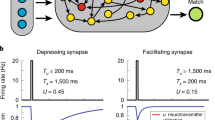
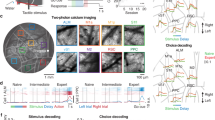
Abstract
Cortical neurons process information on multiple timescales, and areas important for working memory (WM) contain neurons capable of integrating information over a long timescale. However, the underlying mechanisms for the emergence of neuronal timescales stable enough to support WM are unclear. By analyzing a spiking recurrent neural network model trained on a WM task and activity of single neurons in the primate prefrontal cortex, we show that the temporal properties of our model and the neural data are remarkably similar. Dissecting our recurrent neural network model revealed strong inhibitory-to-inhibitory connections underlying a disinhibitory microcircuit as a critical component for long neuronal timescales and WM maintenance. We also found that enhancing inhibitory-to-inhibitory connections led to more stable temporal dynamics and improved task performance. Finally, we show that a network with such microcircuitry can perform other tasks without disrupting its pre-existing timescale architecture, suggesting that strong inhibitory signaling underlies a flexible WM network.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The trained RNN models used in the present study are deposited as MATLAB-formatted data in Open Science Framework, https://osf.io/md4wg. The experimental data used in the study can be obtained from Constantinidis et al.21.
Code availability
The code for the analyses performed in this work is available at https://github.com/rkim35/wmRNN.
References
Murray, J. D. et al. A hierarchy of intrinsic timescales across primate cortex. Nat. Neurosci. 17, 1661–1663 (2014).
Chaudhuri, R., Knoblauch, K., Gariel, M.-A., Kennedy, H. & Wang, X.-J. A large-scale circuit mechanism for hierarchical dynamical processing in the primate cortex. Neuron 88, 419–431 (2015).
Cavanagh, S. E., Wallis, J. D., Kennerley, S. W. & Hunt, L. T. Autocorrelation structure at rest predicts value correlates of single neurons during reward-guided choice. eLife 5, e18937 (2016).
Miller, E. K., Erickson, C. A. & Desimone, R. Neural mechanisms of visual working memory in prefrontal cortex of the macaque. J. Neurosci. 16, 5154–5167 (1996).
Fuster, J. M. & Alexander, G. E. Neuron activity related to short-term memory. Science 173, 652–654 (1971).
Fascianelli, V., Tsujimoto, S., Marcos, E. & Genovesio, A. Autocorrelation structure in the macaque dorsolateral, but not orbital or polar, prefrontal cortex predicts response-coding strength in a visually cued strategy task. Cereb. Cortex 29, 230–241 (2017).
Cavanagh, S. E., Towers, J. P., Wallis, J. D., Hunt, L. T. & Kennerley, S. W. Reconciling persistent and dynamic hypotheses of working memory coding in prefrontal cortex. Nat. Commun. 9, 3498 (2018).
Wasmuht, D. F., Spaak, E., Buschman, T. J., Miller, E. K. & Stokes, M. G. Intrinsic neuronal dynamics predict distinct functional roles during working memory. Nat. Commun. 9, 3499 (2018).
Kim, D. et al. Distinct roles of parvalbumin- and somatostatin-expressing interneurons in working memory. Neuron 92, 902–915 (2016).
Kamigaki, T. & Dan, Y. Delay activity of specific prefrontal interneuron subtypes modulates memory-guided behavior. Nat. Neurosci. 20, 854–863 (2017).
Xu, H. et al. A disinhibitory microcircuit mediates conditioned social fear in the prefrontal cortex. Neuron 102, 668–682 (2019).
Cummings, K. A. & Clem, R. L. Prefrontal somatostatin interneurons encode fear memory. Nat. Neurosci. 23, 61–74 (2019).
Krabbe, S. et al. Adaptive disinhibitory gating by VIP interneurons permits associative learning. Nat. Neurosci. 22, 1834–1843 (2019).
Pfeffer, C. K., Xue, M., He, M., Huang, Z. J. & Scanziani, M. Inhibition of inhibition in visual cortex: the logic of connections between molecularly distinct interneurons. Nat. Neurosci. 16, 1068–1076 (2013).
Tremblay, R., Lee, S. & Rudy, B. GABAergic interneurons in the neocortex: from cellular properties to circuits. Neuron 91, 260–292 (2016).
Medalla, M., Gilman, J. P., Wang, J.-Y. & Luebke, J. I. Strength and diversity of inhibitory signaling differentiates primate anterior cingulate from lateral prefrontal cortex. J. Neurosci. 37, 4717–4734 (2017).
Mongillo, G., Rumpel, S. & Loewenstein, Y. Inhibitory connectivity defines the realm of excitatory plasticity. Nat. Neurosci. 21, 1463–1470 (2018).
Kim, R., Li, Y. & Sejnowski, T. J. Simple framework for constructing functional spiking recurrent neural networks. Proc. Natl Acad. Sci. USA 116, 22811–22820 (2019).
Qi, X.-L., Meyer, T., Stanford, T. R. & Constantinidis, C. Changes in prefrontal neuronal activity after learning to perform a spatial working memory task. Cereb. Cortex 21, 2722–2732 (2011).
Meyer, T., Qi, X.-L., Stanford, T. R. & Constantinidis, C. Stimulus selectivity in dorsal and ventral prefrontal cortex after training in working memory tasks. J. Neurosci. 31, 6266–6276 (2011).
Constantinidis, C., Qi, X.-L. & Meyer, T. Single-neuron spike train recordings from macaque prefrontal cortex during a visual working memory task before and after training. CRCNS https://doi.org/10.6080/K0ZW1HVD (2016).
Stokes, M. G. et al. Dynamic coding for cognitive control in prefrontal cortex. Neuron 78, 364–375 (2013).
Spaak, E., Watanabe, K., Funahashi, S. & Stokes, M. G. Stable and dynamic coding for working memory in primate prefrontal cortex. J. Neurosci. 37, 6503–6516 (2017).
Miller, E. K. & Cohen, J. D. An integrative theory of prefrontal cortex function. Annu. Rev. Neurosci. 24, 167–202 (2001).
Goldman-Rakic, P. S. in Comprehensive Physiology (ed. Terjung, R. L.) 373–417 (American Cancer Society, 2011).
Meyer, T., Qi, X.-L. & Constantinidis, C. Persistent discharges in the prefrontal cortex of monkeys naïve to working memory tasks. Cereb. Cortex 17, i70–i76 (2007).
Yang, G. R., Murray, J. D. & Wang, X.-J. A dendritic disinhibitory circuit mechanism for pathway-specific gating. Nat. Commun. 7, 12815 (2016).
Wang, X.-J. & Yang, G. R. A disinhibitory circuit motif and flexible information routing in the brain. Curr. Opin. Neurobiol. 49, 75–83 (2018).
Mante, V., Sussillo, D., Shenoy, K. V. & Newsome, W. T. Context-dependent computation by recurrent dynamics in prefrontal cortex. Nature 503, 78–84 (2013).
Bouchacourt, F. & Buschman, T. J. A flexible model of working memory. Neuron 103, 147–160 (2019).
Song, H. F., Yang, G. R. & Wang, X.-J. Training excitatory-inhibitory recurrent neural networks for cognitive tasks: a simple and flexible framework. PLoS Comput. Biol. 12, e1004792 (2016).
Miconi, T. Biologically plausible learning in recurrent neural networks reproduces neural dynamics observed during cognitive tasks. eLife 6, e20899 (2017).
Orhan, A. E. & Ma, W. J. A diverse range of factors affect the nature of neural representations underlying short-term memory. Nat. Neurosci. 22, 275–283 (2019).
Yang, G. R., Joglekar, M. R., Song, H. F., Newsome, W. T. & Wang, X.-J. Task representations in neural networks trained to perform many cognitive tasks. Nat. Neurosci. 22, 297–306 (2019).
Kepecs, A. & Fishell, G. Interneuron cell types are fit to function. Nature 505, 318–326 (2014).
Batista-Brito, R. et al. Developmental dysfunction of VIP interneurons impairs cortical circuits. Neuron 95, 884–895.e9 (2017).
Pi, H.-J. et al. Cortical interneurons that specialize in disinhibitory control. Nature 503, 521–524 (2013).
Karnani, M. M. et al. Opening holes in the blanket of inhibition: localized lateral disinhibition by VIP interneurons. J. Neurosci. 36, 3471–3480 (2016).
Wang, X.-J. Probabilistic decision making by slow reverberation in cortical circuits. Neuron 36, 955–968 (2002).
Kim, Y. et al. Brain-wide maps reveal stereotyped cell-type-based cortical architecture and subcortical sexual dimorphism. Cell 171, 456–469.e22 (2017).
Medalla, M. & Barbas, H. Synapses with inhibitory neurons differentiate anterior cingulate from dorsolateral prefrontal pathways associated with cognitive control. Neuron 61, 609–620 (2009).
Wimmer, K., Nykamp, D. Q., Constantinidis, C. & Compte, A. Bump attractor dynamics in prefrontal cortex explains behavioral precision in spatial working memory. Nat. Neurosci. 17, 431–439 (2014).
Maris, E. & Oostenveld, R. Nonparametric statistical testing of EEG and MEG data. J. Neurosci. Methods 164, 177–190 (2007).
Acknowledgements
We are grateful to B. Tsuda, Y. Chen and J. Fleischer for helpful discussions and feedback on the manuscript. We also thank J. Aldana for assistance with computing resources. This work was funded by the National Institute of Mental Health (grant no. F30MH115605-01A1 to R.K.). We also gratefully acknowledge the support of NVIDIA Corporation with the donation of the Quadro P6000 graphics processing unit used for this research. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
R.K. and T.J.S. designed the study and wrote the manuscript. R.K. performed the analyses and simulations.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Neuroscience thanks Dean Buonomano, Timothy Buschman, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–14.
Rights and permissions
About this article
Cite this article
Kim, R., Sejnowski, T.J. Strong inhibitory signaling underlies stable temporal dynamics and working memory in spiking neural networks. Nat Neurosci 24, 129–139 (2021). https://doi.org/10.1038/s41593-020-00753-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-020-00753-w
This article is cited by
-
Synaptic wiring motifs in posterior parietal cortex support decision-making
Nature (2024)
-
Intracortical recordings reveal vision-to-action cortical gradients driving human exogenous attention
Nature Communications (2024)
-
Highly biomimetic spiking neuron using SiGe heterojunction bipolar transistors for energy-efficient neuromorphic systems
Scientific Reports (2024)
-
Multiplexing working memory and time in the trajectories of neural networks
Nature Human Behaviour (2023)
-
Choice selective inhibition drives stability and competition in decision circuits
Nature Communications (2023)