Abstract
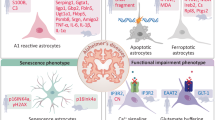
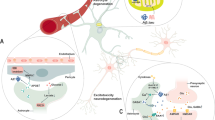
Although the pathological contributions of reactive astrocytes have been implicated in Alzheimer’s disease (AD), their in vivo functions remain elusive due to the lack of appropriate experimental models and precise molecular mechanisms. Here, we show the importance of astrocytic reactivity on the pathogenesis of AD using GiD, a newly developed animal model of reactive astrocytes, where the reactivity of astrocytes can be manipulated as mild (GiDm) or severe (GiDs). Mechanistically, excessive hydrogen peroxide (H2O2) originated from monoamine oxidase B in severe reactive astrocytes causes glial activation, tauopathy, neuronal death, brain atrophy, cognitive impairment and eventual death, which are significantly prevented by AAD-2004, a potent H2O2 scavenger. These H2O2−-induced pathological features of AD in GiDs are consistently recapitulated in a three-dimensional culture AD model, virus-infected APP/PS1 mice and the brains of patients with AD. Our study identifies H2O2 from severe but not mild reactive astrocytes as a key determinant of neurodegeneration in AD.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available in the Mendeley data repository (https://data.mendeley.com/) with DOI https://doi.org/10.17632/8mf35ntz9z.1. The datasets generated for the supplementary figures are available from the corresponding author upon reasonable request. Source data are provided with this paper.
References
Jack, C. R. Jr. et al. Hypothetical model of dynamic biomarkers of the Alzheimer’s pathological cascade. Lancet Neurol. 9, 119–128 (2010).
Sofroniew, M. V. Molecular dissection of reactive astrogliosis and glial scar formation. Trends Neurosci. 32, 638–647 (2009).
Anderson, M. A., Ao, Y. & Sofroniew, M. V. Heterogeneity of reactive astrocytes. Neurosci. Lett. 565, 23–29 (2014).
Ben Haim, L., Carrillo-de Sauvage, M. A., Ceyzériat, K. & Escartin, C. Elusive roles for reactive astrocytes in neurodegenerative diseases. Front. Cell. Neurosci. 9, 278 (2015).
Kraft, A. W. et al. Attenuating astrocyte activation accelerates plaque pathogenesis in APP/PS1 mice. FASEB J. 27, 187–198 (2013).
Kamphuis, W. et al. GFAP and vimentin deficiency alters gene expression in astrocytes and microglia in wild-type mice and changes the transcriptional response of reactive glia in mouse model for Alzheimer’s disease. Glia 63, 1036–1056 (2015).
Sirko, S. et al. Reactive glia in the injured brain acquire stem cell properties in response to Sonic hedgehog. Cell Stem Cell 12, 426–439 (2013).
Chun, H. & Lee, C. J. Reactive astrocytes in Alzheimer’s disease: a double-edged sword.Neurosci. Res. 126, 44–52 (2018).
Chun, H., Marriott, I., Lee, C. J. & Cho, H. Elucidating the interactive roles of glia in Alzheimer’s disease using established and newly developed experimental models. Front. Neurol. 9, 797 (2018).
Ben Haim, L. et al. The JAK/STAT3 pathway is a common inducer of astrocyte reactivity in Alzheimer’s and Huntington’s diseases. J. Neurosci. 35, 2817–2829 (2015).
Allaman, I. et al. Amyloid-β aggregates cause alterations of astrocytic metabolic phenotype: impact on neuronal viability. J. Neurosci. 30, 3326–3338 (2010).
Liddelow, S. A. et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature 541, 481–487 (2017).
Hertz, L., Peng, L. & Dienel, G. A. Energy metabolism in astrocytes: high rate of oxidative metabolism and spatiotemporal dependence on glycolysis/glycogenolysis. J. Cereb. Blood Flow. Metab. 27, 219–249 (2007).
Pomilio, C. et al. Glial alterations from early to late stages in a model of Alzheimer’s disease: evidence of autophagy involvement in Aβ internalization. Hippocampus 26, 194–210 (2016).
Ries, M. & Sastre, M. Mechanisms of Aβ clearance and degradation by glial cells. Front. Aging Neurosci. 8, 160 (2016).
Wyss-Coray, T. et al. Adult mouse astrocytes degrade amyloid-β. in vitro and in situ. Nat. Med. 9, 453–457 (2003).
Korenić, A., Andjus, P., Radenović, L. & Spasojević, I. The role of autophagy and lipolysis in survival of astrocytes under nutrient deprivation. Neurosci. Lett. 595, 128–133 (2015).
Jo, S. et al. GABA from reactive astrocytes impairs memory in mouse models of Alzheimer’s disease. Nat. Med. 20, 886–896 (2014).
Chen, Q. M., Tu, V. C., Wu, Y. & Bahl, J. J. Hydrogen peroxide dose dependent induction of cell death or hypertrophy in cardiomyocytes. Arch. Biochem. Biophys. 373, 242–248 (2000).
Afanas’ev, I. New nucleophilic mechanisms of ROS-dependent epigenetic modifications: comparison of aging and cancer. Aging Dis. 5, 52–62 (2014).
Buch, T. et al. A Cre-inducible diphtheria toxin receptor mediates cell lineage ablation after toxin administration. Nat. Methods 2, 419–426 (2005).
Park, Y. M., Chun, H., Shin, J. I. & Lee, C. J. Astrocyte specificity and coverage of hGFAP-CreERT2 [Tg(GFAP-Cre/ERT2)13Kdmc] mouse line in various brain regions. Exp. Neurobiol. 27, 508–525 (2018).
Gropp, E. et al. Agouti-related peptide-expressing neurons are mandatory for feeding. Nat. Neurosci. 8, 1289–1291 (2005).
Durieux, P. F. et al. D2R striatopallidal neurons inhibit both locomotor and drug reward processes. Nat. Neurosci. 12, 393–395 (2009).
Arruda-Carvalho, M. et al. Posttraining ablation of adult-generated olfactory granule cells degrades odor–reward memories. J. Neurosci. 34, 15793–15803 (2014).
Parkhurst, C. N. et al. Microglia promote learning-dependent synapse formation through brain-derived neurotrophic factor. Cell 155, 1596–1609 (2013).
Ghosh, A. et al. Targeted ablation of oligodendrocytes triggers axonal damage. PLoS ONE 6, e22735 (2011).
Chun, H. et al. Astrocytic proBDNF and Tonic GABA distinguish active versus reactive astrocytes in hippocampus. Exp. Neurobiol. 27, 155–170 (2018).
Maggiorani, D. et al. Monoamine oxidases, oxidative stress, and altered mitochondrial dynamics in cardiac ageing. Oxid. Med. Cell. Longev. 2017, 3017947 (2017).
Shin, J. H. et al. Concurrent blockade of free radical and microsomal prostaglandin E synthase-1-mediated PGE2 production improves safety and efficacy in a mouse model of amyotrophic lateral sclerosis. J. Neurochem. 122, 952–961 (2012).
Borroni, E. et al. Sembragiline: a novel, selective monoamine oxidase type B inhibitor for the treatment of Alzheimer’s disease. J. Pharmacol. Exp. Ther. 362, 413–423 (2017).
Zhu, D. et al. Hydrogen peroxide alters membrane and cytoskeleton properties and increases intercellular connections in astrocytes. J. Cell Sci. 118, 3695–3703 (2005).
Park, J.-H. et al. Newly developed reversible MAO-B inhibitor circumvents the shortcomings of irreversible inhibitors in Alzheimer’s disease.Sci. Adv. 5, eaav0316 (2019).
Popa-Wagner, A., Mitran, S., Sivanesan, S., Chang, E. & Buga, A.-M. ROS and brain diseases: the good, the bad, and the ugly. Oxid. Med. Cell. Longev. 2013, 963520 (2013).
Garwood, C. J., Pooler, A. M., Atherton, J., Hanger, D. P. & Noble, W. Astrocytes are important mediators of Aβ-induced neurotoxicity and tau phosphorylation in primary culture. Cell Death Dis. 2, e167 (2011).
Krstic, D. et al. Systemic immune challenges trigger and drive Alzheimer-like neuropathology in mice. J. Neuroinflammation 9, 151 (2012).
Eidet, J. R., Pasovic, L., Maria, R., Jackson, C. J. & Utheim, T. P. Objective assessment of changes in nuclear morphology and cell distribution following induction of apoptosis. Diagn. Pathol. 9, 92 (2014).
Benzing, W. C., Mufson, E. J. & Armstrong, D. M. Alzheimer’s disease-like dystrophic neurites characteristically associated with senile plaques are not found within other neurodegenerative diseases unless amyloid β-protein deposition is present. Brain Res. 606, 10–18 (1993).
Yang, D.-S. et al. Neuronal apoptosis and autophagy cross talk in aging PS/APP mice, a model of Alzheimer’s disease. Am. J. Pathol. 173, 665–681 (2008).
Augustinack, J. C., Schneider, A., Mandelkow, E.-M. & Hyman, B. T. Specific tau phosphorylation sites correlate with severity of neuronal cytopathology in Alzheimer’s disease. Acta Neuropathol. 103, 26–35 (2002).
Stephenson, R. M. & Andrew, R. J. Amnesia due to β-antagonists in a passive avoidance task in the chick. Pharmacol. Biochem. Behav. 15, 597–604 (1981).
Park, J. et al.A 3D human triculture system modeling neurodegeneration and neuroinflammation in Alzheimer’s disease.Nat. Neurosci. 21, 941–951 (2018).
Schwab, C., Klegeris, A. & McGeer, P. L. Inflammation in transgenic mouse models of neurodegenerative disorders. Biochim. Biophys. Acta 1802, 889–902 (2010).
Ortinski, P. I. et al. Selective induction of astrocytic gliosis generates deficits in neuronal inhibition. Nat. Neurosci. 13, 584–591 (2010).
Haettig, J., Sun, Y., Wood, M. A. & Xu, X. Cell-type specific inactivation of hippocampal CA1 disrupts location-dependent object recognition in the mouse. Learn. Mem. 20, 139–146 (2013).
Woo, J. et al. Functional characterization of resting and adenovirus-induced reactive astrocytes in three-dimensional culture. Exp. Neurobiol. 26, 158–167 (2017).
Liu, P. P., Xie, Y., Meng, X.-Y. & Kang, J.-S. History and progress of hypotheses and clinical trials for Alzheimer’s disease. Signal Transduct. Target. Ther. 4, 29 (2019).
Hawkes, N. Merck ends trial of potential Alzheimer’s drug verubecestat. BMJ 356, j845 (2017).
Roostaei, T. et al. Genome-wide interaction study of brain beta-amyloid burden and cognitive impairment in Alzheimer’s disease. Mol. Psychiatry 22, 287–295 (2017).
Lee, C. J. et al. Astrocytic control of synaptic NMDA receptors. J. Physiol. 581, 1057–1081 (2007).
Kim, Y. et al. Uvrag targeting by Mir125a and Mir351 modulates autophagy associated with Ewsr1 deficiency. Autophagy 11, 796–811 (2015).
Choi, S. H. et al. A three-dimensional human neural cell culture model of Alzheimer’s disease. Nature 515, 274–278 (2014).
Acknowledgements
This research was supported by the Creative Research Initiative Program, Korean National Research Foundation (NRF) (grant no. 2015R1A3A2066619), Brain Research Program through the NRF funded by the Ministry of Science and ICT (grant nos. 2018M3C7A1056682 and 2018M3C7A1056897), a National Research Council of Science & Technology grant by the Korean government (MSIP) (no. CRC-15-04-KIST), grant no. 2E28411 from the Korean Institute of Science and Technology (KIST) and grant no. IBS-R001-D2 from the Institute for Basic Science from the Ministry of Science (to C.J.L.). This study was also supported by a National Institutes of Health grant no. AG054156, grant no. NRF-2016M3C7A1904233 and grant no. 2E26663 from KIST (to H.R.). This work was supported by the Pioneering Funding Award funded by the Cure Alzheimer’s Fund and the NIH (grant no. AG059236-01A1 to H.C.), by the National Honor Scientist Program (grant no. NRF-2012R1A3A1050385 to B.K.K.) and by The Bio & Medical Technology Development Program (grant no. NRF‐2019M3E5D2A01066259 to D.K.).
Author information
Authors and Affiliations
Contributions
H.J.C., H.R. and C.J.L. designed the study, analyzed the data and wrote the manuscript. H.J.C., J.W.L. and Y.M.P. performed and analyzed the IHC in GiD, fGiD and APP/PS1 mice. Y.H.K. performed and analyzed the western blot and MTT assay. Y.J.K. performed and analyzed the experiments in the 3D neurocyte AD model. H.J.C. and J.W.L. performed the microdialysis and in vivo H2O2 assay. W.J.W. performed the IHC and in vitro H2O2 assay. Y.M.P. validated the GFAP-CreERT2 mice. J.W.L., J.H.S., Y.H.J. and S.K. performed the behavioral tests. H.J.C., W.J.W., Y.H.J., J.K.L. and J.S.W. performed the electrophysiological experiments. S.E.L. generated the adenovirus and AAV virus. H.J.I. and Y.J.H. performed the immunostaining and quantitative RT–PCR analysis in the human samples. D.Y.K. and H.C. provided the 3D neurocyte AD model. J.H.S. and B.J.G. provided the AAD-2004 scavenger. S.M.J. and D.K. provided the transgenic animal models. Y.S.K. provided the Aβ. J.H.P. and K.D.P. provided the KDS2010 MAO-B inhibitor. H.S.C. and J.S.S. performed the gene expression analysis. B.K.K. designed the experiments and wrote the manuscript. All authors contributed to the analysis and discussion of the results and reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Neuroscience thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Validation and characterization of GiD system.
a, (left) RT-PCR analysis of diphtheria toxin receptor(DTR) in hippocampus of Gcon and GiDm. (right) RT-PCR analysis of DTR in cultured astrocytes from Gcon and GiD (GiDc). GAPDH, internal control. Experiments were repeated more than twice. b, Measuring passive conductance in astrocytes of Gcon and GiDs. c, Experimental procedures of GiD mice, which GFAP-CreERT2 crossed with iDTR mice. Tam, tamoxifen, 100 mg/kg/day, 5 days; DT, 50 µg/kg/day, 2 days. d, GFAP immunostaining in brain regions of frontal lobe containing cortex, hippocampus, striatum and amygdala of Gcon and GiD mice. Ctx, cortex; Hipp, hippocampus; Str, striatum; Amyg, amygdala. e, Experimental procedures for PI staining in GiDm, CiD mice and KA-injected seizure model mice. DT, 50 µg/kg/day, 2 days; KA, kainic acid, 25 mg/kg. f, PI staining in GiDm, CiD and seizure model mice. g, h, MTT assay in DT-treated GiD astrocyte. DT, 1 ug/ml, 5 days; 3-MA, 3-Methyladenine, 0.5 mM (g), Baf A1, bafilomycin A1, 4 µM (h). Data are presented as mean ± SEM. *P < 0.05; NS: not significant. Additional statistical details are provided in Supplementary Table 1.
Extended Data Fig. 2 Autophagy activation in astrocytes of GiDs and GiDc.
a, Schematic diagram for possible mechanism of DT intoxification in GiD. b, Immunostaining for LC3 and GFAP in cortex of Gcon and GiDs. c, d, Immunostaining for LC3 (c) and SQSTM1 (d) in hippocampus of Gcon and GiDs. e, Experimental timeline for DT treatment in cultured astrocytes from Gcon and GiD (GiDc). f, g, Western blot analysis showing that levels of endogenous SQSTM1 and LC3-II are time-dependently increased in DT-treated GiDc astrocyte. h-s, Autophagic flux assay and densitometry analysis in DT-treated GiDc astrocytes. DT, 1 µg/ml, 8 hrs; E64d+Pep.A, 10 µg/ml E64d plus 10 µg/ml Pep.A, 6 hrs (h-m); CQ, 20 µM, 6 hrs (n-s). Data are presented as mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001; NS: not significant. Additional statistical details are provided in Supplementary Table 1.
Extended Data Fig. 3 MAO-B-mediated astrocytic hypertrophy and lack of neurodegeneration in GiDm brain.
a, Experimental procedures for AAD-2004 and selegiline treatment in GiDm. b,c, Immunostaining and quantification for GFAP in Gcon, GiDm, selegline-treated GiDm and AAD-2004-treated GiDm. d,e, Immunostaining and quantification for NeuN in Gcon and GiDm. Data are presented as mean ± SEM. ***P < 0.001, ****P < 0.0001; NS: not significant. Additional statistical details are provided in Supplementary Table 1.
Extended Data Fig. 4 Astrocytic hypertrophy and neurodegeneration in cortex, striatum and amygdala of GiDs brain.
a, Representative image of immunostaining for GFAP and NeuN in the brain of Gcon and GiDs. b-d, Quantification for the number of cells positive for NeuN in cortex, striatum and amygdala of Gcon and GiDs. Data are presented as mean ± SEM. *P < 0.05, ***P < 0.001, ****P < 0.0001. Additional statistical details are provided in Supplementary Table 1.
Extended Data Fig. 5 Development of focal GiD (fGiD).
a, b, Schematic diagram and experimental procedure for making mild and severe focal GiD, fGiDm and fGiDs, respectively. c, Immunostaining for GFAP and mCherry in CTL, fGiDm and fGiDs. d, Quantification for GFAP intensity in CTL, fGiDm and fGiDs. e, DAPI image in CTL, fGiDm and fGiDs. f, Counting the number of DAPI signals in stratum radiatum of CTL, fGiDm and fGiDs. g, Measuring the size of CA1 region in CTL, fGiDm and fGiDs. h, Comparison of glial activation in hippocampus between fGiDs and GiDs. Data are presented as mean ± SEM. **P < 0.01, ***P < 0.001, ****P < 0.0001; NS: not significant. Additional statistical details are provided in Supplementary Table 1.
Extended Data Fig. 6 Virus-mediated increase of astrocyte’s reactivity and its effects on neurodegeneration and tauopathy in APP/PS1 mice.
a, Schematic diagram and experimental timeline for increasing the reactivity of astrocytes in APP/PS1 mice using Adeno-GFAP-GFP virus. b-d, Immunostaining for GFAP, NeuN and quantification of the mean intensity in uninjected- or virus injected- WT and APP/PS1 mice. Magenta, Adeno-GFAP-GFP virus. e-g, DAB staining and quantification for p-Tau(S199) and p-Tau(S396) in virus injected- WT and APP/PS1 mice. Data are presented as mean ± SEM. *P < 0.05, **P < 0.01, ****P < 0.0001; NS: not significant. Additional statistical details are provided in Supplementary Table 1.
Supplementary information
Supplementary Information
Supplementary Figs. 1–11 and Supplementary Tables 1 and 2.
Source data
Source Data Fig. 2
Unprocessed western blots and/or gels.
Source Data Extended Data Fig. 1
Unprocessed western blots and/or gels.
Source Data Extended Data Fig. 2
Unprocessed western blots and/or gels.
Rights and permissions
About this article
Cite this article
Chun, H., Im, H., Kang, Y.J. et al. Severe reactive astrocytes precipitate pathological hallmarks of Alzheimer’s disease via H2O2− production. Nat Neurosci 23, 1555–1566 (2020). https://doi.org/10.1038/s41593-020-00735-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-020-00735-y
This article is cited by
-
Long-term inhibition of ODC1 in APP/PS1 mice rescues amyloid pathology and switches astrocytes from a reactive to active state
Molecular Brain (2024)
-
Environmental aluminum oxide inducing neurodegeneration in human neurovascular unit with immunity
Scientific Reports (2024)
-
Immunological aspects of central neurodegeneration
Cell Discovery (2024)
-
Relationship Between Reactive Astrocytes, by [18F]SMBT-1 Imaging, with Amyloid-Beta, Tau, Glucose Metabolism, and TSPO in Mouse Models of Alzheimer’s Disease
Molecular Neurobiology (2024)
-
Characterization of monoamine oxidase-B (MAO-B) as a biomarker of reactive astrogliosis in Alzheimer’s disease and related dementias
Acta Neuropathologica (2024)