Abstract
Multiple sclerosis (MS) patients exhibit neuropsychological symptoms in early disease despite the immune attack occurring predominantly in white matter and spinal cord. It is unclear why neurodegeneration may start early in the disease and is prominent in later stages. We assessed cortical microcircuit activity by employing spiking-specific two-photon Ca2+ imaging in proteolipid protein-immunized relapsing-remitting SJL/J mice in vivo. We identified the emergence of hyperactive cortical neurons in remission only, independent of direct immune-mediated damage and paralleled by elevated anxiety. High levels of neuronal activity were accompanied by increased caspase-3 expression. Cortical TNFα expression was mainly increased by excitatory neurons in remission; blockade with intraventricular infliximab restored AMPA spontaneous excitatory postsynaptic current frequencies, completely recovered normal neuronal network activity patterns and alleviated elevated anxiety. This suggests a dysregulation of cortical networks attempting to achieve functional compensation by synaptic plasticity mechanisms, indicating a link between immune attack and early start of neurodegeneration.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors upon request.
Change history
07 November 2018
In the version of this article initially published, Inigo Ruiz de Azua’s name was miscategorized. His given name is Inigo and his surname is Ruiz de Azua. This has been corrected in the HTML coding.
References
Steinman, L. Immunology of relapse and remission in multiple sclerosis. Annu. Rev. Immunol. 32, 257–281 (2014).
Trapp, B. D. & Nave, K. A. Multiple sclerosis: an immune or neurodegenerative disorder? Annu. Rev. Neurosci. 31, 247–269 (2008).
Keegan, B. M. & Noseworthy, J. H. Multiple sclerosis. Annu. Rev. Med. 53, 285–302 (2002).
Peterson, J. W., Bö, L., Mörk, S., Chang, A. & Trapp, B. D. Transected neurites, apoptotic neurons, and reduced inflammation in cortical multiple sclerosis lesions. Ann. Neurol. 50, 389–400 (2001).
De Stefano, N. et al. Evidence of early cortical atrophy in MS: relevance to white matter changes and disability. Neurology 60, 1157–1162 (2003).
Feinstein, A., Magalhaes, S., Richard, J. F., Audet, B. & Moore, C. The link between multiple sclerosis and depression. Nat. Rev. Neurol. 10, 507–517 (2014).
Gold, R., Linington, C. & Lassmann, H. Understanding pathogenesis and therapy of multiple sclerosis via animal models: 70 years of merits and culprits in experimental autoimmune encephalomyelitis research. Brain 129, 1953–1971 (2006).
Ben-Nun, A. et al. From classic to spontaneous and humanized models of multiple sclerosis: impact on understanding pathogenesis and drug development. J. Autoimmun. 54, 33–50 (2014).
MacKenzie-Graham, A. et al. Cortical atrophy in experimental autoimmune encephalomyelitis: in vivo imaging. Neuroimage 60, 95–104 (2012).
Busche, M. A. et al. Clusters of hyperactive neurons near amyloid plaques in a mouse model of Alzheimer’s disease. Science 321, 1686–1689 (2008).
Grienberger, C. et al. Staged decline of neuronal function in vivo in an animal model of Alzheimer’s disease. Nat. Commun. 3, 774 (2012).
Kajikawa, Y. & Schroeder, C. E. How local is the local field potential? Neuron 72, 847–858 (2011).
Rochefort, N. L. et al. Sparsification of neuronal activity in the visual cortex at eye-opening. Proc. Natl. Acad. Sci. USA 106, 15049–15054 (2009).
Stroh, A. et al. Making waves: initiation and propagation of corticothalamic Ca2+ waves in vivo. Neuron 77, 1136–1150 (2013).
Svoboda, K., Denk, W., Kleinfeld, D. & Tank, D. W. In vivo dendritic calcium dynamics in neocortical pyramidal neurons. Nature 385, 161–165 (1997).
Beattie, E. C. et al. Control of synaptic strength by glial TNFα. Science 295, 2282–2285 (2002).
Mc Guire, C., Beyaert, R. & van Loo, G. Death receptor signalling in central nervous system inflammation and demyelination. Trends Neurosci. 34, 619–628 (2011).
Stosiek, C., Garaschuk, O., Holthoff, K. & Konnerth, A. In vivo two-photon calcium imaging of neuronal networks. Proc. Natl. Acad. Sci. USA 100, 7319–7324 (2003).
Vogt, J. et al. Lower motor neuron loss in multiple sclerosis and experimental autoimmune encephalomyelitis. Ann. Neurol. 66, 310–322 (2009).
Crawford, D. K., Mangiardi, M. & Tiwari-Woodruff, S. K. Assaying the functional effects of demyelination and remyelination: revisiting field potential recordings. J. Neurosci. Methods 182, 25–33 (2009).
Jolivel, V. et al. Modulation of dendritic cell properties by laquinimod as a mechanism for modulating multiple sclerosis. Brain 136, 1048–1066 (2013).
Aktas, O. et al. Neuronal damage in autoimmune neuroinflammation mediated by the death ligand TRAIL. Neuron 46, 421–432 (2005).
Aktas, O. et al. Treatment of relapsing paralysis in experimental encephalomyelitis by targeting Th1 cells through atorvastatin. J. Exp. Med. 197, 725–733 (2003).
Wong, A. A. & Brown, R. E. Visual detection, pattern discrimination and visual acuity in 14 strains of mice. Genes Brain Behav. 5, 389–403 (2006).
Chang, B. et al. Retinal degeneration mutants in the mouse. Vision. Res. 42, 517–525 (2002).
Guenthner, C. J., Miyamichi, K., Yang, H. H., Heller, H. C. & Luo, L. Permanent genetic access to transiently active neurons via TRAP: targeted recombination in active populations. Neuron 78, 773–784 (2013).
Black, R. A. et al. A metalloproteinase disintegrin that releases tumour-necrosis factor-alpha from cells. Nature 385, 729–733 (1997).
Mbebi, C. et al. Antibody-bound beta-amyloid precursor protein stimulates the production of tumor necrosis factor-alpha and monocyte chemoattractant protein-1 by cortical neurons. Neurobiol. Dis. 19, 129–141 (2005).
Liu, T. et al. Tumor necrosis factor-alpha expression in ischemic neurons. Stroke 25, 1481–1488 (1994).
Stellwagen, D. & Malenka, R. C. Synaptic scaling mediated by glial TNF-alpha. Nature 440, 1054–1059 (2006).
Neumann, H., Schmidt, H., Cavalié, A., Jenne, D. & Wekerle, H. Major histocompatibility complex (MHC) class I gene expression in single neurons of the central nervous system: differential regulation by interferon (IFN)-gamma and tumor necrosis factor (TNF)-alpha. J. Exp. Med. 185, 305–316 (1997).
Hallermalm, K. et al. Tumor necrosis factor-α induces coordinated changes in major histocompatibility class I presentation pathway, resulting in increased stability of class I complexes at the cell surface. Blood 98, 1108–1115 (2001).
Höftberger, R. et al. Expression of major histocompatibility complex class I molecules on the different cell types in multiple sclerosis lesions. Brain Pathol. 14, 43–50 (2004).
Huh, G. S. et al. Functional requirement for class I MHC in CNS development and plasticity. Science 290, 2155–2159 (2000).
Glynn, M. W. et al. MHCI negatively regulates synapse density during the establishment of cortical connections. Nat. Neurosci. 14, 442–451 (2011).
Methner, A. & Zipp, F. Multiple sclerosis in 2012: novel therapeutic options and drug targets in MS. Nat. Rev. Neurol. 9, 72–73 (2013).
Busche, M. A. et al. Rescue of long-range circuit dysfunction in Alzheimer’s disease models. Nat. Neurosci. 18, 1623–1630 (2015).
Busche, M. A. & Konnerth, A. Neuronal hyperactivity–A key defect in Alzheimer’s disease? BioEssays 37, 624–632 (2015).
Putcha, D. et al. Hippocampal hyperactivation associated with cortical thinning in Alzheimer’s disease signature regions in non-demented elderly adults. J. Neurosci. 31, 17680–17688 (2011).
Zhang, L., Yang, H., Zhao, H. & Zhao, C. Calcium-related signaling pathways contributed to dopamine-induced cortical neuron apoptosis. Neurochem. Int. 58, 281–294 (2011).
Sakry, D. et al. Oligodendrocyte precursor cells modulate the neuronal network by activity-dependent ectodomain cleavage of glial NG2. PLoS Biol. 12, e1001993 (2014).
Korostil, M. & Feinstein, A. Anxiety disorders and their clinical correlates in multiple sclerosis patients. Mult. Scler. 13, 67–72 (2007).
Ribbons, K., Lea, R., Schofield, P. W. & Lechner-Scott, J. Anxiety levels are independently associated with cognitive performance in an australian multiple sclerosis patient cohort. J. Neuropsychiatry Clin. Neurosci. 29, 128–134 (2017).
Paxinos, G. & Franklin, K.B.J. Paxinos and Franklin’s the Mouse Brain in Stereotaxic Coordinates (Elsevier Academic Press, 2012).
Schwalm, M. et al. Cortex-wide BOLD fMRI activity reflects locally-recorded slow oscillation-associated calcium waves. eLife 6, e27602 (2017).
Garaschuk, O. Imaging microcircuit function in healthy and diseased brain. Exp. Neurol. 242, 41–49 (2013).
Nimmerjahn, A., Kirchhoff, F., Kerr, J. N. & Helmchen, F. Sulforhodamine 101 as a specific marker of astroglia in the neocortex in vivo. Nat. Methods 1, 31–37 (2004).
Baker, A. J. et al. Attenuation of the electrophysiological function of the corpus callosum after fluid percussion injury in the rat. J. Neurotrauma 19, 587–599 (2002).
Patel, R. et al. Attenuation of corpus callosum axon myelination and remyelination in the absence of circulating sex hormones. Brain Pathol. 23, 462–475 (2013).
Reeves, T. M., Phillips, L. L. & Povlishock, J. T. Myelinated and unmyelinated axons of the corpus callosum differ in vulnerability and functional recovery following traumatic brain injury. Exp. Neurol. 196, 126–137 (2005).
Siffrin, V. et al. In vivo imaging of partially reversible th17 cell-induced neuronal dysfunction in the course of encephalomyelitis. Immunity 33, 424–436 (2010).
Paterka, M. et al. Gatekeeper role of brain antigen-presenting CD11c+ cells in neuroinflammation. EMBO J. 35, 89–101 (2016).
Spiwoks-Becker, I., Lamberti, R., Tom Dieck, S. & Spessert, R. Evidence for synergistic and complementary roles of Bassoon and darkness in organizing the ribbon synapse. Neuroscience 236, 149–159 (2013).
Luchtman, D. et al. In vivo and in vitro effects of multiple sclerosis immunomodulatory therapeutics on glutamatergic excitotoxicity. J. Neurochem. 136, 971–980 (2016).
Becker, D., Zahn, N., Deller, T. & Vlachos, A. Tumor necrosis factor alpha maintains denervation-induced homeostatic synaptic plasticity of mouse dentate granule cells. Front. Cell. Neurosci. 7, 257 (2013).
Bussey, T. J. et al. The touchscreen cognitive testing method for rodents: how to get the best out of your rat. Learn. Mem. 15, 516–523 (2008).
Rowland, N. E. Food or fluid restriction in common laboratory animals: balancing welfare considerations with scientific inquiry. Comp. Med. 57, 149–160 (2007).
Acknowledgements
This study was supported by the German Research Council (DFG, CRC-TR-128 (to F.Z., S.B., T.K. and A.S.) and CRC-1080 (to A.S., T.M. and F.Z.)). We thank C. Fois, J. Doering, P.-H. Prouvot, M. Schwalm, E. Witsch, F. Aedo-Jury, K. Radyushkin and K. Robohm for support. We would like to thank A. Zymny, C. Liefländer, H. Ehrengard, O. Levai and I. Gravenitz for excellent technical assistance and D. O’Neill, C. Ernest and S. Stroh for proofreading the manuscript.
Author information
Authors and Affiliations
Contributions
E.E. and G.P. contributed equally and are listed in alphabetical order. A.S. and F.Z. designed the study. The corresponding authors were unable to reach E.R.J. for final approval of the author list and contributions statement. E.E. conducted and analyzed cytokine production; E.E. and S.M. conducted EAE, mRNA analyses and infliximab treatment. E.E. performed the EM analysis. G.P. conducted the two-photon Ca2+ imaging experiments. G.P., E.R.J. and Z.B. analyzed the Ca2+ imaging data. T.N. and T.M. performed and analyzed electrophysiology recordings. D.L., E.E. and M.S. conducted the behavioral tests. G.P., E.E., T.K., I.A., I.R.A., B.L., C.F.V., S.B. and J.V. conducted and analyzed the histochemistry, ISH and immunofluorescence. A.S., F.Z., G.P., E.E., D.L., S.B. and T.M. wrote the manuscript. A.S., F.Z. and E.E. edited the text.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
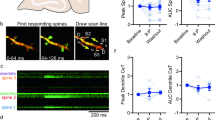
Supplementary Figure 1 Staining of neurons and astrocytes by the synthetic indicator OGB-1.
(a) Cortical neurons stained with Oregon Green BAPTA-1 AM (OGB-1) using multi-cell bolus loading technique. (b) Simultaneously recorded astrocytes counterstained with Sulforhodamine-101 (SR101). (c) An overlay of OGB-1-loaded neurons (green) and astrocytes with SR101 (yellow). Experiment was repeated three times. Scale bar = 25 µm.
Supplementary Figure 2 Immunofluorescence for TNFα in EAE remission.
Coronal slices from remission animals were stained with the following antibodies to label specific cell types for idenfication of the source of TNFα: (a) astrocytic anti-EAAT1, (b) microglial anti-Iba-1, (c) neuronal marker anti-NeuN, (d) anti-GAD67 for GABAergic neurons and (e) anti-CamKII for CamKII+ excitatory neurons; DAPI was used for nuclear staining. (Center) TNFα was stained with anti-TNFα (green), (Right) Merged images of respective stainings are shown. (Scale bars = 30 µm; arrow heads indicate cells which do not express TNFα, arrows indicate cells which are positive for TNFα.) (f) (Left) Magnification of staining for TNFα, scale bar = 1 µm. (Right) Superresolution image (resolution 36 nm) showing the intracellular compartment, which was delineated by CamKII staining (red). Remarkably, TNF alpha (green) immunosignal was present in the intracellular compartment as well as in the extracellular compartment, which is in line with neuronal TNFα secretion. Representative images from 5 animals, scale bar = 500 nm.
Supplementary Figure 3 Immunofluorescence and ISH for TNFα in healthy control animals.
Coronal images of brain slices stained with the following antibodies: (a) anti-NeuN, (b) anti-GAD67, (c) anti-CamKII and DAPI. Note there was less co-expression of TNFα found in anti-NeuN and anti-CamKII staining compared to the EAE-remission mice. (Scale bars = 8 µm; n = 5 mice). (d–g) In-situ hybridization for TNFα in control animals. Stainings for GFAP and neurotrace in sagittal slices. Representative images from 5 animals. (Scale bars overview = 50 µm, inserts = 8 µm).
Supplementary Figure 4 Immunohistochemistry staining for amyloid precursor protein (APP) in remission.
Anti-APP staining in coronal brain slices from remission animals shows no axonal degeneration in the cortex. Representative images from 4 animals. Scale bars overview image = 400 μm, inserts = 50 μm.
Supplementary Figure 5 Distribution of Ca2+ transient frequencies for all experimental groups.
Cumulative non-binned normalized frequency graph of healthy controls, relapse, and remission mice; a significant shift in frequency distribution was found in remission compared to control and relapse (mean is displayed, two-sided K-S, P < 0.001) but not in infliximab-treated animals in remission. Control n = 567 neurons, 6 mice; relapse n = 595 neurons, 6 mice; remission n = 650 neurons, 5 mice, infliximab-treated remission n = 525 neurons, 4 mice.
Supplementary Figure 6 Immunohistochemical staining for myelin-basic-protein (MBP) to assess myelin sheath damage in control, relapse and remission.
(a–c) Anti-MBP staining of sagittal slices of the cortex shows intact myelination in relapse and remission animals, similar to control. The lower images are enlargements of the areas enclosed by squares in the respective upper images. Representative images from 4 animals per group. (Scale bars overview = 500 µm, middle and lower inserts = 100 µm, 50 µm respectively).
Supplementary Figure 7 No significant immune cell infiltration present in the cortex in remission.
(a–c) Histological stainings for Mac-3+ activated macrophages and CD3+ T cells in the brain and spinal cord. No Mac-3+ or CD3+ cells were found in control animals but a few Mac-3+ and CD3+ cells could be visualized mainly in the spinal cord of relapse and remission animals. Scale bars of overview images of brain slices = 400 µm, and inserts = 100 µm; scale bars of overview images of spinal cord slices = 400 µm, and inserts = 50 µm. (d) Quantitative assessment of percentage of Mac-3+ activated macrophages in the cortex, including all cortical layers, revealed a significant increase in Mac-3+ macrophage infiltration only in relapse phase (one-way ANOVA, *P = 0.02), not in remission (left panel). Restricting the analysis to layer II/III only resulted in no significant differences between all three groups (right panel). Control n = 16 cortical slices from 4 animals, relapse n = 14 cortical slices from 4 animals, remission n = 18 cortical slices from 6 animals. (e) Analyzing for CD3+ cells in the whole cortex, no significant differences were observed between all groups, albeit a trend towards higher number in relapse (left panel), and a significant increase in relapse when analyzing only layer II/III (right panel; one-way ANOVA, *P = 0.013). No significant differences were observed in remission for either the whole cortex or only layer II/III. Control n = 10 cortical slices from 4 animals, relapse n = 8 cortical slices from 4 animals, remission n = 12 cortical slices from 4 animals. Data represented as mean and SEM.
Supplementary Figure 8 Microglial morphology in the remission cortex remains unchanged.
(a) Anti-Iba-1 staining for microglial morphology in the visual cortex showed no obvious changes in any group, scale for overview images = 50 µm. (b) Quantification of ramification of microglia revealed no significant differences for shape factor, form factor or solidity (one-way ANOVA) between groups. Control n = 6 cortical slices from 3 animals, relapse n = 6 cortical slices from 3 animals and remission n = 6 cortical slices from 3 animals, box-and-whisker plot indicates the median value (center line), the 25–75th percentiles (box) and the 10–90th percentiles (whiskers).
Supplementary Figure 9 No changes in expression of ion channels Nav1.6 or Kv7.3 in remission.
Exemplary immunohistochemical stainings for Nav1.6 or Kv7.3 and βIII-tubulin (βIII-tub) in the visual cortex of (a) control and (b) SJL-EAE remission animals show no changes of expression patterns. Representative images from 4 animals per group.
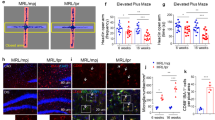
Supplementary Figure 10 Remission phase mice show normal visual discrimination and rotarod performance.
(a) Schematic illustration of visual discrimination task set-up. (b) Pre-training of EAE mice to perform visual discrimination tasks. No difference observed between healthy controls and mice before EAE induction when pre-training was performed. (c) Time course of the correct choice in visual discrimination task in relation to the clinical EAE score during remission (mean ± SEM; healthy controls n = 10; remission phase animals n = 10). (d) Rotarod behavior task. Latencies (s) until the mice fell off for each group are shown. Box-and-whisker plot indicates the median value (center line), the 25 –75th percentiles (box) and the 10–90th percentiles (whiskers); (two-sided t-test, P = 0.8062; healthy controls n = 9; remission phase animals n = 10).
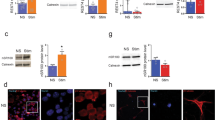
Supplementary Figure 11 Release of TNFα by primary cortical neurons.
(a) qPCR analysis for TNFα of primary cortical neurons stimulated with LPS for 24 h, values are normalized to control (Stim. 1, n = 8; Stim. 2, n = 8; and n = 6 for controls, horizontal line depicts median value, Kruskal-Wallis test, **P < 0.01). (b) Quantitative comparison of TNFα release from primary cortical neurons in controls (unstimulated, n = 16), treated with Stim. 1 (n = 14) and Stim. 2 (n = 20) for 24 h. Data points are normalized to control, median value is indicated, Kruskal-Wallis test, *P < 0.05 and ***P < 0.001. Stim. 1: 0.01-0.1 µg/ml LPS, Stim. 2: 1-10 µg/ml LPS; LPS = lipopolysaccharide.
Supplementary Figure 12 Immunofluorescence for MHC class I in EAE remission animals.
(a-c) Confocal images of brain slices stained with the following antibodies: (a) anti-Iba-1, (b) anti-GFAP, (c) anti-CamKII and DAPI. Astrocytes did not show colocalization with MHC class I (arrow heads). Neurons and microglia showed staining with MHC I (arrows). Representative images from 4 animals. (Scale bars overview = 50 µm, inserts = 8 µm).
Supplementary Figure 13 Scheme of dysregulation of cortical networks in EAE remission.
We did not observe immune cell infiltration or demyelination in the cortex in SJL remission (a). We could demonstrate that mainly excitatory neurons release TNFα (b) and that the spontaneous neuronal network activity increases (c), which might lead to cell death in cells with highest activity (d). TNFα blocking on the other hand ameliorates neuronal activity and behaviour (e).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–13-
Supplementary Video 1 - Two-photon Ca2+ imaging in visual cortex, layer II/III of a control mouse.
Real-time video; recording at 30 Hz of a local microcircuit.
Supplementary Video 2 - Two-photon Ca2+ imaging in visual cortex, layer II/III of an EAE mouse, Relapse.
Real-time video; recording at 30 Hz of a local microcircuit.
Supplementary Video 3 - Two-photon Ca2+ imaging in visual cortex, layer II/III of an EAE mouse, Remission.
Real-time video; recording at 30 Hz of a local microcircuit.
Supplementary Video 4 - Visual discrimination task.
Visual discrimination task for mice as depicted in Supplementary Figure 10.
Rights and permissions
About this article
Cite this article
Ellwardt, E., Pramanik, G., Luchtman, D. et al. Maladaptive cortical hyperactivity upon recovery from experimental autoimmune encephalomyelitis. Nat Neurosci 21, 1392–1403 (2018). https://doi.org/10.1038/s41593-018-0193-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-018-0193-2
This article is cited by
-
Repetitive and compulsive behavior after Early-Life-Pain associated with reduced long-chain sphingolipid species
Cell & Bioscience (2023)
-
Significance of an altered lncRNA landscape in schizophrenia and cognition: clues from a case–control association study
European Archives of Psychiatry and Clinical Neuroscience (2023)
-
NOX4-derived ROS are neuroprotective by balancing intracellular calcium stores
Cellular and Molecular Life Sciences (2023)
-
Network alterations underlying anxiety symptoms in early multiple sclerosis
Journal of Neuroinflammation (2022)
-
Acitretin reverses early functional network degradation in a mouse model of familial Alzheimer’s disease
Scientific Reports (2021)