Abstract
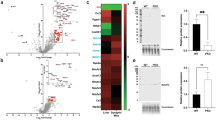
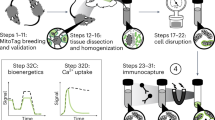
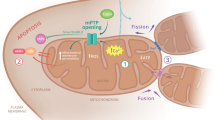
Mitochondria vary in morphology and function in different tissues; however, little is known about their molecular diversity among cell types. Here we engineered MitoTag mice, which express a Cre recombinase-dependent green fluorescent protein targeted to the outer mitochondrial membrane, and developed an isolation approach to profile tagged mitochondria from defined cell types. We determined the mitochondrial proteome of the three major cerebellar cell types (Purkinje cells, granule cells and astrocytes) and identified hundreds of mitochondrial proteins that are differentially regulated. Thus, we provide markers of cell-type-specific mitochondria for the healthy and diseased mouse and human central nervous systems, including in amyotrophic lateral sclerosis and Alzheimer’s disease. Based on proteomic predictions, we demonstrate that astrocytic mitochondria metabolize long-chain fatty acids more efficiently than neuronal mitochondria. We also characterize cell-type differences in mitochondrial calcium buffering via the mitochondrial calcium uniporter (Mcu) and identify regulator of microtubule dynamics protein 3 (Rmdn3) as a determinant of endoplasmic reticulum–mitochondria proximity in Purkinje cells. Our approach enables exploring mitochondrial diversity in many in vivo contexts.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All proteomics data generated within this study are deposited to the ProteomeXchange Consortium via the PRIDE44 partner repository with the dataset identifiers PXD010772, PXD010774, PXD010781 and PXD013380. In addition, Supplementary Data provide information on the proteomics data analysis used for the conclusions in the main and supplementary figures. All additional data that support the findings of this study are available from the corresponding author upon reasonable request. The MitoTag mouse line is available from The Jackson Laboratory as JAX#032675 (Rosa26–CAG–LSL–GFP–OMM). The McuFL/FL and the Rbp4:Cre mouse strains are protected under a material transfer agreement with the MMRRC (C57BL/6N-Mcutm1a(EUCOMM)Hmgu/H; Tg(Rbp4-cre)KL100Gsat/Mmucd), and the Rmdn3−/− mouse strain is protected under a material transfer agreement with the KOMP Repository (KO mouse project 3U01HG004080).
References
MacIver, N. J., Michalek, R. D. & Rathmell, J. C. Metabolic regulation of T lymphocytes. Annu. Rev. Immunol. 31, 259–283 (2013).
Chen, H. & Chan, D. C. Mitochondrial dynamics in regulating the unique phenotypes of cancer and stem cells. Cell Metab. 26, 39–48 (2017).
Misgeld, T. & Schwarz, T. L. Mitostasis in neurons: maintaining mitochondria in an extended cellular architecture. Neuron 96, 651–666 (2017).
Devine, M. J. & Kittler, J. T. Mitochondria at the neuronal presynapse in health and disease. Nat. Rev. Neurosci. 19, 63–80 (2018).
Nunnari, J. & Suomalainen, A. Mitochondria: in sickness and in health. Cell 148, 1145–1159 (2012).
West, A. P., Shadel, G. S. & Ghosh, S. Mitochondria in innate immune responses. Nat. Rev. Immunol. 11, 389–402 (2011).
Pagliarini, D. J. et al. A mitochondrial protein compendium elucidates complex I disease biology. Cell 134, 112–123 (2008).
Shigeoka, T. et al. Dynamic axonal translation in developing and mature visual circuits. Cell 166, 181–192 (2016).
Ghazalpour, A. et al. Comparative analysis of proteome and transcriptome variation in mouse. PLoS Genet. 7, e1001393 (2011).
Heiman, M. et al. A translational profiling approach for the molecular characterization of CNS cell types. Cell 135, 738–748 (2008).
Chen, W. W., Freinkman, E., Wang, T., Birsoy, K. & Sabatini, D. M. Absolute quantification of matrix metabolites reveals the dynamics of mitochondrial metabolism. Cell 166, 1324–1337.e1311 (2016).
Abu-Remaileh, M. et al. Lysosomal metabolomics reveals V-ATPase- and mTOR-dependent regulation of amino acid efflux from lysosomes. Science 358, 807–813 (2017).
Ahier, A. et al. Affinity purification of cell-specific mitochondria from whole animals resolves patterns of genetic mosaicism. Nat. Cell Biol. 20, 352–360 (2018).
Roh, H. C. et al. Simultaneous transcriptional and epigenomic profiling from specific cell types within heterogeneous tissues in vivo. Cell Rep. 18, 1048–1061 (2017).
Hornig-Do, H. T. et al. Isolation of functional pure mitochondria by superparamagnetic microbeads. Anal. Biochem. 389, 1–5 (2009).
Meeusen, S., McCaffery, J. M. & Nunnari, J. Mitochondrial fusion intermediates revealed in vitro. Science 305, 1747–1752 (2004).
Sharma, K. et al. Cell type- and brain region-resolved mouse brain proteome. Nat. Neurosci. 18, 1819–1831 (2015).
Calvo, S. E., Clauser, K. R. & Mootha, V. K. MitoCarta2.0: an updated inventory of mammalian mitochondrial proteins. Nucl. Acids Res. 44, D1251–D1257 (2016).
Kang, J. S. et al. Docking of axonal mitochondria by syntaphilin controls their mobility and affects short-term facilitation. Cell 132, 137–148 (2008).
Sato, K., Yoshida, S., Fujiwara, K., Tada, K. & Tohyama, M. Glycine cleavage system in astrocytes. Brain Res. 567, 64–70 (1991).
Miyake, S. et al. Identification and characterization of a novel mitochondrial tricarboxylate carrier. Biochem. Biophys. Res. Commun. 295, 463–468 (2002).
Fowler, J. S. et al. Monoamine oxidase: radiotracer development and human studies. Methods 27, 263–277 (2002).
Petrova, V. Y., Drescher, D., Kujumdzieva, A. V. & Schmitt, M. J. Dual targeting of yeast catalase a to peroxisomes and mitochondria. Biochem. J. 380, 393–400 (2004).
Fan, J., Li, X., Issop, L., Culty, M. & Papadopoulos, V. ACBD2/ECI2-mediated peroxisome-mitochondria interactions in Leydig cell steroid biosynthesis. Mol. Endocrinol. 30, 763–782 (2016).
Shai, N. et al. Systematic mapping of contact sites reveals tethers and a function for the peroxisome-mitochondria contact. Nat. Commun. 9, 1761 (2018).
Sinha, S., Bheemsetty, V. A. & Inamdar, M. S. A double helical motif in OCIAD2 is essential for its localization, interactions and STAT3 activation. Sci. Rep. 8, 7362 (2018).
Baughman, J. M. et al. Integrative genomics identifies MCU as an essential component of the mitochondrial calcium uniporter. Nature 476, 341–345 (2011).
De Stefani, D., Raffaello, A., Teardo, E., Szabò, I. & Rizzuto, R. A forty-kilodalton protein of the inner membrane is the mitochondrial calcium uniporter. Nature 476, 336–340 (2011).
De Vos, K. J. et al. VAPB interacts with the mitochondrial protein PTPIP51 to regulate calcium homeostasis. Hum. Mol. Genet. 21, 1299–1311 (2012).
Galmes, R. et al. ORP5/ORP8 localize to endoplasmic reticulum-mitochondria contacts and are involved in mitochondrial function. EMBO Rep. 17, 800–810 (2016).
Di Giorgio, F. P., Boulting, G. L., Bobrowicz, S. & Eggan, K. C. Human embryonic stem cell-derived motor neurons are sensitive to the toxic effect of glial cells carrying an ALS-causing mutation. Cell Stem Cell 3, 637–648 (2008).
Schönfeld, P. & Reiser, G. Why does brain metabolism not favor burning of fatty acids to provide energy? Reflections on disadvantages of the use of free fatty acids as fuel for brain. J. Cereb. Blood Flow. Metab. 33, 1493–1499 (2013).
Ebert, D., Haller, R. G. & Walton, M. E. Energy contribution of octanoate to intact rat brain metabolism measured by 13C nuclear magnetic resonance spectroscopy. J. Neurosci. 23, 5928–5935 (2003).
Knobloch, M. et al. A fatty acid oxidation-dependent metabolic shift regulates adult neural stem cell activity. Cell Rep. 20, 2144–2155 (2017).
Eraso-Pichot, A. et al. GSEA of mouse and human mitochondriomes reveals fatty acid oxidation in astrocytes. Glia 66, 1724–1735 (2018).
Harrington, J. L. & Murphy, E. The mitochondrial calcium uniporter: mice can live and die without it. J. Mol. Cell. Cardiol. 78, 46–53 (2015).
Paillard, M. et al. Tissue-Specific mitochondrial decoding of cytoplasmic Ca(2+) signals is controlled by the stoichiometry of MICU1/2 and MCU. Cell Rep. 18, 2291–2300 (2017).
Eisenberg-Bord, M., Shai, N., Schuldiner, M. & Bohnert, M. A tether is a tether is a tether: tethering at membrane contact sites. Dev. Cell 39, 395–409 (2016).
Lv, B. F. et al. Protein tyrosine phosphatase interacting protein 51 (PTPIP51) is a novel mitochondria protein with an N-terminal mitochondrial targeting sequence and induces apoptosis. Apoptosis 11, 1489–1501 (2006).
Rangaraju, V., Lauterbach, M. & Schuman, E. M. Spatially stable mitochondrial compartments fuel local translation during plasticity. Cell 176, 73–84.e15 (2019).
Lewis, T. L. Jr., Kwon, S. K., Lee, A., Shaw, R. & Polleux, F. MFF-dependent mitochondrial fission regulates presynaptic release and axon branching by limiting axonal mitochondria size. Nat. Commun. 9, 5008 (2018).
Alvarez-Castelao, B. et al. Cell-type-specific metabolic labeling of nascent proteomes in vivo. Nat. Biotechnol. 35, 1196–1201 (2017).
Chen, C. L. et al. Proteomic mapping in live drosophila tissues using an engineered ascorbate peroxidase. Proc. Natl Acad. Sci. USA 112, 12093–12098 (2015).
Bayraktar, E. C. et al. MITO-Tag mice enable rapid isolation and multimodal profiling of mitochondria from specific cell types in vivo. Proc. Natl Acad. Sci. USA 116, 303–312 (2019).
Kerschensteiner, M., Reuter, M. S., Lichtman, J. W. & Misgeld, T. Ex vivo imaging of motor axon dynamics in murine triangularis sterni explants. Nat. Protoc. 3, 1645–1653 (2008).
Sugiura, A., Mattie, S., Prudent, J. & McBride, H. M. Newly born peroxisomes are a hybrid of mitochondrial and ER-derived pre-peroxisomes. Nature 542, 251–254 (2017).
Meera, P., Pulst, S. M. & Otis, T. S. Cellular and circuit mechanisms underlying spinocerebellar ataxias. J. Physiol. 594, 4653–4660 (2016).
Zaglia, T. et al. Content of mitochondrial calcium uniporter (MCU) in cardiomyocytes is regulated by microRNA-1 in physiologic and pathologic hypertrophy. Proc. Natl Acad. Sci. USA 114, E9006–E9015 (2017).
Dong, Z. et al. Mitochondrial Ca2+ uniporter is a mitochondrial luminal redox sensor that augments MCU channel activity. Mol. Cell 65, 1014–1028.e1017 (2017).
Henzi, T. & Schwaller, B. Antagonistic regulation of parvalbumin expression and mitochondrial calcium handling capacity in renal epithelial cells. PLoS One 10, e0142005 (2015).
Suomalainen, A. & Battersby, B. J. Mitochondrial diseases: the contribution of organelle stress responses to pathology. Nat. Rev. Mol. Cell Biol. 19, 77–92 (2018).
Yoshii, S. R., Kishi, C., Ishihara, N. & Mizushima, N. Parkin mediates proteasome-dependent protein degradation and rupture of the outer mitochondrial membrane. J. Biol. Chem. 286, 19630–19640 (2011).
Horie, C., Suzuki, H., Sakaguchi, M. & Mihara, K. Characterization of signal that directs C-tail-anchored proteins to mammalian mitochondrial outer membrane. Mol. Biol. Cell 13, 1615–1625 (2002).
Hitz, C., Steuber-Buchberger, P., Delic, S., Wurst, W. & Kühn, R. Generation of shRNA transgenic mice. Methods Mol. Biol. 530, 101–129 (2009).
Ortiz, O., Wurst, W. & Kühn, R. Reversible and tissue-specific activation of MAP kinase signaling by tamoxifen in Braf(V637)ER(T2) mice. Genesis 51, 448–455 (2013).
Hitz, C., Wurst, W. & Kühn, R. Conditional brain-specific knockdown of MAPK using Cre/loxP regulated RNA interference. Nucl. Acids Res. 35, e90 (2007).
Breckwoldt, M. O. et al. Multiparametric optical analysis of mitochondrial redox signals during neuronal physiology and pathology in vivo. Nat. Med. 20, 555–560 (2014).
Gorski, J. A. et al. Cortical excitatory neurons and glia, but not GABAergic neurons, are produced in the Emx1-expressing lineage. J. Neurosci. 22, 6309–6314 (2002).
Rossi, J. et al. Melanocortin-4 receptors expressed by cholinergic neurons regulate energy balance and glucose homeostasis. Cell Metab. 13, 195–204 (2011).
Barski, J. J., Dethleffsen, K. & Meyer, M. Cre recombinase expression in cerebellar purkinje cells. Genesis 28, 93–98 (2000).
Fünfschilling, U. & Reichardt, L. F. Cre-mediated recombination in rhombic lip derivatives. Genesis 33, 160–169 (2002).
Gregorian, C. et al. Pten deletion in adult neural stem/progenitor cells enhances constitutive neurogenesis. J. Neurosci. 29, 1874–1886 (2009).
Gong, S. et al. Targeting Cre recombinase to specific neuron populations with bacterial artificial chromosome constructs. J. Neurosci. 27, 9817–9823 (2007).
Gong, S. et al. A gene expression atlas of the central nervous system based on bacterial artificial chromosomes. Nature 425, 917–925 (2003).
Busche, M. A. et al. Clusters of hyperactive neurons near amyloid plaques in a mouse model of alzheimer’s disease. Science 321, 1686–1689 (2008).
Gurney, M. E. et al. Motor neuron degeneration in mice that express a human Cu,Zn superoxide dismutase mutation. Science 264, 1772–1775 (1994).
Rodriguez, C. I. et al. High-efficiency deleter mice show that FLPe is an alternative to Cre-loxP. Nat. Genet. 25, 139–140 (2000).
Skarnes, W. C. et al. A conditional knockout resource for the genome-wide study of mouse gene function. Nature 474, 337–342 (2011).
Brill, M. S. et al. Branch-specific microtubule destabilization mediates axon branch loss during neuromuscular synapse elimination. Neuron 92, 845–856 (2016).
Kleele, T. et al. An assay to image neuronal microtubule dynamics in mice. Nat. Commun. 5, 4827 (2014).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Wettmarshausen, J. & Perocchi, F. Isolation of functional mitochondria from cultured cells and mouse tissues. Methods Mol. Biol. 1567, 15–32 (2017).
Franko, A. et al. Efficient isolation of pure and functional mitochondria from mouse tissues using automated tissue disruption and enrichment with anti-TOM22 magnetic beads. PLoS One 8, e82392 (2013).
Sims, N. R. & Anderson, M. F. Isolation of mitochondria from rat brain using percoll density gradient centrifugation. Nat. Protoc. 3, 1228–1239 (2008).
Wang, X. et al. Isolation of brain mitochondria from neonatal mice. J. Neurochem. 119, 1253–1261 (2011).
Watkins, P. A., Ferrell, E. V. Jr., Pedersen, J. I. & Hoefler, G. Peroxisomal fatty acid beta-oxidation in HepG2 cells. Arch. Biochem. Biophys. 289, 329–336 (1991).
Demarquoy, J. & Le Borgne, F. Crosstalk between mitochondria and peroxisomes. World J. Biol. Chem. 6, 301–309 (2015).
Wiśniewski, J. R., Zougman, A., Nagaraj, N. & Mann, M. Universal sample preparation method for proteome analysis. Nat. Methods 6, 359–362 (2009).
Rappsilber, J., Ishihama, Y. & Mann, M. Stop and go extraction tips for matrix-assisted laser desorption/ionization, nanoelectrospray, and LC/MS sample pretreatment in proteomics. Anal. Chem. 75, 663–670 (2003).
Cox, J. et al. Accurate proteome-wide label-free quantification by delayed normalization and maximal peptide ratio extraction, termed MaxLFQ. Mol. Cell Proteom. 13, 2513–2526 (2014).
Vizcaino, J. A. et al. update of the PRIDE database and its related tools. Nucl. Acids Res. 44, D447–456 (2016).
Tyanova, S. et al. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Methods 13, 731–740 (2016).
Cox, J. & Mann, M. 1D and 2D annotation enrichment: a statistical method integrating quantitative proteomics with complementary high-throughput data. BMC Bioinform. 13 (Suppl. 16), S12 (2012).
Goldberg, T. et al. LocTree3 prediction of localization. Nucl. Acids Res. 42, W350–W355 (2014).
Wang, J., Vasaikar, S., Shi, Z., Greer, M. & Zhang, B. WebGestalt 2017: a more comprehensive, powerful, flexible and interactive gene set enrichment analysis toolkit. Nucl. Acids Res. 45, W130–W137 (2017).
Kanehisa, M., Goto, S., Sato, Y., Furumichi, M. & Tanabe, M. KEGG for integration and interpretation of large-scale molecular data sets. Nucl. Acids Res. 40, D109–D114 (2011).
Shin, J. B. et al. Molecular architecture of the chick vestibular hair bundle. Nat. Neurosci. 16, 365–374 (2013).
Uchida, Y. & Takahashi, H. Rapid detection of Abeta deposits in APP transgenic mice by Hoechst 33342. Neurosci. Lett. 448, 279–281 (2008).
Tillberg, P. W. et al. Protein-retention expansion microscopy of cells and tissues labeled using standard fluorescent proteins and antibodies. Nat. Biotechnol. 34, 987–992 (2016).
Acknowledgements
We thank M. Budak, N. Budak and S. Taskin for animal husbandry and A. Berghofer, A. Graupner, Y. Hufnagel, K. Wullimann and M. Schetterer for technical and administrative support. We thank U. Fünfschilling (Max Planck Institute Göttingen) for providing Gabra6:Cre mice, D. Crane (Griffith University) for the Pex14 antibody and N. Mizushima (Addgene plasmid #38249) for the pMXs-IP GFP–Omp25 plasmid. We are grateful to L. Jiang for help with bioinformatics analyses, Y.-H. Tai for neonatal virus injection and B. Zott for APPPS1 breeding. We thank L. Godinho, M. Schuldiner and O. Schuldiner for reading an earlier version of this article. This work was supported by the Deutsche Forschungsgemeinschaft through the Munich Center for Systems Neurology (SyNergy; grant no. EXC 1010 to A.K., T.K., S.F.L., T.M., F.P. and W.W.); the Center for Integrated Protein Science Munich (grant no. EXC 114 to A.K. and T.M.); the Collaborative Research Centers (CRC870 to T.M., CRC1054 to T.K. and CRC-TR128 to T.K.); research unit grant no. FOR2290 to S.F.L. and research grant no. Mi694/7-1/8-1 to T.M. F.P. and J.W. were supported by the Emmy-Noether program of the Deutsche Forschungsgemeinschaft (grant no. Pe2053/1-1 to F.P.). Further support came from the European Research Council under the European Union’s Seventh Framework Program (grant no. FP/2007-2013; European Research Council Grant Agreement nos. CoG 616791 to T.M. and CoG 647215 to T.K.) and the German Center for Neurodegenerative Diseases (DZNE Munich to S.F.L., T.M. and W.W.). L.T. was supported by an EMBO Long-Term Fellowship (no. EMBO ALTF 108-2013). Further support came from the Centers of Excellence in Neurodegeneration and the Helmholtz-Israel Program to S.F.L., the Swiss National Science Formation and HHS Foundation to D.M., the German Federal Ministry of Education and Research (BMBF) through ‘T-B interaction in NMO’ to T.K. and ‘Mitochondrial endophenotypes of Morbus Parkinson’ (grant no. 031A430E) to W.W. A.K. is supported by a Senior Hertie Professorship of Neuroscience. T.M. is supported by the Institute for Advanced Study, Technical University of Munich (Focus Group ‘Subcellular Dynamics in Neurons’).
Author information
Authors and Affiliations
Contributions
C.F., L.T. and T.M. devised the study. L.T., T.M., O.O., R.K. and W.W. designed and generated the MitoTag mice, which C.F. and L.T. characterized. C.F. designed the immunocapture protocol (with support from J.W. and F.P.) and performed most of the isolations. S.A.M. and S.F.L. performed sample preparation, mass spectrometry and primary data analysis. C.F. further analyzed the proteomics datasets. L.T. performed bioenergetics measurements and corresponding isolations (with support from J.W. and F.P.). C.F. performed calcium uptake assays (with support from F.P.). C.F., S.H. and T.K. performed and analyzed flow cytometry. C.F. performed western blot analysis and immunofluorescence stainings of candidates. N.S. and C.F. obtained and analyzed electron microscopy data. C.F. characterized the Rmdn3−/− and GC-specific McuFL/FL mouse models. J.H., R.M.K. and A.K. provided pilot experiments for single-cell characterization in the cerebellum. A.K. provided APPPS1 mice. I.W. and D.M. performed immunofluorescence staining on human tissue. C.F., L.T. and T.M. wrote the paper with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information: Nature Neuroscience thanks Robert Friedlander, Daniel McClatchy and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Data
Proteomics data from cell-type-specific mitochondria: Purkinje cells, granule cells and astrocytes.
Rights and permissions
About this article
Cite this article
Fecher, C., Trovò, L., Müller, S.A. et al. Cell-type-specific profiling of brain mitochondria reveals functional and molecular diversity. Nat Neurosci 22, 1731–1742 (2019). https://doi.org/10.1038/s41593-019-0479-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-019-0479-z
This article is cited by
-
Changes in lipid metabolism track with the progression of neurofibrillary pathology in tauopathies
Journal of Neuroinflammation (2024)
-
Mitovesicles secreted into the extracellular space of brains with mitochondrial dysfunction impair synaptic plasticity
Molecular Neurodegeneration (2024)
-
Mitochondrial proteome research: the road ahead
Nature Reviews Molecular Cell Biology (2024)
-
Mitochondrial damage and impaired mitophagy contribute to disease progression in SCA6
Acta Neuropathologica (2024)
-
Ex vivo immunocapture and functional characterization of cell-type-specific mitochondria using MitoTag mice
Nature Protocols (2023)