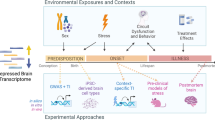
Abstract
Despite extensive study of the neurobiological correlates of post-traumatic stress disorder (PTSD), little is known about its molecular determinants. Here, differential gene expression and network analyses of four prefrontal cortex subregions from postmortem tissue of people with PTSD demonstrate extensive remodeling of the transcriptomic landscape. A highly connected downregulated set of interneuron transcripts is present in the most significant gene network associated with PTSD. Integration of this dataset with genotype data from the largest PTSD genome-wide association study identified the interneuron synaptic gene ELFN1 as conferring significant genetic liability for PTSD. We also identified marked transcriptomic sexual dimorphism that could contribute to higher rates of PTSD in women. Comparison with a matched major depressive disorder cohort revealed significant divergence between the molecular profiles of individuals with PTSD and major depressive disorder despite their high comorbidity. Our analysis provides convergent systems-level evidence of genomic networks within the prefrontal cortex that contribute to the pathophysiology of PTSD in humans.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Code availability
All codes used in this study are freely available online and can be found at https://github.com/mjgirgenti/PTSDCorticalTranscriptomics.
References
Mota, N. et al. Late-life exacerbation of PTSD symptoms in US veterans: results from the National Health and Resilience in Veterans Study. J. Clin. Psychiatry 77, 348–354 (2016).
McLaughlin, K. A. et al. Subthreshold posttraumatic stress disorder in the World Health Organization world mental health surveys. Biol. Psychiatry 77, 375–384 (2015).
Jovanovic, T. et al. Impaired fear inhibition is a biomarker of PTSD but not depression. Depress. Anxiety 27, 244–251 (2010).
Yehuda, R. & LeDoux, J. Response variation following trauma: a translational neuroscience approach to understanding PTSD. Neuron 56, 19–32 (2007).
Koenen, K. C. et al. A high risk twin study of combat-related PTSD comorbidity. Twin Res. 6, 218–226 (2003).
Koenen, K. C. et al. A twin registry study of the relationship between posttraumatic stress disorder and nicotine dependence in men. Arch. Gen. Psychiatry 62, 1258–1265 (2005).
Gelernter, J. et al. Genome-wide association study of post-traumatic stress disorder reexperiencing symptoms in >165,000 US veterans. Nat. Neurosci. 22, 1394–1401 (2019).
Nievergelt, C. M. et al. International meta-analysis of PTSD genome-wide association studies identifies sex- and ancestry-specific genetic risk loci. Nat. Commun. 10, 4558 (2019).
Stein, M. B. et al. Genomic characterization of posttraumatic stress disorder in a large US military veteran sample. Preprint at bioRxiv https://doi.org/10.1101/764001 (2019).
Kessler, R. C. et al. The global burden of mental disorders: an update from the WHO World Mental Health (WMH) surveys. Epidemiol. Psichiat Soc. 18, 23–33 (2009).
Holbrook, T. L., Hoyt, D. B., Stein, M. B. & Sieber, W. J. Gender differences in long-term posttraumatic stress disorder outcomes after major trauma: women are at higher risk of adverse outcomes than men. J. Trauma 53, 882–888 (2002).
Baran, S. E., Armstrong, C. E., Niren, D. C. & Conrad, C. D. Prefrontal cortex lesions and sex differences in fear extinction and perseveration. Learn. Mem. 17, 267–278 (2010).
Goldstein, J. M., Jerram, M., Abbs, B., Whitfield-Gabrieli, S. & Makris, N. Sex differences in stress response circuitry activation dependent on female hormonal cycle. J. Neurosci. 30, 431–438 (2010).
Labonté, B. et al. Sex-specific transcriptional signatures in human depression. Nat. Med. 23, 1102–1111 (2017).
Nees, F., Witt, S. H. & Flor, H. Neurogenetic approaches to stress and fear in humans as pathophysiological mechanisms for posttraumatic stress disorder. Biol. Psychiatry 83, 810–820 (2018).
Young, K. A., Thompson, P. M., Cruz, D. A., Williamson, D. E. & Selemon, L. D. BA11 FKBP5 expression levels correlate with dendritic spine density in postmortem PTSD and controls. Neurobiol. Stress 2, 67–72 (2015).
Meng, L. et al. Trauma-specific grey matter alterations in PTSD. Sci. Rep. 6, 33748 (2016).
Bonne, O. et al. Resting regional cerebral perfusion in recent posttraumatic stress disorder. Biol. Psychiatry 54, 1077–1086 (2003).
Britton, J. C., Phan, K. L., Taylor, S. F., Fig, L. M. & Liberzon, I. Corticolimbic blood flow in posttraumatic stress disorder during script-driven imagery. Biol. Psychiatry 57, 832–840 (2005).
Jovanovic, T. et al. Reduced neural activation during an inhibition task is associated with impaired fear inhibition in a traumatized civilian sample. Cortex 49, 1884–1891 (2013).
Averill, L. A. et al. Glutamate dysregulation and glutamatergic therapeutics for PTSD: evidence from human studies. Neurosci. Lett. 649, 147–155 (2017).
Ardi, Z., Albrecht, A., Richter-Levin, A., Saha, R. & Richter-Levin, G. Behavioral profiling as a translational approach in an animal model of posttraumatic stress disorder. Neurobiol. Dis. 88, 139–147 (2016).
Sun, X., Song, Z., Si, Y. & Wang, J.-H. microRNA and mRNA profiles in ventral tegmental area relevant to stress-induced depression and resilience. Prog. Neuropsychopharmacol. Biol. Psychiatry 86, 150–165 (2018).
Geuze, E. et al. Reduced GABAA benzodiazepine receptor binding in veterans with post-traumatic stress disorder. Mol. Psychiatry 13, 74–83 (2008).
Möller, A. T., Bäckström, T., Nyberg, S., Söndergaard, H. P. & Helström, L. Women with PTSD have a changed sensitivity to GABA-A receptor active substances. Psychopharmacology 233, 2025–2033 (2016).
Rosso, I. M. et al. Insula and anterior cingulate GABA levels in posttraumatic stress disorder: preliminary findings using magnetic resonance spectroscopy. Depress. Anxiety 31, 115–123 (2014).
Sheth, C. et al. Reduced gamma-amino butyric Acid (GABA) and glutamine in the anterior cingulate cortex (ACC) of veterans exposed to trauma. J. Affect. Disord. 248, 166–174 (2019).
Vaiva, G. et al. Relationship between posttrauma GABA plasma levels and PTSD at 1-year follow-up. Am. J. Psychiatry 163, 1446–1448 (2006).
Passos, I. C. et al. Inflammatory markers in post-traumatic stress disorder: a systematic review, meta-analysis, and meta-regression. Lancet Psychiatry 2, 1002–1012 (2015).
Kroes, M. C. W., Rugg, M. D., Whalley, M. G. & Brewin, C. R. Structural brain abnormalities common to posttraumatic stress disorder and depression. J. Psychiatry Neurosci. 36, 256–265 (2011).
Powers, A. et al. Neural correlates and structural markers of emotion dysregulation in traumatized civilians. Soc. Cogn. Affect. Neursci. 12, 823–831 (2017).
Philip, N. S. et al. Network mechanisms of clinical response to transcranial magnetic stimulation in posttraumatic stress disorder and major depressive disorder. Biol. Psychiatry 83, 263–272 (2018).
Duncan, L. E. et al. Largest GWAS of PTSD (N=20 070) yields genetic overlap with schizophrenia and sex differences in heritability. Mol. Psychiatry 23, 666–673 (2017).
Gandal, M. J. et al. Shared molecular neuropathology across major psychiatric disorders parallels polygenic overlap. Science 359, 693–697 (2018).
Dunn, H. A., Patil, D. N., Cao, Y., Orlandi, C. & Martemyanov, K. A. Synaptic adhesion protein ELFN1 is a selective allosteric modulator of group III metabotropic glutamate receptors in trans. Proc. Natl Acad. Sci. USA 115, 5022–5027 (2018).
Stachniak, T. J., Sylwestrak, E. L., Scheiffele, P., Hall, B. J. & Ghosh, A. Elfn1-induced constitutive activation of mGluR7 determines frequency-dependent recruitment of somatostatin interneurons. J. Neurosci. 39, 4461–4474 (2019).
Sun, A. X. et al. Direct induction and functional maturation of forebrain GABAergic neurons from human pluripotent stem cells. Cell Rep. 16, 1942–1953 (2016).
Gusev, A. et al. Integrative approaches for large-scale transcriptome-wide association studies. Nat. Genet. 48, 245–252 (2016).
GTEx Consortium The genotype–tissue expression (GTEx) pilot analysis: multitissue gene regulation in humans. Science 348, 648–660 (2015).
Rytwinski, N. K., Scur, M. D., Feeny, N. C. & Youngstrom, E. A. The co-occurrence of major depressive disorder among individuals with posttraumatic stress disorder: a meta analysis. J. Trauma. Stress 26, 299–309 (2013).
Seney, M. L. et al. Opposite molecular signatures of depression in men and women. Biol. Psychiatry 84, 18–27 (2018).
Seney, M. L., Tripp, A., McCune, S., Lewis, D. A. & Sibille, E. Laminar and cellular analyses of reduced somatostatin gene expression in the subgenual anterior cingulate cortex in major depression. Neurobiol. Dis. 73, 213–219 (2015).
Lewis, D. A. & Sweet, R. A. Schizophrenia from a neural circuitry perspective: advancing toward rational pharmacological therapies. J. Clin. Invest. 119, 706–716 (2009).
Peters, J., Dieppa-Perea, L. M., Melendez, L. M. & Quirk, G. J. Induction of fear extinction with hippocampal–infralimbic BDNF. Science 328, 1288–1290 (2010).
Etkin, A. & Wager, T. D. Functional neuroimaging of anxiety: a meta-analysis of emotional processing in PTSD, social anxiety disorder, and specific phobia. Am. J. Psychiatry 164, 1476–1488 (2007).
Przanowski, P. et al. The signal transducers Stat1 and Stat3 and their novel target Jmjd3 drive the expression of inflammatory genes in microglia. J. Mol. Med. 92, 239–254 (2013).
Licznerski, P. et al. Decreased SGK1 expression and function contributes to behavioral deficits induced by traumatic stress. PLoS Biol. 13, e1002282 (2015).
Holmes, S. E. et al. Altered metabotropic glutamate receptor 5 markers in PTSD: in vivo and postmortem evidence. Proc. Natl Acad. Sci. USA 114, 8390–8395 (2017).
Koenen, K. C. et al. Polymorphisms in FKBP5 are associated with peritraumatic dissociation in medically injured children. Mol. Psychiatry 10, 1058–1059 (2005).
Labonté, B. et al. Gadd45b mediates depressive-like role through DNA demethylation. Sci. Rep. 9, 4615 (2019).
Spitzer, R. L., Williams, J. B. W., Gibbon, M. & First, M. B. The structured clinical interview for DSM-III-R (SCID): I: history, rationale, and description. Arch. Gen. Psychiatry 49, 624–629 (1992).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Jaffe, A. E. et al. Developmental and genetic regulation of the human cortex transcriptome illuminate schizophrenia pathogenesis. Nat. Neurosci. 21, 1117–1125 (2018).
Feng, Y.-Y. et al. RegTools: integrated analysis of genomic and transcriptomic data for discovery of splicing variants in cancer. Preprint at bioRxiv https://doi.org/10.1101/436634 (2020).
Kim, D. et al. TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol. 14, R36 (2013).
Raudvere, U. et al. g:Profiler: a web server for functional enrichment analysis and conversions of gene lists (2019 update). Nucleic Acids Res. 47, W191–W198 (2019).
Langfelder, P. & Horvath, S. WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics 9, 559 (2008).
Zhang, B. & Horvath, S. A general framework for weighted gene co-expression network analysis. Stat. Appl. Genet. Mol. Biol. 4, Article17 (2005).
Dougherty, J. D., Schmidt, E. F., Nakajima, M. & Heintz, N. Analytical approaches to RNA profiling data for the identification of genes enriched in specific cells. Nucleic Acids Res. 38, 4218–4230 (2010).
Xu, X., Wells, A. B., O’Brien, D. R., Nehorai, A. & Dougherty, J. D. Cell type-specific expression analysis to identify putative cellular mechanisms for neurogenetic disorders. J. Neurosci. 34, 1420–1431 (2014).
Zhang, Y. et al. Purification and characterization of progenitor and mature human astrocytes reveals transcriptional and functional differences with mouse. Neuron 89, 37–53 (2016).
Margolin, A. A. et al. ARACNE: an algorithm for the reconstruction of gene regulatory networks in a mammalian cellular context. BMC Bioinformatics 7 Suppl. 1, S7 (2006).
Hu, Y. et al. A statistical framework for cross-tissue transcriptome-wide association analysis. Nat. Genet. 51, 568–576 (2019).
Newman, A. M. et al. Determining cell type abundance and expression from bulk tissues with digital cytometry. Nat. Biotechnol. 37, 773–782 (2019).
Zhu, Y. et al. Spatiotemporal transcriptomic divergence across human and macaque brain development. Science 362, eaat8077 (2018).
Acknowledgements
We thank M. Picciotto, C. Pittenger and A. Che for critically reading the manuscript. We thank R. Terwilliger for technical assistance. We are grateful to the families who donated to this research. This work was supported with resources and use of facilities at the VA Connecticut Health Care System, West Haven, CT, the Central Texas Veterans Health Care System, Temple, TX, the Durham VA Healthcare System, Durham, NC, the VA San Diego Healthcare System, La Jolla, CA, the VA Boston Healthcare System, Boston, MA, USA, and the National Center for PTSD, US Department of Veterans Affairs. The research reported here was supported by the Department of Veterans Affairs, Veteran Health Administration, VISN1 Career Development Award and a Brain and Behavior Research Foundation Young Investigator Award to M.J.G. and by NIMH grants MH093897 and MH105910 to R.S.D. The views expressed here are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs (VA) or the US government.
Author information
Authors and Affiliations
Consortia
Contributions
M.J.G., M.J.F., J.H.K. and R.S.D. conceived the project, designed the experiments and wrote the manuscript. M.J.G. also generated and analyzed all of the data. J.W., D.J. and H.Z. oversaw all bioinformatics analyses for gene expression, network analysis and TWAS. M.B.S. and J.G. contributed GWAS data. D.A.C., K.A.Y., B.R.H. and D.E.W. contributed to the study design. All authors contributed to the preparation of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
J.G. is named as a co-inventor on PCT patent application number 15/878,640 entitled “Genotype-guided dosing of opioid agonists”, filed 24 January 2018. M.B.S. has in the past 3 years been a consultant for Aptinyx, Bionomics, EpiVario, Greenwich Biosciences, Janssen, and Jazz Pharmaceuticals. He also receives payment from the following entities for editorial work: Biological Psychiatry (published by Elsevier), Depression and Anxiety (published by Wiley) and UpToDate. J.H.K. has consulting agreements (less than US$10,000 per year) with the following: AstraZeneca Pharmaceuticals, Biogen, Idec, MA, Biomedisyn Corporation, Bionomics, Limited (Australia), Boehringer Ingelheim International, COMPASS Pathways, Limited, United Kingdom, Concert Pharmaceuticals, Inc., Epiodyne, Inc., EpiVario, Inc., Heptares Therapeutics, Limited (UK), Janssen Research & Development, Otsuka America, Pharmaceutical, Inc., Perception Neuroscience Holdings, Inc., Spring Care, Inc., Sunovion Pharmaceuticals, Inc., Takeda Industries and Taisho Pharmaceutical Co., Ltd. J.H.K. serves on the scientific advisory boards of Bioasis Technologies, Inc., Biohaven Pharmaceuticals, BioXcel Therapeutics, Inc. (Clinical Advisory Board), BlackThorn Therapeutics, Inc., Cadent Therapeutics (Clinical Advisory Board), Cerevel Therapeutics, LLC., EpiVario, Inc., Lohocla Research Corporation, PsychoGenics, Inc.; is on the board of directors of Inheris Biopharma, Inc.; has stock options with Biohaven Pharmaceuticals Medical Sciences, BlackThorn Therapeutics, Inc., EpiVario, Inc. and Terran Life Sciences; and is editor of Biological Psychiatry with income greater than $10,000. R.S.D. has received consulting fees from Taisho, Johnson & Johnson and Naurex; and receives grant support from Taisho, Johnson & Johnson, Naurex, Navitor, Allergan, Lundbeck and Lilly. None of the above listed companies or funding agencies had any influence on the content of this article.
Additional information
Peer review information Nature Neuroscience thanks Douglas Bremner and Michael Gandal for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Transcriptomic concordance comparisons between PTSD and other neuropsychiatric disorders.
a, PTSD transcriptomic profiles were compared with 5 other neuropsychiatric disorders. Individual cortical regions were compared to meta-analyzed transcriptomic signatures for autism spectrum disorder (ASD), schizophrenia (SCZ), bipolar disorder (BP), major depression disorder (MDD(*)), alcohol abuse disorder (AAD), and control disease irritable bowel syndrome (IBS). There was a significant correlation between PTSD and AAD in the dACC. b, Comparison of psychiatric control cohort MDD transcriptomic signature to the 5 psychiatric disorders. Error bars indicate mean ± s.d. **bootstrap P=0.007, *bootstrap P=0.04, n= 9304 genes. P values were calculated with null distribution from bootstrap and not corrected for multiple comparisons.
Extended Data Fig. 2 Extended WGCNA of PTSD combined-sex modules.
a, Extended circos plot from Fig. 2a, including complete regional break down and cell type enrichment for each module. Each slice of the chart represents a coexpression module. The key for the circos plot is on the left. The outermost rectangle is the arbitrary color name. The outermost concentric circles represent degree to which DEGs are contained within the module for each brain region and the inner concentric circles indicate cell-type enrichment (colors reflect corrected FET P-values with key provided). b, Gene ontology analysis of each module with at least 2 GO terms. Top two significant ontologies are plotted.
Extended Data Fig. 3 Sex-specific PTSD associated modules with enrichment of cell-type markers.
Cell type enrichment was found in 22 modules in the combined-sex comparison (a), 23 female specific modules (b), and 23 male specific modules (c).
Extended Data Fig. 4 RNA-seq cell type deconvolution and population size estimate for all regions in PTSD and MDD.
Individual cell type population proportions for control (salmon), MDD (green), and PTSD (blue) in dlPFC(a), OFC(b), dACC(c), and sgPFC(d). ExN, excitatory neuron; IntN, interneuron; Oligo, oligodendrocyte; Astro, astrocyte; OPC, oligodendrocyte precursor cell; Vsmc, vascular smooth muscle cell; Endo, endothelial cell. Error bars indicate mean ± s.e.m. one-way ANOVA by diagnosis and region. Box border maxima and minima are ± 1.5 s.d., box borders are 25% and 75% of quantile and the center is the median.
Extended Data Fig. 5 Ontological terms associated with sex-specific regional transcriptomic changes.
Top gene ontology (GO) enrichments for all differentially expressed features across female OFC (a), female sgPFC (b) and male dlPFC (c) including cell compartment, molecular function, and biological function ontologies.
Extended Data Fig. 6 Extended WGCNA of PTSD sex-specific modules and gene ontologies.
Extended circos plot from Fig. 4f (a) and g(b), including complete regional break down and cell type enrichment for each module. Each slice of the chart represents a coexpression module. The key for the circos plot is on the left. The outermost rectangle is the arbitrary color name. The outermost concentric circles represent degree to which DEGs are contained within the module for each brain region and the inner concentric circles indicate cell-type enrichment (colors reflect corrected FET P-values with key provided). Gene ontology analysis of each female-specific modules (c) and male-specific modules (d) with at least 2 GO terms. Top two significant ontologies are plotted.
Extended Data Fig. 7 Differential expression profiles in humans with MDD reveal sex-specific transcriptomic profiles across PFC subregions.
a, Volcano plots display differential regulation of genes in PFC of all MDD cases. b, Volcano plots display sex-specific DEGs in females (top) and males (bottom) across PFC subregions. Blue dots indicate down regulation and red dots indicate up regulation compared to control.
Extended Data Fig. 8 Module correlation between PTSD and MDD gene co-expression modules.
The quantile of correlation among 2000 permutations was calculated for each PTSD and MDD module. Blue line indicates modules above the 97.5% quantile that are significantly convergent and the red line indicates the bottom 2.5% quantile that are significantly divergent. In the combined sex comparison(a) there are 16 convergent modules and 12 divergent modules, in females there are 9 convergent and 19 divergent modules(b), and in males there are 15 convergent and 5 divergent modules(c).
Extended Data Fig. 9 Sex- and cell-type specific network convergence of PTSD and MDD.
a, Female and male module association across PTSD (y-axis) and MDD (x-axis). Plots show β values of module eigengene association with disease. Cell type marker enrichment of combined sex (b), female (c) and male (d) modules in combined disease network analysis.
Supplementary information
Supplementary Information
Supplementary Figs. 1–3, and consortia authors for the Traumatic Stress Group and the MVP.
Supplementary Tables
Supplementary Tables 1–10.
Supplementary Software 1
All code used in the manuscript.
Rights and permissions
About this article
Cite this article
Girgenti, M.J., Wang, J., Ji, D. et al. Transcriptomic organization of the human brain in post-traumatic stress disorder. Nat Neurosci 24, 24–33 (2021). https://doi.org/10.1038/s41593-020-00748-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-020-00748-7
This article is cited by
-
Unraveling the cell-type-specific molecular pathways of PTSD: integrating GWAS with brain genomic profiling and in vitro modeling
Neuropsychopharmacology (2024)
-
Fear memory regulation by the cAMP signaling pathway as an index of reexperiencing symptoms in posttraumatic stress disorder
Molecular Psychiatry (2024)
-
Synaptotagmin-4 induces anhedonic responses to chronic stress via BDNF signaling in the medial prefrontal cortex
Experimental & Molecular Medicine (2024)
-
Effects of sex and gender on the etiologies and presentation of select internalizing psychopathologies
Translational Psychiatry (2024)
-
Genome-wide association analyses identify 95 risk loci and provide insights into the neurobiology of post-traumatic stress disorder
Nature Genetics (2024)