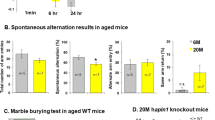
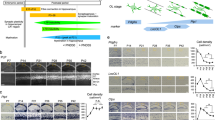
Abstract
Cognitive decline remains an unaddressed problem for the elderly. We show that myelination is highly active in young mice and greatly inhibited in aged mice, coinciding with spatial memory deficits. Inhibiting myelination by deletion of Olig2 in oligodendrocyte precursor cells impairs spatial memory in young mice, while enhancing myelination by deleting the muscarinic acetylcholine receptor 1 in oligodendrocyte precursor cells, or promoting oligodendroglial differentiation and myelination via clemastine treatment, rescues spatial memory decline during aging.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Data from this study are available from the corresponding author upon reasonable request.
References
Gazzaley, A., Cooney, J. W., Rissman, J. & D’Esposito, M. Top-down suppression deficit underlies working memory impairment in normal aging. Nat. Neurosci. 8, 1298–1300 (2005).
Garde, E. et al. Relation between age-related decline in intelligence and cerebral white-matter hyperintensities in healthy octogenarians: a longitudinal study. Lancet 356, 628–634 (2000).
Hughes, E. G. & Orthmann-Murphy, J. L. Myelin remodeling through experience-dependent oligodendrogenesis in the adult somatosensory cortex. Nat. Neurosci. 21, 696–706 (2018).
Young, K. M. et al. Oligodendrocyte dynamics in the healthy adult CNS: evidence for myelin remodeling. Neuron 77, 873–885 (2013).
Hill, R. A., Li, A. M. & Grutzendler, J. Lifelong cortical myelin plasticity and age-related degeneration in the live mammalian brain. Nat. Neurosci. 21, 683–695 (2018).
Lasiene, J. et al. Age-related myelin dynamics revealed by increased oligodendrogenesis and short internodes. Aging Cell 8, 201–213 (2009).
McKenzie, I. A. et al. Motor skill learning requires active central myelination. Science 346, 318–322 (2014).
Zatorre, R. J., Fields, R. D. & Johansen-Berg, H. Plasticity in gray and white: neuroimaging changes in brain structure during learning. Nat. Neurosci. 15, 528–536 (2012).
Hughes, E. G., Kang, S. H., Fukaya, M. & Bergles, D. E. Oligodendrocyte progenitors balance growth with self-repulsion to achieve homeostasis in the adult brain. Nat. Neurosci. 16, 668–676 (2013).
Kang, S. H. et al. NG2+ CNS glial progenitors remain committed to the oligodendrocyte lineage in postnatal life and following neurodegeneration. Neuron 68, 668–681 (2010).
Wang, F. et al. Enhancing oligodendrocyte myelination rescues synaptic loss and improves functional recovery after chronic hypoxia. Neuron 99, 689–701e5 (2018).
Mei, F. et al. Accelerated remyelination during inflammatory demyelination prevents axonal loss and improves functional recovery. eLife 5, 1–21 (2016).
Mei, F. et al. Stage-specific deletion of Olig2 conveys opposing functions on differentiation and maturation of oligodendrocytes. J. Neurosci. 33, 8454–8462 (2013).
Safaiyan, S. et al. Age-related myelin degradation burdens the clearance function of microglia during aging. Nat. Neurosci. 19, 995–998 (2016).
Cantuti-Castelvetri, L. & Fitzner, D. Defective cholesterol clearance limits remyelination in the aged central nervous system. Science 359, 684–688 (2018).
Shen, S. et al. Age-dependent epigenetic control of differentiation inhibitors is critical for remyelination efficiency. Nat. Neurosci. 11, 1024–1034 (2008).
Morrison, J. H. & Baxter, M. G. The ageing cortical synapse: hallmarks and implications for cognitive decline. Nat. Rev. Neurosci. 13, 240–250 (2012).
Mei, F. et al. Micropillar arrays as a high-throughput screening platform for therapeutics in multiple sclerosis. Nat. Med. 20, 954–960 (2014).
Ruckh, J. M. et al. Rejuvenation of regeneration in the aging central nervous system. Cell Stem Cell 10, 96–103 (2012).
Nave, K. A. Myelination and the trophic support of long axons. Nat. Rev. Neurosci. 11, 275–283 (2010).
Vorhees, C. V. & Williams, M. T. Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat. Protoc. 1, 848–858 (2006).
Acknowledgements
We thank J.J. Lawrence (Texas Tech University) and S. Tonegawa (MIT) for the Chrm1 floxed mice and Q.R. Lu (University of Cincinnati) for the Olig2 floxed mice. We appreciate the assistance of N.-X. Huang in image processing. This work was supported by the National Natural Science Foundation of China (grant nos. 31970916, 31771120), the Chongqing Education Commission Fund (CXQT19009) and the Chongqing Outstanding Young Investigator Fund Project (grant no. cstc2019jcyjjqx0001); and by the AMU Foundation (grant no. 2018JCZX02) to F.M., Beijing Natural Science Foundation (grant no. 7172156) to Z.-F.L., the NSFC (grant nos. 31671117, 31921003) to L.X., the National Institutes of Health/National Institute of Neurological Disorders and Stroke (grant nos. R01NS062796, R01NS097428, R01NS095889), the Adelson Medical Research Foundation: ANDP (grant no. A130141) and the Rachleff Family Endowment to J.R.C.
Author information
Authors and Affiliations
Contributions
F.W., S.-Y.R., J.-F.C., F.M., K.L., R.-X.L., Z.-F.L. and L.X. performed experiments. F.W., S.-Y.R., J.-F.C., L.X., R.-X.L., Z.-F.L., B.H., J.-Q.N., J.R.C. and F.M. provided reagents. F.M., S.-Y.R., J.R.C. and F.W. provided intellectual contributions. F.W., S.-Y.R., J.-F.C., F.M. and J.R.C. analyzed the data and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Age-related myelin and oligodendrocyte loss.
a, Representative images and quantification of mean MBP intensity, n = 3 biologically independent mice, one-way ANOVA followed by post hoc Tukey test was used (F(2,6) = 31.35, p < 0.001, TUKEY (4 M versus 13 M): p = 0.002, TUKEY (13 M versus 18 M): p = 0.001). Scale bar = 50 μm; b, Representative images and quantification of CC1 positive cells, n = 4 biologically independent mice, one-way ANOVA followed by post hoc Tukey test was used (F(2,9) = 8.466, p = 0.009, TUKEY (4 M versus 13 M): p = 0.026, TUKEY (13 M versus 18 M): p = 0.010), scale bar = 100 μm; c, Representative images and quantification of Cspg4 positive cells, n = 4 biologically independent mice, one-way ANOVA followed by post hoc Tukey test was used (F(2,9) = 0.086, p = 0.919), scale bar = 100 μm. Points represented individual animals. Error bars represent mean ± s.e.m. *p < 0.05, **p < 0.01.
Extended Data Fig. 2 Identification of mGFP positive cells.
a, Schematic illustration showing mGFP expression in the Cspg4-CreERt; Mapt-mGFP mice; b–d, Immunostaining indicates that mGFP positive cell bodies are CC1 positive (b) and mGFP positive segments are co-localized with MBP (c) and associated with NF200 positive axons (d). These experiments were repeated 3 times independently with similar results, scale bar = 20 μm.
Extended Data Fig. 3 Stability of myelin sheaths in PLP-CreERt; mT/mG mice.
a, Representative image showing mGFP positive areas are co-localized with MBP positive myelin sheaths in the cortex of PLP-CreERt; mT/mG mice, 10 days after recombination at 3 months. These experiments were repeated 3 times independently with similar results. Scale bar = 50 μm; b, Schematic diagram displaying the time course of tamoxifen induction and histology, and representative images and quantification of the mGFP positive areas in the layers I-III of the cortex 1 or 10 months after recombination at 3 months. These experiments were repeated 3 times with similar results, n = 3 biologically independent mice for each group, two-tailed unpaired t test was used (t(4) = 3.327, p = 0.0292). Scale bar = 100 μm. Points represented individual animals. Error bars represent mean ± s.e.m. *p < 0.05.
Extended Data Fig. 4 Recombination efficiency in 4- and 13-month old Cspg4-CreERt; mT/mG mice.
Representative images and quantification of the percentage of mGFP (green) positive cells in the Cspg4 (red) positive cells (OPCs) by immunostaining, 10 days after 4 consecutive days of tamoxifen treatment. Arrows indicating Cspg4/mGFP double positive cells, n = 3 biologically independent mice for each group, two-tailed unpaired t test was used (t(4) = 0.2774, p = 0.7953), scale bar = 100 μm. Points represented individual animals. Error bars represent mean ± s.e.m.
Extended Data Fig. 5 Expression of mGFP positive cells in the Cspg4-CreERt; mT/mG CNS.
a, Schematic illustration showing the mGFP expression pattern in the Cspg4-CreERt; mT/mG mice and representative image showing mGFP (green)/Cspg4 (gray) double positive OPCs, putative mGFP positive OLs (arrowhead) and mGFP (green)/mTomato (red) double positive pericytes and blood vessels (right lower panels, arrows), Scale bar: 20 μm; b, Immunostaining showing mGFP (green)/CC1 (red) double positive OLs (arrowheads), Scale bar: 20 μm (left panel); 10 μm (middle and right panel); c, mGFP (green)/MBP (red) double positive myelin sheaths (arrows), sale bar = 10 μm. These experiments were repeated 3 times independently with similar results.
Extended Data Fig. 6 Segregating mGFP positive pericytes, OPCs and OLs in the Cspg4-CreERt; mT/mG brains.
a, The blood vessels (red) /pericytes (green) (mT/mG double positive) are segregated and deducted from the mGFP channel, and the Cspg4 (red)/mGFP (green) double positive OPCs are pseudo-colored in red, Cspg4 immunostaining in Cspg4-CreERt; The experiments were repeated 3 times independently with similar results. Scale bar: 20μm (left panels on the top); 50μm (left panels on the bottom); 0.4 mm (right panels); b, Schematic diagram displaying the time course of tamoxifen induction and histology in the Cspg4-CreERt; mT/mG mice, and representative images of Cspg4 + /mGFP + OPCs (red) and Cspg4-/mGFP + new OLs (green, arrows) in the corpus callosum at 4-, 8- and 13 months, 10 days after induction and quantification of Cspg4 + /mGFP + OPC density and Cspg4-/mGFP + new myelin area, n = 3 biologically independent mice for each group, one-way ANOVA was used followed by post hoc Tukey test (Cspg4-/mGFP+area: F(2,6) = 97.587, p < 0.001, TUKEY (4 M versus 13 M): p = 0.001, TUKEY (8 M versus 13 M): p = 0.010; Cspg4+/mGFP+cell: F(2,6) = 0.066, p = 0.937). Scale bar = 0.4 mm. Points represented individual animals. Error bars represent mean ± s.e.m. **p < 0.01.
Extended Data Fig. 7 MBP expression in hippocampus at 13- or 18- months of age.
Representative images and quantification of mean MBP intensity in the CA2 region of the hippocampus. N = 3 biologically independent mice for each group, two-tailed unpaired t test was used (t(4) = 3.730, p = 0.0203). Scale bar = 100 μm. Points represented individual animals. Error bars represent mean ± s.e.m. *p < 0.05.
Extended Data Fig. 8 OPCs, OLs and microglia in Olig2 cKO brains.
a, Representative images and quantification of Iba1, Cspg4, CC1 positive cells in the motor cortex of Olig2 cKO and littermate controls, n = 3 biologically independent mice for the Iba1 and CC1 immunostaining in each group, and n = 4 biologically independent mice for the Cspg4 immunostaining in each group, two-tailed unpaired t test were used (Iba1:t(4) = 0.1257, p = 0.9060; Cspg4:t(6) = 0.2827, p = 0.7869; CC1:t(4) = 0.4201, p = 0.6960); b, Schematic diagram displaying the time course of tamoxifen induction and the water maze test. The Morris water maze test showing latency to platform in acquisition phase and numbers of platform crossing of the Olig2 fl/ + (n = 9 biologically independent mice) and Olig2 + / + (n = 10 biologically independent mice) mice. Two-way repeated ANOVA was used for the latencies to platform (F(1,17) = 0.130, p = 0.723), two-tailed unpaired t test was used for number of platform crossing in the target quadrant (t(17) = 0.03205, p = 0.9748). Scale bar: 100 μm. Points represented individual animals. Error bars represent mean ± s.e.m.
Extended Data Fig. 9 Chrm1 deletion in OPCs does not affect blood vessel density and microglia activation.
a, Schematic diagram displaying the time course of tamoxifen induction and histology; b, Representative images and quantification of mGFP positive myelin (yellow arrowheads) in the cortex of Chrm1 cKO mice and age-matched wildtypes, scale bar = 0.2 mm; c, Representative images and quantification of CD31 positive area in the cortex of Chrm1 cKO mice and age-matched wildtypes, Scale bar = 50μm; d, Representative images and quantification of Iba1 positive microglia in the cortex of Chrm1 cKO mice and age-matched wildtypes, Scale bar = 50 μm, n = 3 biologically independent mice for each group for all experiments, two-tailed unpaired t test were used for each group in all experiments (mGFP:t(4) = 11.37, p < 0.001; CD31:t(4) = 1.269, p = 0.2733; Iba1:t(4) = 0.2773, p = 0.7953); Inserted images showing 3D Iba1 positive cells, scale bar = 10 μm. Points represented individual animals. Error bars represent mean ± s.e.m. ***p < 0.001.
Supplementary information
Rights and permissions
About this article
Cite this article
Wang, F., Ren, SY., Chen, JF. et al. Myelin degeneration and diminished myelin renewal contribute to age-related deficits in memory. Nat Neurosci 23, 481–486 (2020). https://doi.org/10.1038/s41593-020-0588-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-020-0588-8
This article is cited by
-
Microglia regulation of central nervous system myelin health and regeneration
Nature Reviews Immunology (2024)
-
Clemastine and metformin extend the window of NMDA receptor surface expression in ageing oligodendrocyte precursor cells
Scientific Reports (2024)
-
Long-term in vivo three-photon imaging reveals region-specific differences in healthy and regenerative oligodendrogenesis
Nature Neuroscience (2024)
-
Functional myelin in cognition and neurodevelopmental disorders
Cellular and Molecular Life Sciences (2024)
-
Myelin in Alzheimer’s disease: culprit or bystander?
Acta Neuropathologica Communications (2023)