hz and l, 20 Hz. m, mPFC spike frequency with posterior vermis Gi inhibition. ****P < 0.0001; ***P < 0.001; **P < 0.01; *P < 0.05. P values complete animal numbers can be found in Supplementary Table 2. Statistical analysis was done with the Wilcoxon matched pairs signed ranks test.
Supplementary information
Supplementary Information
Supplementary Tables 1–5.
Rights and permissions
About this article
Cite this article
Kelly, E., Meng, F., Fujita, H. et al. Regulation of autism-relevant behaviors by cerebellar–prefrontal cortical circuits. Nat Neurosci 23, 1102–1110 (2020). https://doi.org/10.1038/s41593-020-0665-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-020-0665-z
This article is cited by
-
Prebiotic diet normalizes aberrant immune and behavioral phenotypes in a mouse model of autism spectrum disorder
Acta Pharmacologica Sinica (2024)
-
Population-wide cerebellar growth models of children and adolescents
Nature Communications (2024)
-
Cerebellar state estimation enables resilient coupling across behavioural domains
Scientific Reports (2024)
-
Behaviour-correlated profiles of cerebellar-cerebral functional connectivity observed in independent neurodevelopmental disorder cohorts
Translational Psychiatry (2024)
-
A cerebro-cerebellar network for learning visuomotor associations
Nature Communications (2024)