Abstract
Comorbid depressive symptoms (CDS) in chronic pain are a common health problem, but the neural circuit mechanisms underlying these symptoms remain unclear. Here we identify a novel pathway involving 5-hydroxytryptamine (5-HT) projections from the dorsal raphe nucleus (5-HTDRN) to somatostatin (SOM)-expressing and non-SOM interneurons in the central nucleus of the amygdala (CeA). The SOMCeA neurons project directly to the lateral habenula, an area known involved in depression. Inhibition of the 5-HTDRN→SOMCeA pathway produced depression-like behavior in a male mouse model of chronic pain. Activation of this pathway using pharmacological or optogenetic approaches reduced depression-like behavior in these mice. Human functional magnetic resonance imaging data showed that compared to healthy controls, functional connectivity between the CeA-containing centromedial amygdala and the DRN was reduced in patients with CDS but not in patients in chronic pain without depression. These findings indicate that a novel 5-HTDRN→SOMCeA→lateral habenula pathway may mediate at least some aspects of CDS.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon request.
Change history
01 October 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Bair, M. J., Robinson, R. L., Katon, W. & Kroenke, K. Depression and pain comorbidity: a literature review. Arch. Intern. Med. 163, 2433–2445 (2003).
McWilliams, L. A., Goodwin, R. D. & Cox, B. J. Depression and anxiety associated with three pain conditions: results from a nationally representative sample. Pain 111, 77–83 (2004).
Simon, G. E., VonKorff, M., Piccinelli, M., Fullerton, C. & Ormel, J. An international study of the relation between somatic symptoms and depression. New Engl. J. Med. 341, 1329–1335 (1999).
Arnow, B. A. et al. Comorbid depression, chronic pain, and disability in primary care. Psychosom. Med 68, 262–268 (2006).
Goyal, M. et al. Meditation programs for psychological stress and well-being: a systematic review and meta-analysis. JAMA Intern. Med. 174, 357–368 (2014).
Turk, D. C. & Gatchel, R. J. Psychological Approaches to Pain Management: A Practitioner’s Handbook (Guilford Publications, 2018).
Campbell, L. C., Clauw, D. J. & Keefe, F. J. Persistent pain and depression: a biopsychosocial perspective. Biol. Psychiatry 54, 399–409 (2003).
Strigo, I. A., Simmons, A. N., Matthews, S. C., Arthur, D. & Paulus, M. P. Association of major depressive disorder with altered functional brain response during anticipation and processing of heat pain. Arch. Gen. Psychiatry 65, 1275–1284 (2008).
Martin, S. L. et al. 5-HT modulation of pain perception in humans. Psychopharmacology 234, 2929–2939 (2017).
Kuner, R. Central mechanisms of pathological pain. Nat. Med. 16, 1258–1266 (2010).
Kim, Y. S. et al. Central terminal sensitization of TRPV1 by descending serotonergic facilitation modulates chronic pain. Neuron 81, 873–887 (2014).
Lee, Y. C., Nassikas, N. J. & Clauw, D. J. The role of the central nervous system in the generation and maintenance of chronic pain in rheumatoid arthritis, osteoarthritis and fibromyalgia. Arthritis Res. Ther. 13, 211 (2011).
Basbaum, A. I., Bautista, D. M., Scherrer, G. & Julius, D. Cellular and molecular mechanisms of pain. Cell 139, 267–284 (2009).
Bushnell, M. C., Ceko, M. & Low, L. A. Cognitive and emotional control of pain and its disruption in chronic pain. Nat. Rev. Neurosci. 14, 502–511 (2013).
Ren, J. et al. Anatomically defined and functionally distinct dorsal raphe serotonin sub-systems. Cell 175, 472–487 (2018).
Commons, K. G. Ascending serotonin neuron diversity under two umbrellas. Brain Struct. Funct. 221, 3347–3360 (2016).
Hamilton, J. P., Siemer, M. & Gotlib, I. H. Amygdala volume in major depressive disorder: a meta-analysis of magnetic resonance imaging studies. Mol. Psychiatry 13, 993–1000 (2008).
Amat, J. et al. Medial prefrontal cortex determines how stressor controllability affects behavior and dorsal raphe nucleus. Nat. Neurosci. 8, 365 (2005).
Tye, K. M. et al. Amygdala circuitry mediating reversible and bidirectional control of anxiety. Nature 471, 358–362 (2011).
Li, H. et al. Experience-dependent modification of a central amygdala fear circuit. Nat. Neurosci. 16, 332–339 (2013).
Neugebauer, V., Li, W., Bird, G. C. & Han, J. S. The amygdala and persistent pain. Neuroscientist 10, 221–234 (2004).
Han, S., Soleiman, M. T., Soden, M. E., Zweifel, L. S. & Palmiter, R. D. Elucidating an affective pain circuit that creates a threat memory. Cell 162, 363–374 (2015).
Penzo, M. A. et al. The paraventricular thalamus controls a central amygdala fear circuit. Nature 519, 455–459 (2015).
Haubensak, W. et al. Genetic dissection of an amygdala microcircuit that gates conditioned fear. Nature 468, 270–276 (2010).
Hieronymus, F., Lisinski, A., Nilsson, S. & Eriksson, E. Efficacy of selective serotonin reuptake inhibitors in the absence of side effects: a mega-analysis of citalopram and paroxetine in adult depression. Mol. Psychiatry 23, 1731–1736 (2017).
Barbui, C., Furukawa, T. A. & Cipriani, A. Effectiveness of paroxetine in the treatment of acute major depression in adults: a systematic re-examination of published and unpublished data from randomized trials. CMAJ 178, 296–305 (2008).
Gonchar, Y., Wang, Q. & Burkhalter, A. Multiple distinct subtypes of GABAergic neurons in mouse visual cortex identified by triple immunostaining. Front Neuroanat. 1, 3 (2007).
Scott, M. M. et al. A genetic approach to access serotonin neurons for in vivo and in vitro studies. Proc. Natl Acad. Sci. USA 102, 16472–16477 (2005).
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci. 13, 133–140 (2010).
Marcinkiewcz, C. A. et al. Serotonin engages an anxiety and fear-promoting circuit in the extended amygdala. Nature 537, 97–101 (2016).
Mogil, J. S. Animal models of pain: progress and challenges. Nat. Rev. Neurosci. 10, 283–294 (2009).
Dworkin, R. H. et al. Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain 132, 237–251 (2007).
Decosterd, I. & Woolf, C. J. Spared nerve injury: an animal model of persistent peripheral neuropathic pain. Pain 87, 149–158 (2000).
Shabel, S. J., Proulx, C. D., Piriz, J. & Malinow, R. Mood regulation. GABA/glutamate co-release controls habenula output and is modified by antidepressant treatment. Science 345, 1494–1498 (2014).
Li, B. et al. Synaptic potentiation onto habenula neurons in the learned helplessness model of depression. Nature 470, 535–539 (2011).
Knowland, D. et al. Distinct ventral pallidal neural populations mediate separate symptoms of depression. Cell 170, 284–297 e218 (2017).
Hikosaka, O. The habenula: from stress evasion to value-based decision-making. Nat. Rev. Neurosci. 11, 503–513 (2010).
Tervo, D. G. et al. A designer AAV variant permits efficient retrograde access to projection neurons. Neuron 92, 372–382 (2016).
Bzdok, D., Laird, A. R., Zilles, K., Fox, P. T. & Eickhoff, S. B. An investigation of the structural, connectional, and functional subspecialization in the human amygdala. Hum. Brain Mapp. 34, 3247–3266 (2013).
Amunts, K. et al. Cytoarchitectonic mapping of the human amygdala, hippocampal region and entorhinal cortex: intersubject variability and probability maps. Anat. Embryol. 210, 343–352 (2005).
Ehnvall, A., Mitchell, P. B., Hadzi-Pavlovic, D., Malhi, G. S. & Parker, G. Pain during depression and relationship to rejection sensitivity. Acta Psychiatr. Scand. 119, 375–382 (2009).
Howland, R. H. et al. Factors predicting reduced antidepressant response: experience with the SNRI duloxetine in patients with major depression. Ann. Clin. Psychiatry 20, 209–218 (2008).
Cortes-Altamirano, J. L. et al. Review: 5-Ht1, 5-Ht2, 5-Ht3, And 5-Ht7 receptors and their role in the modulation of pain response in the central nervous system. Curr. Neuropharmacol. 16, 210–221 (2018).
Savitz, J., Lucki, I. & Drevets, W. C. 5-HT1A receptor function in major depressive disorder. Prog. Neurobiol. 88, 17–31 (2009).
Asmundson, G. J. & Katz, J. Understanding the co-occurrence of anxiety disorders and chronic pain: state-of-the-art. Depress. Anxiety 26, 888–901 (2009).
Pascual-Leone, A., Rubio, B., Pallardo, F. & Catala, M. D. Rapid-rate transcranial magnetic stimulation of left dorsolateral prefrontal cortex in drug-resistant depression. Lancet 348, 233–237 (1996).
Taghva, A. S., Malone, D. A. & Rezai, A. R. Deep brain stimulation for treatment-resistant depression. World Neurosurg. 80, S27 e17–24 (2013).
Wickersham, I. R. et al. Monosynaptic restriction of transsynaptic tracing from single, genetically targeted neurons. Neuron 53, 639–647 (2007).
Wall, N. R., Wickersham, I. R., Cetin, A., De La Parra, M. & Callaway, E. M. Monosynaptic circuit tracing in vivo through Cre-dependent targeting and complementation of modified rabies virus. Proc. Natl Acad. Sci. USA 107, 21848–21853 (2010).
Zhang, Z., Cai, Y.-Q., Zou, F., Bie, B. & Pan, Z. Z. Epigenetic suppression of GAD65 expression mediates persistent pain. Nat. Med. 17, 1448 (2011).
Hamilton, M. A rating scale for depression. J. Neurol. Neurosurg. Psychiatry 23, 56–62 (1960).
Melzack, R. The short-form McGill pain questionnaire. Pain 30, 191–197 (1987).
Treede, R.-D. et al. A classification of chronic pain for ICD-11. Pain 156, 1003 (2015).
Murphy, K., Birn, R. M., Handwerker, D. A., Jones, T. B. & Bandettini, P. A. The impact of global signal regression on resting state correlations: are anti-correlated networks introduced? Neuroimage 44, 893–905 (2009).
Saad, Z. S. et al. Trouble at rest: how correlation patterns and group differences become distorted after global signal regression. Brain Connect. 2, 25–32 (2012).
Jenkinson, M., Bannister, P., Brady, M. & Smith, S. Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage 17, 825–841 (2002).
Acknowledgements
We thank X. Yu and Y. Yang for providing Ai9 (RCL-tdT) mice. We thank F. Xu and T. Xue for technical support. All data are available in the main text or the Supplementary Data. Support for this study was provided by the National Natural Science Foundation of China (grant nos. 81870877, 91732303 and 91849119 to Z.Z.); the National Key Research and Development Program of China (grant no. 2016YFC1305900 to Z.Z.); the Strategic Priority Research Program of the Chinese Academy of Sciences (grant no. XDB02010000 to Z.Z.) and the National Natural Science Foundation of China (grant no. 31600851 to W.T. and grant no. 81600964 to Y.J.).
Author information
Authors and Affiliations
Contributions
W.Z., Y.J., Q.M. and X.Zhu designed the studies, performed most of the experiments and data analysis, and wrote the draft manuscript. T.B., Y.T., Y.M., L.W., H.Z., B.-S.Q., X.Zhang, Y.L. and W.X. conducted the fMRI experiments and data analyses and wrote the text of the final manuscript. W.T., H.W., Jie Li and N.Z. conducted some of the molecular and behavioral experiments. X.L., M.-H.L., Juan Li and H.X. designed the viral tracing experiments in CR-Cre, PV-Ce and SOM-Cre mice. Patients with chronic pain or depression were evaluated by certified doctors Y.M., Y.T., W.X. and K.W. J.Z., K.W., G.R.L., L.X. and Z.Z. were involved in the overall design of the study and the revision of the final manuscript. Z.Z. was involved in the overall design of the project, individual experiments, data analysis and the writing of the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information: Nature Neuroscience thanks Rohini Kuner, Manuel Mameli and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Mapping of CeA GABA neuron inputs.
a, Schematic of viral injection in the CeA of GAD2-Cre mice. b, Representative images of viral expression within the CeA (left) and DsRed signals in the BLA and paraventricular thalamus (PVT) (right). Scale bars: 500 µm (left), 50 µm (right). c,d, DsRed signals in the posterior thalamus (Po), parafascicular thalamic nucleus (PF, c), and DRN (d). Scale bars: 300 µm (left), 50 µm (right). Aq, aqueduct. e,f, CeA injection of Cre-dependent helper virus (AAV-DIO-TVA-GFP, AAV-DIO-RVG, green) and RV-EnvA-ΔG-DsRed (red) in CR-Cre mice (e) or PV-Cre mice (f). No DsRed signals were identified in the DRN. Scale bars: 200 µm (left), 50 µm (right). CR, calretinin; PV, parvalbumin. g, Observed mCherry fibers projected from 5-HTDRN neurons in the CeA. Scale bar: 200 µm. h, Sample traces of action potentials evoked by 473 nm light (blue bars) recorded from DRN mCherry+ neurons in acute slices. i, Representative images of DRN infusion of AAV-DIO-ChR2-mCherry in Pet1-Cre mice, and mCherry+ neurons in the DRN that co-localized with the 5-HT neuronal marker TPH2. Scale bar: 200 µm. j, Statistical data of mCherry+ neurons in the DRN that co-localized with TPH2 (n = 9 slices from three mice). Data are shown as box and whisker plots [medians, quartiles (boxes) and ranges min to max (whiskers)]. For b-i, the experiment was independently repeated five times with similar results obtained.
Supplementary Figure 2 Effects of the GABAAR blocker picrotoxin on 5-HTDRN→SOMCeA synaptic transmission.
a, Schematic of the DRN injection of AAV-hSyn-ChR2-EYFP in SOM-tdTOM mice and the recording configuration in acute slices. b, A representative image of viral expression within the DRN. The experiment was independently repeated five times with similar results obtained. Scale bar, 250 µm. c, A sample trace and summarized data of hyperpolarized potentials of SOMCeA neurons evoked by photostimulation in the presence of picrotoxin (n = 5 cells, t4 = 0.03337, P = 0.975). Significance was assessed by two-tailed paired Student’s t test in c. The data are expressed as the mean ± s.e.m. ns, no significance.
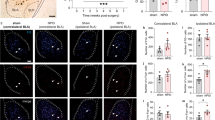
Supplementary Figure 3 SOMCeA neurons are innervated by 5-HTDRN neurons.
a,b, Representative images of c-Fos expression in the CeA after optical inhibition of 5-HTDRN→SOMCeA circuit of mice with the DRN infusion of eNpHR3.0-EYFP (a) or EYFP (b). The experiment was independently repeated four times with similar results obtained. Scale bars, 500 μm (left), 50 μm (right). c, Summarized data of c-Fos-positive neurons in the CeA from the indicated groups (n = 9 slices from four mice each group; t16 = 9.119, P < 0.0001). d,e, Representative images (d) and summarized data (e) of c-Fos-positive neurons that co-localized with SOM immunofluorescence in the CeA (n = 9 slices from three mice). Scale bar, 50 μm. f,g, Sample traces (f) and summarized data (g) of the spontaneous action potential firing rate showing the effect of optical activation of 5-HTDRN terminals in the CeA with or without perfusion of WAY (n = 10 cells each group; F1,18 = 10.69, P = 0.0043). h, Representative images and percentage of 5-HT2AR immunoreactivity in non-SOM neurons in the CeA (n = 9 slice from three mice). Scale bar, 50 μm. For e and h, data are shown as box and whisker plots [medians, quartiles (boxes) and ranges min to max (whiskers)]. Significance was assessed by two-tailed unpaired Student’s t test in c, and two-way RM ANOVA with post hoc comparison between groups in g. The data are expressed as the mean ± s.e.m. ***P < 0.001. ns, no significance.
Supplementary Figure 4 Behavioural phenotypes in mice treated with CFA or SNI.
a, Time course of CFA-induced sensory pain (saline, n = 8 mice; CFA, n = 13 mice; F8,169 = 4.181, P = 0.0001). b, Performance of sham 2W and SNI 2W mice in SPT, TST, OFT, and EPM (sham 2W, n = 10 mice; SNI 2W, n = 15 mice; SPT, t23 = 0.0424, P = 0.9666; TST, t23 = 0.490, P = 0.6291; OFT, t23 = 1.388, P = 0.1783; EPM, t23 = 0.4599, P = 0.6499). c, Performance of mice treated with CFA or saline for two or three weeks in SPT, TST, OFT, and EPM (n = 12 mice per group; SPT, F3,44 = 4.261, P = 0.01; TST, F3,44 = 6.006, P = 0.0016; OFT, F3,44 = 4.677, P = 0.0064; EPM, F3,44 = 4.489, P = 0.0078). d, e,Typical traces of 5-HT detected by electrochemical detection combined with HPLC (d) and summarized data of 5-HT concentration in the CeA from the indicated groups (e, n = 8 mice each group; F3,28 = 3.525, P = 0.0277). f, Summarized data of the resting membrane potential (Vrest) recorded from SOMCeA neurons in the indicated groups (Sham 6W, n = 12 cells; SNI 6W, n = 15 cells; t25 = 1.524, P = 0.1401). Significance was assessed by two-way RM ANOVA with post hoc comparison between groups in a, two-tailed unpaired Student’s t test in b, f, and ordinary one-way ANOVA with post hoc comparison between groups in c, e. The data are expressed as the mean ± s.e.m. *P < 0.05; **P < 0.01; ***P < 0.001. ns, no significance.
Supplementary Figure 5 Inhibition of the 5-HTDRN→CeA pathway produces depressive-like behaviors.
a, Behavioural effects of optogenetic activation of 5-HTDRN terminals in the CeA in response to WAY (mCherry + ASCF and mCherry + WAY, n = 10 mice per group; ChR2 + ACSF and ChR2 + WAY, n = 9 mice per group; F3,34 = 15.83, P < 0.0001; pre vs light post hoc comparison, mCherry + ASCF, P > 0.9; ChR2 + ACSF, P < 0.0001; ChR2 + WAY, P > 0.9; mCherry + WAY, P = 0.3947). b, Pain threshold in Pet1-Cre mice with DRN infusion of AAV-DIO-ChR2-mCherry (ChR2-mCherry) or control virus (mCherry) after photostimulation in the CeA (n = 9 mice per group; F2,32 = 5.03, P = 0.0126). c, Schematic of viral injection and optical manipulation. d, Sample trace (left) and statistical data (right) of currents recorded from AAV-DIO-eNpHR3.0-EYFP-expressing 5-HTDRN neurons via photostimulation (594 nm) in acute slices from Pet1-Cre mice (n = 9 cells, one sample t test, t8 = −12.564, P < 0.0001). e, Statistic charts showing the behavioural effects of inhibition of 5-HTDRN terminals in the CeA (EYFP, n = 9 mice; eNpHR3.0-EYFP, n = 10 mice; SPT, t17 = 2.815, P = 0.0119; TST, F1,17 = 24.35, P = 0.0001; pre vs light post hoc comparison, EYFP, P = 0.1810, eNpHR3.0-EYFP, P = 0.0001). Significance was assessed by two-way RM ANOVA with post hoc comparison between groups in a, b, e (TST), and two-tailed unpaired Student’s t test in e (SPT). The data are expressed as the mean ± s.e.m. *P < 0.05; **P < 0.01; ***P < 0.001. ns, no significance.
Supplementary Figure 6 Behavioural effects of activation of the 5-HTDRN→SOMCeA circuit in mice treated with chronic stress.
a, Performance of chronic restraint stress (CRS) and control (CON) mice in SPT, TST, OFT, and EPM (n = 10 per group; SPT, t18 = 2.669, P = 0.0188; TST, t18 = 2.865, P = 0.0103; OFT, t18 = 2.366, P = 0.0322; EPM, t18 = 2.375, P = 0.0289). b, Summarized data of 5-HT concentration in the CeA from CRS and CON mice (n = 6 per group; t10 = 0.6872, P = 0.5076). c, Performance of social defeat stress (SDS, n = 15) and control (CON, n = 10) mice in SPT, TST, OFT, and EPM (SPT, t23 = 3.139, P = 0.0046; TST, t23 = 2.603, P = 0.0159; OFT, t23 = 5.282, P < 0.0001; EPM, t23 = 4.354, P = 0.0002). d, Summarized data of 5-HT concentration in the CeA from SDS or CON mice (n = 6 per group; t10 = 0.5819, P = 0.5735). e, Statistical data showing the effect of optical activation of 5-HTDRN terminals in the CeA in CRS mice (CRS + mCherry, n = 10 mice; CRS + ChR2-mCherry, n = 8 mice; SPT, t16 = 0.5517, P = 0.5888; TST, F2,32 = 1.661, P = 0.2059). f, Statistical data showing the effect of optical activation of 5-HTDRN terminals in the CeA in SDS mice. (n = 8 per group; SPT, t16 = 0.1535, P = 0.8802; TST, F2,28 = 0.474, P = 0.6274). Significance was assessed by two-tailed unpaired Student’s t test in a-d, e (SPT), f (SPT), and two-way RM ANOVA with post hoc comparison between groups in e (TST), f (TST). The data are expressed as the mean ± s.e.m. *P < 0.05; **P < 0.01; ***P < 0.001. ns, no significance.
Supplementary Figure 7 Outputs of SOMCeA neurons.
a, Representative images of mCherry expression in SOM-Cre mice after CeA injection of AAV-DIO-ChR2-mCherry. Scale bars: 300 µm (left), 50 µm (right). b–f, mCherry signals in the lateral periaqueductal gray matter (LPAG, b), the nucleus accumbens (AcbC, c), the medial geniculate nucleus (MG, d), the bed nucleus of the stria terminalis (BNST, e), and the substantia nigra pars reticulata (SNR, f). Scale bars: 500 µm (left), 50 µm (right). The experiment was independently repeated five times with similar results obtained.
Supplementary Figure 8 Activation of the SOMCeA→LHb pathway produces depressive-like behaviours.
a, Schematic of the Cre-dependent retrograde trans-monosynaptic rabies virus tracing strategy. b, Representative images of viral expression within the LHb of the GAD2-Cre mice (left). No DsRed signal was observed in the CeA (right). The experiment was independently repeated with similar results in n = 5 mice. Scale bars: 100 µm (left), 200 µm (right). c,d, Representative images (c) and percentage (d) of DsRed signals from the LHb co-localized with 5-HT1ARs in the CeA (n = 9 slices from three mice). Scale bar: 50 µm. e, Representative images and percentage of DsRed signals from the LHb co-localized with 5-HT2ARs in the CeA (n = 9 slices from three mice). Scale bar: 50 µm. f, Schematic of viral injection for CeA-LHb tracing. g,h, Typical images of GFP expression in the LHb of mice with CeA infusion of AAV2/1-hSyn-Cre (g, Scale bars: 300 µm left, 50 µm right), which co-localized with VGLUT2 (h, Scale bar: 50 µm). The experiment was independently repeated with similar results in n = 9 slices from three mice. i, Schematic of viral injection and optical manipulation in the LHb. j, Behavioural effects of photostimulation in the LHb of SOM-Cre mice with CeA injection of AAV-DIO-ChR2-mCherry or control virus (ChR2-mCherry, n = 10 mice; mCherry, n = 8 mice; SPT, t16 = 2.285, P = 0.0398; TST, F2,32 = 3.572, P = 0.0398; pre vs light post hoc comparison, ChR2-mCherry, P = 0.0004; mCherry, P > 0.9). k, Behavioural effects of optical inhibition of SOMCeA terminals in the LHb of SNI 6W SOM-Cre mice with CeA injection of AAV-DIO-eNpHR3.0-EYFP (eNpHR3.0-EYFP) or control virus EYFP (eNpHR3.0-EYFP, n = 9; EYFP, n = 10; F2,34 = 7.809, P = 0.0016; pre vs light post hoc comparison, eNpHR3.0-EYFP, P < 0.0001; EYFP, P > 0.9). For d, e and h, data are shown as box and whisker plots [medians, quartiles (boxes) and ranges min to max (whiskers)]. Significance was assessed by two-tailed unpaired Student’s t test in j (SPT), and two-way RM ANOVA with post hoc comparison between groups in j (TST), k. The data are expressed as the mean ± s.e.m. *P < 0.05; ***P < 0.001. ns, no significance.
Supplementary Figure 9 A model of the disinhibitory 5-HTDRN→SOMCeA→GluLHb circuit in CDS.
In the chronic pain state, a disinhibitory process occurs within the CeA, which involves decreased 5-HTDRN inputs that mediate inhibition of SOMCeA neurons (red) through 5-HT1AR, but mediate excitation of non-SOM interneurons (NSCeA, green) through 5-HT2AR. Both alterations lead to even higher excitation of SOMCeA neurons. As a result, the SOMCeA glutamatergic outputs (red) to the LHb are enhanced, which presumably causes CDS. DRN, dorsal raphe nucleus; LHb, lateral habenula; CeA, central nucleus of the amygdala; 5-HT, 5-hydroxytryptamine; 5-HT1ARs, 5-HT1A receptors; 5-HT2ARs, 5-HT2A receptors; GABA, gamma-aminobutyric acid; Glu, glutamate; AMPARs, α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors.
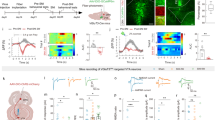
Supplementary Figure 10 Altered functional connectivity with the right CM in CDS patients.
a, Location of the subregions of the human right amygdala. Red, centromedial amygdala (CM); green, laterobasal nuclei; blue, superficial nuclei. b,c, fMRI images showing brain areas with significant differences in functional connectivity with the right CM in CDS patients (n = 22) compared with depressed patients without chronic pain (b, n = 27) and healthy controls (c, n = 27). d,e, Brain areas showing altered functional connectivity with the right CM in depressed patients without chronic pain (d) and chronic pain patients without depression (e, n = 29) compared with healthy controls. f, Statistical values of CM-DRN functional connectivity in the indicated groups (F3,101 = 13.72, P < 0.0001). g, The Pearson correlation analysis of the functional connectivity of the DRN-CM pathway and the HAMD score in CDS and MDD patients (CDS, P = 0.0169, r = -0.5036; MDD, P = 0.2029, r = -0.253). h, Summarized data of the HAMD scores of CDS and MDD patients (t47 = 0.9976, P = 0.3236). MDD, major depression disorder without pain; CP, chronic pain without depression. The numbers beneath the images refer to the coordinates in the Montreal Neurological Institute space. The red circles in each image represent the regions that contain the DRN. The t-score bars are shown at the right of images. Significance was assessed by one-way ANOVA with post hoc comparison between groups in f, and two-tailed unpaired Student’s t test in h. The data are expressed as the mean ± s.e.m. **P < 0.01; ***P < 0.001. ns, no significance.
Supplementary Figure 11 Altered functional connectivity with the left CM in CDS patients.
a–d, fMRI images showing brain areas with significant differences in functional connectivity with the amygdala compared with the indicated groups. e–h, fMRI images showing brain areas with significant differences in functional connectivity with the left CM compared with the indicated groups. The numbers beneath the images refer to the coordinates in the Montreal Neurological Institute space. The red circles in each image represent the regions that contain the DRN. The t-score bars are shown at the right of images.
Supplementary information
Rights and permissions
About this article
Cite this article
Zhou, W., Jin, Y., Meng, Q. et al. A neural circuit for comorbid depressive symptoms in chronic pain. Nat Neurosci 22, 1649–1658 (2019). https://doi.org/10.1038/s41593-019-0468-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-019-0468-2
This article is cited by
-
High frequency deep brain stimulation of the dorsal raphe nucleus prevents methamphetamine priming-induced reinstatement of drug seeking in rats
Translational Psychiatry (2024)
-
Somatosensory cortex and central amygdala regulate neuropathic pain-mediated peripheral immune response via vagal projections to the spleen
Nature Neuroscience (2024)
-
CRF regulates pain sensation by enhancement of corticoaccumbal excitatory synaptic transmission
Molecular Psychiatry (2024)
-
Network analysis of monoamines involved in anxiety-like behavior in a rat model of osteoarthritis
Pharmacological Reports (2024)
-
Electroacupuncture Exerts Analgesic Effects by Restoring Hyperactivity via Cannabinoid Type 1 Receptors in the Anterior Cingulate Cortex in Chronic Inflammatory Pain
Molecular Neurobiology (2024)