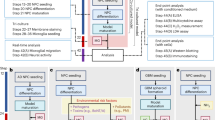
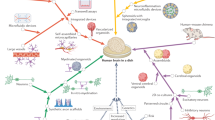
Abstract
Alzheimer’s disease (AD) is characterized by beta-amyloid accumulation, phosphorylated tau formation, hyperactivation of glial cells, and neuronal loss. The mechanisms of AD pathogenesis, however, remain poorly understood, partially due to the lack of relevant models that can comprehensively recapitulate multistage intercellular interactions in human AD brains. Here we present a new three-dimensional (3D) human AD triculture model using neurons, astrocytes, and microglia in a 3D microfluidic platform. Our model provided key representative AD features: beta-amyloid aggregation, phosphorylated tau accumulation, and neuroinflammatory activity. In particular, the model mirrored microglial recruitment, neurotoxic activities such as axonal cleavage, and NO release damaging AD neurons and astrocytes. Our model will serve to facilitate the development of more precise human brain models for basic mechanistic studies in neural–glial interactions and drug discovery.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Alzheimer’s Association. 2016 Alzheimer’s disease facts and figures. Alzheimers Dement. 12, 459–509 (2016).
Tanzi, R. E. & Bertram, L. Twenty years of the Alzheimer’s disease amyloid hypothesis: a genetic perspective. Cell 120, 545–555 (2005).
Karran, E. & De Strooper, B. The amyloid cascade hypothesis: are we poised for success or failure? J. Neurochem. 139, 237–252 (2016). Suppl 2.
Armstrong, R. A. A critical analysis of the ‘amyloid cascade hypothesis’. Folia Neuropathol. 52, 211–225 (2014).
Paquet, D. et al. Efficient introduction of specific homozygous and heterozygous mutations using CRISPR/Cas9. Nature 533, 125–129 (2016).
Tubsuwan, A. et al. Generation of induced pluripotent stem cells (iPSCs) from an Alzheimer’s disease patient carrying a L150P mutation in PSEN-1. Stem Cell Res. 16, 110–112 (2016).
Moore, S. et al. APP metabolism regulates tau proteostasis in human cerebral cortex neurons. Cell Rep. 11, 689–696 (2015).
Muratore, C. R. et al. The familial Alzheimer’s disease APPV717I mutation alters APP processing and Tau expression in iPSC-derived neurons. Hum. Mol. Genet. 23, 3523–3536 (2014).
Sproul, A. A. et al. Characterization and molecular profiling of PSEN1 familial Alzheimer’s disease iPSC-derived neural progenitors. PLoS One 9, e84547 (2014).
Kondo, T. et al. Modeling Alzheimer’s disease with iPSCs reveals stress phenotypes associated with intracellular Aβ and differential drug responsiveness. Cell Stem Cell 12, 487–496 (2013).
Yagi, T. et al. Establishment of induced pluripotent stem cells from centenarians for neurodegenerative disease research. PLoS One 7, e41572 (2012).
Koch, P. et al. Presenilin-1 L166P mutant human pluripotent stem cell-derived neurons exhibit partial loss of γ-secretase activity in endogenous amyloid-β generation. Am. J. Pathol. 180, 2404–2416 (2012).
Shi, Y. et al. A human stem cell model of early Alzheimer’s disease pathology in Down syndrome. Sci. Transl. Med. 4, 124ra29 (2012).
Israel, M. A. et al. Probing sporadic and familial Alzheimer’s disease using induced pluripotent stem cells. Nature 482, 216–220 (2012).
Choi, S. H. et al. A three-dimensional human neural cell culture model of Alzheimer’s disease. Nature 515, 274–278 (2014).
Kim, Y. H. et al. A 3D human neural cell culture system for modeling Alzheimer’s disease. Nat. Protoc. 10, 985–1006 (2015).
D’Avanzo, C. et al. Alzheimer’s in 3D culture: challenges and perspectives. BioEssays 37, 1139–1148 (2015).
Choi, S. H., Kim, Y. H., Quinti, L., Tanzi, R. E. & Kim, D. Y. 3D culture models of Alzheimer’s disease: a road map to a “cure-in-a-dish”. Mol. Neurodegener. 11, 75 (2016).
Heneka, M. T. et al. NLRP3 is activated in Alzheimer’s disease and contributes to pathology in APP/PS1 mice. Nature 493, 674–678 (2013).
Griciuc, A. et al. Alzheimer’s disease risk gene CD33 inhibits microglial uptake of amyloid beta. Neuron 78, 631–643 (2013).
Butovsky, O. et al. Identification of a unique TGF-β-dependent molecular and functional signature in microglia. Nat. Neurosci. 17, 131–143 (2014).
Crehan, H., Hardy, J. & Pocock, J. Blockage of CR1 prevents activation of rodent microglia. Neurobiol. Dis. 54, 139–149 (2013).
Guerreiro, R. et al. TREM2 variants in Alzheimer’s disease. N. Engl. J. Med. 368, 117–127 (2013).
Xu, Q., Li, Y., Cyras, C., Sanan, D. A. & Cordell, B. Isolation and characterization of apolipoproteins from murine microglia. Identification of a low density lipoprotein-like apolipoprotein J-rich but E-poor spherical particle. J. Biol. Chem. 275, 31770–31777 (2000).
Zhang, Z.-N. et al. Layered hydrogels accelerate iPSC-derived neuronal maturation and reveal migration defects caused by MeCP2 dysfunction. Proc. Natl. Acad. Sci. USA 113, 3185–3190 (2016).
Han, S. et al. Three-dimensional extracellular matrix-mediated neural stem cell differentiation in a microfluidic device. Lab Chip 12, 2305–2308 (2012).
Tischbirek, C., Birkner, A., Jia, H., Sakmann, B., & Konnerth, A. Deep two-photon brain imaging with a red-shifted fluorometric Ca2+ indicator. Proc. Natl. Acad. Sci. USA 112, 11377–11382 (2015).
El Khoury, J. B. et al. CD36 mediates the innate host response to β-amyloid. J. Exp. Med. 197, 1657–1666 (2003).
El Khoury, J. et al. Ccr2 deficiency impairs microglial accumulation and accelerates progression of Alzheimer-like disease. Nat. Med. 13, 432–438 (2007).
Brion, J. P. Neurofibrillary tangles and Alzheimer’s disease. Eur. Neurol. 40, 130–140 (1998).
Li, S.-Q. et al. Deficiency of macrophage migration inhibitory factor attenuates tau hyperphosphorylation in mouse models of Alzheimer’s disease. J. Neuroinflamm. 12, 177 (2015).
Spangenberg, E. E. et al. Eliminating microglia in Alzheimer’s mice prevents neuronal loss without modulating amyloid-β pathology. Brain 139, 1265–1281 (2016).
Papageorgiou, I. E. et al. TLR4-activated microglia require IFN-? to induce severe neuronal dysfunction and death in situ. Proc. Natl. Acad. Sci. USA 113, 212–217 (2016).
Browne, T. C. et al. IFN-γ production by amyloid β-specific Th1 cells promotes microglial activation and increases plaque burden in a mouse model of Alzheimer’s disease. J. Immunol. 190, 2241–2251 (2013).
Garwood, C. J., Pooler, A. M., Atherton, J., Hanger, D. P. & Noble, W. Astrocytes are important mediators of Aβ-induced neurotoxicity and tau phosphorylation in primary culture. Cell Death Dis. 2, e167 (2011).
White, J. A., Manelli, A. M., Holmberg, K. H., Van Eldik, L. J. & Ladu, M. J. Differential effects of oligomeric and fibrillar amyloid-β 1-42 on astrocyte-mediated inflammation. Neurobiol. Dis. 18, 459–465 (2005).
Xiao, B. G. & Link, H. IFN-gamma production of adult rat astrocytes triggered by TNF-alpha. Neuroreport 9, 1487–1490 (1998).
Hashioka, S., Klegeris, A., Schwab, C., Yu, S. & McGeer, P. L. Differential expression of interferon-γ receptor on human glial cells in vivo and in vitro. J. Neuroimmunol. 225, 91–99 (2010).
Cho, H. et al. Microfluidic chemotaxis platform for differentiating the roles of soluble and bound amyloid-β on microglial accumulation. Sci. Rep. 3, 1823 (2013).
Smits, H. A. et al. Amyloid-β-induced chemokine production in primary human macrophages and astrocytes. J. Neuroimmunol. 127, 160–168 (2002).
Baruch, K. et al. Breaking immune tolerance by targeting Foxp3(+) regulatory T cells mitigates Alzheimer’s disease pathology. Nat. Commun. 6, 7967 (2015).
Haynes, S. E. et al. The P2Y12 receptor regulates microglial activation by extracellular nucleotides. Nat. Neurosci. 9, 1512–1519 (2006).
Dou, Y. et al. Microglial migration mediated by ATP-induced ATP release from lysosomes. Cell Res. 22, 1022–1033 (2012).
Perez de Lara, M. J. & Pintor, J. Presence and release of ATP from the retina in an Alzheimer’s disease model. J. Alzheimers Dis. 43, 177–181 (2015).
Johansson, J. U. et al. Suppression of inflammation with conditional deletion of the prostaglandin E2 EP2 receptor in macrophages and brain microglia. J. Neurosci. 33, 16016–16032 (2013).
Hong, S. et al. Complement and microglia mediate early synapse loss in Alzheimer mouse models. Science 352, 712–716 (2016).
Acknowledgements
We thank L. Quinti (MGH) for sharing preliminary results regarding 3D neuron/microglia co-culture systems, M. Busche (MGH) for helpful guidance regarding the calcium imaging, S.H. Choi (MGH) for helpful discussion regarding the data interpretation, and Y.J. Kang (UNCC) for critically reviewing our manuscript. This work was supported by the NIH/NIA (P01 AG015379 and RF1 AG048080 to D.Y.K. and R.E.T.; R01 AG014713 to D.Y.K.), the Pioneering Funding Award funded by Cure Alzheimer’s Fund (CAF; to H.C., D.Y.K., R.E.T.), BrightFocus Foundation (D.Y.K.), the Duke Energy Special Initiatives funded by Charlotte Research Institute (CRI; to H.C.), and the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2015R1A6A3A03019848, to J.P.).
Author information
Authors and Affiliations
Contributions
J.P. and H.C. designed, fabricated, and tested devices; designed experiments; performed immunostaining and statistical quantification; generated figures; and wrote and edited the manuscript. I.W. performed immunostaining and statistical quantification. C.D. generated human AD NPCs derived from iPSCs and measured APP and Aβ levels. D.D. and I.M. helped with confocal imaging and immunostaining, and wrote and edited the manuscript. D.Y.K., R.E.T., and H.C. conceived the ideas and directed the work, including all experiments and data analysis, and wrote and edited the manuscript. All authors read and edited the manuscript extensively.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Generation of FACS-sorted ReN cells with FAD mutations.
FACS sorting of ReN cell VM human neural stem (ReN) cells that were stably transfected with polycistronic GFP lentiviral vector. The cells were then enriched based on GFP and signals by FACS (above black-dotted lines, the selected ranges of cells for the experiments GFP, area intensity of GFP signal). ReN cells stably expressing GFP alone (ReN-G), APPSL-GFP (ReN-GA). (Numberdevice = 5; All experiments were repeated ≥ 3 times, Green: GFP; scale bars: 25 μm).
Supplementary Figure 2 3D reconstructed human AD neuron/astrocyte expressing GFP and APPSL mutation in microfluidic device.
The depth is 400 um. (Numberdevice = 5; All experiments were repeated ≥ 3 times, green: GFP; scale bars: 100 μm).
Supplementary Figure 3 3-week differentiated ReN cells with dendritic marker of MAP2 and dendritic spine formation.
(a) 3-week differentiated ReN cells immunstained with dendritic marker of MAP2 (yellow) with nucleus stain (blue). (b) dendritic spine formation observed in week 3 neuron cells that were stably transfected with polycistronic GFP (Numberdevice = 5 in a and b; All experiments were repeated ≥ 3 times, scale bars: 50 μm (a), 5 μm (b)).
Supplementary Figure 4 Astrocyte marker staining of S100, S100A6 and S100β at Week 6.
(Numberdevice = 5; All experiments were repeated ≥ 3 times, Scale bars: 100 μm).
Supplementary Figure 5 Focal plane of neurons and astrocytes used to measure the Ca2 + transients in the cell indicated by the red dotted line.
Somata of stained neurons had a characteristic ring-shape appearance with the surrounding cytoplasm (Numberdevice = 5; All experiments were repeated ≥ 3 times, Scale bar: 100 μm).
Supplementary Figure 6 Pie charts fractions of hyperactive neurons (red) in week 3 and week 9.
(Numberneurons = 50; All experiments were repeated ≥ 3 times at each condition, t = 2.31, d.f. = 7.71, P = 0.032).
Supplementary Figure 7 Released amount of Abeta40 and Abeta42 ratio in week 9.
Conditioned media from each individual device from 3D Neu + AC (blue), 2D Neu + AC AD (purple) and 3D Neu + AC AD (red) were measured and quantified by dividing Abeta40 or 42 at week0 by showing the released amount of soluble Abeta40 and Abeta42 in week 9. Graph showed increased amount of soluble Abeta40 in conditioned media while Abeta42 was slightly decreased by comparison with Week 0. Two-way ANOVA revealed significant differences in between 3D Neu + AC vs 3D Neu + AC AD and 2D Neu + AC AD vs 3D Neu + AC AD (all **p < 0.0001, F (2, 8) = 100.8), All experiments were repeated ≥ 3 times. Each data point is the average ± SEM. of five different devices.
Supplementary Figure 8 Analysis of SDS soluble Abeta levels in the enriched model by western blotting.
6E10-Abeta detected ~4 kDa a monomer band in the medium collected from the 3D Neu + AC, 2D Neu + AC AD and 3D Neu + AC AD cells. The 6E10 antibody also detected sAPP (~100 kDa), which would represent the amounts of full-length APPs in these cell lines (n = 3 per each sample).
Supplementary Figure 9 Aggregated for of pTau in AD neuron.
Phosphorylated tau (pTau) at Ser202/Thr206 was confirmed by immunofluorescent staining (pTau antibody AT8) in both neuritic and soma bodies of 3D Neu + AC + MG AD (Numberdevice = 5; All experiments were repeated ≥ 3 times, green: GFP, scale bars: 50 μm).
Supplementary Figure 10 Schematic representation for the definition of the recruitment index, R.I.
Microglia recruitment was quantified by comparing the fraction of cells inside the central chamber (CC) to the total number of cells in the corresponding angular chamber device (AC). The R.I. is calculated as R.I. = (CCDay i - CCDay 0) / ACDay i, where Day i is the number of cells in the central chamber and ADDay i is the number of cells in angular chamber. The recruitment index, R.I., is obtained by normalizing with the subtraction at ‘CCDay 0’ cell numbers.
Supplementary Figure 11 CCL2 dependent recruitment of microglial cells.
Microglial cell recruitment significantly reduced in AD neuron/astrocyte co-culture condition with neutralizing Anti-CCL2. Two-way ANOVA revealed significant differences in between 2D Neu + AC + MG AD vs 2D Neu + AC + MG AD + Anti-CCL2 (**p < 0.0020, F (1, 4) = 51.09) and 3D Neu + AC + MG AD vs 3D Neu + AC + MG AD + Anti-CCL2 (***p < 0.0007, F (1, 4) = 86.39), All experiments were repeated ≥ 3 times. Each data point is the average ± SEM. of five different devices.
Supplementary Figure 12 ATP measurement of different brain model.
Extracellular released ATP was measured from conditioned media in different type of brain model at week 0 and week 9. Two-way ANOVA revealed significant differences in between 3D Neu + AC vs 3D Neu + AC AD in week 9 (**p < 0.2389, F (1, 4) = 1.912) and 2D Neu + AC + MG vs 3D Neu + AC + MG AD in week 9 (***p < 0.0001, F (1, 4) = 732.3). (Numberdevice = 5; All experiments were repeated ≥ 3 times, blue: 3D Neu + AC, purple: 2D Neu + AC AD, red: 3D Neu + AC AD).
Supplementary Figure 13 Membrane kit assay for the cytokine measurement.
. 36 cytokine/chemokine were measured with different conditions 3D Neu + AC + MG, 3D Neu + AC AD and 3D Neu + AC + MG AD. (Numberdevice = 5, All experiments were repeated ≥ 3 times,).
Supplementary Figure 14 Axotomy mediated by microglia.
Time-lapse images showed that microglia (red) mediated neurite cleavage (green) in co-culture condition. (Numberdevice = 5; All experiments were repeated ≥ 3 times, Scale bar: 20 μm).
Supplementary Figure 15 Microglial cells are colocalized with 3D Neu + AC + MG AD showed large area disruption of neuron/astrocyte.
(Numberdevice = 5; All experiments were repeated ≥ 3 times, Scale bar: 100 μm).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–15
Supplementary Video 1
Reconstructed 3D confocal image
Supplementary Video 2
Whole device rotation
Supplementary Video 3
Calcium imaging of AD neuron/astrocyte
Supplementary Video 4
Calcium imaging of AD neuron/astrocyte in week 3
Supplementary Video 5
Calcium imaging of AD neuron/astrocyte in week 9
Supplementary Video 6
Microglial expansion
Supplementary Video 7
Microglia mediated axotomy
Supplementary Video 8
Microglia mediated neurite retraction
Rights and permissions
About this article
Cite this article
Park, J., Wetzel, I., Marriott, I. et al. A 3D human triculture system modeling neurodegeneration and neuroinflammation in Alzheimer’s disease. Nat Neurosci 21, 941–951 (2018). https://doi.org/10.1038/s41593-018-0175-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-018-0175-4
This article is cited by
-
Mutual interaction of neurons and astrocytes derived from iPSCs with APP V717L mutation developed the astrocytic phenotypes of Alzheimer’s disease
Inflammation and Regeneration (2024)
-
Modeling the neuroimmune system in Alzheimer’s and Parkinson’s diseases
Journal of Neuroinflammation (2024)
-
iPSC-derived PSEN2 (N141I) astrocytes and microglia exhibit a primed inflammatory phenotype
Journal of Neuroinflammation (2024)
-
Advanced patient-specific microglia cell models for pre-clinical studies in Alzheimer’s disease
Journal of Neuroinflammation (2024)
-
Neuropathogenesis-on-chips for neurodegenerative diseases
Nature Communications (2024)